- Home
- »
- Clinical Diagnostics
- »
-
Real-time PCR, Digital PCR, And End-point PCR Market Report 2033GVR Report cover
![Real-time PCR, Digital PCR, And End-point PCR Market Size, Share & Trends Report]()
Real-time PCR, Digital PCR, And End-point PCR Market (2026 - 2033) Size, Share & Trends Analysis Report By Technology (Quantitative, Digital, End-point), By Product (Consumables & Reagents, Instruments, Software & Services), By Application, By Region, And Segment Forecasts
- Report ID: GVR-1-68038-552-6
- Number of Report Pages: 120
- Format: PDF
- Historical Range: 2021 - 2024
- Forecast Period: 2026 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
Real-time PCR, Digital PCR, And End-point PCR Market Summary
The global real-time PCR, digital PCR, and end-point PCR market size was valued at USD 10.58 billion in 2025 and is expected to reach USD 21.31 billion by 2033, growing at a CAGR of 9.2% from 2026 to 2033. Market growth is driven by the rising prevalence of infectious diseases and cancer, increasing demand for early and accurate diagnostics, and the growing adoption of precision medicine.
Key Market Trends & Insights
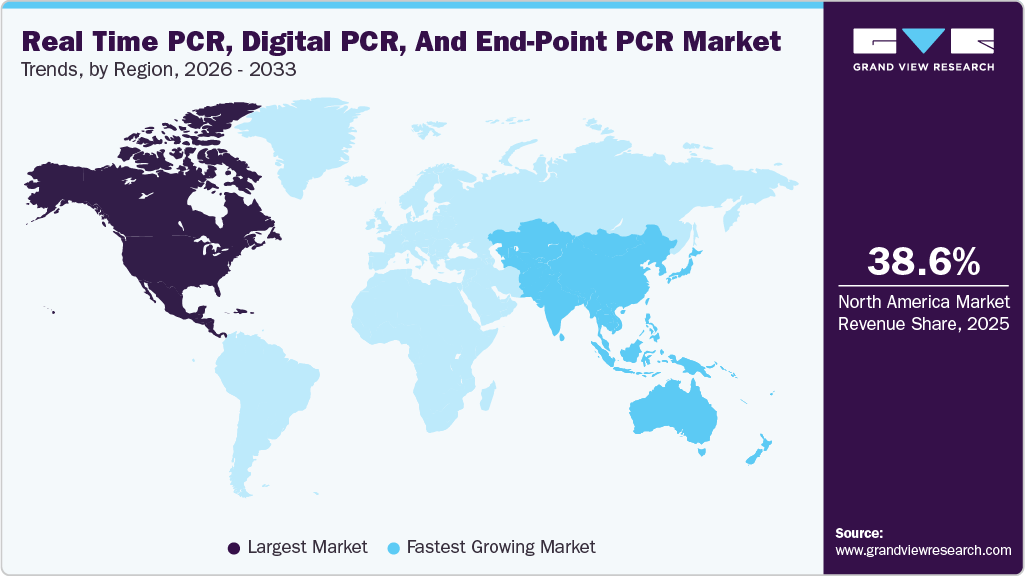
- North America real-time PCR, digital PCR, and end-point PCR market dominated global industry in 2025, with a revenue share of 38.58%.
- The U.S. real-time PCR, digital PCR, and end-point PCR market represents the largest share in North America.
- By product, the consumables and reagents segment held the largest revenue share of over 62.88% in 2025.
- By technology, the quantitative PCR (qPCR) segment accounted for the largest revenue share of over 79.21% in 2025.
- By application, the research segment accounted for the largest revenue share of 45.95% in 2025.
Market Size & Forecast
- 2025 Market Size: USD 10.58 Billion
- 2033 Projected Market Size: USD 21.31 Billion
- CAGR (2026-2033): 9.2%
- North America: Largest market in 2025
- Asia Pacific: Fastest Market
Technological advancements such as multiplex PCR, digital PCR for high sensitivity, are further accelerating adoption. According to a CDC report December 2025, congenital heart defects affect approximately 1% of births annually in the U.S., highlighting the growing need for prenatal genetic testing. PCR technologies enable early detection of genetic variations, supporting timely diagnosis and treatment decisions.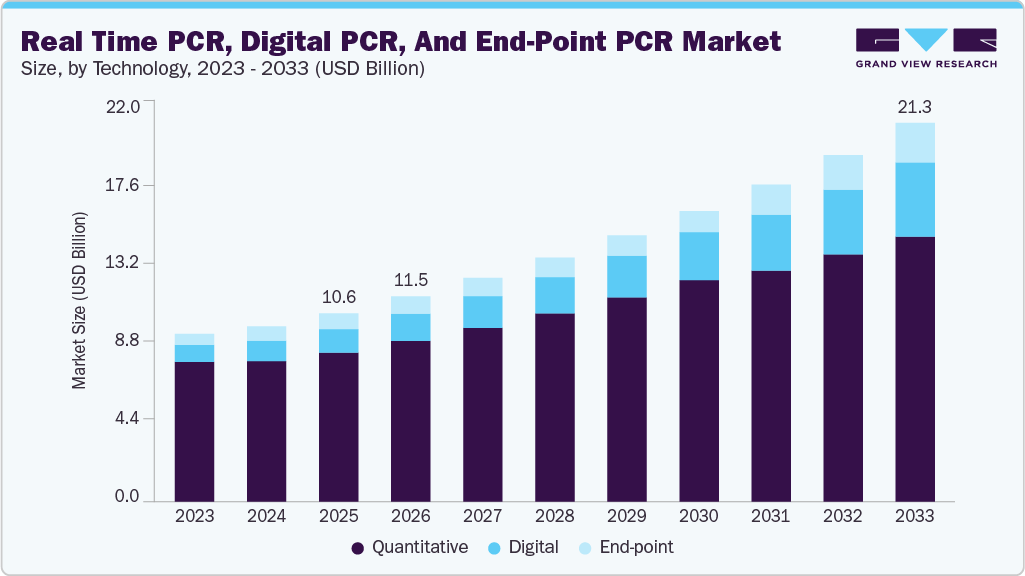
The growing incidence of sexually transmitted infections (STIs) such as HIV and HPV, along with rising cases of chronic, infectious, and genetic disorders, including Alzheimer’s disease, Streptococcal infections, Turner syndrome, and Parkinson’s disease, is significantly expanding the target patient population, thereby driving the growth of real-time PCR, digital PCR, and end-point PCR markets. These conditions require highly sensitive and accurate molecular diagnostic tools for early detection and disease monitoring. For instance, according to a WHO report in July 2025, approximately 1.3 million people globally acquired HIV, highlighting the continued burden of infectious diseases. In addition, European surveillance data reported over 105,922 HIV diagnoses in 2024, with several countries observing increasing trends. Such rising disease incidence is accelerating the demand for PCR-based diagnostic solutions.
The rapid adoption of digital PCR (dPCR) for precision medicine is a key driver of market growth. Unlike conventional PCR methods, dPCR enables absolute quantification without the need for standard curves, offering ultra-high sensitivity and the ability to detect rare genetic mutations with greater accuracy. This makes it particularly valuable in oncology, infectious disease monitoring, and genetic testing, where precise detection is critical for early diagnosis and treatment selection. In addition, dPCR provides improved reproducibility and reliability, supporting its increasing use in clinical and research settings. For instance, in July 2025, Bio-Rad expanded its Droplet Digital PCR portfolio with the launch of the QX Continuum and QX700 systems, which support over 400,000 assays. This advancement reinforces dPCR’s role in precision diagnostics and accelerates its adoption across advanced healthcare applications.
Technological advancements in multiplexing and assay innovation are significantly improving the efficiency and throughput of PCR-based diagnostics, thereby driving market growth. Advanced multiplex PCR systems enable the simultaneous detection of multiple pathogens in a single test, reducing turnaround time, operational costs, and sample requirements. For instance, in November 2025, FlashDx introduced a fully automated multiplex real-time PCR system based on semi-solid phase real-time PCR (sSPRT) technology, enabling a seamless sample-to-result workflow and rapid multi-pathogen detection. In addition, a study published in January 2026 developed a multiplex RT-qPCR assay capable of detecting 18 respiratory viruses simultaneously, demonstrating high sensitivity up to 99.81% and specificity 100%, significantly improving diagnostic throughput, further enhancing diagnostic accuracy, and laboratory productivity.
Moreover, the growth of point-of-care (POC) and decentralized testing is significantly driving the PCR market, supported by increasing demand for portable, rapid, and easy-to-use diagnostic systems. Miniaturized PCR devices enable faster turnaround times and facilitate testing in clinics, airports, and remote settings, strengthening pandemic preparedness and on-site diagnostics. Simultaneously, the rising demand for high-throughput, automated systems is encouraging laboratories to adopt advanced platforms that process large sample volumes with minimal human intervention, thereby reducing errors and improving efficiency.
Market Concentration & Characteristics
The real-time PCR, digital PCR, and end-point PCR market demonstrates a high degree of innovation, driven by advancements in automation, multiplexing, and high-throughput capabilities. Emerging platforms are increasingly designed to deliver faster, more accurate, and user-friendly diagnostic solutions. For instance, in 2025, Roche launched the cobas 5800 System, a compact molecular diagnostic platform that enables automated PCR testing with faster turnaround times and improved laboratory efficiency. In addition, in 2025, Seegene introduced the CURECA system, a fully automated PCR platform that integrates the entire workflow from sample preparation to result analysis, allowing continuous, hands-free operation. These innovations enhance workflow efficiency, reduce manual intervention, and improve diagnostic precision. As a result, PCR technologies are evolving into scalable, integrated solutions, accelerating their adoption across clinical diagnostics, research applications, and decentralized healthcare settings.
The real-time PCR, digital PCR, and end-point PCR market is witnessing a moderate to high level of M&A activity, as key players are actively acquiring innovative companies to strengthen their technological capabilities and expand their product portfolios. These strategic acquisitions are primarily focused on enhancing digital PCR, multiplexing capabilities, and automation to meet the growing demand for precision diagnostics. For instance, in June 2025, Bio-Rad Laboratories completed the acquisition of Stilla Technologies, a developer of next-generation digital PCR systems, to expand its capabilities in high-precision genetic analysis and clinical diagnostics. This acquisition enabled Bio-Rad to integrate advanced multiplexing and high throughput dPCR technologies into its portfolio, reinforcing its leadership in the digital PCR segment. For instance, in February 2023, QIAGEN acquired a majority stake in Verogen to expand its presence in PCR-based and next-generation sequencing workflows, strengthening its capabilities in human identification and molecular diagnostics applications.

Regulatory frameworks play a key role in shaping the real-time PCR, digital PCR, and end-point PCR market by ensuring the safety, accuracy, and reliability of molecular diagnostic tests. Regulatory bodies such as the U.S. Food and Drug Administration (FDA), European Medicines Agency (EMA), and World Health Organization (WHO) enforce stringent approval and validation requirements for PCR-based assays and instruments, which can extend development timelines and increase costs. However, these organizations have also introduced expedited approval pathways for molecular diagnostics targeting infectious diseases and emerging health threats, enabling faster market access. In addition, evolving guidelines for quality control and standardization are improving test reliability and supporting broader adoption across clinical and decentralized healthcare settings.
Regional expansion in the real-time PCR, digital PCR, and end-point PCR market is accelerating as companies strengthen their presence across both developed and emerging regions to capture rising diagnostic demand. North America dominates the market, driven by advanced healthcare infrastructure, high adoption of molecular diagnostics, and the strong presence of key industry players. Meanwhile, increasing healthcare investments, growing awareness of early disease detection, and expanding laboratory infrastructure in the Asia Pacific and Latin America are supporting market growth in these regions. For instance, in 2025, several diagnostic companies expanded their operations and distribution networks in the Asia Pacific to meet the rising demand for molecular and point-of-care testing. Such expansion strategies are improving global access to PCR technologies and driving overall market growth.
Technology Insights
The quantitative PCR (qPCR) segment accounted for the largest revenue share, over 79.21%, in 2025, owing to its high sensitivity, accuracy, and rapid turnaround time. qPCR is widely used in gene expression analysis, pathogen detection, viral load monitoring, SNP genotyping, and microRNA profiling, making it a cornerstone technology in clinical diagnostics and research. The segment’s growth is driven by continuous technological advancements, including multiplexing, automation, and integration with digital tools for enhanced data analysis. For instance, in January 2025, Thermo Fisher Scientific received FDA clearance for its Applied Biosystems TaqPath COVID-19 diagnostic PCR kit, strengthening its clinical diagnostic portfolio. In addition, in January 2026, Thermo Fisher launched the TaqMan Cells-to-CT HepatoExpress Kit, enabling faster gene expression analysis by eliminating RNA extraction steps and reducing workflow time to nearly 75 minutes.
The digital PCR (dPCR) segment is expected to register the fastest CAGR over the forecast period, driven by advancements in droplet-based and chip-based technologies that enable ultra-sensitive and precise nucleic acid quantification. dPCR offers advantages such as absolute quantification, high reproducibility, and detection of rare mutations, making it increasingly valuable in oncology, infectious disease monitoring, and genetic testing. The segment’s growth is further supported by strategic initiatives and product innovations by key market players. For instance, in July 2025, Bio-Rad expanded its Droplet Digital PCR portfolio with the launch of QX Continuum and QX700systems, enhancing high-throughput and multiplexing capabilities. In addition, in April 2025, QIAGEN advanced its QIAcuity digital PCR platform with new assay kits, supporting applications in cell and gene therapy. These developments are accelerating adoption and expanding dPCR applications.
Product Insights
Consumables and reagents segment held the largest revenue share of over 62.88% in 2025 and it is anticipated to grow at the fastest rate over the forecast period. Consumables & reagents influence the overall performance of an assay. They help create standardized workflows to support researchers in various fields, such as food testing, cancer & infectious diseases detection, and forensics. The global burden of diseases has increased over the past few years, and the pandemic has contributed largely to high demand for consumables & reagents. Development of novel reagents has increased exponentially, which can be attributed to the demand for polymerase chain reaction reagents in diverse applications. For instance, in 2023, F. Hoffmann-La Roche Ltd introduced the VirSNiP SARS-CoV-2 Spike F486P assay to detect emerging Omicron sub-variants for research use. In addition, increasing adoption of multiplex and high-throughput PCR workflows is further accelerating demand for specialized consumables and reagents.
Moreover, the rising prevalence of infectious and chronic diseases, along with the growing demand for early and accurate diagnostics, is driving a significant increase in the demand for PCR consumables and reagents. The expanding adoption of molecular diagnostics, particularly in point-of-care and decentralized testing settings, is further driving demand. In addition, continuous technological advancements such as multiplex PCR, automation, and integration with digital platforms are boosting reagent utilization across clinical and research applications. Increasing investments in genomics, precision medicine, and drug development are also contributing to higher consumption of PCR reagents. Furthermore, the growing focus on high-throughput testing and standardized workflows in laboratories is accelerating the demand for advanced and application-specific consumables and reagents.
Application Insights
The research segment accounted for the largest revenue share of 45.95% in 2025, driven by the extensive use of PCR technologies in genomics, molecular biology, and life science research. The growing focus on gene expression analysis, next-generation sequencing (NGS) workflows, and biomarker discovery has significantly increased the demand for high-throughput and precise PCR solutions. Additionally, rising investments in academic and research institutions, along with increased funding for genomics and infectious disease research, are supporting segment dominance. The integration of advanced technologies, such as digital and real-time PCR, further enhances sensitivity and accuracy, making PCR an indispensable tool in research applications. Moreover, the expansion of personalized medicine and ongoing advancements in genetic and oncology research continue to fuel the adoption of PCR-based techniques across research laboratories globally.
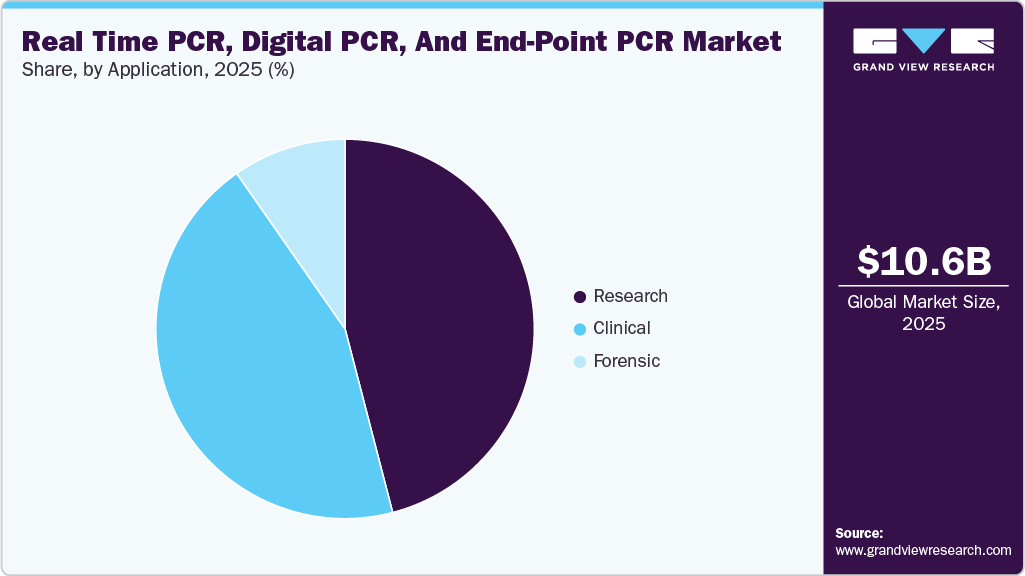
The forensic segment is expected to register the fastest growth over the forecast period, driven by the increasing adoption of PCR technologies in criminal investigations and human identification. PCR offers high sensitivity, specificity, and rapid processing, enabling accurate analysis of trace and degraded DNA samples. Advanced techniques such as real-time and digital PCR allow amplification of low-quality DNA, improving reliability compared to traditional methods like RFLP analysis. Additionally, PCR-based analysis of STRs and VNTR loci, combined with high-resolution electrophoresis, enhances identification accuracy. For instance, in May 2024, QIAGEN expanded its QIAcuity digital PCR applications into forensic workflows, supporting precise DNA quantification and analysis of challenging samples. Furthermore, increasing integration of automated and high-throughput PCR systems in forensic laboratories is improving efficiency, thereby accelerating segment growth.
Regional Insights
North America real-time PCR, digital PCR, and end-point PCR market dominated the global market and accounted for the largest revenue share of 38.58%in 2025, driven by advanced healthcare infrastructure, high adoption of molecular diagnostics, and strong presence of leading companies. The region benefits from significant investments in precision medicine, genomics, and infectious disease testing, which continue to drive demand for high-sensitivity PCR technologies, particularly digital PCR. The widespread use of PCR in oncology, infectious disease diagnostics, and research applications further strengthens market growth. In addition, favorable reimbursement policies and supportive regulatory frameworks encourage rapid adoption of innovative platforms. The region is also witnessing increasing demand for automated and high-throughput PCR systems to manage large testing volumes, further reinforcing its leadership position in the global market.
U.S. Real-time PCR, Digital PCR, and End-point PCR Market Trends
The real-time PCR, digital PCR, and end-point PCR market in the U.S. represents the largest share in North America, supported by strong funding for molecular diagnostics and continuous technological advancements in PCR platforms. The growing burden of infectious diseases, cancer, and genetic disorders is driving demand for accurate and early diagnostic solutions. The country is at the forefront of adopting digital PCR for precision medicine applications, including liquid biopsy and genetic testing. In addition, increasing demand for point-of-care PCR testing and decentralized diagnostics is expanding market reach beyond traditional laboratory settings. The presence of major market players and ongoing research collaborations further accelerates innovation. Regulatory support for rapid diagnostic approvals also contributes to the widespread adoption of advanced PCR technologies across clinical and research environments.
Europe Real-time PCR, Digital PCR, and End-point PCR Market Trends
Europe real-time PCR, digital PCR, and end-point PCR market represents a significant market, supported by well-established healthcare systems and strong emphasis on early disease detection. The region is witnessing increasing adoption of real-time and digital PCR technologies for infectious disease surveillance, oncology diagnostics, and genetic testing. Government initiatives promoting molecular diagnostics and rising investments in healthcare infrastructure are further driving market growth. In addition, the demand for multiplex PCR assays is increasing due to the need for efficient multi-pathogen detection. The region also focuses on standardization and quality assurance in diagnostic testing, enhancing the reliability of PCR-based solutions. Increasing collaborations between research institutions and diagnostic companies are fostering innovation and expanding the application scope of PCR technologies across clinical and research settings.
The real-time PCR, digital PCR, and end-point PCR market in UK is driven by strong public healthcare infrastructure and increasing focus on early and accurate diagnostics. The adoption of advanced PCR technologies is supported by national health initiatives and investments in genomics and precision medicine. Real-time PCR remains widely used in clinical diagnostics, while digital PCR is gaining traction in research and specialized applications. The country is also focusing on expanding point-of-care testing capabilities to improve access to rapid diagnostics. In addition, increasing demand for multiplex assays is supporting efficient detection of multiple pathogens. The UK continues to invest in molecular diagnostic capabilities to strengthen preparedness for infectious disease outbreaks and improve overall healthcare outcomes.
The real-time PCR, digital PCR, and end-point PCR market in Germany is, driven by its strong biotechnology and diagnostics industry. The country has a well-developed laboratory infrastructure and high adoption of advanced molecular diagnostic technologies. Increasing demand for PCR-based testing in infectious diseases, oncology, and genetic research is supporting market growth. Germany is also witnessing rising adoption of automated and high-throughput PCR systems to improve laboratory efficiency. The presence of leading diagnostic companies and research institutions fosters continuous innovation in PCR technologies. In addition, the country emphasizes quality standards and regulatory compliance, ensuring high reliability and accuracy of PCR-based diagnostics across clinical and research applications.
Asia Pacific Real-time PCR, Digital PCR, and End-point PCR Market Trends
The Asia Pacific real-time PCR, digital PCR, and end-point PCR market is expected to witness the fastest growth of 11.01% driven by increasing healthcare investments, rising disease burden, and improving diagnostic infrastructure. Growing awareness of early disease detection and expanding access to molecular diagnostics are key factors supporting market expansion. Countries in the region are increasingly adopting real-time and digital PCR technologies for infectious disease testing and genetic analysis. In addition, the demand for cost-effective and rapid diagnostic solutions is driving the adoption of point-of-care PCR systems. Government initiatives to strengthen healthcare systems and improve laboratory capabilities are further accelerating market growth, making Asia Pacific a key focus area for market players.
The real-time PCR, digital PCR, and end-point PCR market in Japan is characterized by advanced healthcare infrastructure and strong focus on technological innovation. The country has high adoption of molecular diagnostics, particularly in infectious disease testing and oncology. Digital PCR is gaining traction due to its high precision and suitability for genetic analysis and personalized medicine. Japan is also investing in automation and high-throughput diagnostic systems to enhance laboratory efficiency. The presence of established diagnostic companies and strong research capabilities supports continuous development of advanced PCR solutions. In addition, the country emphasizes early diagnosis and preventive healthcare, further driving the demand for reliable and high-sensitivity PCR technologies.
The real-time PCR, digital PCR, and end-point PCR market in China, driven by expanding healthcare infrastructure and increasing investments in molecular diagnostics. The country has a large patient population and rising prevalence of infectious and chronic diseases, which is fueling demand for PCR-based testing. China is also focusing on domestic manufacturing of diagnostic technologies, supporting the availability of cost-effective PCR systems. Increasing adoption of real-time PCR and growing interest in digital PCR for precision medicine are further contributing to market growth. Government initiatives to improve healthcare access and strengthen disease surveillance systems are accelerating the adoption of advanced PCR technologies across clinical and research settings.
Latin America Real-time PCR, Digital PCR, and End-point PCR Market Trends
Latin America real-time PCR, digital PCR, and end-point PCR market is witnessing steady growth, supported by improving healthcare infrastructure and increasing awareness of early disease diagnosis. The rising prevalence of infectious diseases is a key factor driving demand for PCR-based diagnostics in the region. Countries are gradually adopting advanced molecular diagnostic technologies, including real-time PCR, to enhance disease detection capabilities. In addition, the growing need for rapid, accurate diagnostic solutions is driving the adoption of point-of-care PCR systems. Government efforts to strengthen healthcare systems and expand laboratory capabilities are driving market growth. However, cost constraints and limited access to advanced technologies may slightly hinder growth in certain areas.
Middle East & Africa (MEA) PCR, digital PCR, and end-point PCR Market Trends
The Middle East and Africa real-time PCR, digital PCR, and end-point PCR market is growing due to increasing investments in healthcare infrastructure and rising demand for advanced diagnostic solutions. The region is focusing on improving access to molecular diagnostics, particularly for infectious disease detection and outbreak management. Real-time PCR is widely used, while digital PCR adoption is gradually increasing. Government initiatives to enhance healthcare systems and strengthen laboratory networks are supporting market growth. In addition, the demand for portable and point-of-care diagnostic solutions is rising, especially in remote and underserved areas. Despite challenges such as limited resources in some regions, increasing awareness and investment are expected to drive the gradual adoption of PCR technologies. For instance, in September 2025, Co-Diagnostics signed a strategic MOU to establish a joint venture in Saudi Arabia to manufacture and distribute its Co-Dx PCR platform across the Middle East and North Africa, strengthening regional access to molecular diagnostics.
Key Real-time PCR, Digital PCR, and End-point PCR Company Insights
The real-time PCR, digital PCR, and end-point PCR market is led by established players such as Thermo Fisher Scientific, Bio-Rad Laboratories, F. Hoffmann-La Roche Ltd, and QIAGEN, driven by strong product portfolios, global presence, and continuous innovation in real-time and digital PCR platforms. These companies maintain leadership through strategic acquisitions, partnerships, and collaborations to expand technological capabilities and market reach. Emerging players such as Seegene Inc. are gaining traction with fully automated and multiplex PCR solutions. Such strategies enhance competitiveness, accelerate innovation, and support market expansion across clinical, research, and decentralized diagnostic settings.
Key Real-time PCR, Digital PCR, And End-point PCR Companies:
The following key companies have been profiled for this study on the real-time PCR, digital PCR, and end-point PCR market
- Abbott
- Qiagen
- Bio-Rad Laboratories Inc.
- Agilent Technologies, Inc.
- Thermo Fisher Scientific, Inc.
- GE Healthcare
- bioMérieux
- F. Hoffmann-La Roche Ltd
- Fluidigm Corporation
Recent Developments
-
In February 2026, Co-Diagnostics’ joint venture CoSara received regulatory approval from CDSCO to manufacture and commercialize the CoSara PCR Pro real-time PCR instrument in India, supporting decentralized PCR testing expansion. Approval enables localized production, reducing dependency on imports and improving the affordability and accessibility of advanced molecular diagnostics.
-
In January 2026, HLB Panagene entered into a collaboration with RocGene to distribute molecular diagnostics solutions, including real-time PCR platforms, enhancing market penetration and access to advanced PCR technologies.
-
In April 2026, Thermo Fisher Scientific launched the Applied Biosystems PowerFlex Thermal Cycler, a next-generation PCR system designed to enhance flexibility, precision, and productivity in modern molecular laboratories.
-
In December 2025, Co-Diagnostics introduced advancements in its Co-Dx PCR platform with integrated AI and cloud-based connectivity, enabling real-time data analysis and enhancing point-of-care diagnostic capabilities. This innovation improves diagnostic accuracy and reduces reliance on skilled personnel by automating data interpretation. It also supports decentralized testing by enabling remote access and faster clinical decision-making across healthcare settings.
-
In April 2025, Molbio Diagnostics launched an indigenously developed HPV test on its Truenat real-time PCR platform for cervical cancer screening, supporting large-scale and decentralized testing in resource-limited settings. This development enhances early disease detection and expands access to molecular diagnostics in underserved regions. It also reduces dependency on centralized laboratories, improving screening efficiency and overall healthcare outcomes.
Real-time PCR, Digital PCR, And End-point PCR Market Report Scope
Report Attribute
Details
Market size value in 2026
USD 11.54 billion
Revenue forecast in 2033
USD 21.31 billion
Growth rate
CAGR of 9.2% from 2021 to 2033
Base year for estimation
2025
Historical data
2021 - 2024
Forecast period
2026 - 2033
Quantitative units
Revenue in USD billion/million, and CAGR from 2021 to 2033
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Technology, application, product, region
Regional scope
North America; Europe; Asia Pacific; Latin America; MEA
Country scope
U.S.; Canada; Germany; UK; France; Italy; Spain; Denmark; Sweden; Norway; China; Japan; India; Australia; South Korea; Thailand; Brazil; Mexico; Argentina; South Africa; Saudi Arabia; UAE; Kuwait
Key companies profiled
Abbott; Qiagen; Bio-Rad Laboratories Inc.; Agilent Technologies, Inc.; Thermo Fisher Scientific, Inc.; GE Healthcare; bioMérieux; F. Hoffmann-La Roche Ltd; Fluidigm Corporation
Customization scope
Free report customization (equivalent up to 8 analyst’s working days) with purchase. Addition or alteration to country, regional & segment scope.
Pricing and purchase options
Avail of customized purchase options to meet your exact research needs. Explore purchase options
Global Real-time PCR, Digital PCR, And End-point PCR Market Report Segmentation
This report forecasts revenue growth and provides an analysis on the latest trends in each of the sub-segments from 2018 to 2030. For the purpose of this report, Grand View Research has segmented the global real time PCR, digital PCR, and end-point PCR market report on the basis of technology, application, product, and regions.

-
Technology Outlook (Revenue, USD Million, 2021 - 2033)
-
Quantitative
-
Consumables & Reagents
-
Instruments
-
Software & Services
-
-
Digital
-
Consumables & Reagents
-
Instruments
-
Software & Services
-
-
End-point
-
Consumables & Reagents
-
Instruments
-
Software & Services
-
-
-
Product Outlook (Revenue, USD Million, 2021 - 2033)
-
Consumables & Reagents
-
Instruments
-
Software & Services
-
-
Application Outlook (Revenue, USD Million, 2021 - 2033)
-
Clinical
-
Pathogen Testing
-
Oncology testing
-
Blood Screening
-
Liquid Biopsy
-
dPCR-based Non-Invasive Prenatal Testing (NIPT)
-
Others
-
-
Research
-
Stem Cell Research
-
DNA Cloning & Sequencing
-
Recombinant DNA Technology
-
Rare Mutation Detection
-
Gene Expression
-
Single Cell Analysis
-
Microbiome Analysis
-
Copy Number Variation Analysis
-
Library Quantification (NGS)
-
Characterization of Low-fold Changes in mRNA and miRNA Expression.
-
Species Identification
-
GMO Detection
-
Others
-
-
Forensic
-
-
Regional Outlook (Revenue, USD Million, 2021 - 2033)
-
North America
-
U.S.
-
Canada
-
Mexico
-
-
Europe
-
UK
-
Germany
-
France
-
Italy
-
Spain
-
Denmark
-
Sweden
-
Norway
-
-
Asia Pacific
-
Japan
-
China
-
India
-
Australia
-
South Korea
-
Thailand
-
-
Latin America
-
Brazil
-
Argentina
-
-
Middle East & Africa
-
South Africa
-
Saudi Arabia
-
UAE
-
Kuwait
-
-
Frequently Asked Questions About This Report
b. The global real-time PCR, endpoint PCR, and digital PCR market size was estimated at USD 7.68 billion in 2023 and is expected to reach USD 5.59 billion in 2024.
b. The global real-time PCR, endpoint PCR, and digital PCR market is anticipated to grow at a compound annual growth rate of 4.2% from 2024 to 2030 to reach USD 7.18 billion by 2030.
b. The consumables and reagents segment held the largest market share over 62.62% in terms of value in 2023. This can be attributed to the increased demand for COVID-19 testing and technological advancements in PCR techniques.
b. Some key players operating in the real-time PCR, endpoint PCR, and digital PCR market include Bio-Rad Laboratories Inc.; Thermo Fisher Scientific, Inc.; Qiagen; Abbott; Agilent Technologies, Inc.; BIOMÉRIEUX; Fluidigm Corporation.; and F. Hoffmann-La Roche Ltd.
b. Key factors that are driving the real-time PCR, endpoint PCR, and digital PCR market growth include the high prevalence of target diseases and rapid technological advancements such as the development of high-performance and superior qPCR and dPCR systems.
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










