- Home
- »
- Medical Devices
- »
-
U.S. Automated Medical Washer Disinfector Market, 2033GVR Report cover
![U.S. Automated Medical Washer Disinfector Market Size, Share & Trends Report]()
U.S. Automated Medical Washer Disinfector Market (2025 - 2033) Size, Share & Trends Analysis Report By Product Type (Floor Standing, Benchtop), By Capacity, By Chamber Type, By Device Classification, By End Use, And Segment Forecasts
- Report ID: GVR-4-68040-667-8
- Number of Report Pages: 110
- Format: PDF
- Historical Range: 2021 - 2024
- Forecast Period: 2025 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
Market Size & Trends
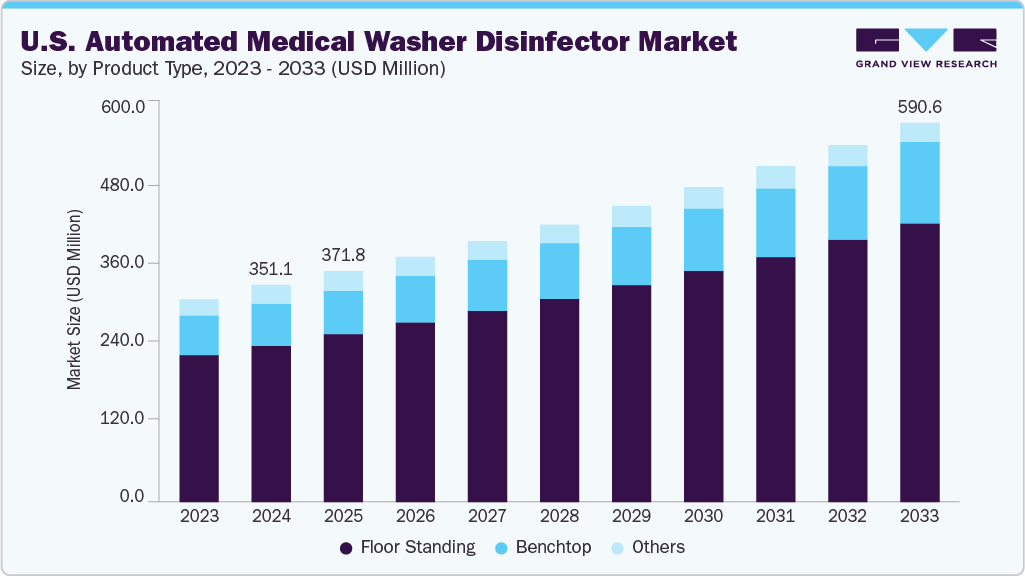
The U.S. automated medical washer disinfector market size was estimated at USD 351.11 million in 2024 and is expected to grow at a CAGR of 5.96% from 2025 to 2033. The market growth is attributed to the potent combination of high surgical volumes, stringent infection control regulations, and advancements in healthcare infrastructure. With the continued growth of ambulatory surgical centers (ASCs), there is a rising need for efficient, high-throughput instrument reprocessing systems. Regulatory agencies such as the FDA, CDC, and OSHA mandate compliance with strict decontamination protocols, pushing healthcare facilities to adopt validated, automated washer-disinfectors that ensure consistent cleaning, thermal disinfection, and traceability. Additionally, the U.S. healthcare system’s growing focus on patient safety, sustainability, and digital integration has spurred demand for smart washer disinfectors that reduce manual labor, conserve water and energy, and support real-time cycle monitoring, making them essential in modern sterile processing environments.
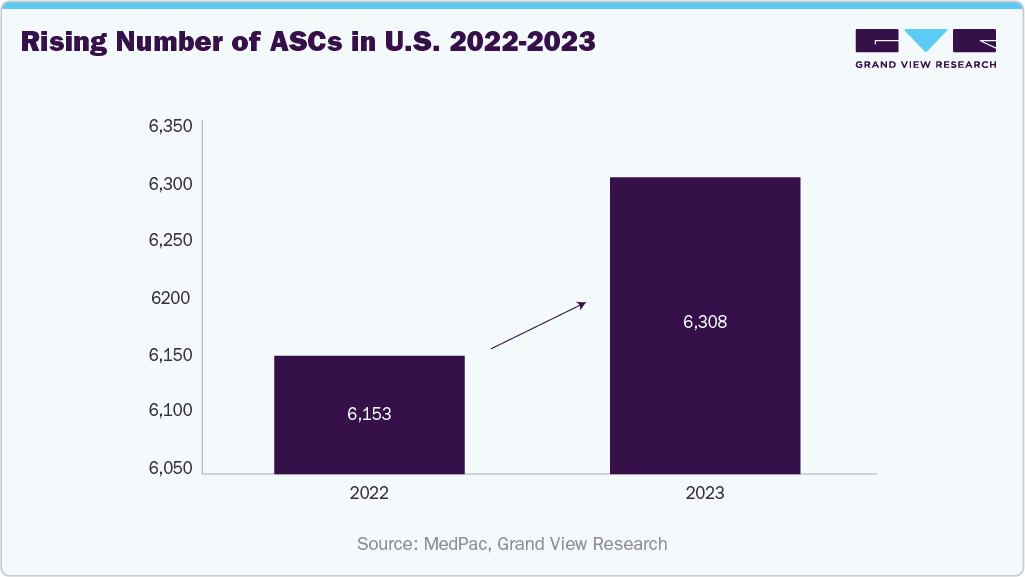
The rapid expansion of ambulatory surgical centers (ASCs) in the U.S. has emerged as a major driver of the automated medical washer-disinfector market, as more surgical procedures shift from inpatient to outpatient settings. ASCs increasingly perform same-day, minimally invasive surgeries, creating a critical need for compact, efficient, and high-performing washer disinfectors that can handle frequent instrument turnover within limited space. These facilities prioritize equipment that offers fast cycle times, reduced water and detergent usage, and ease of use, making benchtop and medium-capacity systems particularly attractive. Additionally, ASCs are held to the same infection control standards as hospitals, necessitating validated cleaning protocols and traceable disinfection processes, further fueling demand for automated solutions. As the ASC model becomes more prominent due to its cost-effectiveness and patient convenience, it significantly boosts the need for scalable, regulation-compliant washer disinfectors across the U.S. market.
The consistently high volume of surgical procedures in the U.S. is a major driver for the growth of the automated medical washer disinfector market, as hospitals and surgical centers require efficient, reliable systems to manage the rapid reprocessing of instruments. According to the American College of Rheumatology in 2024, over 1.3 million hip and knee replacement surgeries were performed annually in the U.S., in addition to hundreds of thousands of other elective and emergency surgeries. This immense procedural load places significant pressure on Central Sterile Supply Departments (CSSDs) to maintain instrument availability while ensuring strict infection control compliance.
Automated washer disinfectors are essential in this environment as they offer standardized cleaning, thermal disinfection, and fast turnaround times, helping facilities maintain surgical schedules without compromising patient safety. Their ability to process large instrument sets with minimal human error also aligns with regulatory expectations and supports efficiency in high-throughput settings.
Cosmetic Surgery Procedures 2023
Category
Procedure
2023
2022
% Change
Breast
Breast augmentation (implant placement for primary/revisions)
304,181
298,568
2%
Breast implant removals (augmentation patients only)
41,115
37,679
9%
Breast lift (mastopexy)
153,600
143,364
7%
Breast reduction (aesthetic patients only)
76,031
71,364
7%
Breast reduction in males (gynecomastia surgery)
25,888
24,517
6%
Body
Buttock augmentation with fat grafting (Brazilian butt lift)
29,383
28,638
3%
Buttock implants
1,234
1,164
6%
Buttock lift
7,748
7,338
6%
Labiaplasty
10,631
10,433
2%
Liposuction
347,782
325,669
7%
Lower body lift
10,947
10,445
5%
Thigh lift
9,600
9,421
2%
Tummy tuck (abdominoplasty)
170,110
161,948
5%
Upper arm lift (brachioplasty)
23,058
21,429
8%
Face
Buccal fat pad removal
4,866
4,543
7%
Cheek implant (malar augmentation)
8,825
8,238
7%
Chin augmentation (mentoplasty)
5,484
5,403
1%
Ear surgery (otoplasty)
4,817
4,713
2%
Eyelid surgery (blepharoplasty)
120,747
115,261
5%
Facelift (rhytidectomy)
78,482
72,668
8%
Facial fat grafting
34,216
33,877
1%
Forehead lift
13,518
13,318
2%
Liposuction (submental)
23,667
22,285
6%
Neck lift
22,007
21,575
2%
Nose reshaping (rhinoplasty)
47,307
44,503
6%
Total
All Procedures Combined
1,575,244
1,498,361
5%
SourceAmerican Society of Plastic Surgeons, Grand View Research
The increasing prevalence of healthcare-associated infections (HAIs) in the U.S. is a significant factor driving the demand for automated medical washer disinfectors. HAIs compromise patient safety and impose significant financial and reputational burdens on healthcare institutions. According to a June 2024 report by the NIH, approximately 1.7 million hospital patients in the U.S. acquire HAIs annually, resulting in around 98,000 deaths. Hospitals are under growing pressure to adopt validated, standardized cleaning and disinfection technologies to reduce infection risks. Automated washer-disinfectors offer consistent thermal disinfection, traceable cycles, and minimal human error, making them essential for meeting infection control protocols mandated by agencies like the CDC and FDA.
Technological Advancements
Company Name
Product Launch
KOLs
Olympus
In June 2023, Olympus has announced the launch of its latest endoscope reprocessing solution, the Olympus ETD, available in two versions: ETD Basic and ETD Premium. This new addition to the company’s Infection Prevention portfolio is designed to enhance the efficiency and sustainability of endoscope reprocessing, while delivering high-performance cleaning and disinfection outcomes. Developed with a focus on operational ease and environmental responsibility, the ETD models aim to support healthcare facilities in achieving improved infection control standards.
"As a leading global medical technology company in endoscopy, patient safety along the entire care pathway is our top priority. Our new Olympus ETD washer disinfector helps healthcare professionals provide the best possible care for their patients by delivering cleaning and disinfection results that exceed standard requirements,” said Oliver Burghardt, Director Olympus EMEA Medical Endoscopy Group."
Source: Olympus, Grand View Research
Market Concentration & Characteristics
The U.S. automated medical washer disinfector industry is experiencing high innovation, fueled by ongoing technological advancements to enhance efficiency, infection control compliance, and sustainability in healthcare settings. Recent innovations include integrating smart cycle tracking, IoT-enabled remote diagnostics, touchscreen user interfaces, and automated detergent dosing systems, all of which improve workflow accuracy and reduce the risk of human error. U.S. manufacturers also prioritize eco-conscious features, such as reduced water and chemical consumption, heat recovery technology, and compact system designs for ambulatory surgical centers (ASCs) and space-limited facilities. Developing multi-chamber washer-disinfectors for high-throughput processing and their seamless integration with digital hospital systems, including traceability and data reporting platforms, demonstrates the market’s commitment to innovation, regulatory compliance, and operational excellence.
Regulations play a pivotal role in shaping the U.S. automated medical washer-disinfector market, driving the adoption of systems that ensure standardized, traceable, and validated reprocessing of medical instruments. Oversight by agencies such as the U.S. Food and Drug Administration (FDA) and the Centers for Disease Control and Prevention (CDC) mandates strict compliance with infection prevention protocols, including the use of washer-disinfectors that meet performance standards under 21 CFR Part 820 and ISO 15883. Facilities must document and validate each cleaning and disinfection cycle, which has led to a surge in demand for automated systems equipped with digital tracking and reporting capabilities.

The U.S. automated medical washer disinfector market has experienced high merger and acquisition (M&A) activity, primarily driven by the need for technological integration, portfolio expansion, and market consolidation among major players. Leading infection control and sterilization companies have acquired smaller, specialized firms to broaden their offerings and accelerate the development of digitally connected, eco-efficient washer disinfectors. For instance, the acquisition of Cantel Medical by Steris in recent years bolstered Steris’s presence in the reprocessing space with complementary endoscope washer-disinfector technologies. M&A activity has enabled companies to access new distribution networks, expand into ambulatory care and outpatient surgical centers, and increase R&D capabilities.
In the U.S. automated medical washer disinfector industry, product substitutes for automated medical washer disinfectors include manual cleaning methods, ultrasonic cleaners, and steam sterilizers (autoclaves) with limited washing capabilities. While manual cleaning using brushes, enzymatic detergents, and soaking trays is still employed in low-volume or resource-constrained settings, it highly depends on human consistency. It often lacks validation and traceability, posing a risk to infection control. Ultrasonic cleaners, commonly used for delicate instruments, provide effective debris removal but do not offer complete disinfection or meet regulatory sterilization requirements on their own.
The market is highly concentrated among large hospitals and central sterile supply departments (CSSDs), which account for most system installations due to their high surgical volumes and strict infection control requirements. These institutions rely on advanced washer-disinfectors to manage daily reprocessing of thousands of surgical instruments across multiple departments, necessitating robust, high-capacity systems with validated cycles and digital traceability. In parallel, the growing number of ambulatory surgical centers (ASCs) and outpatient clinics has contributed to rising demand for compact and mid-capacity units, especially as these facilities expand same-day procedure capabilities. While smaller facilities and specialty practices represent a lower market share, their adoption gradually increases as awareness of regulatory compliance and efficiency benefits spreads.
Product Type Insights
The floor standing segment held the largest market share in 2024.This growth is primarily driven by the widespread adoption of floor-standing washer-disinfectors in large hospitals and central sterile supply departments (CSSDs). These systems are specifically engineered to manage high volumes of surgical instruments, trays, and containers, offering superior capacity, customizable cleaning cycles, and adherence to stringent disinfection standards. Their robust performance, ability to integrate with automated loading and conveyor systems, and compatibility with diverse instrument types make them highly suitable for high-throughput, mission-critical environments. Moreover, the rising number of surgical procedures and ongoing expansion of hospital sterilization infrastructure have amplified the need for durable, full-scale reprocessing systems, solidifying the market dominance of the floor-standing segment.
The benchtop segment is expected to witness significant growth over the forecast period. The growing demand from small to mid-sized healthcare facilities, such as ambulatory surgical centers (ASCs), dental clinics, endoscopy units, and laboratories, is fueling the adoption of compact, benchtop washer-disinfectors. These systems provide a cost-effective and space-efficient solution for environments with lower reprocessing volumes but equally high standards for infection control. As outpatient facilities increasingly perform minimally invasive procedures, the need for validated and automated cleaning solutions that do not require the infrastructure or investment of large-scale floor-standing units is rising rapidly, driving strong growth in this segment.
Capacity Insights
The large capacity (>200 L) segment held the largest share of the U.S. automated medical washer disinfector market in 2024. These large-capacity systems are engineered to efficiently manage high volumes of surgical instruments, trays, and containers, making them indispensable in high-throughput healthcare environments where speed, consistency, and reliability are critical. Their ability to perform multiple reprocessing cycles daily, combined with advanced automation, reduced turnaround times, and validated disinfection protocols, makes them ideally suited for facilities handling many procedures. Moreover, their compatibility with loading trolleys, automated conveyor systems, and digital monitoring platforms enhances workflow efficiency, minimizes manual handling, and supports compliance with rigorous infection control and regulatory standards.
The medium capacity (100-200 L) segment is expected to witness the fastest CAGR of 6.98% over the forecast period. This growth is primarily driven by increasing demand from mid-sized hospitals, ambulatory surgical centers (ASCs), and specialty clinics that seek an optimal balance between processing capacity and space efficiency. Medium-capacity washer-disinfectors provide a versatile solution, offering sufficient throughput to manage substantial instrument loads while maintaining a compact footprint suitable for facilities with limited space or infrastructure. As more healthcare providers shift toward decentralized sterilization models and outpatient surgical procedures continue to rise, these systems are becoming a preferred choice due to their cost-effectiveness, energy efficiency, and ability to meet strict infection control standards without requiring extensive operational resources.
Chamber Type Insights
The single chamber segment dominated the U.S. automated medical washer disinfector industry in 2024, and is expected to witness the fastest CAGR over the forecast period. Single-chamber washer disinfectors are widely favored for their simplicity, reliability, and cost-effectiveness. They are the go-to solution for central sterile supply departments (CSSDs), ambulatory surgical centers (ASCs), and clinics managing routine instrument reprocessing. These units’ streamline operations by performing cleaning, disinfection, and drying in a single cycle, minimizing the need for manual handling and supporting uninterrupted workflow. Their compact design, ability to handle a variety of medical instruments, and compliance with international standards such as ISO 15883 further enhance their appeal. As more healthcare facilities invest in modernizing their sterilization infrastructure, the versatility, space efficiency, and lower operating costs of single-chamber systems have firmly established their leading position in the market.
Device Classification Insights
The critical segment dominated the automated medical washer disinfector market in 2024 is expected to witness the fastest CAGR over the forecast period. This dominance is driven by the rising number of complex surgical procedures and the growing emphasis on infection control and patient safety within operating rooms and specialized clinical departments. Critical medical instruments demand validated, high-level cleaning and thermal disinfection capabilities that automated washer-disinfectors are uniquely equipped to provide. As global healthcare regulations become more stringent and awareness of healthcare-associated infections (HAIs) continues to rise, hospitals and surgical centers increasingly prioritize investments in advanced washer disinfectors that offer traceability, regulatory compliance documentation, and high-throughput performance to meet evolving clinical and operational standards.
End Use Insights
The hospitals segment dominated the U.S. automated medical washer disinfector market in 2024. This dominance is primarily attributed to the high volume of surgical procedures, the presence of central sterile supply departments (CSSDs), and strict compliance with infection control protocols in hospital settings. Hospitals require robust, high-capacity washer-disinfectors capable of efficiently reprocessing a wide range of instruments used across various departments, including surgery, endoscopy, and intensive care units. In response to increasing regulatory scrutiny and the rising threat of healthcare-associated infections (HAIs), hospitals are prioritizing the adoption of automated systems that ensure standardized cleaning, validated thermal disinfection, and comprehensive digital traceability. Additionally, ongoing infrastructure upgrades and government-driven infection prevention initiatives across both public and private healthcare networks are driving significant investment in modern washer-disinfector technologies.
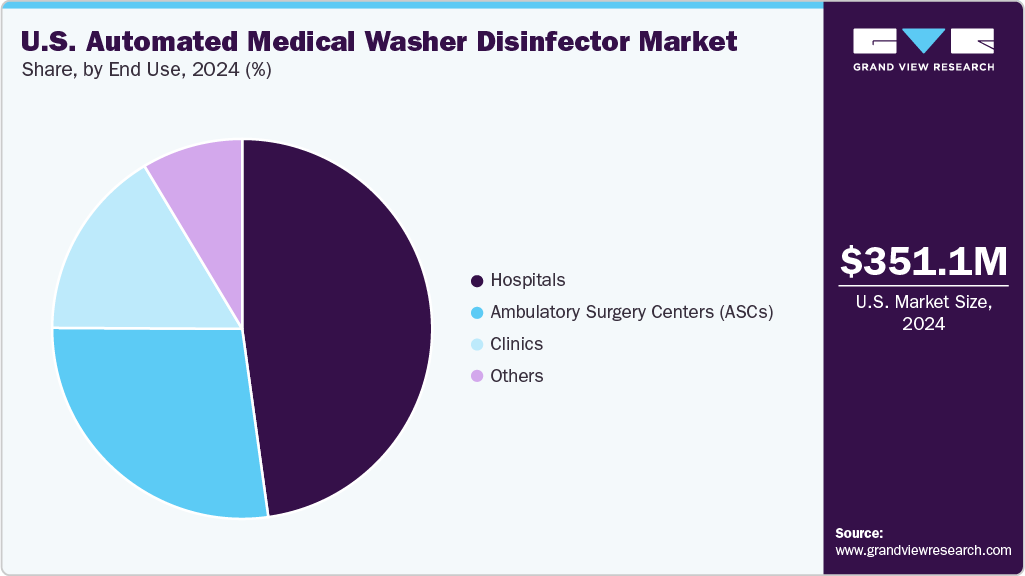
However, ambulatory surgical centers segment is projected to witness fastest CAGR over the forecast period. This can be attributed to the rapid expansion of same-day, minimally invasive procedures and the growing shift of surgeries from hospitals to outpatient settings. ASCs increasingly require compact, efficient, and cost-effective washer-disinfectors that meet sterilization standards while operating within smaller spaces and tighter budgets. As regulatory bodies emphasize infection control in all healthcare environments, ASCs invest in automated reprocessing solutions that offer reliable cleaning cycles, faster turnaround, and lower resource consumption. This, combined with the increasing number of ASCs across both developed and emerging regions, is accelerating demand for mid-sized and benchtop washer disinfector systems tailored to their operational needs, fueling rapid market expansion in this segment.
Key U.S. Automated Medical Washer Disinfector Company Insights
Some of the key companies include Steelco S.p.A; STERIS; Getinge; HOYA Corporation; Olympus; Tuttnauer; CYTENA GmbH; Spire Integrated Solutions.; MMM Group; Midmark Corporation, among others. They provide a broad range of breast conserving surgery solutions through their strong distribution and supply channels across the world. Leading companies are involved in new product launch, strategic collaborations, mergers & acquisitions, and regional expansions to gain the maximum revenue share in the industry. Mergers & acquisitions help companies to expand their businesses and market presence.
Key U.S. Automated Medical Washer Disinfector Companies:
- Steelco S.p.A
- STERIS
- Getinge
- HOYA Corporation
- Olympus
- Tuttnauer
- CYTENA GmbH
- Spire Integrated Solutions.
- MMM Group
- Midmark Corporation
- Ecolab Inc.
Recent Developments
-
In June 2023, Olympus has announced the launch of its latest endoscope reprocessing solution, the Olympus ETD, available in two versions: ETD Basic and ETD Premium. This new addition to the company’s Infection Prevention portfolio is designed to enhance the efficiency and sustainability of endoscope reprocessing, while delivering high-performance cleaning and disinfection outcomes. Developed with a focus on operational ease and environmental responsibility, the ETD models aim to support healthcare facilities in achieving improved infection control standards.
-
In April 2024, Getinge has introduced the GEW 888 neo, a next-generation cGMP washer specifically engineered to enhance cleanroom efficiency within the biopharmaceutical industry. Designed to meet the industry's most stringent regulatory and operational standards, the GEW 888 neo offers advanced performance while optimizing space utilization in cleanroom environments, making it a valuable solution for facilities focused on compliance, productivity, and cleanliness.
U.S. Automated Medical Washer Disinfector Market Report Scope
Report Attribute
Details
Market size value in 2025
USD 371.78 million
Revenue forecast in 2033
USD 590.62 million
Growth rate
CAGR of 5.96% from 2025 to 2033
Actual data
2021 - 2024
Forecast period
2025 - 2033
Quantitative units
Revenue in USD million and CAGR from 2025 to 2033
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Product type, capacity, chamber type, device classification, end use
Country scope
U.S.
Key companies profiled
Steelco S.p.A; STERIS; Getinge; HOYA Corporation; Olympus; Tuttnauer; CYTENA GmbH; Spire Integrated Solutions.; MMM Group; Midmark Corporation; Ecolab Inc.
Customization scope
Free report customization (equivalent up to 8 analysts working days) with purchase. Addition or alteration to country, regional & segment scope.
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
U.S. Automated Medical Washer Disinfector Market Report Segmentation
This report forecasts revenue growth and provides an analysis of the latest trends in each of the sub-segments from 2021 to 2033. For this study, Grand View Research has segmented the U.S. automated medical washer disinfector market report based on product type, capacity, chamber type, device classification, and end use:
-
Product Type Outlook (Revenue, USD Million, 2021 - 2033)
-
Floor Standing
-
Benchtop
-
Others
-
-
Capacity Outlook (Revenue, USD Million, 2021 - 2033)
-
Small Capacity (<100 L)
-
Medium Capacity (100-200 L)
-
Large Capacity (>200 L)
-
-
Chamber Type Outlook (Revenue, USD Million, 2021 - 2033)
-
Single Chamber
-
Multi Chamber
-
-
Device Classification Outlook (Revenue, USD Million, 2021 - 2033)
-
Critical
-
Semi Critical
-
-
End Use Outlook (Revenue, USD Million, 2021 - 2033)
-
Hospitals
-
Ambulatory Surgical Centers (ASCs)
-
Clinics
-
Others
-
Frequently Asked Questions About This Report
b. The global automated medical washer disinfector market size was estimated at USD 351.11 million in 2024 and is expected to reach USD 371.78 million in 2025.
b. The global automated medical washer disinfector market is expected to grow at a compound annual growth rate of 5.96% from 2025 to 2033 to reach USD 590.62 million by 2033.
b. Floor standing dominated the automated medical washer disinfector market with a share of 72.81% in 2024. This is attributable to the widespread adoption of floor-standing washer-disinfectors in large hospitals and central sterile supply departments (CSSDs).
b. Some key players operating in the automated medical washer disinfector market include Steelco S.p.A; STERIS; Getinge; HOYA Corporation; Olympus; Tuttnauer; CYTENA GmbH; Spire Integrated Solutions.; MMM Group; Midmark Corporation
b. Key factors that are driving the market growth include the high surgical volumes, stringent infection control regulations, and advancements in healthcare infrastructure. With the continued growth of ambulatory surgical centers (ASCs), there is a rising need for efficient, high-throughput instrument reprocessing systems.
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










