- Home
- »
- Medical Devices
- »
-
Transcatheter Heart Valve Market Size, Industry Report, 2033GVR Report cover
![Transcatheter Heart Valve Market Size, Share & Trends Report]()
Transcatheter Heart Valve Market (2025 - 2033) Size, Share & Trends Analysis Report By Application (Transcatheter Aortic Valve, Transcatheter Pulmonary Valve, Transcatheter Mitral Valve), By Technology (Self-Expanded, Balloon Expanded), By Region, And Segment Forecasts
- Report ID: GVR-1-68038-096-5
- Number of Report Pages: 120
- Format: PDF
- Historical Range: 2021 - 2024
- Forecast Period: 2025 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
Transcatheter Heart Valve Market Summary
The global transcatheter heart valve market size was estimated at USD 5.49 billion in 2024 and projected to reach USD 21.24 billion by 2033, growing at a CAGR of 16.49% from 2025 to 2033. The rising prevalence of heart valve diseases, particularly in the aging population, is driving the demand for transcatheter heart valve (THV) procedures, such as TAVR, especially for conditions such as aortic stenosis and mitral regurgitation.
Key Market Trends & Insights
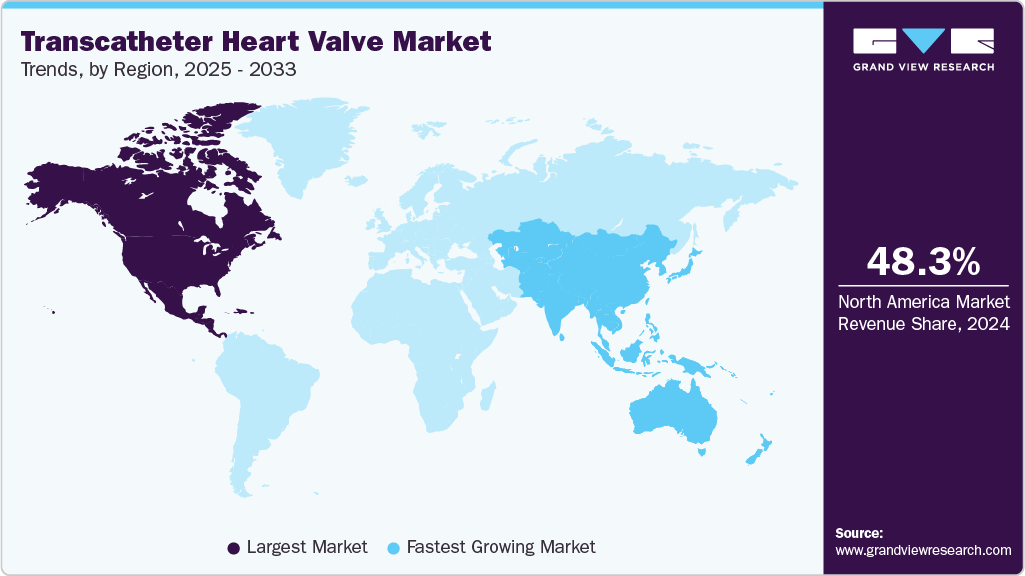
- The North America transcatheter heart valve market held the largest share of 48.30% in 2024.
- The transcatheter heart valve industry in the U.S. is expected to grow significantly from 2025 to 2033.
- By application, the transcatheter aortic valve segment held the highest market share of 81.67% in 2024.
- By technology, the self-expanded transcatheter valve segment held the highest market share in 2024.
Market Size & Forecast
- 2024 Market Size: USD 5.49 Billion
- 2033 Projected Market Size: USD 21.24 Billion
- CAGR (2025-2033): 16.49%
- North America: Largest Market in 2024
- Asia Pacific: Fastest Growing Market
Key factors propelling the growth of the transcatheter heart valve industry include continuous improvements in THV design, advancements in imaging and delivery systems, and the increasing preference for minimally invasive surgeries, which offer shorter recovery times, reduced complications, and lower healthcare costs compared to traditional open-heart surgery.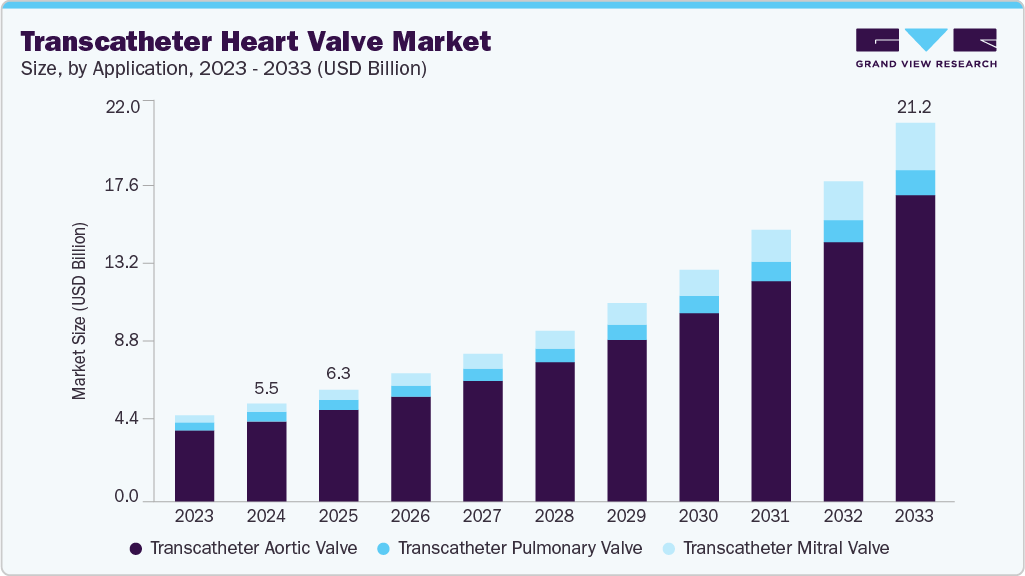
In September 2024, the CDC reported that 75% of adults in the U.S. have limited knowledge about heart valve disease. This lack of awareness extends to high-risk groups, particularly individuals aged 65 and older. Each year, over 5 million people in the U.S. receive a diagnosis of heart valve disease, contributing to more than 25,000 fatalities attributed to this condition annually.
The growing prevalence of heart valve diseases, particularly in the aging population, significantly drives the demand for transcatheter heart valve (THV) procedures. Conditions such as aortic stenosis and mitral regurgitation are becoming more common, necessitating advanced treatment solutions such as THVs. In September 2024, the CDC reported that 75% of adults in the U.S. have limited knowledge about heart valve disease. This lack of awareness extends to high-risk groups, particularly individuals aged 65 and older. Each year, over 5 million people in the U.S. receive a diagnosis of heart valve disease, contributing to more than 25,000 fatalities attributed to this condition annually.

Continuous improvements in THV design, including enhanced valve durability, ease of implantation, and improved patient outcomes, are crucial drivers. Advancements in imaging techniques and valve delivery systems have also significantly improved procedural success and expanded the patient pool eligible for THV procedures. In August 2024, Boston Scientific announced that its ACURATE Prime Aortic Valve System has received CE mark approval. This state-of-the-art transcatheter aortic valve replacement system features a new valve size to serve patients with larger anatomies and is indicated for individuals at low, intermediate, and high risk experiencing severe aortic stenosis. The system features a self-expanding design and a refined deployment mechanism, enhancing the accuracy of valve positioning to improve overall patient outcomes.
There is an increasing demand for minimally invasive surgeries, including transcatheter valve replacement (TAVR), due to their shorter recovery times, reduced complication risks, and lower overall healthcare costs compared to traditional open-heart surgery. This shift towards non-invasive procedures is a critical factor propelling the market. In April 2024, Abbott announced FDA approval for its TriClip system, a transcatheter edge-to-edge repair technology for treating tricuspid regurgitation (TR). This first-of-its-kind device enables less invasive treatment, enhancing patient outcomes without the need for high-risk open-heart surgery. Clinical trials showed significant improvements in TR severity and quality of life for patients treated with TriClip.
Key Clinical Trials in the Transcatheter Heart Valve Market, by Region
Trial
Company/
Product
Summary
Primary Completion Date
Study Completion Date (Estimated)
Results
Market Outlook
PORTICO IDE
Portico transcatheter aortic valve (Abbott )
A head-to-head randomized trial comparing Abbott’s Portico device against an already commercialized TAVR system in the U.S. The study is designed to measure both efficacy and safety endpoints.
October 2019
December 2026
The Portico valve was non-inferior to the control valve, with a slightly higher 30-day safety event rate (13.8% vs 9.6%) and comparable 1-year effectiveness (14.8% vs 13.4%).
Positive results strengthen Abbott’s competitive position in the U.S. TAVR market, supporting Portico adoption alongside Navitor for broader patient coverage.
VANTAGE
Navitor TAVI system (Abbott)
This single-arm clinical investigation is designed to evaluate Abbott’s Navitor TAVR system in patients diagnosed with severe, symptomatic aortic stenosis. The trial focuses on safety outcomes and device performance.
June 2025
February 2026
Navitor met all safety and effectiveness primary endpoints, supporting an expanded CE Mark indication in Europe for low- and intermediate-risk severe aortic stenosis patients
Successful outcomes position Navitor as a growth driver in Europe and potential U.S. expansion candidate, improving Abbott’s portfolio differentiation in the intermediate- and low-risk segment.
PROGRESS
SAPIEN 3 Ultra Delivery System with SAPIEN 3 Ultra THV (Edwards Lifesciences)
A randomized study assessing Edwards Lifesciences’ SAPIEN 3 valve in patients with moderate, calcific aortic stenosis-a population not traditionally eligible for TAVR. The trial aims to establish safety and efficacy in this earlier-stage disease segment.
December 2019
Upgrade report license to gain access to the complete analysis clinical trials
Upgrade report license to gain access to the complete analysis. Of clinical trials
Technological advancements in transcatheter heart valve therapy have significantly expanded both the safety profile and patient population suitable for intervention. Key developments include refined delivery systems with lower profiles for minimally invasive access, improved imaging and planning tools (including 3D CT, real-time imaging, and AI-driven simulation), and new valve materials and frame designs that enhance durability, reduce paravalvular leak, and simplify future interventions. For example, the deployment of the SAPIEN 3 Ultra RESILIA valve by Edwards Lifesciences incorporates RESILIA bovine-pericardial leaflet tissue designed to resist calcification and allow dry storage, thereby improving long-term durability and ease of handling.
Recent Product Launches in the Transcatheter Heart Valve Market
Company
Month and Year
Initiative
Medtronic
October 2025
Medtronic launched the Avalus Ultra surgical aortic valve in India, enhancing its cardiac surgery portfolio with a next-generation bioprosthesis designed for ease of implantation, durable performance, and support for future valve-in-valve procedures.
Edwards Lifesciences Corporation
September 2025
Edwards Lifesciences Corporation launched its next-generation balloon-expandable transcatheter heart valve platform in India, offering a minimally invasive, durable solution for patients with aortic stenosis who are not candidates for open-heart surgery, incorporating calcification-resistant tissue technology to reduce structural valve deterioration and enhance long-term outcomes.
Market Concentration & Characteristics
The degree of innovation in the transcatheter heart valve industry is currently high, driven by continuous advancements in valve design, materials, and delivery mechanisms. Companies focus on improving valve durability, reducing procedural complications, and enhancing patient outcomes through cutting-edge technologies, such as advanced imaging systems and robotic-assisted surgery. In April 2024, Foldax, Inc. announced that its TRIA heart valves have exceeded 200 patient life years in human recipients. Designed to improve management of heart valve disease, these valves utilize a proprietary polymer, LifePolymer, which enhances durability and minimizes calcification.
The level of merger and acquisition activities in the transcatheter heart valve market is considered medium. Key players in the medical device industry are acquiring or merging with smaller companies that have innovative technologies or a strong market presence in the cardiovascular space. These acquisitions allow larger companies to enhance their product portfolios and expand into new market segments. In August 2024, Edwards Lifesciences Corporation acquired JC Medical, gaining its J-Valve TAVR technology for treating severe symptomatic native aortic regurgitation. This acquisition continued the company's strategic focus on structural heart disease and expanded its portfolio of transcatheter therapies.

The impact of regulations on the transcatheter heart valve industry is rated as high, with strict oversight from bodies such as the U.S. FDA and European Medicines Agency (EMA). Regulatory approval processes can significantly influence market entry and product development timelines. In November 2025, Anteris Technologies received FDA clearance to initiate its PARADIGM global pivotal trial of the DurAVR transcatheter heart valve for the treatment of severe calcific aortic stenosis. The randomized trial will enroll 1,000 patients across the U.S., Europe, and Canada, comparing DurAVR against approved TAVR devices, with results supporting future FDA PMA and CE Mark submissions.
Product expansion within the transcatheter heart valve industry is evaluated as high, with companies constantly expanding their product lines to address different types of heart valve diseases and patient needs. This includes the development of new valve sizes, materials, and designs, as well as exploring novel delivery systems. The focus on expanding into new therapeutic areas, such as mitral and tricuspid valve interventions, is also contributing to the growth and diversification of THV offerings.
The regional expansion of the transcatheter heart valve industry is classified as medium. Companies are increasingly focusing on penetrating emerging markets in Asia-Pacific, Latin America, and the Middle East. While these regions present significant growth opportunities due to rising healthcare demands and improving healthcare infrastructure, they also face challenges, such as regulatory hurdles, cultural differences, and cost barriers, which slow down the pace of market penetration compared to more established markets such as North America and Europe. However, growing awareness and investments in healthcare are expected to accelerate expansion over time.
Application Insights
The transcatheter aortic valve segment held the highest market share of 81.67% in 2024, driven by rising healthcare expenditures, a higher prevalence of chronic heart disease, and an increase in cases of severe aortic stenosis. Advancements in tricuspid valve interventions and neochord systems are expected to increase usage rates during the forecast period. In May 2024, MicroPort CardioFlow’s VitaFlow Liberty Transcatheter Aortic Valve received CE certification, marking it as the first Chinese TAVI solution approved in the EU. With over 47 million patients worldwide suffering from aortic valve diseases, this second-generation product enhances design features for better performance and safety. It has been introduced in nearly 700 hospitals across various countries, addressing the growing demand for TAVI treatments.
The transcatheter mitral valve (TMV) segment is anticipated to grow at the fastest CAGR over the forecast period due to the demand for less invasive treatments for mitral valve diseases. TMV procedures provide significant advantages over traditional open-heart surgery, offering faster recovery times and reducing patient risks. As innovation progresses, TMV devices are becoming more refined, enhancing patient outcomes. In July 2024, Edwards Lifesciences announced its acquisition of Innovalve Bio Medical Ltd., enhancing its transcatheter mitral valve replacement technologies. This acquisition follows an initial investment in 2017 and aims to address significant unmet needs in structural heart treatment. Innovalve will join Edwards’ transcatheter mitral and tricuspid therapies group, with the closing expected by the end of 2024.
Technology Insights
In 2024, the self-expanded transcatheter valve segment led the transcatheter heart valve market with the largest share of 63.97%. This segment predominantly consists of first- and second-generation transcatheter heart valves, contributing to its significant market presence. In April 2024, Allegheny General Hospital led an international clinical trial on transcatheter aortic valve replacements (TAVRs), comparing self-expanding and balloon-expandable valves in patients with aortic stenosis. The study found that self-expanding valves demonstrated superior blood flow results after one year. Notably, nearly 90% of participants were women, addressing the historical underrepresentation of women in cardiovascular trials. Aortic stenosis affects over 20% of older Americans, posing significant health risks if untreated.
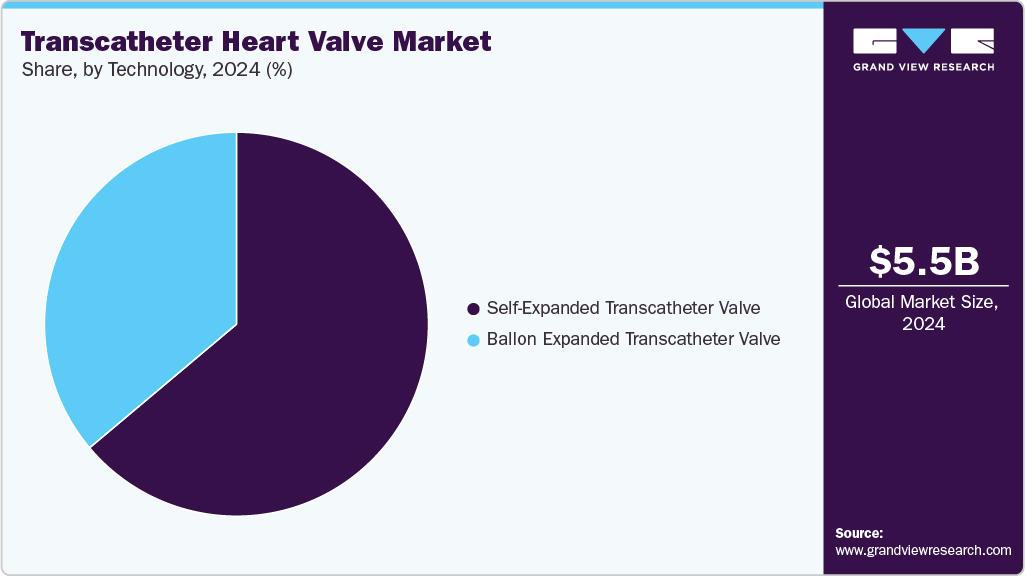
Advancements in technology, increased awareness of balloon-expandable valve systems, and improved clinical outcomes drive rapid growth in the balloon-expanded transcatheter valve segment, projected to experience the highest CAGR during the forecast period. Innovations in techniques and technology allow valve repairs and replacements through mini-sternotomies and mini-thoracotomies. These methods utilize smaller incisions and longer surgical instruments, resulting in faster wound healing, reduced pain, quicker recovery, and earlier hospital discharge than traditional incisions. In September 2024, Venus Medtech made a significant advancement in transcatheter aortic valve replacement technology. The company achieved a milestone by executing the first two procedures of its newly designed balloon-expandable valve system, Venus-Vitae, during an international clinical trial named Venus-Vitae SMART-ALIGN at Prince of Wales Hospital in Hong Kong, China.
Regional Insights
North America's transcatheter heart valve market dominated with a 48.30% share in 2024, driven by advanced healthcare infrastructure, high awareness levels, and strong healthcare spending. The region benefits from cutting-edge medical technologies and a significant demand for minimally invasive procedures. The U.S. leads the market in the adoption of THV technologies, largely due to key industry players, favorable reimbursement policies, and robust healthcare systems. In October 2024, Edwards Lifesciences announced results from the EARLY TAVR Trial, which studied asymptomatic severe aortic stenosis (AS). The trial found that patients undergoing early intervention with transcatheter aortic valve replacement (TAVR) had significantly better outcomes than those receiving clinical surveillance, with 26.8% of TAVR patients experiencing adverse events compared to 45.3% in the surveillance group over a median follow-up of 3.8 years.
U.S. Transcatheter Heart Valve Market Trends
In the U.S., the transcatheter heart valve industry is characterized by the country’s strong healthcare system, a high incidence of heart valve diseases, and significant research and development in the medical device sector. Regulatory bodies, such as the FDA, have proactively approved transcatheter valve technologies, promoting market growth. In May 2024, 4C Medical Technologies, Inc. received Breakthrough Device designation from the U.S. FDA for its AltaValve System, a transcatheter mitral valve replacement device. This designation helps expedite the review process, allowing for quicker access to the technology for patients if approved.
Europe Transcatheter Heart Valve Market Trends
In Europe, the transcatheter heart valve industry is anticipated to grow significantly over the forecast period. Europe represents a key market for THV devices, with countries such as Germany, France, and the UK seeing rapid adoption of these technologies. The region’s healthcare infrastructure is well-developed, with strong government and private sector investment in cardiovascular health. Europe's favorable regulatory environment, which includes the approval of THVs under the EU Medical Device Regulation (MDR), enables quicker product launches. In August 2025, Abbott received CE Mark in Europe for an expanded indication of its Navitor TAVI system, allowing minimally invasive treatment of symptomatic, severe aortic stenosis in low- and intermediate-risk patients, supported by positive outcomes from the VANTAGE trial.
The transcatheter heart valve market in the UK has witnessed significant growth in the THV market, particularly in the transcatheter aortic valve replacement (TAVR) for aortic stenosis. The National Health Service (NHS) has played a crucial role in adopting THV devices by integrating them into its treatment protocols. In July 2024, Abbott launched the Navitor Vision valve in the UK and Ireland, a minimally invasive device for transcatheter aortic valve implantation (TAVI). Designed for patients with severe aortic stenosis at high surgical risk, it enhanced visualization during the procedure to ensure precise implantation. TAVI served as a less invasive alternative to open-heart surgery, providing an effective treatment option for nearly 300,000 affected individuals in the UK.
France’s transcatheter heart valve market is characterized by its advanced healthcare infrastructure, high incidence of heart valve diseases, and strong government support for cardiovascular treatment innovation. The adoption of THVs, particularly TAVR, has been significant, driven by the positive clinical outcomes and cost-effectiveness of these procedures. France's healthcare system provides excellent access to high-quality medical services, facilitating the widespread use of advanced devices such as THVs.In March 2025, French registry data from FRANCE 2 and FRANCE TAVI showed that fewer than 1 in 50 TAVI patients required reintervention within 8 years, with most redo procedures occurring within the first year.
Asia Pacific Transcatheter Heart Valve Market Trends
The Asia Pacific transcatheter heart valve industry is experiencing significant growth driven by an increasing number of patients diagnosed with cardiovascular diseases due to changing lifestyles, urbanization, and an aging population. While the market is still in the early stages of adoption compared to North America and Europe, it is growing rapidly. In December 2024, the VitaFlow Liberty Transcatheter Aortic Valve and Retrievable Delivery System received marketing approval from the Korean Ministry of Food and Drug Safety. This electric retrievable TAVI system features a hybrid-density self-expanding stent and advanced design elements that enhance support and reduce paravalvular leaks. Its innovative delivery system enables precise valve release and retrieval, while offering 360° flexibility for enhanced surgical precision.
The Japan transcatheter heart valve market is experiencing significant growth driven by the country's high incidence of heart valve diseases, particularly in its elderly population. Japan's advanced healthcare infrastructure and strong regulatory framework have contributed to the adopting THV procedures. In November 2023, Meril Life Sciences announced a partnership with Japan Lifeline to promote its Myval Octacor transcatheter heart valve in Japan, pending regulatory approval. The valve is designed for aortic replacements, offering a wider size range and better placement accuracy.
The transcatheter heart valve market in India is witnessing robust growth because India is one of the fastest-growing markets for transcatheter heart valves in Asia, driven by a rapidly increasing burden of cardiovascular diseases, rising healthcare awareness, and improving healthcare access. In October 2024, the Sree Chitra Tirunal Institute inaugurated a Centre of Excellence for minimally invasive cardiovascular devices, including transcatheter aortic valves. The center aims to improve patient outcomes for high-risk individuals and support India's 'Make in India' initiatives through collaboration with the medical device industry.
Latin America Transcatheter Heart Valve Market Trends
The transcatheter heart valve industry in Latin America is experiencing a notable shift driven by an emerging market for transcatheter heart valves, with Brazil and Argentina representing the largest markets. The region is witnessing increasing demand for advanced medical devices, particularly in countries with improving healthcare systems and rising cardiovascular disease rates. In November 2022, Venus Medtech advanced its First-in-Man (FIM) studies for the Venus PowerX transcatheter aortic valve replacement system, completing three procedures in Argentina. This marks the device's earliest clinical use outside China, demonstrating its potential value in addressing clinical demand.
The Brazil transcatheter heart valve market is characterized by the country's significant burden of cardiovascular diseases, particularly in its aging population, and Brazil's healthcare system has increasingly adopted minimally invasive techniques such as TAVR. Despite challenges related to healthcare disparities and high treatment costs, Brazil is expected to continue growing in the adoption of THV technologies, particularly in large urban centers, with increasing government and private sector support for cardiovascular health. In May 2025, MicroPort CardioFlow’s VitaFlow Liberty TAVI system received ANVISA approval in Brazil, marking its commercial launch. The device features a retrievable design with enhanced deployment precision, catering to the growing Brazilian TAVI market.
Middle East & Africa Transcatheter Heart Valve Market Trends
The transcatheter heart valve industry in Middle East and Africa is relatively small but growing, driven by increasing healthcare investments, rising awareness of cardiovascular diseases, and improving medical infrastructure. Countries such as Saudi Arabia, the UAE, and South Africa are leading the way in adopting advanced cardiovascular treatments, including THV procedures. In October 2024, Cleveland Clinic Abu Dhabi achieved a significant milestone by completing 500 Transcatheter Aortic Valve Implantation (TAVI) procedures. Since introducing TAVI in the UAE in 2015, the Heart, Vascular & Thoracic Institute has transformed the treatment of aortic stenosis, offering a minimally invasive alternative to open-heart surgery and enhancing outcomes for higher-risk patients.
The transcatheter heart valve market in Saudi Arabia is witnessing robust growth fueled by investments in healthcare and the adoption of advanced medical technologies. The country has a high prevalence of cardiovascular diseases, and the healthcare system is actively adopting minimally invasive treatments such as TAVR. In January 2024, MicroPort CardioFlow progressed in registering the VitaFlow Liberty Transcatheter Aortic Valve and Retrievable Delivery System in Saudi Arabia. This milestone was a key step towards enhancing cardiac care in the region.
Key Transcatheter Heart Valve Company Insights
Key companies in the transcatheter heart valve market are actively pursuing various strategic initiatives to enhance their market presence. These initiatives include investing in research and development to innovate and improve the properties of the transcatheter heart valve, which aims to achieve better clinical outcomes and enhance patient comfort. To meet the diverse needs of the global market, these players focus on product diversification, offering a range of cements suitable for various dental procedures.
Key Transcatheter Heart Valve Companies:
The following are the leading companies in the transcatheter heart valve market. These companies collectively hold the largest market share and dictate industry trends.
- Medtronic
- Abbott
- Braile Biomédica
- Meril Life Sciences Pvt. Ltd., Inc.
- Edwards Lifesciences Corporation
- MicroPort Scientific Corporation
- Venus Medtech (Hangzhou) Inc.
- JenaValve
- Biosensors International Group, Ltd
- HLT Medical (Bracco)
Recent Developments
-
In September 2025, Medtronic launched its Evolut FX+ transcatheter aortic valve in India, following approval by the CDSCO in June 2025. The valve offers enhanced coronary access and proven hemodynamic performance, improving treatment options for patients with severe aortic stenosis.
-
In June 2025, SIMS Hospital in Chennai successfully carried out India’s first hybrid procedure combining transcatheter aortic valve replacement (TAVR) with a Frozen Elephant Trunk (FET) graft on a 61-year-old patient who had previously undergone a Bentall operation.
-
In February 2025, Eisenhower Health was the first hospital in the U.S. to serve as an Edwards Benchmark Case Observation Site, reflecting its record of nearly 1,000 procedures with a 99% success rate and its role in advancing minimally invasive cardiac care.
-
In December 2024, Sahajanand Medical Technologies (SMT), a global innovator in cardiovascular solutions, introduced its Hydra Transcatheter Aortic Valve Replacement system in Mexico. This launch represents a significant advancement in SMT’s commitment to providing state-of-the-art, minimally invasive cardiac treatments to patients across the globe.
-
In November 2024, Meril Life Sciences unveiled its Myval Octapro Transcatheter Heart Valve (THV) at major cardiology conferences, highlighting its advancements in valve replacement technology. The new valve is designed to improve procedural outcomes and patient compatibility. Meril’s ongoing research and innovation reflect its dedication to enhancing care in structural heart disease.
-
In November 2024, Abbott introduced its experimental balloon-expandable transcatheter aortic valve implantation (TAVI) system for patients with severe aortic stenosis. This new system aims to facilitate AI-assisted procedures and provide a less invasive treatment option for individuals at high risk of undergoing open-heart surgery. Using a balloon, the TAVI device expands a new valve inside the narrowed aortic valve, serving as a modern alternative to conventional methods.
-
In March 2024, Medtronic revealed that the FDA approved its Evolut FX+ transcatheter aortic valve replacement (TAVR) system for patients with symptomatic severe aortic stenosis. This updated system features a diamond-shaped frame that offers enhanced coronary access while maintaining the high valve performance the Evolut platform is known for. If left untreated, severe aortic stenosis can severely impact patient health and lead to heart failure within two years.
Transcatheter Heart Valve Market Report Scope
Report Attribute
Details
Market size value in 2025
USD 6.26 billion
Revenue forecast in 2033
USD 12.24 billion
Growth rate
CAGR of 16.49% from 2025 to 2033
Actual data
2021 - 2024
Forecast period
2025 - 2033
Quantitative units
Revenue in USD million/billion and CAGR from 2025 to 2033
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Application, technology, region
Regional scope
North America; Europe; Asia Pacific; Latin America; MEA
Country scope
U.S.; Canada; Mexico; UK; Germany; France; Italy; Spain; Sweden; Denmark; Norway; China; Japan; India; Australia; South Korea; Thailand; Brazil; Argentina; Saudi Arabia; South Africa; UAE; Kuwait
Key companies profiled
Medtronic; Abbott; Braile Biomédica; Meril Life Sciences Pvt. Ltd., Inc.; Edwards Lifesciences Corporation; MicroPort Scientific Corporation; Venus Medtech (Hangzhou) Inc.; JenaValve; Biosensors International Group, Ltd; HLT Medical (Bracco)
Customization scope
Free report customization (equivalent up to 8 analyst’s working days) with purchase. Addition or alteration to country, regional & segment scope
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
Global Transcatheter Heart Valve Market Report Segmentation
This report forecasts revenue growth at the global, regional, and country levels and provides an analysis of the latest industry trends in each of the sub-segments from 2021 to 2033. For this study, Grand View Research has segmented the global transcatheter heart valve market report based on application, technology, and region:

-
Application Outlook (Revenue, USD Million, 2021 - 2033)
-
Transcatheter Aortic Valve
-
Transcatheter Pulmonary Valve
-
Transcatheter Mitral Valve
-
-
Technology Outlook (Revenue, USD Million, 2021 - 2033)
-
Balloon Expanded Transcatheter Valve
-
Self-Expanded Transcatheter Valve
-
-
Regional Outlook (Revenue, USD Million, 2021 - 2033)
-
North America
-
U.S.
-
Canada
-
Mexico
-
-
Europe
-
Germany
-
UK
-
France
-
Italy
-
Spain
-
Denmark
-
Sweden
-
Norway
-
-
Asia Pacific
-
China
-
Japan
-
India
-
South Korea
-
Australia
-
Thailand
-
-
Latin America
-
Brazil
-
Argentina
-
-
Middle East and Africa (MEA)
-
South Africa
-
Saudi Arabia
-
UAE
-
Kuwait
-
-
About the authors:
Author: GVR Medical Devices Research Team | Last Updated:
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.









