- Home
- »
- Medical Devices
- »
-
North America Cardiac Leads Market, Industry Report, 2033GVR Report cover
![North America Cardiac Leads Market Size, Share & Trends Report]()
North America Cardiac Leads Market (2025 - 2033) Size, Share & Trends Analysis Report By Leads (Pacing Leads, Defibrillator Leads), By End Use (Hospitals, Ambulatory Surgery Centers), By Country (U.S., Canada, Mexico), And Segment Forecasts
- Report ID: GVR-4-68040-819-5
- Number of Report Pages: 120
- Format: PDF
- Historical Range: 2021 - 2024
- Forecast Period: 2025 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
North America Cardiac Leads Market Summary
The North America cardiac leads market size was estimated at USD 1.82 billion in 2024 and is projected to reach USD 2.52 billion by 2033, growing at a CAGR of 3.70% from 2025 to 2033. The rising prevalence of cardiovascular diseases (CVDs) in the U.S., including heart failure and arrhythmias, is being driven by multiple factors, notably sedentary lifestyles, obesity, and associated comorbidities.
Key Market Trends & Insights
- The U.S. cardiac leads market accounted for the largest share of 83.89% in North America in 2024.
- Based on leads, the defibrillator leads segment held the largest share in 2024.
- Based on end use, hospitals held the largest share in 2024.
Market Size & Forecast
- 2024 Market Size: USD 1.82 Billion
- 2033 Projected Market Size: USD 2.52 Billion
- CAGR (2025-2033): 3.70%
- U.S.: Largest market in 2024
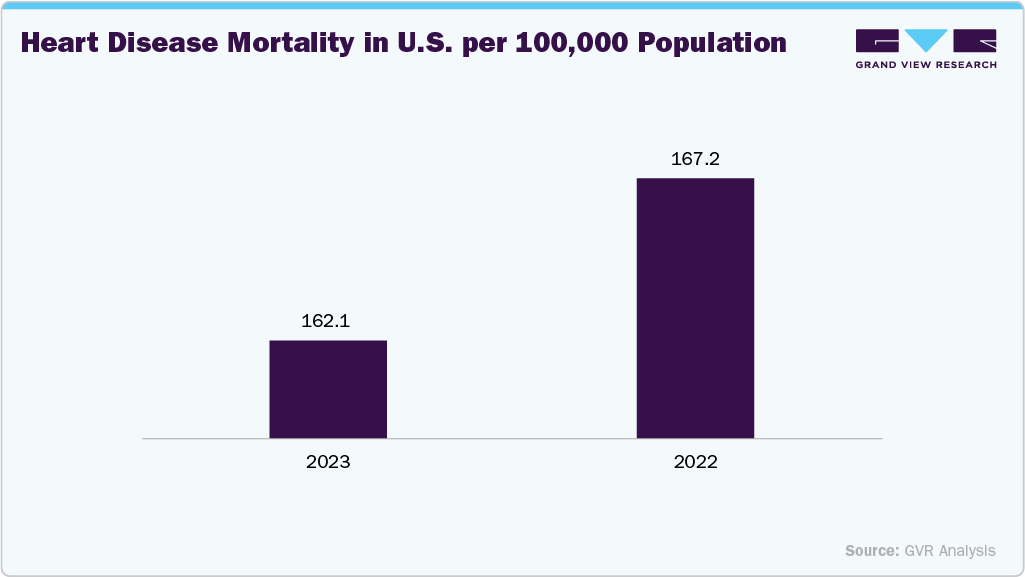
According to the America Heart Association, in January 2024, the U.S. reported 931,578 deaths linked to cardiovascular disease, nearly 3,000 higher than the previous year, with the age-adjusted death rate climbing to 233.3 per 100,000 people, reflecting a 4.0% increase. This escalating disease load is contributing to a higher number of patients requiring device-based cardiac interventions, thereby strengthening demand for pacing leads and defibrillator leads essential for effective rhythm management.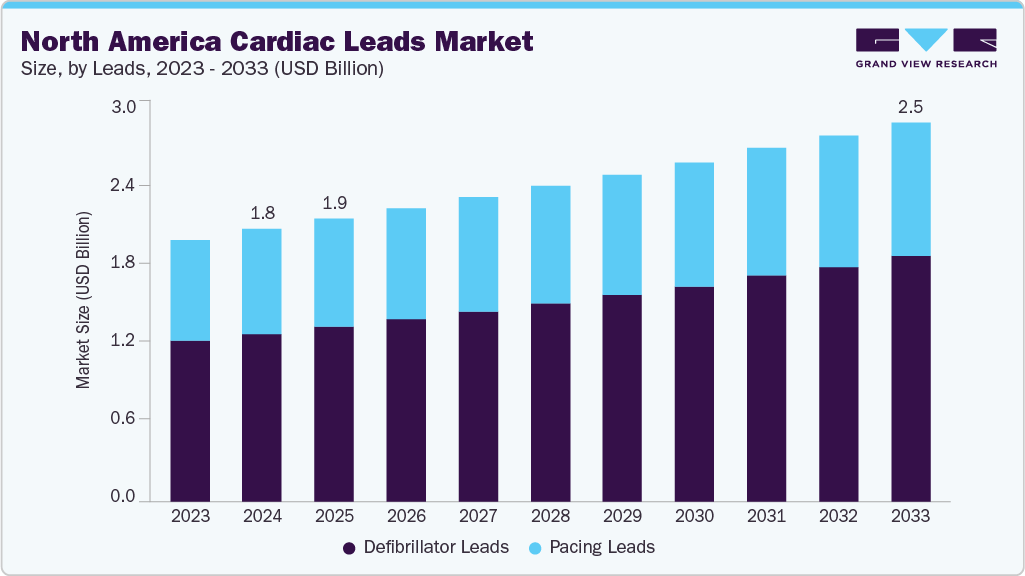
Atrial fibrillation continues to be widely underdiagnosed in the U.S., often progressing silently until symptoms become clinically significant. According to the American Medical Association article published in February 2025, more than 5 million American adults are estimated to be living with AFib, and this number is expected to exceed 12 million by 2030, reflecting a rapidly growing arrhythmia burden across the country. While the patient population expands, hospitals and electrophysiology centers are seeing elevated demand for device-based rhythm stabilization, including pacemakers, conduction-system pacing solutions, and implantable defibrillators. Since these systems rely on high-quality cardiac leads to ensure accurate sensing and consistent electrical therapy, the increasing prevalence of AFib is directly strengthening the need for advanced pacing and defibrillator lead technologies throughout the U.S. and broader North America cardiac leads industry.
Key Facts on Cardiovascular Disease and Related Events in the U.S. (2024)
Statistic
Value
Total CVD deaths per day
2,552
Deaths from heart disease per day (including heart attacks)
1,905
Average time between heart attacks
Every 40 seconds
New heart attacks each year
Approximately 605,000
Recurrent heart attacks each year
Approximately 200,000
Silent heart attacks
Estimated 170,000
Average age at first heart attack (males)
65.6 years
Average age at first heart attack (females)
72.0 years
Source: American Heart Association, Inc. in January 2024 & GVR
The rising prevalence of sedentary lifestyles across North America is intensifying the region’s cardiovascular disease burden, leading to higher incidences of heart failure, arrhythmias, and sudden cardiac events that necessitate device-based interventions. According to the Harvard Gazette article published in November 2024, research from Massachusetts General Hospital found that individuals spending over 10.6 hours per day in sedentary activities faced a 40-60% greater risk of heart failure and cardiovascular death, even when meeting recommended exercise levels.
This growing risk profile is translating into a larger patient pool requiring pacing and defibrillation therapies. Subsequently, demand for pacing leads, including standard right ventricular (RV) and left ventricular (LV) leads as well as advanced conduction system pacing (CSP) or left bundle branch area pacing (LBBAP) leads rising alongside increased implantation of defibrillator leads used in both traditional and LV configurations. While sedentary behavior continues to contribute to cardiac morbidity across North America, the expanding clinical adoption of pacemakers and implantable defibrillator systems is expected to drive sustained growth in the North America cardiac leads industry.

Technologically advanced product launch drives the growth of the market. For instance, in September 2024, Boston Scientific Corporation (NYSE: BSX) recently secured FDA approval for an expanded use of its INGEVITY + pacing leads, allowing them to support conduction system pacing (CSP) and sensing within the left bundle branch area (LBBA) when paired with single- or dual-chamber pacemakers. These leads ultra-thin wires designed to deliver electrical impulses from an implanted device to the heart can now be used in a more physiologic pacing strategy. Left bundle branch area pacing offers an alternative to conventional right ventricular pacing for patients with symptomatic bradycardia, a condition marked by abnormally slow heart rhythms. Placing the lead within the heart’s intrinsic conduction network, this method more closely replicates natural electrical activation, helping maintain coordinated ventricular contraction. Clinically, this approach has the potential to preserve heart function over time and may lessen the long-term risk of pacing-related heart failure compared with traditional right ventricular pacing.

The rising prevalence of heart disease management across the U.S. is strengthening the need for advanced cardiac monitoring and rhythm-support technologies, including high-performance cardiac leads. While national prevention programs enhance early detection of arrhythmias, conduction disorders, and other cardiac abnormalities, more patients are being identified at stages where device-based intervention becomes essential. This growing clinical focus on timely diagnosis and more effective long-term management is driving higher adoption of pacemakers, defibrillators, and conduction-system pacing solutions, thereby creating sustained demand for reliable, durable, and technologically advanced cardiac leads in the market.
Case Study:
-
“Performance of an active fixation stylet‑driven lead in left bundle branch area pacing: Results from INSIGHT‑LBBA”
Patient / Lead Scope:
-
According to the NCBI article published in August 2025, a 1,122 single‑ or dual‑chamber pacemaker patients in 8 U.S. sites (mean age ~76 years, 43% female) who received an active‑fixation stylet-driven lead for left bundle branch area pacing (LBBAP) using the lead model (stylet‑driven) under investigation.
Key Findings:
-
Success (left bundle or left septal capture) achieved in 95.6% of patients (1,073 of 1,122).
-
Lead-related complication-free (“LRC‑free”) rate at 3 months was 97.7% and remained ~97.2% at 24 months.
-
Electrical performance: At 3 months, 98.8% of pacing capture thresholds (PCT) were ≤2 V at 0.4 ms; 94.8% had sensed R‑wave amplitudes ≥5 mV.
-
External validation cohort (864 LBBAP implants) showed concordant results.
-
This is clearly focused on a pacing lead designed forconduction system pacing (CSP) / LBBAP, aligning with your “Pacing Leads → CSP/LBBAP Leads” sub‑segment.
-
The large U.S. cohort across multiple centres suggests meaningful real-world performance data in the North American context.
-
Because lead performance (success rate, thresholds, complication-free rates) is strong, it supports market arguments such as value proposition, adoption of advanced pacing leads, and technology differentiation.
Considerations:
-
Although commercial regulatory indication status isn’t detailed here, the lead under study is designed explicitly for LBBAP - indicating a shift in the lead market toward CSP.
-
The data are robust, but follow-up in many cases is relatively short (median ~302 days in the study). Long-term durability (5‑- 10 years) has yet to be established.
Key Facts on Cardiovascular Disease and Related Events in the U.S. (2024)
Key Case‑Study Factors
Market Impact
Conducted at multiple U.S. centers using active‑fixation stylet‑driven leads for left bundle branch area pacing (LBBAP).
Confirms the reliability and effectiveness of CSP/LBBAP pacing leads, supporting adoption in the North American industry.
Demonstrated high lead success and stable performance during follow‑up.
Builds clinical trust in pacing leads, encouraging hospital and clinician preference, driving market growth.
Focused on the U.S. patient population with commercially relevant leads.
Provides region‑specific evidence for North America, strengthening market forecasts and supporting investment in advanced leads.
Source: American Heart Association, Inc. in January 2024 & GVR
Market Concentration & Characteristics
The industry exhibits a significant degree of innovation, driven by advancements in defibrillator leads and pacing leads. For instance, in April 2025, Medtronic introduced the OmniaSecure lumenless defibrillation lead, featuring a smaller diameter and advanced placement capabilities in physiological locations like the LBB area. This reduces device footprint, enhances patient safety, and improves therapy delivery for both traditional defibrillator and LV lead applications.
Companies in North America actively pursue mergers, acquisitions, and strategic partnerships at a medium level to strengthen technological capabilities and market reach. For instance, in November 2024, Merit Medical Systems completed the acquisition of Cook Medical’s lead‑management portfolio (devices and accessories for pacemaker/defibrillator lead removal/replacement) for approximately USD 210 million.
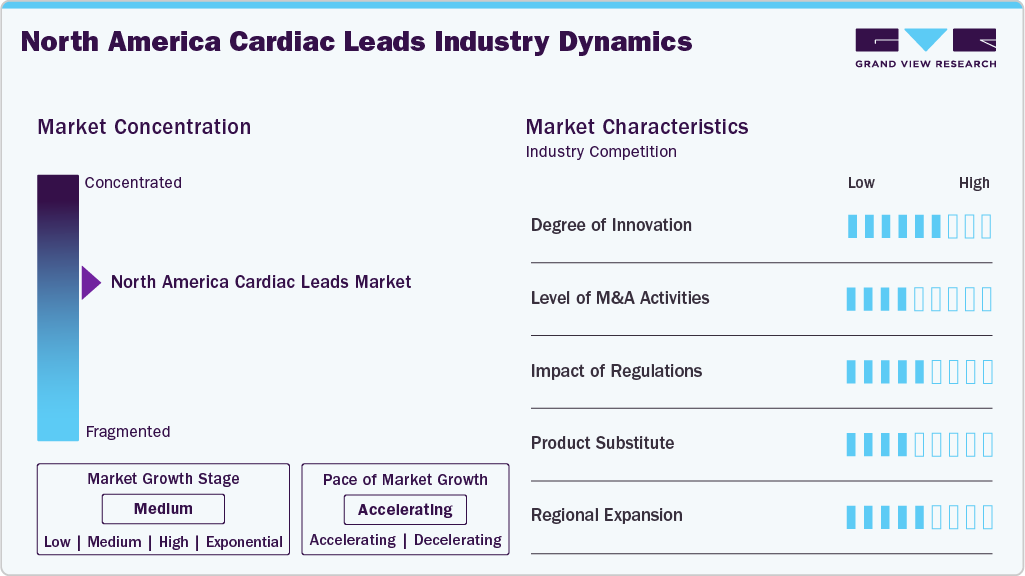
The industry is governed by stringent regulatory frameworks, primarily led by the U.S. Food & Drug Administration (FDA). Compliance with these regulations ensures device safety, clinical efficacy, and cybersecurity, while also influencing development costs, clinical trial requirements, and market entry timelines. Regulatory oversight covers all lead sub-segments, including pacing leads (standard RV/LV and conduction system pacing/LBBAP) and defibrillator leads (traditional and LV leads). Agencies are increasingly accommodating emerging technologies, such as AI-enabled diagnostics, remote monitoring, and advanced device designs, which can streamline clinical workflows and enhance patient care. Adherence to regulatory standards is key for maintaining market approval, ensuring product reliability, and facilitating the adoption of next-generation cardiac leads in North America.
Key players in the market are actively expanding their product portfolios to address the growing demand for advanced cardiac rhythm management therapies. Manufacturers are introducing innovations such as miniaturized pacing leads, conduction system pacing (CSP) / left bundle branch area pacing (LBBAP) leads, and lumenless defibrillator leads, as well as MRI-compatible and leadless designs. These product expansions aim to improve patient outcomes, enhance procedural efficiency, and provide more physiologic pacing options for a broader range of patients.
Key players in the industry are strategically expanding within the U.S. and Canada at medium strength to capture growing demand and address rising cardiovascular disease prevalence. Expansion initiatives focus on enhancing distribution networks, clinician training programs, and remote patient monitoring services to improve access to therapy, adherence, and clinical outcomes. Companies are also introducing advanced pacing and defibrillator leads, including standard RV/LV, CSP/LBBAP, and traditional/LV defibrillator leads, to meet diverse patient needs and support the adoption of next-generation cardiac rhythm management solutions across North America.
Product Insights
By Leads, the defibrillator leads segment accounted for the largest revenue share of 61.29% in 2024 and is expected to grow at the fastest CAGR over the forecast period. Segment growth is driven by increasing CVD incidence and the launch of technologically advanced products. For instance, in October 2023, Medtronic plc, a key global healthcare technology company, obtained approval from the U.S. Food and Drug Administration (FDA) for its Aurora EV-ICD MRI SureScan (Extravascular Implantable Cardioverter-Defibrillator) and the Epsila EV MRI SureScan defibrillation lead. These devices are designed to treat life-threatening fast heart rhythms that can result in sudden cardiac arrest (SCA). The Aurora EV-ICD system is a first-of-its-kind solution that combines the protective benefits of traditional transvenous ICDs with a lead positioned under the breastbone, outside the heart and veins. The system delivers essential therapies, including defibrillation, anti-tachycardia pacing (ATP), and backup pacing to prevent pauses, all through a device comparable in size, shape, and durability to conventional transvenous ICDs.
The pacing leads segment is expected to grow at a significant CAGR over the forecast period. Segment growth is driven by the increasing prevalence of cardiovascular diseases, particularly arrhythmias and heart failure, as well as the introduction of technologically advanced products. For instance, in September 2024, BIOTRONIK recently secured U.S. FDA labeling approval for its Selectra 3D delivery catheter when paired with the Solia S pacing lead, formally authorizing the system for use in left bundle branch area pacing (LBBAP).

This milestone marks the first FDA-approved stylet-driven pacing lead and dedicated catheter combination explicitly designed for conduction system pacing in the U.S. market. The Solia S lead and Selectra 3D system have already been widely adopted globally, and the newly expanded approval supports their use in procedures that aim to activate the heart’s native conduction pathways, offering a more physiologic alternative to traditional right-ventricular pacing. This development enhances the availability of advanced CSP/LBBAP-focused pacing options across North America, reflecting the growing clinical demand for next-generation conduction-system pacing solutions.
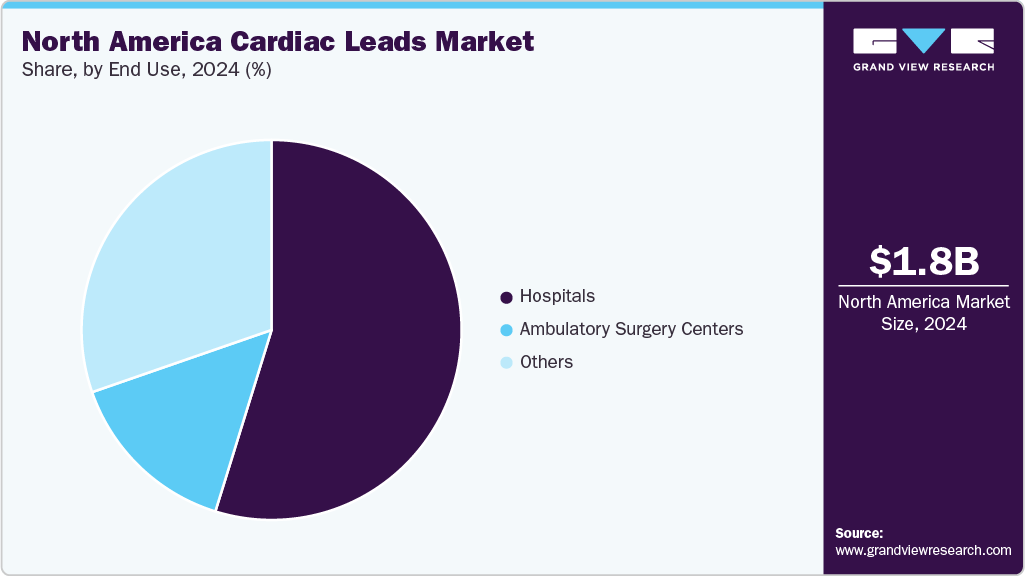
End Use Insights
By end use, the hospitals segment accounted for the largest revenue share of 54.79% in 2024. Hospitals are the primary setting for the implantation and management of cardiac leads in North America. These facilities provide the specialized infrastructure of electrophysiology labs, device implantation teams, and post‑operative monitoringthat is necessary for procedures involving pacing leads (standard RV/LV and CSP/LBBAP) and defibrillator leads (traditional and LV). Hospitals also serve as early adopters of advanced medical technologies and play a key role in physician training, device follow-up programs, and managing repeat or upgrade procedures.
U.S. Hospital Database 2024
Hospital Category
Number of Hospitals
Total Number of All U.S. Hospitals
6,093
Number of U.S. Community Hospitals
5,112
Number of Nongovernment Not-for-Profit Community Hospitals
2,978
Number of Investor-Owned (For-Profit) Community Hospitals
1,214
Number of State and Local Government Community Hospitals
920
Number of Federal Government Hospitals
207
Number of Nonfederal Psychiatric Hospitals
654
Other Hospitals
120
Source: American Hospital Association & GVR
The ambulatory surgery centers segment is expected to grow at the fastest CAGR over the forecast period. Ambulatory Surgery Centers (ASCs) are becoming increasingly crucial in cardiac rhythm management, particularly for patients undergoing low-risk procedures such as pacemaker or loop recorder implantations. The shift toward ASCs is part of a broader effort to reduce the load on inpatient hospital services, lower procedural costs, and maintain high-quality clinical outcomes. These facilities typically manage stable patients and rely on minimally invasive techniques, supported by portable ECG systems and device programming tools, which enable the efficient implantation of cardiac devices and the performance of diagnostic procedures. For instance, in February 2024, the American College of Cardiology launched its CV ASC Registry Suite to monitor the growing volume of cardiac procedures conducted in outpatient centers. The registry tracks interventions, including diagnostic catheterizations, as well as pacemaker and defibrillator implantations, providing critical insights into procedural trends and outcomes in the ASC setting.
Top 5 U.S. States by Number of Ambulatory Surgical Centers (ASCs) in 2025
Sr
State
Number of ASCs
1
California
894
2
Florida
517
3
Texas
497
4
Georgia
423
5
Maryland
347
Source: Ambulatory Surgery Center Association & GVR
Country Insights
The U.S. cardiac leads market dominated the region with a share of 83.89% in 2024. This growth is driven by the country’s robust healthcare infrastructure, a rising elderly population, and the rapid adoption of advanced, technology-driven products. Innovations such as extended battery life, biocompatible materials, miniaturized designs, and leadless systems, along with a growing number of regulatory approvals, have further fueled market expansion. According to the CDC article published in October 2024, in the U.S., cardiovascular disease remains a leading public health concern, claiming a life approximately every 34 seconds. This highlights the growing burden of cardiac conditions such as arrhythmias, heart failure, and coronary artery disease, which collectively place significant pressure on the healthcare system. These trends underscore the rising need for advanced cardiac devices and associated leads to manage heart rhythm disorders effectively.
Canada Cardiac Leads Market Trends
The Canada cardiac leads market is expected to grow over the forecast period. This expansion is primarily driven by the increasing incidence of cardiovascular disease (CVD) in the country. According to a Government of Canada report published in October 2024, cardiovascular diseases remain a significant public health concern, ranking as the second leading cause of death after cancer. Currently, approximately 2.4 million Canadians live with heart-related conditions, placing considerable pressure on the healthcare system. The high prevalence of cardiac disorders not only contributes significantly to the nation’s overall disease burden but also represents a leading cause of years of life lost, underscoring the rising demand for advanced cardiac devices and associated leads to manage arrhythmias and other heart rhythm disorders effectively.
Mexico Cardiac Leads Market Trends
The Mexico cardiac leads market is growing over the forecast period, driven by the rising incidence of cardiovascular disease (CVD) in the country. According to a Springer Nature Limited report published in January 2025, approximately 82% of CVD-related deaths in Mexico are associated with unhealthy lifestyle habits, alongside other contributing factors. Studies also indicate that individuals with even a single CVD risk factor have a 23% higher risk of mortality compared to those with no risk factors, regardless of age, gender, socioeconomic status, or comorbidities. This high prevalence of heart-related conditions highlights the increasing demand for effective cardiac interventions, including advanced cardiac devices and associated leads, to manage arrhythmias and other rhythm disorders, improve cardiac function, and enhance patient outcomes across the Mexican population.
Key North America Cardiac Leads Company Insights
Key players operating in the North America cardiac leads market are undertaking various initiatives to strengthen their presence and increase the reach of their products and services. Strategies such as expansion activities and partnerships are key in propelling the market growth.
Key Cardiac Leads Companies:
- Abbott
- Medtronic
- Boston Scientific Corporation
- Biotronik
- Schiller
- Koninklijke Philips N.V.
- MicroPort Scientific Corporation
- Stryker
- Merit Medical Systems
- Lepu Medical Technology (Beijing) Co., Ltd.
Recent Developments
-
In September 2025, BIOTRONIK launched Solia CSP S, a pacing lead combining a fixed screw design with a stylet-driven implantation approach. It is designed specifically for conduction system pacing (CSP) to simplify implant handling while maintaining optimal outcomes.
-
In May 2024, Boston Scientific Corporation announced positive six-month data from the MODULAR ATP trial of its mCRM System. The system combines a leadless pacemaker with a subcutaneous ICD to deliver anti-tachycardia pacing without transvenous leads. Results met safety and efficacy endpoints, with strong signal communication and no ATP-related discomfort.
-
In March 2024, MicroPort Scientific Corporation completed the first U.S. implantation of the ALIZEA Bluetooth-enabled pacemaker system at PIH Health Downey Hospital. The device, featuring 13-year projected longevity and AutoMRI mode, was paired with VEGA leads and supported by remote monitoring via SmartView Connect.
-
In February 2023, Abbott entered into a definitive agreement to acquire Cardiovascular Systems, Inc., a medical device company.
North America Cardiac Leads Market Report Scope
Report Attribute
Details
Market size value in 2025
USD 1.88 billion
Revenue forecast in 2033
USD 2.52 billion
Growth rate
CAGR of 3.70% from 2025 to 2033
Actual Data
2021 - 2024
Forecast period
2025 - 2033
Quantitative units
Revenue in USD million/billion and CAGR from 2025 to 2033
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Leads, end use, country
Regional scope
North America
Country scope
U.S.; Canada; Mexico
Key companies profiled
Abbott; Medtronic; Boston Scientific Corporation; Biotronik; Schiller; Koninklijke Philips N.V.; MicroPort Scientific Corporation; Stryker; Merit Medical Systems, Lepu Medical Technology (Beijing) Co., Ltd.
Customization scope
Free report customization (equivalent up to 8 analysts working days) with purchase. Addition or alteration to country, regional & segment scope.
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options North America Cardiac Leads Market Report Segmentation
This report forecasts revenue growth at the regional and country levels and provides an analysis of the latest industry trends in each sub-segment from 2021 to 2033. For this study, Grand View Research has segmented the North America cardiac leads market report based on lead, end use, and region:
-
Leads Outlook (Volume in ‘000 Units; Revenue in USD Million, 2021 - 2033)
-
Pacing Leads
-
Standard RV/LV Leads
-
Conduction System Pacing (CSP) / LBBAP Leads
-
-
Defibrillator Leads
-
Traditional Leads
-
LV Leads
-
-
-
End Use Outlook (Volume in ‘000 Units; Revenue in USD Million, 2021 - 2033)
-
Hospitals
-
Ambulatory Surgery Centers
-
Others
-
-
Country Outlook (Volume in ‘000 Units; Revenue in USD Million, 2021 - 2033)
-
U.S.
-
Canada
-
Mexico
-
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










