- Home
- »
- Medical Devices
- »
-
Rare Disease Clinical Trials Market, Industry Report, 2030GVR Report cover
![Rare Disease Clinical Trials Market Size, Share, & Trends Report]()
Rare Disease Clinical Trials Market (2025 - 2030) Size, Share, & Trends Analysis Report By Phase (Phase I, Phase II, Phase III, Phase IV), By Therapeutic Area (Oncology, Cardiovascular), By Sponsor, By Region, And Segment Forecasts
- Report ID: GVR-4-68040-031-1
- Number of Report Pages: 100
- Format: PDF
- Historical Range: 2018 - 2023
- Forecast Period: 2025 - 2030
- Industry: Healthcare
- Report Summary
- Table of Contents
- Interactive Charts
- Methodology
- Download FREE Sample
-
Download Sample Report
Rare Disease Clinical Trials Market Summary
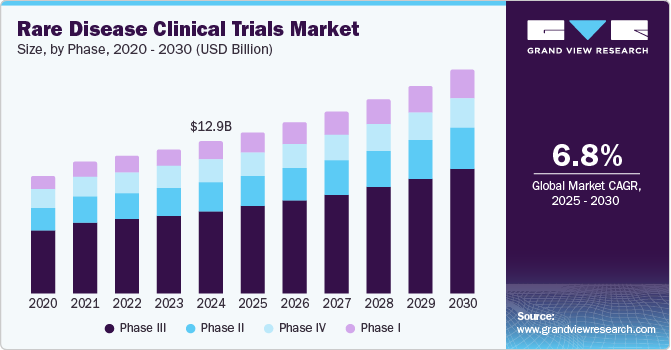
The global rare disease clinical trials market size was estimated at USD 12,961.6 million in 2024 and is projected to reach USD 19,019.9 million by 2030, growing at a CAGR of 6.8% from 2025 to 2030. The increasing prevalence of rare diseases has enhanced the demand for specialized treatments and research efforts.
Key Market Trends & Insights
- In terms of region, North America was the largest revenue generating market in 2024.
- Country-wise, China is expected to register the highest CAGR from 2025 to 2030.
- In terms of segment, phase iii accounted for a revenue of USD 7,446.7 million in 2024.
- Phase I is the most lucrative phase segment registering the fastest growth during the forecast period.
Market Size & Forecast
- 2024 Market Size: USD 12,961.6 million
- 2030 Projected Market Size: USD19,019.9 million
- CAGR (2025-2030): 6.8%
- North America: Largest market in 2024
Regulatory agencies offer incentives such as tax credits, fast-track approvals, and market exclusivity to encourage drug development. Moreover, pharmaceutical and biotechnology companies are investing heavily in rare disease treatments due to limited competition and high-profit potential. Advancements in gene and cell therapies are also driving innovation, providing new treatment options for complex conditions. Patient advocacy groups and research collaborations are further supporting drug development efforts. These factors have contributed to the steady expansion of the rare disease clinical trials industry.
The increasing adoption of artificial intelligence and biomarker-driven research is expected to enhance drug discovery and trial efficiency. Decentralized clinical trials, supported by remote monitoring and digital health tools, improve patient participation and data collection. In addition, collaborations between biotech firms, research institutions, and patient organizations accelerate the development of new therapies. Adaptive trial designs help optimize protocols, making clinical studies more efficient and cost-effective. A greater focus on personalized medicine will lead to targeted treatments with improved outcomes. Hence, these advancements are expected to shape the future of the rare disease clinical trials industry.
The increasing use of real-world evidence and patient registries strengthens clinical trial data and regulatory decision-making. Digital health technologies, including wearables and electronic health records, provide valuable insights into treatment efficacy, and expanding financial support from venture capital firms and non-profit organizations drives innovation in drug development. In addition, improved regulatory frameworks and international cooperation help create a more favorable research environment. The shift toward patient-centric approaches also enhances trial designs and therapeutic outcomes. These trends will further support the growth of the market.
Phase Insights
The phase III segment dominated the market with the largest revenue share of 54.2% in 2024, driven by the high number of rare disease trials reaching late-stage development. Complex rare disease treatments often require extensive phase III trials to assess long-term safety and efficacy. Growing investments from pharmaceutical companies in advanced therapies have further fueled phase III expansions. Moreover, increasing patient recruitment efforts and collaboration with specialized research centers have enhanced trial efficiency. The rise in targeted therapies and gene-based treatments has increased the need for large-scale phase III studies. Regulatory incentives encouraging rare disease research have also contributed to the growing volume of late-stage trials. The demand for robust clinical data to support regulatory approvals has reinforced the dominance of this segment in the rare disease clinical trials industry.
The phase I segment is projected to grow at the highest CAGR of 8.1% over the forecast period, fueled by the rising focus on novel drug development for rare diseases. Advances in genetic research have led to an increasing number of first-in-human trials targeting rare conditions. The adoption of precision medicine has accelerated the demand for early-phase trials to evaluate personalized treatments, and the emergence of innovative therapeutic modalities, such as gene and cell therapies, has driven the need for more phase I studies. Enhanced clinical trial designs, including adaptive trials, have improved the efficiency of early-stage research. Moreover, the growing participation of biotech companies in rare disease trials has expanded the phase I pipeline. Increased investments in preclinical research have facilitated the transition of promising drug candidates into early-stage trials.
Therapeutic Area Insights
The oncology segment dominated the market with the largest revenue share in 2024, which can be attributed to the high prevalence of rare cancers requiring specialized treatment options. The development of targeted therapies and immunotherapies has led to a surge in clinical trials for rare malignancies. Moreover, advances in genomic profiling have enabled precise identification of rare cancer subtypes, supporting more focused drug development. The rising demand for personalized medicine has intensified research efforts in rare cancers. The growing interest in breakthrough therapies has driven significant investment in this segment. Increased collaboration between research institutions and pharmaceutical companies has accelerated trial enrollment. The complexity of rare cancers necessitates extensive clinical studies, contributing to the segment's dominance.
The infectious disease segment is projected to grow at the highest CAGR over the forecast period, due to the emergence of rare infectious conditions requiring urgent medical solutions. The rise of antimicrobial resistance has intensified the need for novel therapeutics targeting rare pathogens. Advances in vaccine research have spurred the development of new preventive strategies for rare infectious diseases and expanding global surveillance efforts have improved the identification of previously overlooked rare infections. Increased funding for infectious disease research has accelerated clinical trial activity in this segment. The integration of innovative trial designs, such as decentralized studies, has enhanced the efficiency of infectious disease trials. In addition, rising awareness of rare infectious conditions has encouraged industry investment in this rapidly growing segment.
Sponsor Insights
The pharmaceutical and biopharmaceutical companies segment dominated the market with the largest revenue share in 2024, driven by the increasing focus on developing breakthrough therapies for rare diseases. The potential for high-value orphan drug approvals has attracted significant investment from industry leaders. Expanding research collaborations with academic institutions have strengthened the rare disease trial ecosystem, and advances in biotechnology have enabled the development of innovative therapies, including gene and RNA-based treatments. The competitive landscape has encouraged pharmaceutical companies to prioritize rare disease research as part of long-term growth strategies. Rising demand for targeted treatment options has further stimulated clinical trial activity in this segment. The complexity of rare disease therapies necessitates large-scale trials, reinforcing the dominance of pharmaceutical companies in the rare disease clinical trials industry.
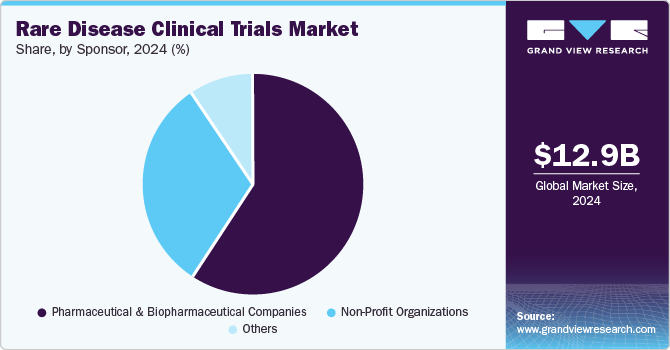
The non-profit organizations segment is projected to grow at a significant CAGR over the forecast period, fueled by increasing support for rare disease research. Strong engagement from patient advocacy groups has facilitated funding for clinical trials targeting underserved conditions. Moreover, collaborations between non-profit organizations and academic researchers have expanded the scope of early-stage rare disease trials. The need for unbiased research has driven greater investment in investigator-initiated studies, and public awareness initiatives have enhanced trial recruitment efforts, improving patient participation rates. Non-profit funding models have provided essential resources for trials addressing ultra-rare conditions. Hence, the growing emphasis on bridging research gaps in neglected diseases has strengthened the role of non-profit organizations in the rare disease clinical trials industry.
Regional Insights
North America rare disease clinical trials market held the highest revenue share of 42.6% in 2024, driven by the strong presence of leading biotechnology and pharmaceutical companies. The region's advanced healthcare infrastructure has supported the efficient execution of complex clinical trials. High levels of investment in biomedical research have accelerated rare disease drug development, and established networks of specialized research centers have facilitated patient recruitment for rare disease studies. The increasing adoption of innovative trial designs, such as decentralized and adaptive trials, has improved research efficiency. Furthermore, strong industry-academic collaborations have contributed to the rapid advancement of rare disease therapies. The rising prevalence of rare conditions has further intensified clinical research efforts across North America.
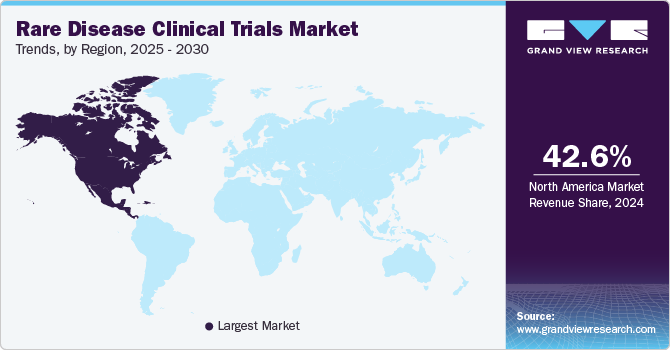
U.S. Rare Disease Clinical Trials Market Trends
The U.S. rare disease clinical trials market dominated North America in 2024 due to the high concentration of biopharmaceutical companies leading rare disease research. The country’s extensive network of specialized clinical trial sites has enhanced research capabilities. Increasing investments in precision medicine have accelerated the development of rare disease therapies, and advanced regulatory frameworks have streamlined the approval process for orphan drugs. Strong collaborations between industry stakeholders and research institutions have boosted trial efficiency. In addition, the availability of cutting-edge research technologies has improved the success rate of rare disease trials. Expanding patient advocacy initiatives also supported greater clinical trial participation.
Europe Rare Disease Clinical Trials Market Trends
Europe rare disease clinical trials market held a substantial share in 2024, driven by the growing focus on rare disease research. Strong industry partnerships with academic institutions have advanced clinical trial capabilities. The increasing prevalence of rare disorders has intensified research efforts across multiple therapeutic areas, and high investment in biotechnology has accelerated the development of innovative treatment options. Growing patient awareness and engagement have also improved trial enrollment rates. The region’s well-established healthcare infrastructure has supported the execution of complex clinical studies. Moreover, the rising demand for advanced therapies has fueled rare disease trials in Europe.
Asia Pacific Rare Disease Clinical Trials Market Trends
Asia Pacific rare disease clinical trials market is expected to register the highest CAGR of 8.4% over the forecast period, which can be attributed to the rapid expansion of biotechnology research in the region. Increasing investments from multinational pharmaceutical companies have strengthened clinical trial activity. Moreover, the growing prevalence of rare diseases has driven demand for novel treatment options. Expanding research collaborations between international and regional institutions have enhanced trial capabilities, and the adoption of digital health technologies has improved patient recruitment and monitoring. Hence, rising participation from emerging biotech firms has boosted early-stage rare disease trials. The region’s increasing healthcare expenditure has further supported the growth of rare disease clinical research.
China rare disease clinical trials market dominated the Asia Pacific region with a significant revenue share in 2024. This share is attributable to the increasing investment in biopharmaceutical innovation in the country. Expanding research partnerships with global pharmaceutical companies have strengthened trial infrastructure, and the rising burden of rare diseases has created a strong demand for advanced treatment options. Moreover, improvements in clinical trial efficiency have accelerated drug development timelines. The rising adoption of precision medicine approaches has supported targeted research for rare conditions. Enhanced patient awareness and engagement have also improved clinical trial participation. The country’s expanding biopharmaceutical sector has further fueled rare disease clinical research initiatives.
Key Rare Disease Clinical Trials Company Insights
Some key companies operating in the market are Takeda Pharmaceutical Company Limited; F. Hoffmann-La Roche Ltd; Pfizer, Inc; AstraZeneca; and Novartis AG. Companies are undertaking strategic initiatives, such as mergers, acquisitions, and product launches, to expand their market presence and address the evolving healthcare demands in rare disease clinical trials market.
-
Takeda Pharmaceutical Company offers a range of innovative therapies and solutions for rare disease clinical trials, focusing on hematology, immunology, metabolic disorders, and genetic diseases. Its product portfolio includes enzyme replacement therapies like Elaprase (for Hunter syndrome) and Replagal (for Fabry disease) and hereditary angioedema treatments such as Takhzyro and Cinryze. Takeda also provides advanced plasma-derived therapies like Gammagard and conducts research on gene therapies for rare conditions. Its clinical trial solutions leverage patient-centric approaches, digital health technologies, and global expertise to accelerate drug development. Takeda aims to enhance access, improve patient outcomes, and drive innovation in rare disease treatment.
-
F. Hoffmann-La Roche Ltd offers cutting-edge therapies, diagnostics, and clinical trial solutions for rare diseases, focusing on neurology, hematology, oncology, and metabolic disorders. Its product portfolio includes Hemlibra (for hemophilia A), Evrysdi (for spinal muscular atrophy), and Enspryng (for neuromyelitis optica spectrum disorder). Roche also develops precision medicine solutions and companion diagnostics to enhance rare disease research. The company’s clinical trial services incorporate real-world data, AI-driven analytics, and decentralized trial models to optimize patient recruitment and study efficiency. Roche aims to advance personalized treatments and improve rare disease patient care globally through these innovations.
Key Rare Disease Clinical Trials Companies:
The following are the leading companies in the rare disease clinical trials market. These companies collectively hold the largest market share and dictate industry trends.
- Takeda Pharmaceutical Company Limited
- F. Hoffmann-La Roche Ltd
- Pfizer Inc
- AstraZeneca
- Novartis AG
- Laboratory Corporation of America Holdings (LabCorp)
- IQVIA
- Charles River Laboratories
- ICON plc
- Parexel International (MA) Corporation
Recent Developments
-
In November 2023, Takeda announced that the U.S. FDA approved ADZYNMA (ADAMTS13, recombinant-krhn) as the unique and exclusive recombinant ADAMTS13 enzyme replacement therapy for treating Congenital Thrombotic Thrombocytopenic Purpura (cTTP).
-
In December 2022, Pfizer announced positive top-line results from its Phase 3 BENEGENE-2 study evaluating fidanacogene elaparvovec, an investigational gene therapy for moderately severe to severe hemophilia B in adult males. The study met its primary endpoint, demonstrating superiority in reducing the annualized bleeding rate compared to the standard prophylaxis regimen with Factor IX.
Rare Disease Clinical Trials Market Report Scope
Report Attribute
Details
Market size value in 2025
USD 13.69 billion
Revenue forecast in 2030
USD 19.02 billion
Growth rate
CAGR of 6.8% from 2025 to 2030
Base year for estimation
2024
Historical data
2018 - 2023
Forecast period
2025 - 2030
Report updated
March 2025
Quantitative units
Revenue in USD billion and CAGR from 2025 to 2030
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, trends
Segments covered
Phase, Therapeutic Area, Sponsor, and Region
Regional scope
North America; Europe; Asia Pacific; Latin America; Middle East & Africa
Country scope
U.S., Canada, Mexico, UK, Germany, France, Italy, Spain, Denmark, Sweden, Norway, Japan, China, India, South Korea, Australia, Thailand, Brazil, Argentina, South Africa, Saudi Arabia, UAE, Kuwait.
Key companies profiled
Takeda Pharmaceutical Company Limited; F. Hoffmann-La Roche Ltd; Pfizer Inc; AstraZeneca; Novartis AG; Laboratory Corporation of America Holdings (LabCorp); IQVIA; Charles River Laboratories; ICON plc; Parexel International (MA) Corporation.
Customization scope
Free report customization (equivalent to 8 analysts working days) with purchase. Addition or alteration to country, regional & segment scope.
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
Global Rare Disease Clinical Trials Market Report Segmentation
This report forecasts global, regional, and country revenue growth and analyzes the latest industry trends in each sub-segment from 2018 to 2030. For this study, Grand View Research has segmented the global rare disease clinical trials market based on phase, therapeutic area, sponsor, and region:
-
Phase Outlook (Revenue, USD Billion, 2018 - 2030)
-
Phase I
-
Phase II
-
Phase III
-
Phase IV
-
-
Therapeutic Area Outlook (Revenue, USD Billion, 2018 - 2030)
-
Oncology
-
Cardiovascular Disorders
-
Neurological Disorders
-
Infectious Disease
-
Genetic Disorders
-
Autoimmune and Inflammation
-
Hematologic Disorders
-
Musculoskeletal Disorders
-
Others
-
-
Sponsor Outlook (Revenue, USD Billion, 2018 - 2030)
-
Pharmaceutical & Biopharmaceutical Companies
-
Non-Profit Organizations
-
Others
-
-
Regional Outlook (Revenue, USD Billion, 2018 - 2030)
-
North America
-
U.S.
-
Canada
-
Mexico
-
-
Europe
-
UK
-
Germany
-
France
-
Italy
-
Spain
-
Denmark
-
Sweden
-
Norway
-
-
Asia Pacific
-
Japan
-
China
-
India
-
South Korea
-
Australia
-
Thailand
-
-
Latin America
-
Brazil
-
Argentina
-
-
Middle East & Africa
-
South Africa
-
Saudi Arabia
-
UAE
-
Kuwait
-
-
Frequently Asked Questions About This Report
b. The oncology segment held a market share of 34.5 by therapeutic area in 2024. The growing investment in the research & development of activities for cancer-based rare diseases is one of the prominent factors supporting the segment's growth.
b. Some key players operating in the rare disease clinical trials market include Parexel International Corporation, Icon PLC, Charles River Laboratories Inc., IQVIA, and a few others.
b. Increasing investments across the R&D activities for rare diseases, and the growing prevalence of rare diseases especially in pediatrics, etc are a few of the factors supporting the market growth.
b. The global rare disease clinical trials market size was estimated at USD 12.96 billion in 2024 and is expected to reach USD 13.69 billion in 2025.
b. The global rare disease clinical trials market is expected to grow at a compound annual growth rate of 6.80% from 2025 to 2030 to reach USD 19.02 billion by 2030.
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










