- Home
- »
- Clinical Diagnostics
- »
-
North America Point Of Care Diagnostics Market Size Report, 2030GVR Report cover
![North America Point Of Care Diagnostics Market Size, Share & Trends Report]()
North America Point Of Care Diagnostics Market (2023 - 2030) Size, Share & Trends Analysis Report By Product (Infectious Diseases, Glucose Testing, Cardiac Markers), By Type, By End-use (Clinics, Home Care, Hospitals), By Country, And Segment Forecasts
- Report ID: 978-1-68038-967-8
- Number of Report Pages: 180
- Format: PDF
- Historical Range: 2018 - 2021
- Forecast Period: 2023 - 2030
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
Report Overview
The North America point of care diagnostics market size was valued at USD 17.53 billion in 2022 and is expected to expand at a compound annual growth rate (CAGR) of 6.2% from 2023 to 2030. Point-of-care (POC) diagnosis refers to performing diagnostic tests at the site of the patient. The growing geriatric population and the ability of POC diagnostic tests to deliver immediate results are likely to fuel the demand for POC diagnostic tests. Furthermore, a rise in funding from the government & private institutions is a key factor driving the market’s growth. An increase in the adoption of mobile diagnostic devices in the region is also one of the key factors driving demand for POC testing.
The COVID-19 outbreak has positively impacted the market. This can primarily be attributed to the urgent need for manufacturing and introduction of rapid & portable diagnostic tests, systems, and accessories in the market that can presently assist in scaling up of COVID-19 testing. Moreover, the advent of rapid data-sharing platforms and solutions is expected to contribute to the development of COVID-19 point-of-care technologies, supplementing the market growth. Companies are actively working toward the development of rapid testing solutions. For instance, in November 2022, Sense Biodetection entered into a collaboration with Bio Nuclear Diagnostics to distribute Sense’s Veros POC, an instrument-free molecular COVID-19 test, in Canada.
Furthermore, increased funding from NIH, private foundations such as the Bill & Melinda Gates Foundation, and the U.S. department of defense (DOD), is anticipated to fuel the market growth. For instance, in November 2021, Hyperfine, Inc., announced receiving USD 3.3 million in funding from the Bill & Melinda Gates Foundation to broaden the usage of portable MRI technology in various countries. Similarly, in August 2022, Nanopath, Inc. received funding of USD 10 million from Medtech Convergence Fund and Norwest Venture Partners with participation from Green D Ventures and Gingerbread Capital. The funding would help develop point-of-care diagnostics for women’s health.
In the U.S., the population aged 65 and older is estimated to double from 52 million in 2018 to 95 million by 2060, which is likely to increase demand for POC diagnostics. According to projections, there will be a 23% increase in the overall population. Aging increases the risk of diseases such as cardiovascular diseases and cancer. Therefore, an increase in the geriatric population is expected to boost the demand for continuous monitoring via facilities requiring point-of-care diagnostics, such as home healthcare and assisted living healthcare facilities.
Product Insights
The infectious diseases segment accounted for 25.3% of the total market share in 2022, owing to increasing cases of dengue fever, enteric, malaria, syphilis, tuberculosis, and others in the region. The significant demand for rapid tests has encouraged market players to offer point-of-care solutions to decentralized regions. For instance, in March 2020, Abbott launched a molecular POC test that could detect the presence of coronavirus in less than five minutes.
The cardiac markers segment is anticipated to witness the fastest CAGR in the coming years owing to due to increase in acceptance of sensitivity and premium-priced POC Troponin testing. The growing trend toward value-based and efficient healthcare service delivery, along with stringent finances, is strengthening the market for point-of-care cardiac diagnostics.Furthermore, in emergency care settings, the majority of medical professionals prefer POC cardiac testing to monitor the state of patients experiencing chest discomfort or other heart illness symptoms.
End-use Insights
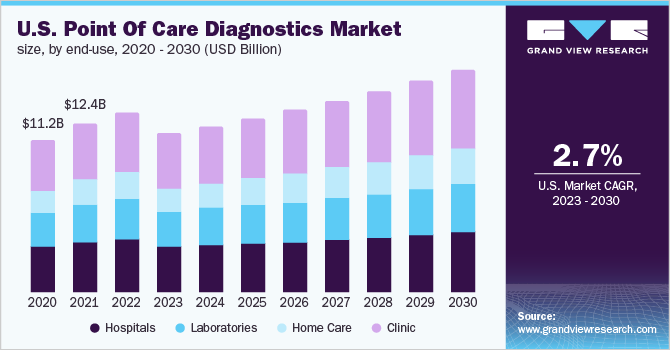
The hospitals segment dominated the North America point-of-care diagnostics market in 2022, capturing a revenue share of 37.68%. POC is beneficial for healthcare personnel as it provides mobility to the diagnosis system and allows reaching patients effectively. The development of wireless communication and miniaturized devices is a boon for hospital-based POC diagnostics as these provide single-level access throughout the hospital.
The laboratories segment is projected to witness the fastest growth during the forecast period owing to the rise in the need for early identification of many diseases. In addition, each year, more than 7 billion clinical laboratory examinations are performed in the U.S., as per an article published by the American Clinical Laboratory Association. The market is predicted to increase due to the presence of a strong distribution network of companies, such as Quest Diagnostics Incorporated, Siemens Healthineers, Laboratory Corporation of America Holdings, and Abbott, among others.
Type Insights
The POC devices that are developed have different medical diagnostic applications, such as cancer, infectious diseases, and pregnancy. Physicians& patients use POC tests to screen diseases, confirm diagnoses, and design suitable therapeutic approaches based on the patient’s health. For instance, in July 2020, BD launched a portable, Rapid POC Antigen Test which detects COVID-19 infection within 15 minutes. It has expanded access to COVID-19 testing.
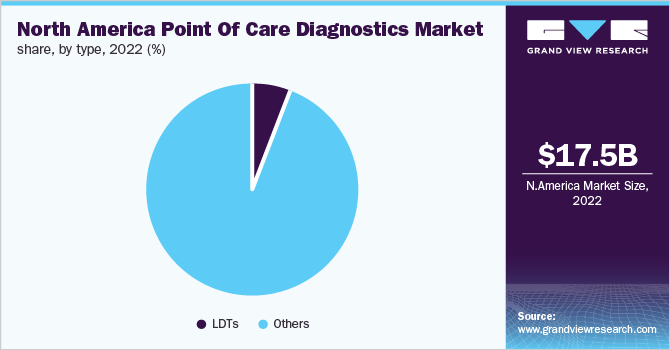
The LDT segment is anticipated to witness the fastest growth rate in the coming years with a CAGR of 6.2%. Laboratory-developed tests are testing services developed in patient care at clinical laboratories, academics, and hospitals. These services are not commercially marketed and manufactured, but are developed, performed, designed, validated, and interpreted by certified professionals in a laboratory. These are used to meet clinical demands and are instrumental for early diagnosis, monitoring, and guidance of patient treatment.
Country Insights
The U.S. dominated the North America point-of-care market in 2022 due to the presence of key players such as Abbott, BIOMERIEUX, BD, Siemens Healthineers AG, QIAGEN, Quidel Corporation, and Quest Diagnostics is positively influencing the market growth. For instance, in November 2020, in the U. S., QIAGEN started offering a portable digital test that allows labs to detect SARS-CoV-2 antigens in persons with current illnesses in 2 to 15 minutes.
Canada is estimated to witness the fastest growth rate over the coming years. Usage of POC diagnostic devices in these regions is increasing owing to the increasing prevalence of target diseases such as cardio-metabolic disorders, infectious diseases, and increasing cases of drug abuse. Increasing demand for rapid and early diagnosis leading to better treatment alternatives and presence of health-conscious population are also considered as other factors fueling industrial growth. People in this region are also shifting their focus towards self-testing devices as compared to frequent laboratory testing visits, thus influencing industrial growth.
Key Companies & Market Share Insights
Key companies operating in the POC diagnostics market are focusing on strategic partnerships, collaborations, and geographical expansion, in economically and emerging favorable regions. For Instance in May 2020, Nova Biomedical received approval from Health Canada for its StatStrip Glucose Hospital Meter System to be used in hospitals and other healthcare settings, as well as intensive care units. The StatStrip Glucose & StatStrip Glucose or Ketone meters and test strips are POC glucose monitoring systems designed to detect & monitor dysglycemia in hospitals. Some prominent players in the North America point of care diagnostics market include:
-
F. Hoffmann-La Roche AG
-
QIAGEN
-
Danaher Corporation
-
BD
-
bioMérieux SA
-
Abbott
-
Siemens Healthineers A.G.
-
Zoetis, Inc.
-
Instrumentation Laboratory
-
Nova Biomedical
-
Trividia Health, Inc.
-
Quidel Corporation
-
Trinity Biotech
-
Sekisui Diagnostics
-
OraSure Technologies, Inc.
-
NIPRO
-
Spectral Medical, Inc.
North America Point Of Care Diagnostics Market Report Scope
Report Attribute
Details
Market size value in 2023
USD 19.01 billion
Revenue forecast in 2030
USD 28.46 billion
Growth rate
CAGR of 6.2% from 2023 to 2030
Base year for estimation
2022
Historical data
2018 - 2021
Forecast period
2023 - 2030
Quantitative units
Revenue in USD million and CAGR from 2023 to 2030
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Product, end-use, type, country
Country scope
U.S., Canada
Key companies profiled
F. Hoffmann-La Roche AG; QIAGEN; Danaher Corporation; BD; bioMérieux SA; Abbott; Siemens Healthineers A.G.; Zoetis, Inc.; Instrumentation Laboratory; Nova Biomedical; Trividia Health, Inc.; Quidel Corporation; Trinity Biotech; Sekisui Diagnostics; OraSure Technologies, Inc.; NIPRO, Spectral Medical, Inc.
Customization scope
Free report customization (equivalent up to 8 analyst’s working days) with purchase. Addition or alteration to country, regional & segment scope.
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
North America Point Of Care Diagnostics Market Segmentation
This report forecasts revenue growth at the regional and country levels and provides an analysis of the latest industry trends in each of the sub-segments from 2018 to 2030. For this study, Grand View Research has segmented the North America point of care diagnostics market report based on product, end-use, type, and country:
-
Product Outlook (Revenue, USD Million, 2018 - 2030)
-
Glucose Testing
-
By End-use
-
Hospitals
-
Laboratories
-
Home Care
-
Clinic
-
Physician Office
-
Pharmacy & Retail Clinics
-
Non-practice Clinics
-
Urgent Care Clinics
-
Others
-
Rural Health Clinic
-
Federally Qualified Health Clinic (FQHC)
-
-
-
-
By Disease
-
Diabetes
-
Others
-
-
-
HbA1c Testing
-
By End-use
-
Hospitals
-
Laboratories
-
Home Care
-
Clinic
-
Physician Office
-
Pharmacy & Retail Clinics
-
Non-practice Clinics
-
Urgent Care Clinics
-
Others
-
Rural Health Clinic
-
Federally Qualified Health Clinic (FQHC)
-
-
-
-
By Disease
-
Diabetes
-
Others
-
-
Coagulation
-
By End-use
-
Hospitals
-
Laboratories
-
Home Care
-
Clinic
-
Physician Office
-
Pharmacy & Retail Clinics
-
Non-practice Clinics
-
Urgent Care Clinics
-
Others
-
Rural Health Clinic
-
Federally Qualified Health Clinic (FQHC)
-
-
-
-
By Disease
-
Thrombosis
-
Hemophilia
-
Others
-
-
-
Fertility/Pregnancy
-
By End-use
-
Hospitals
-
Laboratories
-
Home Care
-
Clinic
-
Physician Office
-
Pharmacy & Retail Clinics
-
Non-practice Clinics
-
Urgent Care Clinics
-
Others
-
Rural Health Clinic
-
Federally Qualified Health Clinic (FQHC)
-
-
-
-
By Disease
-
LH- Ovulation Elite Test
-
HCG- Pregnancy Tests
-
Fertility Tests
-
Others
-
-
-
Infectious Diseases
-
By End-use
-
Hospitals
-
Laboratories
-
Home Care
-
Clinic
-
Physician Office
-
Pharmacy & Retail Clinics
-
Non-practice Clinics
-
Urgent Care Clinics
-
Others
-
Rural Health Clinic
-
Federally Qualified Health Clinic (FQHC)
-
-
-
-
By Disease
-
HIV POC
-
Clostridium difficile POC
-
HBV POC
-
Pneumonia or Streptococcus-associated infections
-
Respiratory syncytial virus (RSV) POC
-
HPV POC
-
Influenza/Flu POC
-
HCV POC
-
MRSA POC
-
TB and drug-resistant TB POC
-
HSV POC
-
COVID-19
-
Other Infectious Diseases
-
-
-
Cardiac Markers
-
By End-use
-
Hospitals
-
Laboratories
-
Home Care
-
Clinic
-
Physician Office
-
Pharmacy & Retail Clinics
-
Non-practice Clinics
-
Urgent Care Clinics
-
Others
-
Rural Health Clinic
-
Federally Qualified Health Clinic (FQHC)
-
-
-
-
By Disease
-
Acute Coronary Syndrome
-
Myocardial Infarction
-
Congestive Heart Failure
-
Others
-
-
By Marker
-
Troponin
-
CK-MB
-
Myoglobin
-
BNP and NT-proBNP
-
Others
-
-
-
Thyroid Stimulating Hormone
-
By End-use
-
Hospitals
-
Laboratories
-
Home Care
-
Clinic
-
Physician Office
-
Pharmacy & Retail Clinics
-
Non-practice Clinics
-
Urgent Care Clinics
-
Others
-
Rural Health Clinic
-
Federally Qualified Health Clinic (FQHC)
-
-
-
-
By Disease
-
Hyperthyroidism
-
Hypothyroidism
-
Hematology
-
-
By End-use
-
Hospitals
-
Laboratories
-
Home Care
-
Clinic
-
Physician Office
-
Pharmacy & Retail Clinics
-
Non-practice Clinics
-
Urgent Care Clinics
-
Others
-
Rural Health Clinic
-
Federally Qualified Health Clinic (FQHC)
-
-
-
-
By Disease
-
Anemia
-
Leukemia
-
Others
-
-
-
Primary Care Systems
-
By End-use
-
Hospitals
-
Laboratories
-
Home Care
-
Clinic
-
Physician Office
-
Pharmacy & Retail Clinics
-
Non-practice Clinics
-
Urgent Care Clinics
-
Others
-
Rural Health Clinic
-
Federally Qualified Health Clinic (FQHC)
-
-
Decentralized Clinical Chemistry
-
-
-
By End-use
-
Hospitals
-
Laboratories
-
Home Care
-
Clinic
-
Physician Office
-
Pharmacy & Retail Clinics
-
Non-practice Clinics
-
Urgent Care Clinics
-
Others
-
Rural Health Clinic
-
Federally Qualified Health Clinic (FQHC)
-
-
-
-
-
Feces
-
By End-use
-
Hospitals
-
Laboratories
-
Home Care
-
Clinic
-
Physician Office
-
Pharmacy & Retail Clinics
-
Non-practice Clinics
-
Urgent Care Clinics
-
Others
-
Rural Health Clinic
-
Federally Qualified Health Clinic (FQHC)
-
-
-
-
By Disease
-
Colorectal Cancer
-
GI Disorders
-
Others
-
-
-
Lipid Testing
-
By End-use
-
Hospitals
-
Laboratories
-
Home Care
-
Clinic
-
Physician Office
-
Pharmacy & Retail Clinics
-
Non-practice Clinics
-
Urgent Care Clinics
-
Others
-
Rural Health Clinic
-
Federally Qualified Health Clinic (FQHC)
-
-
-
-
By Disease
-
Hyperlipidemia
-
Cardiovascular Diseases
-
Others
-
-
-
Cancer Marker
-
By End-use
-
Hospitals
-
Laboratories
-
Home Care
-
Clinic
-
Physician Office
-
Pharmacy & Retail Clinics
-
Non-practice Clinics
-
Urgent Care Clinics
-
Others
-
Rural Health Clinic
-
Federally Qualified Health Clinic (FQHC)
-
-
-
-
By Disease
-
Breast Cancer
-
Lung Cancer
-
Colon Cancer
-
Prostate Cancer
-
Colorectal Cancer
-
Skin Cancer
-
Kidney Cancer
-
Bladder Cancer
-
Others
-
-
By Marker
-
ALK gene rearrangements and overexpression
-
Alpha-fetoprotein (AFP)
-
BCL2 gene rearrangement
-
Bladder Tumor Antigen (BTA)
-
BRCA1 and BRCA2 gene mutations
-
BRAF V600 mutations
-
CA15-3/CA27.29
-
Carcinoembryonic antigen (CEA)
-
PSA
-
Others
-
-
-
Blood Gas/Electrolytes
-
By End-use
-
Hospitals
-
Laboratories
-
Home Care
-
Clinic
-
Physician Office
-
Pharmacy & Retail Clinics
-
Non-practice Clinics
-
Urgent Care Clinics
-
Others
-
Rural Health Clinic
-
Federally Qualified Health Clinic (FQHC)
-
-
-
-
-
Ambulatory Chemistry
-
By End-use
-
Hospitals
-
Laboratories
-
Home Care
-
Clinic
-
Physician Office
-
Pharmacy & Retail Clinics
-
Non-practice Clinics
-
Urgent Care Clinics
-
Others
-
Rural Health Clinic
-
Federally Qualified Health Clinic (FQHC)
-
-
-
-
-
Drug Abuse Testing
-
By End-use
-
Hospitals
-
Laboratories
-
Home Care
-
Clinic
-
Physician Office
-
Pharmacy & Retail Clinics
-
Non-practice Clinics
-
Urgent Care Clinics
-
Others
-
Rural Health Clinic
-
Federally Qualified Health Clinic (FQHC)
-
-
-
-
-
Urinalysis/Nephrology
-
By End-use
-
Hospitals
-
Laboratories
-
Home Care
-
Clinic
-
Physician Office
-
Pharmacy & Retail Clinics
-
Non-practice Clinics
-
Urgent Care Clinics
-
Others
-
Rural Health Clinic
-
Federally Qualified Health Clinic (FQHC)
-
-
-
-
By Disease
-
UTI
-
Kidney Disease
-
Diabetes
-
Others
-
-
-
-
-
End-use Outlook (Revenue, USD Million, 2018 - 2030)
-
Hospitals
-
Laboratories
-
Home Care
-
Self-testing
-
Self-collection
-
-
Clinic
-
Physician Office
-
Pharmacy & Retail Clinics
-
Non-practice Clinics
-
Urgent Care Clinics
-
Others
-
Rural Health Clinic
-
Federally Qualified Health Clinic (FQHC)
-
-
-
-
Type Outlook (Revenue, USD Million, 2018 - 2030)
-
LDTs
-
Others
-
-
Country Outlook (Revenue, USD Million, 2018 - 2030)
-
U.S.
-
Canada
-
Frequently Asked Questions About This Report
b. The North America point of care diagnostics market size was estimated at USD 17.53 billion in 2022 and is expected to reach USD 19.01 billion in 2020.
b. The North America point of care diagnostics market is expected to grow at a compound annual growth rate of 6.2% from 2023 to 2030 to reach USD 28.46 billion by 2030.
b. Infectious diseases segment dominated the North America point of care diagnostics market with a share of 27.8% in 2022. This is attributable to rising cases of infectious diseases in the region coupled with growing R&D activities for the infectious disease diagnosis.
b. Some key players operating in the North America point-of-care diagnostics market include Spectral Diagnostics, Abbott Laboratories, Nova Biomedical, Abaci’s Inc., Siemens Healthcare, Danaher Corporation, Instrumentation Laboratory, Trinity Biotech, Qiagen N.V., BioMerieux, Johnson & Johnson, Roche Diagnostics, Alere Inc., and Nipro Diagnostics
b. Key factors that are driving the market growth include rising incidence rate of chronic and infectious diseases coupled with increased funding from government and private institutes for research and development activities.
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










