- Home
- »
- Medical Devices
- »
-
U.S. Breast Biopsy Devices Market Size Report, 2033GVR Report cover
![U.S. Breast Biopsy Devices Market Size, Share & Trends Report]()
U.S. Breast Biopsy Devices Market (2026 - 2033) Size, Share & Trends Analysis Report By Product Type (Biopsy Guidance Systems, Biopsy Drivers & Handpieces, Biopsy Tables, Biopsy Consumables), By Procedure Type, By Guidance Technology, By End Use, And Segment Forecasts
- Report ID: GVR-4-68040-865-5
- Number of Report Pages: 110
- Format: PDF
- Historical Range: 2021 - 2025
- Forecast Period: 2026 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
U.S. Breast Biopsy Devices Market Summary
The U.S. breast biopsy devices market size was estimated at USD 1.18 billion in 2025 and is anticipated to reach USD 1.60 billion by 2033, growing at a CAGR of 3.9% from 2026 to 2033. The market growth driven by the rising incidence of breast cancer and the consequent emphasis on early detection and accurate diagnosis, which increases the volume of biopsy procedures required in screening and diagnostic pathways.
Key Market Trends & Insights
- By product type, the biopsy consumables segment led the market with the largest revenue share in 2025.
- By procedure type, the core needle biopsy (CNB) segment led the market with the largest revenue share in 2025.
- By guidance technology, the ultrasound-guided segment led the market with the largest revenue share in 2025.
- By end use, the hospitals segment led the market with the largest revenue share in 2025.
Market Size & Forecast
- 2025 Market Size: USD 1.18 Billion
- 2033 Projected Market Size: USD 1.60 Billion
- CAGR (2026-2033): 3.9%
High awareness among patients and clinicians, supported by structured screening programs and favorable reimbursement landscapes, further propels device adoption. In addition, technological advancements, including minimally invasive core-needle and vacuum-assisted systems, integration with advanced imaging modalities (e.g., ultrasound, MRI, 3D mammography), and AI-enhanced diagnostic workflows, are expanding clinical utility, improving procedural accuracy and patient comfort, and encouraging broader clinical integration.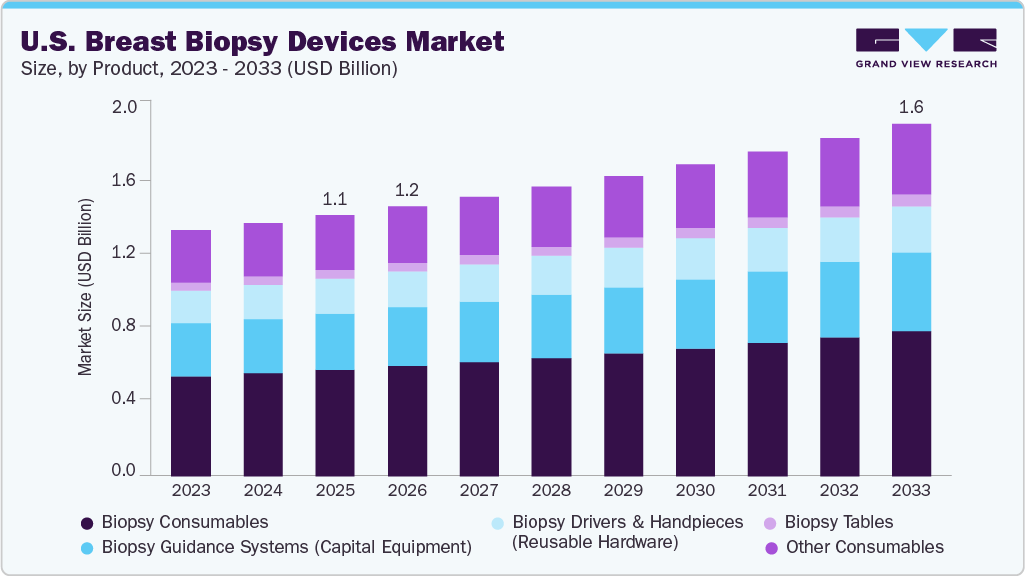
The rising prevalence of breast cancer in the U.S. directly drives growth in the breast biopsy devices market because a higher number of new and suspected cancer cases increases the demand for diagnostic confirmation through tissue sampling. In the U.S., breast cancer is the most commonly diagnosed cancer among women, with hundreds of thousands of new invasive and non-invasive cases reported annually, prompting more routine screening and follow-up biopsies when abnormalities are detected on imaging. As incidence rates climb and screening programs expand, healthcare providers perform more biopsies to confirm diagnoses, guide treatment decisions, and rule out malignancy, thereby boosting utilization of core-needle, vacuum-assisted, and image-guided breast biopsy devices.
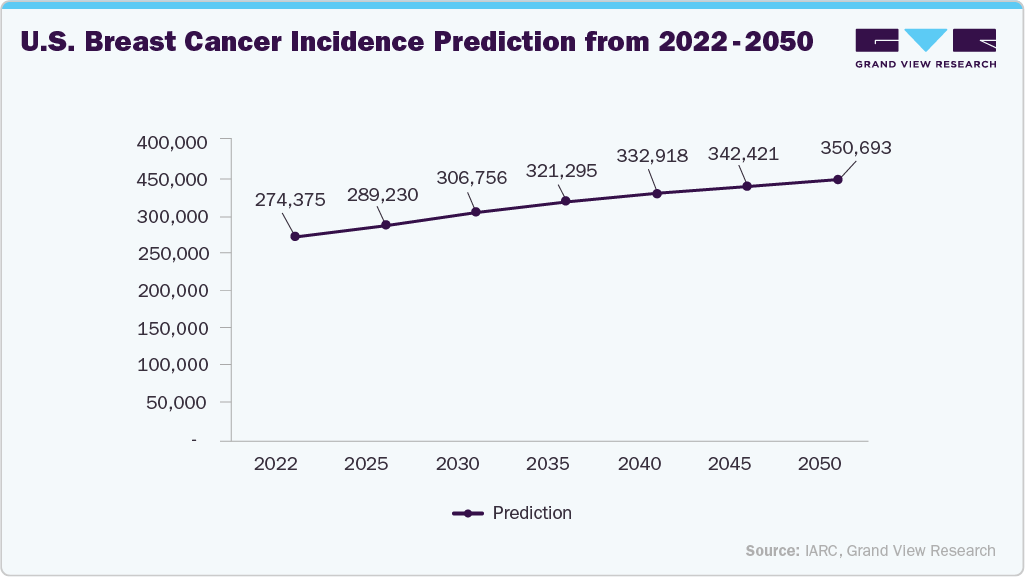
As screening programs such as mammography and MRI expand, more suspicious lesions are detected, necessitating tissue sampling via core-needle or vacuum-assisted biopsy. This sustained rise in diagnostic volumes increases demand for advanced, image-guided biopsy systems in hospitals and ambulatory centers. The link between higher case detection and device innovation is evident in recent industry developments, for instance, in February 2026, Mammotome, a part of Danaher Corporation, introduced the Mammotome Prima MR Dual Vacuum-Assisted Breast Biopsy System, the first system designed to be positioned inside the MRI scanner room at the patient’s side during biopsy. By improving clinician coordination and workflow efficiency in MRI-guided procedures, such innovations address the growing procedural burden driven by rising breast cancer prevalence, thereby accelerating technology adoption and overall market growth in the U.S. breast biopsy devices sector.
Rising awareness of early breast cancer detection, driven by organizations such as the National Breast Cancer Foundation, Inc. (NBCF) and the American Cancer Society, is significantly driving the U.S. breast biopsy devices market by increasing participation in early screening programs such as mammography and MRI. Their nationwide campaigns, educational outreach, and Breast Cancer Awareness Month activities emphasize the importance of early detection, highlighting that breast cancer detected at a localized stage has a very high five-year survival rate, which encourages more women to undergo routine screening. As screening volumes rise, more suspicious lesions are identified that require confirmatory tissue diagnosis via core-needle or vacuum-assisted biopsy, thereby increasing procedural volumes and boosting demand for advanced, image-guided breast biopsy devices across hospitals and diagnostic centers in the U.S.
The rising adoption of advanced breast biopsy systems and ongoing technological advancements are driving the U.S. breast biopsy devices industry’s growth by enhancing diagnostic precision, workflow efficiency, and patient comfort, thereby encouraging wider clinical adoption across hospitals and imaging centers. Innovations such as improved vacuum-assisted technologies, MRI- and ultrasound-guided compatibility, automated tissue acquisition, and ergonomic system designs enable clinicians to obtain more accurate samples with fewer complications and shorter procedure times. These improvements not only increase physician confidence and patient preference for minimally invasive procedures but also support higher procedural throughput in high-volume diagnostic settings. As healthcare providers adopt technologically advanced systems to improve outcomes and operational efficiency, demand for next-generation breast biopsy devices continues to grow, thereby accelerating overall market growth.
New Product Launch
Company
Product Launch
Kols
Mammotome
In February 2026, Mammotome, a subsidiary of Danaher Corporation, introduced the Mammotome Prima MR Dual Vacuum-Assisted Breast Biopsy System, the first breast biopsy system specifically engineered for placement inside the MRI scanner room at the patient’s side during procedures. This innovative design strengthens clinical coordination and communication while keeping attention centered on patient care by positioning the vacuum-assisted biopsy device directly within the MRI suite.
"Our commitment to advancing breast care drives every innovation we bring to market," said Scott Treml, Mammotome Vice President and General Manager. "As the first of its kind in-room solution, the Mammotome Prima™ MR system empowers clinicians with greater efficiency and confidence, ultimately improving the patient experience."
BD
In January 2026, BD, a global medical technology leader, announced that the U.S. Food and Drug Administration (FDA) has granted 510(k) clearance for the EnCor EnCompass Breast Biopsy and Tissue Removal System. This advanced multi-modality breast biopsy system is designed to offer clinicians greater flexibility across various breast imaging platforms, supporting accurate diagnosis and effective tissue removal in the management of breast disease.
"This milestone for our new breast biopsy system marks a meaningful advancement in breast health, playing a critical role in aiding the early detection and diagnosis of breast disease," said Rima Alameddine, worldwide president, Peripheral Intervention at BD. "This innovation underscores our commitment to partnering with clinical leaders to deliver patient-centered solutions. Guided by our vision to transform breast health, we remain focused on developing technologies that empower providers and inspire confidence in care."
Source: Company Websites, Grand View Research
Market Concentration & Characteristics
The U.S. breast biopsy devices market demonstrates a high degree of innovation, driven by continuous advancements in minimally invasive technologies, multi-modality imaging integration, and workflow optimization. Manufacturers such as BD (Becton, Dickinson and Company) and Mammotome are introducing next-generation vacuum-assisted and image-guided systems compatible with ultrasound, stereotactic, and MRI platforms, improving diagnostic accuracy and procedural efficiency. Innovations focus on enhanced tissue acquisition, reduced patient discomfort, ergonomic system design, and improved sample quality for precise pathology assessment. In addition, integration with digital imaging and advanced guidance technologies supports real-time targeting and streamlined clinical workflows. This strong pipeline of technologically differentiated products, supported by regulatory clearances from the U.S. Food and Drug Administration, reflects a dynamic and innovation-intensive market environment in the U.S.
The U.S. breast biopsy devices industry demonstrates a moderate to high level of M&A activity, primarily driven by strategic consolidation aimed at expanding product portfolios, strengthening minimally invasive biopsy capabilities, and enhancing imaging-guided intervention technologies. Market participants pursue acquisitions to access innovative platforms, proprietary vacuum-assisted systems, and advanced tissue sampling technologies that improve diagnostic precision and workflow efficiency. M&A transactions also support geographic expansion, supply chain integration, and cross-selling opportunities across breast imaging and interventional segments. This ongoing consolidation trend increases competitive intensity, accelerates technology commercialization, and contributes to the overall structural evolution and market growth.
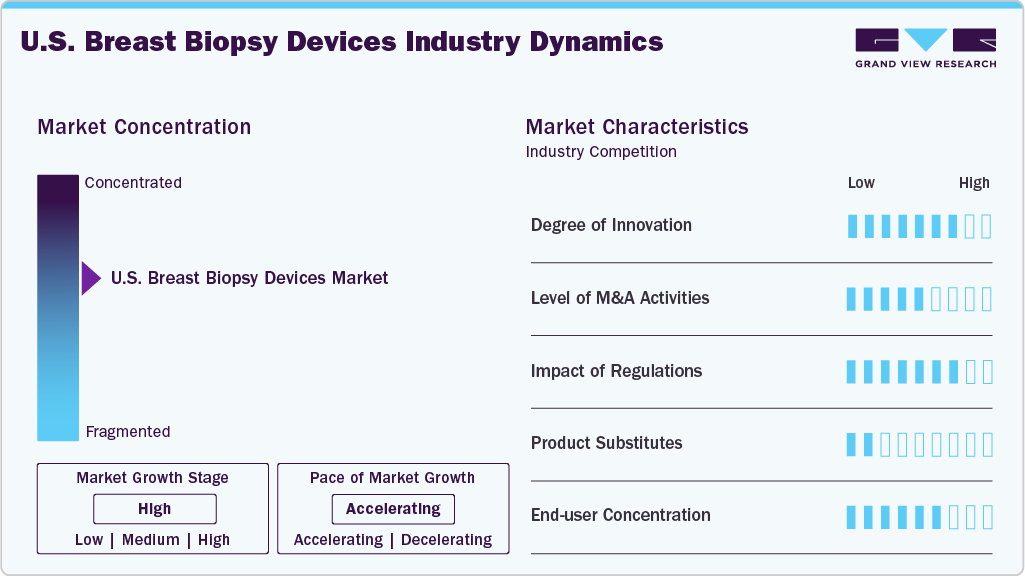
Regulatory changes in the U.S. are shaping the breast biopsy devices market by enhancing the standardization of breast cancer screening and follow-up protocols, which, in turn, can drive higher procedural volumes and technology adoption. The U.S. Food and Drug Administration has updated mammography regulations under the Mammography Quality Standards Act to require facilities to include breast density information in all mammogram reports and to strengthen oversight of quality and patient communication, a change fully implemented in 2024 that affects more than 40 million women screened annually and encourages clinicians to consider further imaging when density complicates interpretation. In addition, the American College of Radiology released the BI-RADS v2025 Manual, updating reporting standards for mammography, ultrasound, MRI, and contrast-enhanced modalities to improve consistency in lesion characterization and management guidance. These regulatory and guideline updates improve detection sensitivity and clarity in risk stratification, leading to more appropriate referrals for diagnostic biopsies and supporting broader use of advanced breast biopsy systems across imaging modalities.
In the U.S. breast biopsy devices market, product substitutes are limited but include non-invasive or less invasive diagnostic approaches that can delay or reduce the need for tissue sampling in select cases. Advanced imaging modalities such as digital mammography, 3D tomosynthesis, breast ultrasound, and breast MRI can sometimes provide sufficient diagnostic confidence for short-term imaging follow-up instead of immediate biopsy, particularly for probably benign lesions. In addition, emerging liquid biopsy and blood-based biomarker tests are being explored for cancer detection, though they currently do not replace tissue biopsy for definitive diagnosis. Despite these alternatives, histopathological examination of tissue obtained through core-needle or vacuum-assisted biopsy remains the gold standard for confirming breast cancer, limiting the long-term substitution threat in the U.S. market.
Product Insights
The biopsy consumables segment dominated the U.S. breast biopsy devices market in 2025, driven by recurring demand for single-use components required for every procedure. Disposable items such as core biopsy needles, vacuum-assisted probes, localization wires, and procedure trays generate consistent revenue across hospitals, ambulatory surgical centers, and diagnostic imaging facilities. The growing volume of minimally invasive and image-guided breast biopsies across the United States has further reinforced the dominance of consumables, as each procedure necessitates new sterile components.
Biopsy guidance is expected to grow at the fastest CAGR during the forecast period, supported by increasing demand for high-precision, image-guided interventions. Advancements in 3D tomosynthesis, breast MRI, and AI-assisted targeting technologies are improving lesion detection and procedural accuracy. As U.S. providers continue investing in workflow efficiency and enhanced diagnostic outcomes, adoption of advanced guidance platforms is accelerating.
Procedure Type Insights
The core needle biopsy (CNB) segment dominated the U.S. breast biopsy devices industry in 2025, driven by its strong clinical acceptance as the standard method for diagnosing suspicious breast lesions. CNB offers high diagnostic reliability, minimal invasiveness, cost efficiency, and compatibility with multiple imaging modalities such as ultrasound and stereotactic systems, making it widely used in outpatient and hospital settings.
The vacuum-assisted biopsy (VAB) segment is expected to grow at the fastest CAGR during the forecast period. VAB enables the retrieval of larger, more contiguous tissue samples with fewer insertions, thereby improving diagnostic confidence and reducing the need for repeat procedures. Its expanding use for microcalcifications and difficult-to-access lesions, along with integration into advanced imaging platforms, is supporting rapid adoption across U.S. diagnostic centers.
Guidance Technology Insights
The ultrasound-guided biopsy segment dominated the U.S. breast biopsy devices market in 2025 due to its widespread availability, real-time imaging, lack of radiation exposure, and cost-effectiveness. Ultrasound-guided procedures are commonly performed in outpatient imaging centers and are highly effective for sampling visible or palpable lesions.
The mammography/stereotactic-guided segment is expected to grow at the fastest CAGR during the forecast period, driven by increased detection of non-palpable lesions and microcalcifications through digital mammography and 3D tomosynthesis. Continuous improvements in targeting precision and imaging resolution are further enhancing the clinical adoption of stereotactic biopsy systems in the U.S.
End Use Insights
Hospitals dominated the U.S. breast biopsy devices market in 2025 due to advanced imaging infrastructure, multidisciplinary oncology teams, and the ability to manage complex or high-risk cases. Hospitals perform a significant proportion of MRI- and stereotactic-guided biopsies, which require specialized equipment and clinical expertise. Their comprehensive cancer care capabilities and integration with pathology services further strengthen patient preference for hospital-based diagnostic procedures, thereby contributing to the segment’s leading market share.
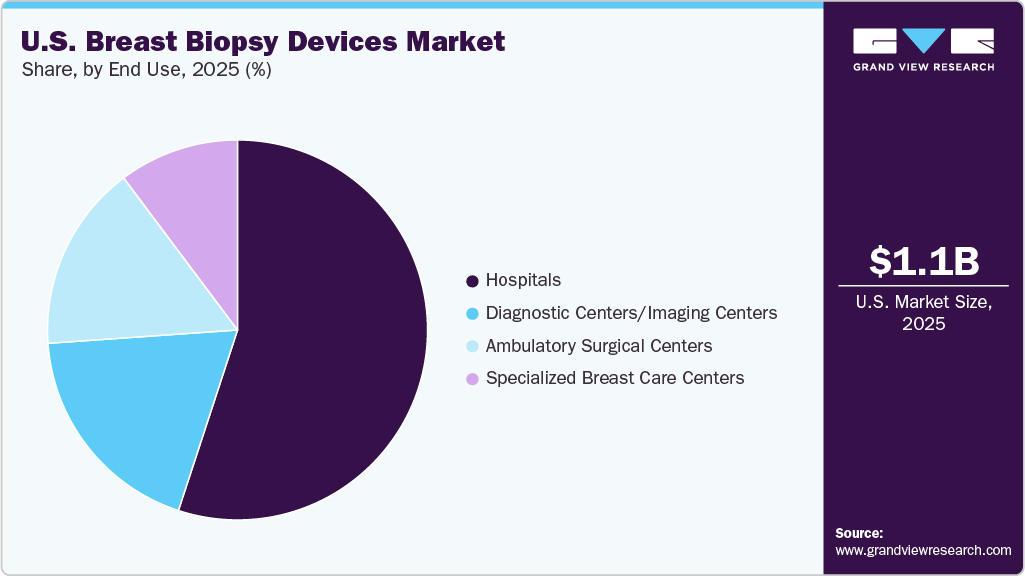
The ambulatory surgical centers (ASCs) are expected to grow at the fastest CAGR during the forecast period. The increasing shift toward minimally invasive procedures and same-day diagnostic services has strengthened the preference for outpatient care settings. ASCs provide cost-efficient treatment options, reduced wait times, and optimized procedural workflows, making them appealing to both patients and healthcare providers. As the U.S. healthcare system continues to prioritize operational efficiency and alleviate hospital capacity constraints, the volume of breast biopsy procedures performed in ASCs is expected to grow substantially.
Key U.S. Breast Biopsy Devices Company Insights
The U.S. breast biopsy devices market is led by established manufacturers such as Hologic, Inc., Mammotome (Danaher); BD; Argon Medical; Merit Medical Systems; GE HealthCare; Cook Medical; and MOLLI Surgical (Stryker), supported by their strong product portfolios and nationwide distribution.
Key U.S. Breast Biopsy Devices Companies:
- Hologic, Inc.
- Mammotome (Danaher)
- BD
- Argon Medical
- Merit Medical Systems
- GE HealthCare
- Cook Medical
- MOLLI Surgical (Stryker)
- Planmed Oy
- Siemens Medical Solutions USA, Inc.
- Fujifilm Healthcare
- INRAD
- Resitu Medical
- TransMed7, LLC.
Recent Developments
-
In February 2026, Mammotome, a subsidiary of Danaher Corporation, introduced the Mammotome Prima MR Dual Vacuum-Assisted Breast Biopsy System, the first breast biopsy system specifically engineered for placement inside the MRI scanner room at the patient’s side during procedures. This innovative design strengthens clinical coordination and communication while keeping attention centered on patient care by positioning the vacuum-assisted biopsy device directly within the MRI suite.
-
In January 2026, BD (Becton, Dickinson and Company) announced that the U.S. Food and Drug Administration (FDA) granted 510(k) clearance for the EnCor EnCompass Breast Biopsy and Tissue Removal System. The advanced multi-modality breast biopsy system is designed to provide clinicians with flexibility across various breast imaging modalities for the diagnosis of breast disease. This clearance strengthens BD’s portfolio in minimally invasive breast biopsy solutions.
-
In September 2025, Resitu announced that it received 510(k) clearance from the U.S. Food and Drug Administration (FDA) for its first breast biopsy device, RESL09. The company stated that the milestone marks a significant achievement in advancing its innovative breast biopsy technology for clinical use in the U.S.
-
In January 2025, Siemens Healthineers announced the first U.S. installation of its Mammomat B.brilliant mammography system. Marking the company’s first fully redesigned mammography platform in over a decade, the system features advanced 3D image acquisition and reconstruction technology. It also supports full-field digital mammography (2D imaging), breast biopsy procedures, and titanium-contrast-enhanced mammography, thereby enhancing diagnostic capabilities in breast imaging.
U.S. Breast Biopsy Devices Market Report Scope
Report Attribute
Details
Market size value in 2026
USD 1.22 billion
Revenue forecast in 2033
USD 1.60 billion
Growth rate
CAGR of 3.9% from 2026 to 2033
Actual data
2021 - 2025
Forecast period
2026 - 2033
Quantitative units
Revenue in USD million/billion and CAGR from 2026 to 2033
Segments covered
Product, procedure type, guidance technology, end use
Regional scope
U.S.
Report coverage
Revenue, competitive landscape, growth factors, and trends
Key companies profiled
Hologic, Inc.; Mammotome (Danaher); BD; Argon Medical; Merit Medical Systems; GE HealthCare; Cook Medical; MOLLI Surgical (Stryker); Planmed Oy; Siemens Medical Solutions USA, Inc.; Fujifilm Healthcare; INRAD; Resitu Medical; TransMed7, LLC.
Customization scope
Free report customization (equivalent to 8 analysts working days) with purchase. Addition or alteration to country, regional & segment scope
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
U.S. Breast Biopsy Devices Market Report Segmentation
This report forecasts revenue growth at the country levels and provides an analysis of the latest industry trends in each of the sub-segments from 2021 to 2033. For the purpose of this study, Grand View Research has segmented the U.S. breast biopsy devices market report based on product, procedure type, guidance technology, and end use:
-
Product Type Outlook (Revenue, USD Million, 2021 - 2033)
-
Biopsy Guidance Systems
-
Biopsy Drivers & Handpieces
-
Vacuum-Assisted Biopsy (VAB) Drivers
-
-
Core Needle Biopsy (CNB) Guns
-
Automated
-
Semi-Automated
-
-
Biopsy Tables
-
Prone Tables
-
Upright/Add-on Tables
-
-
Biopsy Consumables
-
Needles & Probes
-
Site Markers & Clips
-
By Shape/Design
-
Standard Geometric Markers
-
Ring
-
Cross
-
Ribbon
-
Others
-
-
Advanced Anchoring Markers
-
Anchor
-
Wing, "U" Shape
-
Q" Shape
-
-
3D / Expandable Markers
-
3D Ribbon
-
3D Sphere
-
3D Mesh
-
-
-
Localization Systems
-
Traditional Wires
-
Wireless Seeds-RFID/Magnetic
-
-
-
Other Consumables
-
-
Procedure Type Outlook (Revenue, USD Million, 2021 - 2033)
-
Vacuum-Assisted Biopsy (VAB)
-
Core Needle Biopsy (CNB)
-
Fine Needle Aspiration Biopsy (FNAB)
-
Surgical (Open) Biopsy
-
Liquid Biopsy
-
-
Guidance Technology Outlook (Revenue, USD Million, 2021 - 2033)
-
Ultrasound-Guided
-
Mammography / Stereotactic-Guided
-
MRI-Guided
-
-
End Use Outlook (Revenue, USD Million; 2021 - 2033)
-
Hospitals
-
Ambulatory Surgical Centers (ASCs)
-
Specialized Breast Care Centers
-
Diagnostic Centers/Imaging Centers
-
Frequently Asked Questions About This Report
b. The U.S. breast biopsy devices market size was estimated at USD 1.18 billion in 2025 and is expected to reach USD 1.22 billion in 2026
b. The U.S. breast biopsy devices market is expected to grow at a compound annual growth rate of 3.9% from 2026 to 2033 to reach USD 1.60 billion by 2033.
b. Biopsy consumables dominated the U.S. breast biopsy devices market with a share of 40.7% in 2025. This is attributable to the recurring demand for single-use components required in every procedure. Disposable items such as core biopsy needles, vacuum-assisted probes, localization wires, and procedure trays generate consistent revenue across hospitals, ambulatory surgical centers, and diagnostic imaging facilities.
b. Some key players operating in the U.S. breast biopsy devices market include Hologic, Inc.; Mammotome (Danaher); BD; Argon Medical; Merit Medical Systems; GE HealthCare; Cook Medical; MOLLI Surgical (Stryker); Planmed Oy; Siemens Medical Solutions USA, Inc.; Fujifilm Healthcare; INRAD; Resitu Medical; TransMed7, LLC.
b. Key factors that are driving the market growth include the rising incidence of breast cancer and the consequent emphasis on early detection and accurate diagnosis, which increases the volume of biopsy procedures required in screening and diagnostic pathways. High awareness among patients and clinicians, supported by structured screening programs and favorable reimbursement landscapes, further propels device adoption.
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










