- Home
- »
- Clinical Diagnostics
- »
-
U.S. Blood-based Biomarker For Alzheimer’s Disease Diagnostics Market Report, 2033GVR Report cover
![U.S. Blood-based Biomarker For Alzheimer’s Disease Diagnostics Market Size, Share & Trends Report]()
U.S. Blood-based Biomarker For Alzheimer’s Disease Diagnostics Market (2025 - 2033) Size, Share & Trends Analysis By Type (Amyloid-related Markers, Tau-related markers, Neurodegeneration), By Technology, By End Use, And Segment Forecasts
- Report ID: GVR-4-68040-722-3
- Number of Report Pages: 150
- Format: PDF
- Historical Range: 2021 - 2024
- Forecast Period: 2025 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
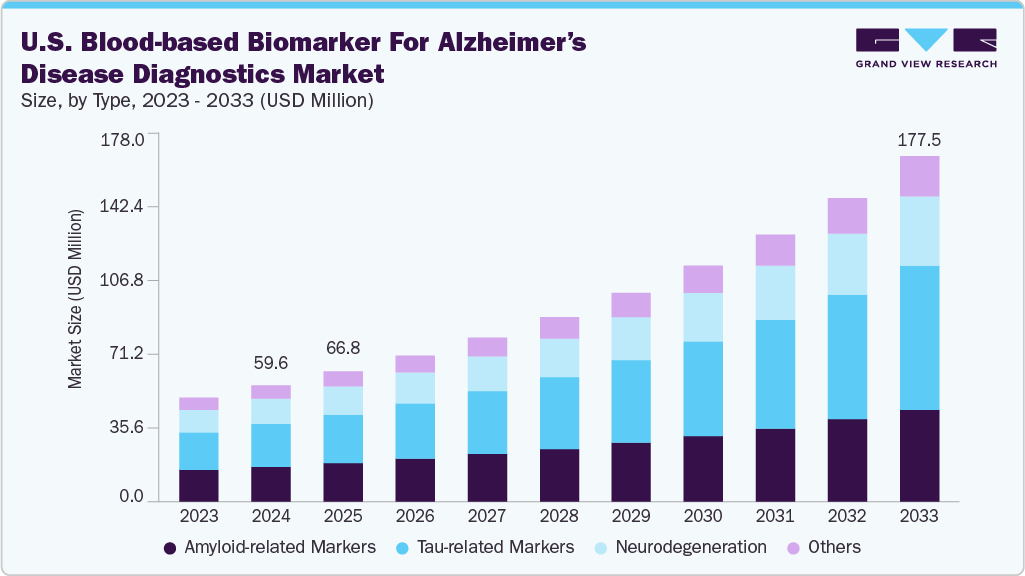
Market Size & Trends
The U.S. blood-based biomarker for alzheimer’s disease diagnostics market size was estimated at USD 59.64 million in 2024 and is expected to grow at a CAGR of 14.60% from 2025 to 2033. This growth is fueled by rising Alzheimer’s prevalence, increasing demand for non-invasive diagnostics, and regulatory approvals such as the FDA’s clearance of blood-based IVD assays. Technological advancements in ultrasensitive immunoassays and multiplex testing further support adoption, positioning blood biomarkers as essential tools in early detection and treatment pathways.
One of the most significant growth drivers for the U.S. blood-based biomarker market is the approval of FDA-cleared IVD assays. A prime example is the Lumipulse G pTau217/β-Amyloid 1-42 Plasma Ratio, which gained marketing authorization in 2025 (Grand View Research; GlobeNewswire). Regulatory recognition not only lowers clinical adoption barriers but also validates the reliability of blood-based tests in practice. At the same time, advances in ultra-sensitive immunoassays and AI-powered multiplex testing are enhancing diagnostic accuracy. These technologies help reduce dependence on costly, invasive methods such as PET imaging and lumbar punctures. The combination of regulatory approval and innovation is creating strong momentum, boosting confidence among healthcare providers, payers, and patients alike
Demographic trends are another critical driver. As the U.S. population ages, the prevalence of dementia is rising rapidly-about 4% of individuals aged 65+ are affected, and the rate climbs to 13% among those over 85 (Grand View Research; GlobeNewswire). This has created an urgent need for scalable, early diagnostic solutions. Blood-based biomarkers address this gap by offering less invasive testing, enabling earlier intervention, and streamlining patient care pathways. They also support the shift toward precision medicine, where biologically driven diagnostics guide therapy selection and monitoring. Importantly, anti-amyloid drugs now require biomarker confirmation of pathology, making blood tests an essential first-line triage tool for patients being evaluated for treatment.
In line with these developments, the Alzheimer’s Association introduced its first clinical practice guideline (CPG) for Alzheimer’s and other dementias at AAIC 2025 in Toronto. This landmark guideline focuses specifically on the use of blood-based biomarker tests by specialists to assess Alzheimer’s pathology in individuals with cognitive impairment. It highlights the importance of early, accurate detection and provides clinicians with evidence-based resources to ensure patients can access the right treatments as quickly as possible. These recommendations form part of ALZPro, the Association’s centralized hub designed to provide dementia professionals with practical tools, resources, and ongoing support.

The newly released CPG represents a transformative step for specialty care. Published in Alzheimer’s & Dementia: The Journal of the Alzheimer’s Association, it provides brand-agnostic, evidence-based recommendations designed to standardize the use of blood biomarkers in clinical practice. Built on a systematic review process using robust and transparent methodology, the guideline ensures scientific rigor while promoting broader adoption of these diagnostics. Furthermore, the Alzheimer’s Association has committed to regularly updating the guidance as new data emerges, ensuring that clinicians and patients alike continue to benefit from the latest scientific advances in Alzheimer’s detection and care.
Market Concentration & Characteristics
The U.S. market demonstrates a high degree of innovation, with breakthroughs in ultra-sensitive immunoassays, multiplex biomarker panels, and AI-driven analytics enhancing diagnostic accuracy. Innovations such as p-tau217/Aβ42 ratio tests have shifted blood biomarkers from research into mainstream clinical use. Continuous R&D ensures evolving assays can detect pathology earlier, improving patient outcomes and supporting the transition to precision-based Alzheimer’s care across diverse healthcare settings.
Mergers, acquisitions, and partnerships are accelerating market growth as established diagnostics firms acquire biotech innovators developing novel Alzheimer’s biomarker assays. Strategic deals expand portfolios, integrate proprietary technologies, and fast-track commercialization in the U.S. market. Collaborations between diagnostics players, pharma companies, and academic institutions further drive innovation, particularly as blood-based tests become integral to clinical trial recruitment and therapy monitoring.
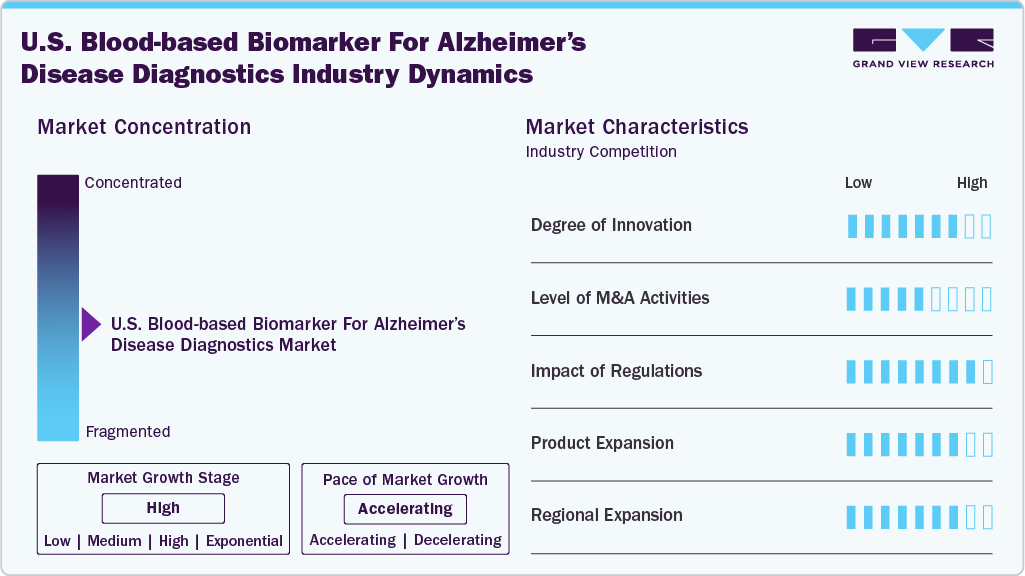
Regulation plays a transformative role, with the FDA’s 2025 clearance of the Lumipulse p-tau217/Aβ42 plasma ratio test marking the first blood-based IVD approval for Alzheimer’s. Such milestones legitimize biomarker use in U.S. clinical practice and encourage payer discussions. Breakthrough Device designations also accelerate development timelines, strengthening market confidence and clinical adoption nationwide.
Product expansion is reshaping the U.S. market, with companies broadening offerings from amyloid and tau assays to multiplex panels including GFAP and NfL. New assays support differential diagnosis, disease staging, and treatment monitoring. Expanding beyond single biomarkers ensures clinicians have comprehensive diagnostic tools, positioning blood-based tests as central to Alzheimer’s patient management strategies.
Within the U.S., expansion is driven by the spread of blood-based Alzheimer’s diagnostics from specialized research centers into community hospitals, clinical labs, and memory care clinics. Nationwide test launches by providers such as Labcorp and Quest Diagnostics increase accessibility. Regional adoption is supported by payer engagement, state-level coverage mandates, and integration into local dementia care programs.
Type Insights
On the basis of type, Tau-related markers accounted for the largest market share of 37.08% in 2024, driven by strong clinical evidence linking phosphorylated tau (p-tau) proteins-particularly p-tau181, p-tau217, and p-tau231-to early Alzheimer’s pathology. These markers have demonstrated higher diagnostic specificity compared to traditional amyloid-based tests, making them critical for distinguishing Alzheimer’s from other forms of dementia. The FDA’s 2025 clearance of Fujirebio’s Lumipulse G pTau217/β-Amyloid 1-42 ratio assay has further accelerated adoption, providing the first regulatory-recognized blood test aiding in the identification of amyloid pathology through tau measurement. Rising demand is supported by the growing use of anti-amyloid therapies, which require biomarker confirmation for treatment eligibility, positioning tau assays as a key triage tool in clinical practice. With Alzheimer’s prevalence climbing in the U.S. and healthcare systems seeking scalable, non-invasive solutions, tau-related blood markers are projected to see robust growth throughout the forecast period.
The amyloid-related markers segment is anticipated to grow steadily, as amyloid beta (Aβ) pathology remains a hallmark feature of the disease. Blood-based assays measuring the Aβ42/40 ratio have shown strong correlation with amyloid PET imaging, offering a cost-effective, less invasive alternative for early detection. The increasing use of anti-amyloid therapies, such as lecanemab and donanemab, which require confirmed amyloid pathology for treatment initiation, is also driving demand for amyloid-based blood tests. As the U.S. population ages and Alzheimer’s prevalence rises, amyloid biomarkers are becoming an essential first-line screening tool, enabling clinicians to efficiently identify patients for confirmatory testing, timely intervention, and enrollment in disease-modifying treatment programs.
Technology Insights
Immunoassays held the largest market share in 2024, driven by its reliability, scalability, and clinical familiarity. Immunoassays-particularly ELISA, chemiluminescent assays, and single-molecule arrays (Simoa)-enable ultra-sensitive detection of Alzheimer’s-related biomarkers such as p-tau217, p-tau181, Aβ42/40, and neurofilament light (NfL) at very low plasma concentrations. Their widespread availability across hospital labs, clinical laboratories, and reference centers makes them highly suitable for large-scale deployment. Recent innovations in high-throughput and multiplex immunoassays have enhanced diagnostic accuracy and reduced turnaround times, making them increasingly attractive for routine clinical use.
The next-generation platforms segment in the U.S. is expected to grow at the fastest rate over the forecast period, driven by the need for more precise, scalable, and comprehensive diagnostic tools. These platforms leverage mass spectrometry, digital immunoassays, and multiplex testing technologies to simultaneously measure multiple biomarkers, including p-tau217, Aβ42/40, GFAP, and NfL, from a single blood sample. This multi-marker approach provides deeper insights into disease staging and progression while enhancing diagnostic specificity compared to single-biomarker assays. In addition, integration with AI-driven data analytics is improving predictive accuracy and patient stratification, especially for clinical trials and precision medicine applications. The increasing focus on early intervention and monitoring response to anti-amyloid therapies further supports the adoption of these advanced platforms. With academic institutions, biotech startups, and diagnostics leaders accelerating innovation, next-generation platforms are set to significantly expand clinical adoption and reshape Alzheimer’s disease management in the U.S.
End Use Insights
Hospitals dominated the U.S. blood-based biomarker for Alzheimer’s disease diagnostics market in revenue in 2024, reflecting their central role in diagnostic services and patient care. These facilities serve as the primary hubs for test deployment, supported by their established infrastructure, advanced equipment, and integration into clinical workflows. Clinical labs also benefit from economies of scale, offering high-throughput testing essential to meet the growing demand as Alzheimer’s prevalence rises in the U.S. Furthermore, their ability to support early diagnosis, differential diagnosis, and treatment monitoring aligns with new clinical guidelines, such as those released by the Alzheimer’s Association in 2025, promoting the integration of blood-based biomarkers into routine dementia care. This positions labs as pivotal growth drivers in the market.
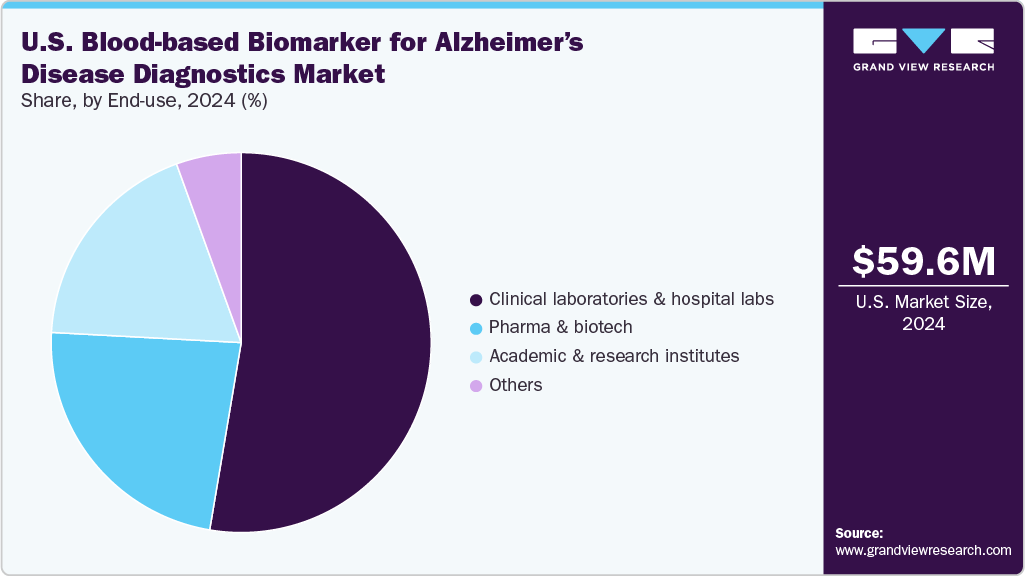
The others segment in the U.S. is expected to grow at the fastest rate over the forecast period. Memory clinics and senior care centers are increasingly adopting blood-based biomarker tests to enable community-level early detection, addressing the rising burden of dementia in aging populations. CROs are leveraging these assays for clinical trial recruitment and monitoring, particularly for emerging anti-amyloid and neuroprotective therapies that require biomarker confirmation for enrollment. Diagnostic centers are also integrating blood-based tests as non-invasive, cost-effective adjuncts to imaging methods such as PET scans. Furthermore, the shift toward personalized medicine and decentralized testing supports adoption across diverse settings, ensuring faster triage and care access. This segment’s growth highlights the expanding reach of blood biomarkers into real-world, patient-centered care pathways.
Key U.S. Blood-based Biomarker for Alzheimer’s Disease Diagnostics Company Insights
In the U.S. blood-based biomarker for Alzheimer’s disease diagnostics market, major players include Fujirebio, Labcorp, Quest Diagnostics, C2N Diagnostics, Quanterix, F. Hoffmann-La Roche Ltd, Siemens Healthineers, Abbott Laboratories, Johnson & Johnson, Diadem, Araclon Biotech, and BioArctic. These companies are advancing FDA-cleared and CLIA-certified blood tests, expanding precision diagnostic offerings. With investments in immunoassays, AI-driven multiplex platforms, and clinical trial partnerships, they are shaping the market’s rapid growth. Their focus on early detection, scalable testing, and therapy-aligned diagnostics strengthens adoption across U.S. healthcare systems.
Key U.S. Blood-based Biomarker for Alzheimer’s Disease Diagnostics Companies:
- F. Hoffmann-La Roche Ltd
- Quanterix
- Fujirebio
- C2N Diagnostics
- Labcorp
- Quest Diagnostics Incorporated
- Siemens Healthineers
- Diadem srl
- Abbott
- BioArctic
- Grifols
Recent Developments
-
In May 2025, research from the Wisconsin Registry for Alzheimer’s Prevention (WRAP) contributed to the launch of the first FDA-cleared blood test for Alzheimer’s disease. On May 16, 2025, the U.S. Food and Drug Administration (FDA) cleared an in vitro diagnostic (IVD) assay designed to detect amyloid - a key protein linked to Alzheimer’s disease - in the blood. Validation of the test included data from studies conducted at the University of Wisconsin-Madison, including WRAP, marking a significant step forward in advancing Alzheimer’s disease diagnosis.
-
In January 2025, researchers reported that healthcare providers improved their ability to assess the likelihood of Alzheimer’s disease (AD) in patients when using the PrecivityAD2™ blood biomarker (BBM) test from C2N Diagnostics, LLC. The test helps detect amyloid plaques in the brain, a key pathological feature of AD. The shift in clinician-reported probability of AD was also linked to meaningful enhancements in patient care, including adjustments to AD medication plans and decisions regarding further brain amyloid testing.
-
In July 2024, Biogen Inc., Beckman Coulter, Inc., and Fujirebio announced a collaboration aimed at identifying and developing blood-based biomarkers for tau pathology in the brain. The partnership also seeks to clinically advance and potentially commercialize new tests to detect tau pathology in Alzheimer’s disease (AD). Creating tau-specific blood-based biomarkers that can measure a patient’s tau burden could offer vital insights into the biological processes driving AD and support the development of next-generation therapies targeting tau pathology.
U.S. Blood-based Biomarker For Alzheimer’s Disease Diagnostics Market Report Scope
Report Attribute
Details
Market size value in 2025
USD 66.82 million
Revenue forecast in 2033
USD 177.48 million
Growth rate
CAGR of 14.60% from 2025 to 2033
Actual data
2021 - 2024
Forecast period
2025 - 2033
Quantitative units
Revenue in USD million/billion and CAGR from 2025 to 2033
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Type, technology, end use
Key companies profiled
F. Hoffmann-La Roche Ltd; Quanterix; Fujirebio; C2N Diagnostics; Labcorp; Quest Diagnostics Incorporated; Siemens Healthineers; Diadem srl; Abbott; BioArctic; Grifols
Customization scope
Free report customization (equivalent up to 8 analyst’s working days) with purchase. Addition or alteration to segment scope.
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
U.S. Blood-based Biomarker For Alzheimer’s Disease Diagnostics Market Report Segmentation
This report forecasts revenue growth at country level and provides an analysis on the latest trends in each of the sub-segments from 2021 to 2033. For the purpose of this report, Grand View Research has segmented the U.S. blood-based biomarker for alzheimer’s disease diagnostics market based on type, technology, and end use:
-
Type Outlook (USD Million, 2021 - 2033)
-
Amyloid-related markers
-
Tau-related markers
-
Neurodegeneration
-
Others
-
-
Technology Outlook (USD Million, 2021 - 2033)
-
Immunoassays
-
Mass spectrometry-based assays
-
Next-generation platforms
-
Others
-
-
End Use Outlook (USD Million, 2021 - 2033)
-
Clinical laboratories & hospital labs
-
Pharma & biotech
-
Academic & research institutes
-
Others
-
Frequently Asked Questions About This Report
b. The U.S. blood-based biomarker for Alzheimer’s disease diagnostics market size was estimated at USD 59.64 million in 2024 and is expected to reach USD 66.82 million in 2025.
b. The U.S. blood-based biomarker for Alzheimer’s disease diagnostics market is expected to grow at a compound annual growth rate of 14.60% from 2025 to 2033 to reach USD 177.48 million by 2033.
b. On the basis of type, Tau-related markers accounted for the largest market share of 37.08% in 2024, driven by strong clinical evidence linking phosphorylated tau (p-tau) proteins—particularly p-tau181, p-tau217, and p-tau231—to early Alzheimer’s pathology.
b. Some key players operating in the U.S. blood-based biomarker for Alzheimer’s disease diagnostics market include F. Hoffmann-La Roche Ltd, Quanterix, Fujirebio, C2N Diagnostics, Labcorp, Quest Diagnostics Incorporated, Siemens Healthineers, Diadem srl, Abbott, BioArctic, and Grifols o Siemens Healthineers o Diadem srl o Abbott o BioArctic o Grifols
b. The growth is fueled by rising Alzheimer’s prevalence, increasing demand for non-invasive diagnostics, and regulatory approvals such as the FDA’s clearance of blood-based IVD assays.
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










