- Home
- »
- Medical Devices
- »
-
Pulmonary Drug Delivery Systems Market Report Size, 2030GVR Report cover
![Pulmonary Drug Delivery Systems Market Size, Share & Trends Report]()
Pulmonary Drug Delivery Systems Market (2024 - 2030) Size, Share & Trends Analysis Report By Product (Inhalers, Nebulizers, Accessories), By Application (Asthma, Cystic Fibrosis), By End-use, By Region And Segment Forecasts
- Report ID: GVR-1-68038-587-8
- Number of Report Pages: 100
- Format: PDF
- Historical Range: 2018 - 2022
- Forecast Period: 2024 - 2030
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
Pulmonary Drug Delivery Systems Market Summary
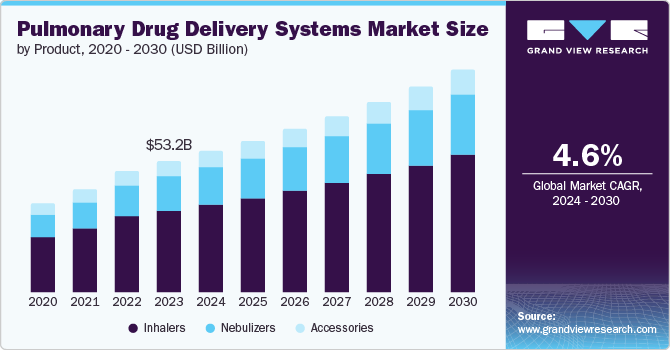
The global pulmonary drug delivery systems market size was estimated at USD 53.24 billion in 2023 and is projected to reach USD 72.33 billion by 2030, growing at a CAGR of 4.6% from 2024 to 2030. The growth is attributed to the rising prevalence of chronic respiratory diseases such as chronic obstructive pulmonary disease (COPD), asthma, and cystic fibrosis.
Key Market Trends & Insights
- North America pulmonary drug delivery systems market dominated the market with largest revenue share of 32.0% in 2023.
- The U.S. pulmonary drug delivery systems market dominated the North America market with a share of 77.7% in 2023.
- Based on application, the chronic obstructive pulmonary disease (COPD) segment accounted for the largest market share in 2023.
- Based on product, the inhalers segment accounted for the largest market share of 62.8% in 2023.
- Based on end use, the home care segment is expected to register the fastest CAGR during the forecast period.
Market Size & Forecast
- 2023 Market Size: USD 53.24 Billion
- 2030 Projected Market USD 72.33 Billion
- CAGR (2024-2030): 4.6%
- North America: Largest market in 2023
The significant increase in occurrence and rate of respiratory diseases has resulted in growth in the number of patients, which has subsequently raised the demand for these systems.
The increasing rate of air pollution lead to rise in risk of lung infections such as bronchitis and pneumonia. Polluted air can lead to difficulty in breathing and results in various severe lung problems. For instance, according to World Health Organization (WHO), it is estimated that environmental air pollution is responsible for approximately 16% of lung cancer deaths, around 17% of ischemic heart disease and stroke fatalities, 25% of COPD deaths, and about 26% of respiratory infection deaths. Pulmonary drug delivery systems provide direct contact with the epithelium, enhancing effectiveness in treating local conditions and offering potential for treating systemic illnesses.
Furthermore, it is expected that technological advancements in the health sector is also driving the demand for pulmonary drug delivery systems. Limitations of traditional treatments, such as the late onset of action and low effectiveness, are projected to encourage the development of novel targeted drug delivery devices, thereby boosting growth during the forecast period. The development of modern-day inhalers, e.g., pressurised metered-dose inhalers (pMDIs), modern-day inhalers, e.g., pressurised metered-dose inhalers (pMDIs), soft mist inhalers (SMIs), and jet and vibrating mesh nebulisers (VMNs) have propelled the market growth.
Product Insights
The inhalers segment accounted for the largest market share of 62.8% in 2023. Unlike injections which circulate in the bloodstream, inhalers administer medicine straight to the lungs. As the medication is delivered straight to the lungs, it enables faster onset of action and may decrease side effects elsewhere in the body. The easy portability offered by inhalers, minimum preparation for use, its small size make it convenient for patients to easily carry and use medication all day.
The nebulizers segment is expected to register the fastest CAGR of 5.4% during the forecast period. The increasing use of nebulizers among patients who have difficulty in using other inhalation devices, such as young children, elderly individuals, and those with coordination issues is the major factor driving the market growth. According to a study published in National Institutes of Health, the use of nebulizers in young children is widespread, with a prevalence between 33% and 71% in children under the age of 12.
Application Insights
The Chronic Obstructive Pulmonary Disease (COPD) segment accounted for the largest market share in 2023. COPD is a chronic disease that requires continuous treatment and care. Pulmonary drug delivery systems provide different choices for prolonged treatment, such as corticosteroids for managing inflammation, enhancing airflow and inhalers for bronchodilators. Although there is no cure for COPD, the use of pulmonary drug delivery helps in controlling the symptoms of pulmonary diseases and enhance quality of life.
The asthma segment is projected to grow at the fastest CAGR over the forecast period. Asthma patients look for treatment options that are easy to carry and use, in their everyday routines. The new inhaler technology and inhalation therapy that uses Dry Powder Inhaler (DPIs) and Metered Dose Inhaler (MDIs) are considered the basis of asthma management. The demand and utilization of devices like MDIs and DPIs for treating asthma are on the rise, owing to the high number of people affected by asthma, thereby propelling the segment growth.
End-use Insights
The hospitals & clinics segment dominated the market in 2023. The treatment offered by doctors in hospital and clinics helps in speedy recovery of patients. For patients who are diagnosed with serious respiratory conditions, necessitates careful observation and accurate medication administration. Nebulizers, frequently used in medical facilities, are highly effective in administering high doses of medication quickly. This makes them essential for treating COPD exacerbations, sudden asthma attacks, and other critical conditions.
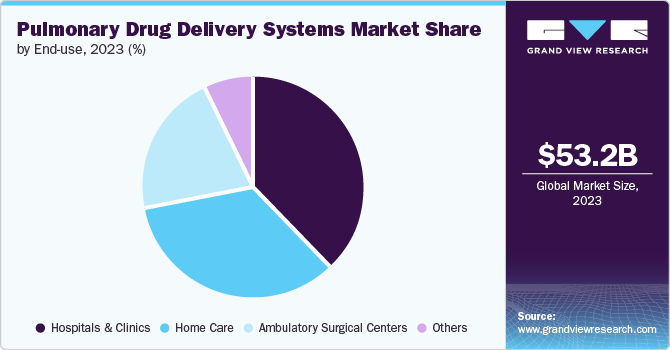
The home care segment is expected to register the fastest CAGR during the forecast period. The growing geriatric population is attributed to the rising demand for home healthcare. Additionally, the increasing prevalence of chronic diseases is becoming more common. Patients can effectively control these conditions at home by using pulmonary drug delivery systems with the right training and guidance. The convenience offered by home healthcare services is driving the segment growth.
Regional Insights
North America pulmonary drug delivery systems market dominated the market with largest revenue share of 32.0% in 2023. The increasing cases of chronic diseases such as asthma and CODP is attributing to the market growth in the region. According to National Center for Health Statistics, percentage of adults who received a diagnosis of COPD, emphysema, or chronic bronchitis was 4.6% in 2022.
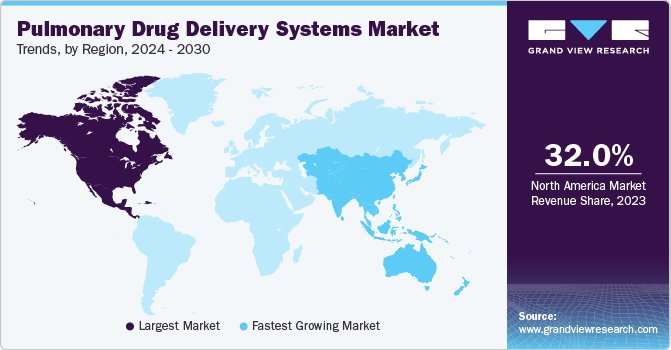
U.S. Pulmonary Drug Delivery Systems Market Trends
U.S. pulmonary drug delivery systems market dominated the North America market with a share of 77.7% in 2023 due to emergence of pulmonary chronic diseases and the rising fatality rate. According to the Center for Disease Control and Prevention, the number of deaths due to chronic lower respiratory diseases including asthma was 147,382 in 2022. In comparison to traditional methods such as shots or pills, pulmonary drugs offers faster onset of action driving the market growth.
Europe Pulmonary Drug Delivery Systems Market Trends
Europe pulmonary drug delivery systems market identified as a lucrative region in 2023 due to the increasing geriatric population in the region. As people age, their chances of experiencing chronic respiratory illnesses increase, which fuels the growth of pulmonary drug delivery market. In addition, manufacturers are introducing innovative inhaler technologies, including dry powder inhalers and smart inhalers that have dose tracking and medication reminders.These developments enhance ease of use and better patient adherence to treatment plans.
The UK pulmonary drug delivery systems market is expected to grow rapidly in the coming years due to increasing penetration of home healthcare. Home healthcare reduces medical expenses and hospital stays, while providing patients with a more comfortable environment. Pulmonary medication delivery systems are ideal for facilitating this shift to care at home. Moreover, increased public awareness of the benefits of pulmonary drug delivery systems, including better adherence to medication and potentially fewer side effects in comparison to oral drugs, is fueling market expansion.
Asia Pacific Pulmonary Drug Delivery Systems Market Trends
Asia Pacific pulmonary drug delivery systems market anticipated to witness significant growth. The continuous increase in the number of asthmatic and allergic rhinitis patients are factors expected to propel the market growth. In addition, governments of Asia Pacific countries are initiating initiatives to increase knowledge about respiratory illnesses and enhance availability of medical care.
The China pulmonary drug delivery systems market is expected to grow rapidly in the coming years. As compared to developed regions, China has a large and unexplored market for pulmonary drug delivery systems. This offers a substantial opportunity for manufacturers and healthcare providers to expand in this region.
Key Pulmonary Drug Delivery Systems Company Insights
Some key companies in pulmonary drug delivery systems market include 3M, Cipla, AstraZeneca, Koninklijke Philips N.V., OMRON Healthcare, Inc., and others. Companies concentrate on addressing market gaps, creating new products, focusing on specific areas and engaging in mergers & acquisitions to increase their market presence.
-
AstraZeneca is a multinational pharmaceutical and biotechnology company. The company focuses on chronic respiratory conditions like asthma and COPD, as well as immune-related disorders like lupus and provide drug methods and combinations with the goal of reaching disease modification and clinical remission.
Key Pulmonary Drug Delivery Systems Companies:
The following are the leading companies in the pulmonary drug delivery systems market. These companies collectively hold the largest market share and dictate industry trends.
- 3M
- AstraZeneca
- GSK plc.
- Novartis AG
- Koninklijke Philips N.V.
- Boehringer Ingelheim International GmbH.
- Teva Pharmaceutical Industries Ltd.
- OMRON Healthcare, Inc.
- PARI GmbH
- Cipla
Recent Developments
- In April 2024, Launch Therapeutics, Inc. and Teva Pharmaceutical Industries Ltd entered into a clinical collaboration agreement to accelerate Teva’s ICS-SABA (TEV-‘248) clinical research program.
Pulmonary Drug Delivery Systems Market Report Scope
Report Attribute
Details
Market size value in 2024
USD 55.24 billion
Revenue forecast in 2030
USD 72.33 billion
Growth Rate
CAGR of 4.6% from 2024 to 2030
Base year for estimation
2023
Historical data
2018 - 2022
Forecast period
2024 - 2030
Quantitative units
Revenue in USD million/billion and CAGR from 2024 to 2030
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Product, application, end-use, region
Regional scope
North America; Europe; Asia Pacific; Latin America; MEA
Country scope
U.S.; Canada; Mexico; UK; Germany; France; Italy; Spain; Denmark; Sweden; Norway; China; Japan; India; South Korea; Australia; Thailand; Brazil; Argentina; Saudi Arabia; Kuwait; UAE; South Africa
Key companies profiled
3M; AstraZeneca; GSK plc.; Novartis AG; Koninklijke Philips N.V.; Boehringer Ingelheim International GmbH.; Teva Pharmaceutical Industries Ltd.; OMRON Healthcare, Inc.; PARI GmbH; Cipla
Customization scope
Free report customization (equivalent up to 8 analysts working days) with purchase. Addition or alteration to country, regional & segment scope.
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
Global Pulmonary Drug Delivery Systems Market Report Segmentation
This report forecasts revenue growth at global, regional, and country levels and provides an analysis of the latest industry trends in each of the sub-segments from 2018 to 2030. For this study, Grand View Research has segmented the global pulmonary drug delivery systems market report based on product, application, end-use, and region:

-
Product Outlook (Revenue, USD Million, 2018 - 2030)
-
Inhalers
-
Nebulizers
-
Accessories
-
-
Application Outlook (Revenue, USD Million, 2018 - 2030)
-
Asthma
-
Cystic Fibrosis
-
Chronic Obstructive Pulmonary Disease (COPD)
-
Allergic Rhinitis
-
Pulmonary Arterial Hypertension
-
Others
-
-
End-use Outlook (Revenue, USD Million, 2018 - 2030)
-
Hospitals & Clinics
-
Ambulatory Surgical Centers (ASCs)
-
Home Care
-
Others
-
-
Regional Outlook (Revenue, USD Million, 2018 - 2030)
-
North America
-
U.S.
-
Canada
-
Mexico
-
-
Europe
-
Germany
-
UK
-
France
-
Italy
-
Spain
-
Denmark
-
Sweden
-
Norway
-
-
Asia Pacific
-
China
-
Japan
-
India
-
South Korea
-
Australia
-
Thailand
-
-
Latin America
-
Brazil
-
Argentina
-
-
Middle East and Africa (MEA)
-
KSA
-
UAE
-
South Africa
-
Kuwait
-
-
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










