- Home
- »
- Biotechnology
- »
-
U.S. Cell Culture Media & Cell Lines Market Size Report, 2033GVR Report cover
![U.S. Cell Culture Media & Cell Lines Market Size, Share & Trends Report]()
U.S. Cell Culture Media & Cell Lines Market (2025 - 2033) Size, Share & Trends Analysis Report By Product (Traditional Cell Line, Classical Media), By Application (Biopharmaceutical Production, Diagnostics), By End Use, And Segment Forecasts
- Report ID: GVR-4-68040-680-4
- Number of Report Pages: 90
- Format: PDF
- Historical Range: 2021 - 2024
- Forecast Period: 2025 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
Market Size & Trends
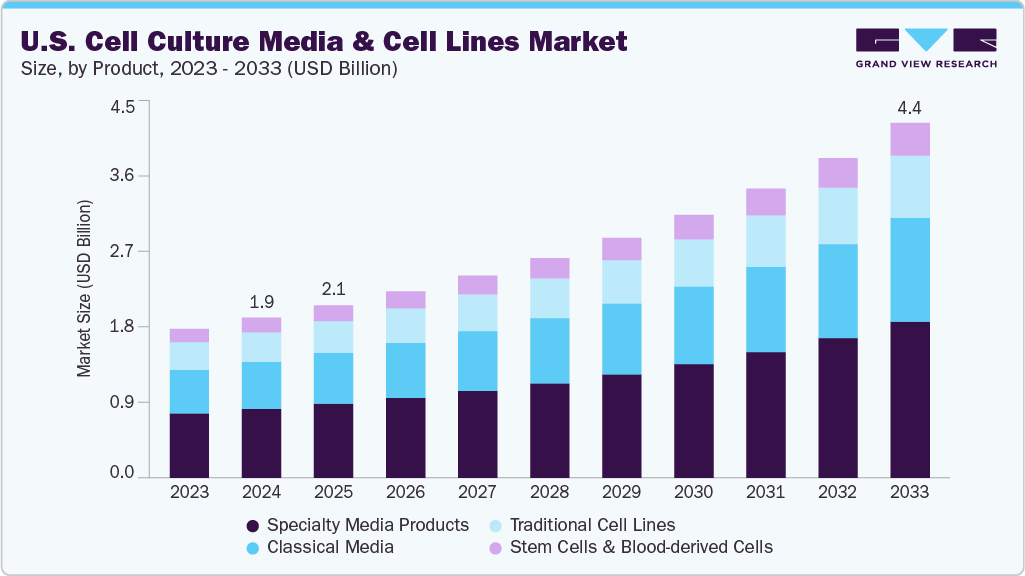
The U.S. cell culture media & cell lines market size was valued at USD 1.97 billion in 2024 and is anticipated to reach USD 4.36 billion by 2033, growing at a CAGR of 9.45% from 2025 to 2033. This growth is driven by increasing demand for biopharmaceutical production, advancements in regenerative medicine, and rising investments in research and development activities. Moreover, the expanding application of cell culture technologies in drug discovery and personalized medicine will fuel market expansion further over the forecast period.
Advancements in Regenerative Medicine
Advancements in regenerative medicine have emerged as a significant driver of the industry. Regenerative medicine aims to repair or replace damaged tissues and organs using cell-based therapies, often involving stem cells and specialized cell lines. The success of these therapies depends on the ability to culture and maintain cells under highly controlled conditions, which requires advanced and carefully formulated culture media. Researchers and biotech companies invest heavily in developing novel regenerative treatments for various situations, including neurodegenerative diseases, cardiovascular disorders, and orthopedic injuries. This growing focus on regenerative therapies has fueled demand for high-quality cell culture products that support efficient cell growth, differentiation, and viability.
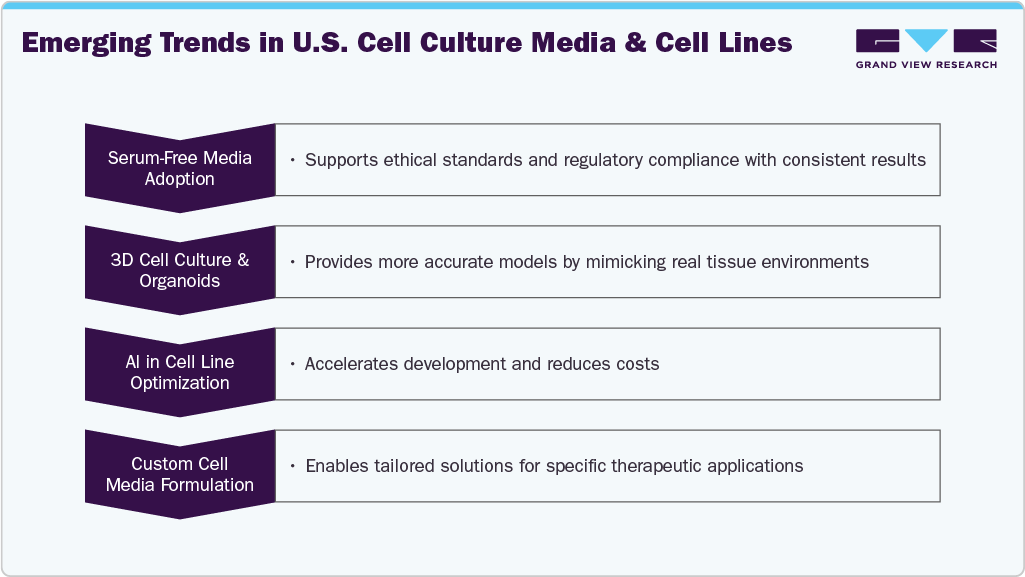
The industry is evolving with innovations that improve compliance, accuracy, cost-efficiency, and customization. The diagram below highlights key trends driving this transformation.
Moreover, the increasing number of clinical trials and regulatory approvals related to regenerative medicine products underscores the need for scalable and reproducible cell culture processes. Manufacturers are adopting innovative serum-free and chemically defined media formulations and well-characterized cell lines to meet these stringent quality and consistency standards. These advancements enhance therapeutic efficacy and facilitate mass production required for commercialization. As regenerative medicine continues to evolve and gain traction in research and clinical settings, it will remain a key catalyst for growth in the U.S. cell culture media and cell lines market throughout the forecast period.
Advancements In Technology And Data Analytics
The drug discovery and development sector is crucial to the U.S. cell culture media and cell lines industry. Pharmaceutical and biotechnology companies rely heavily on cell culture technologies during the early stages of drug development, including target identification, screening, and toxicity testing. High-quality cell culture media are essential to maintain cell health and functionality during these experiments, directly impacting drug development pipelines' reliability and success rate. For instance, in April 2025, Applied StemCell submitted a Type II Drug Master File (DMF) to the U.S. FDA for its ActiCells GMP Human Induced Pluripotent Stem Cell (hiPSC) line. This innovation allows for consistent, reliable expression of one or more genes, offering cell therapy developers an efficient and scalable solution for engineering iPSCs to meet their specific therapeutic needs.
Moreover, as the pharmaceutical industry faces increasing pressure to reduce development timelines and costs, the demand for efficient and scalable cell culture solutions has surged. Innovations such as three-dimensional (3D) cell cultures and high-throughput screening platforms further amplify the need for specialized media and diverse cell lines tailored to specific research requirements. Personalized medicine also plays a role, where patient-derived cell lines are used to test drug responses, helping develop targeted therapies with higher efficacy and fewer side effects. Collectively, these trends contribute to robust demand for advanced cell culture media and cell lines, making drug discovery and development a significant growth engine for the U.S. market.
Market Concentration & Characteristics
The degree of innovation in cell culture media and cell lines is a key driver propelling the U.S. market forward. Advances such as serum-free, chemically defined, and xeno-free media formulations have enhanced consistency, safety, and scalability, addressing critical challenges in biopharmaceutical production and research. Developing genetically engineered and customized cell lines tailored to specific applications has expanded the scope of disease modeling, drug testing, and cell therapy. For instance, in March 2021, Thermo Fisher Scientific launched Gibco Human Plasma Like Medium (HPLM) in the USA, offering a cGMP, physiologically relevant culture medium containing over 60 plasma-level metabolites and salts. This continuous innovation enables more efficient and reliable in vitro models, ultimately accelerating market growth by meeting the evolving needs of pharmaceutical companies, research institutions, and regenerative medicine sectors.
The level of mergers and acquisitions (M&A) activity in the industry significantly influences its growth trajectory. Strategic acquisitions and partnerships enable companies to expand their product portfolios, integrate advanced technologies, and strengthen their market presence. For instance, in April 2023, AnaBios Corporation, based in San Diego, acquired Cell Systems, a human primary cell and cell culture media company located in Kirkland, Washington. This acquisition enhances AnaBios' portfolio of human tissue and cells, providing researchers with a broader array of biologically relevant tools to expedite drug discovery and deepen understanding of cell biology. Cell Systems offers various human primary cells, including endothelial cells from the brain, liver, kidney, eye, and lung. Moreover, consolidation helps organizations achieve economies of scale, reduce costs, and improve operational efficiencies, which are critical in a highly competitive and rapidly evolving industry. The increasing frequency of such transactions reflects growing investor confidence and a robust market outlook, driving further innovation and expansion in the sector.
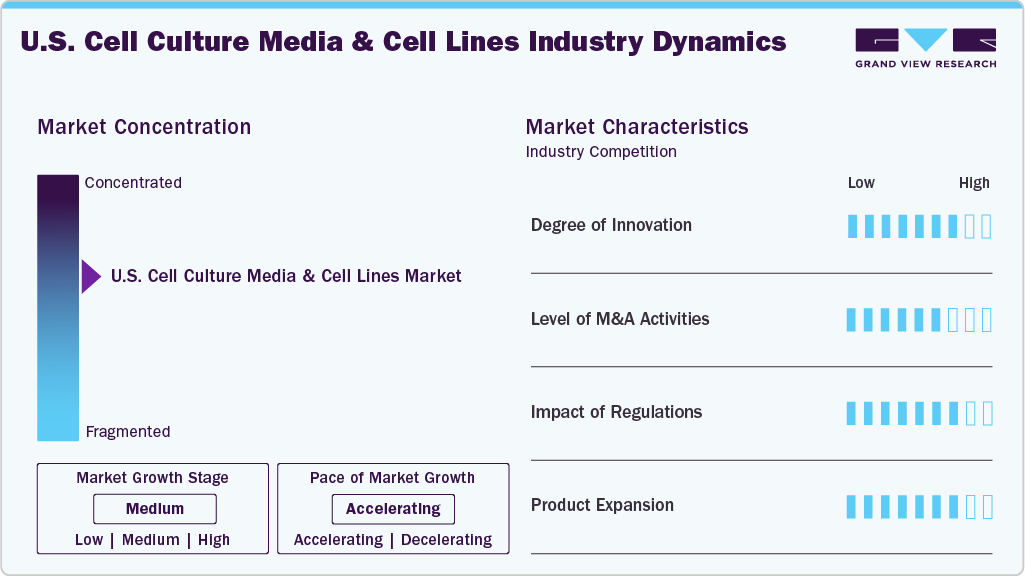
Regulatory frameworks are pivotal in shaping the market, acting as a driver and a gatekeeper of growth. Stringent guidelines set by the FDA and other regulatory bodies ensure the safety, quality, and consistency of products used in drug development, biomanufacturing, and clinical applications. These regulations encourage manufacturers to adopt high-quality, compliant materials and processes, such as using serum-free or chemically defined media and well-characterized cell lines, which, in turn, stimulate innovation and standardization across the industry. As regulatory oversight continues to evolve in response to emerging technologies like gene editing and regenerative medicine, it is expected to shape market dynamics further and encourage the adoption of advanced, regulation-ready cell culture solutions.
Product expansion is pivotal in driving the growth of the industry. Companies are broadening their service portfolios as the demand for customized, scalable, and efficient research and manufacturing solutions increases. These expanded services help clients accelerate time-to-market, reduce operational burdens, and improve research outcomes, particularly in the biopharmaceutical, academic, and regenerative medicine sectors. For instance, in February 2025, Cellistic launched Allo Chassis, ready-to-use, immune-cloaked iPSC cell lines derived from CD34+ and CD4+ T-cell primary cells. These cell lines facilitate cost-effective cell therapy development by reducing timelines and costs. Allo Chassis enables therapeutic developers to create product-specific master cell banks efficiently, supporting applications in regenerative medicine, immune-evasive CAR-T and NK cell therapies, tissue engineering, and organ transplantation. By integrating value-added services alongside core product offerings, providers are enhancing customer loyalty and tapping into new revenue streams and competitive advantages. This trend reflects a shift toward more comprehensive, solution-based business models that cater to evolving scientific and commercial needs.
Product Insights
The specialty media products segment dominated the industry in 2024 with a 43.08% share. This rapid growth is primarily driven by the increasing need for customized and optimized culture media that support the development of specific cell types, including stem cells, primary cells, and genetically engineered cells. Moreover, advancements in biopharmaceutical research, regenerative medicine, and cell therapy fuel demand for specialized media formulations that enhance cell viability, productivity, and functionality. The rising focus on precision medicine and complex cell-based assays further contributes to the expansion of this segment.
The stem cells & blood-derived cells segment is expected to register a significant CAGR throughout the forecast period. This dominance is attributed to the growing focus on regenerative medicine, increasing research activities in hematology and immunology, and the rising adoption of stem cell therapies. Moreover, advancements in blood-derived cell technologies and the expanding application of these cells in drug discovery and personalized medicine have further propelled market growth in this segment.
Application Insights
Biopharmaceutical production held the dominant market share of 46.02% in 2024. The segment's leading position is attributed to the growing demand for monoclonal antibodies, vaccines, and recombinant proteins, which rely heavily on efficient and high-quality cell culture systems. Increasing investments in biopharmaceutical research and manufacturing facilities, along with advancements in cell line development and media optimization, further drive the market expansion.
Tissue engineering and regenerative medicine are projected to grow at the fastest CAGR during the forecast period in the U.S. cell culture media and cell lines industry. Increasing research activities focused on developing innovative therapies for tissue repair and regeneration, and the rising adoption of stem cell technologies are driving this rapid growth. Moreover, advancements in biomaterials and 3D cell culture techniques are accelerating the demand for specialized media and cell lines tailored to these applications.
End Use Insights
Large biopharmaceutical companies dominated the industry in 2024, accounting for the largest share of 31.46%. This dominance is driven by these companies' substantial investments in research and development, advanced manufacturing capabilities, and extensive product pipelines requiring reliable and high-quality cell culture media and cell lines. Their strong focus on biopharmaceutical production, including monoclonal antibodies and vaccines, further reinforces their leading position in the market.
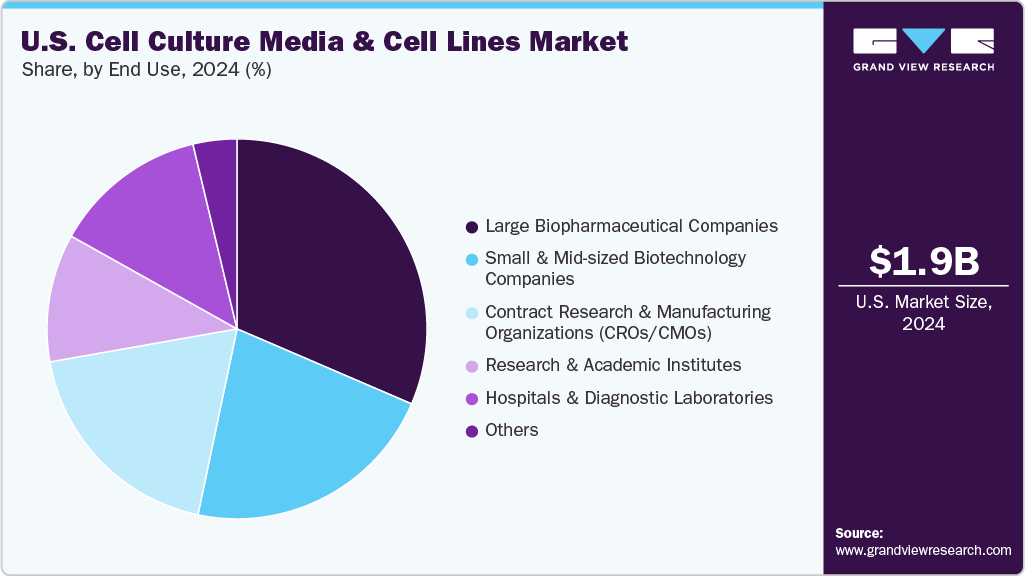
The contract research and manufacturing organizations (CROs/CMOs) segment is expected to witness the fastest growth rate in the market during the forecast period. This growth is fueled by the increasing outsourcing of biopharmaceutical research and production activities to specialized service providers, enabling companies to reduce costs and accelerate time-to-market. Moreover, CROs and CMOs are expanding their cell line development and bioprocessing capabilities, driving higher demand for advanced cell culture media and cell lines tailored to diverse client needs. For instance, in January 2024, Bruker made a strategic minority investment in CRO, NovAliX, strengthening its portfolio and expanding drug discovery and development technologies capabilities.
Key U.S. Cell Culture Media And Cell Lines Companies Insights
Key players operating in the U.S. cell culture media and cell lines market are undertaking various initiatives to strengthen their presence and increase the reach of their products and services. Strategies such as expansion activities and partnerships are key in propelling the market growth.
Specialized providers such as STEMCELL Technologies, PromoCell GmbH, ATCC, and AllCells contribute significantly to the U.S. market by offering primary and immortalized cell lines, specialized media formulations, and tools tailored for specific cell types and experimental needs. These companies are particularly valued for their ability to provide reliable, research-grade, and clinical-grade products supporting cutting-edge immunology, oncology, neuroscience, and more studies.
These organizations actively address key industry challenges such as the demand for serum-free and chemically defined media, improved cell viability, and enhanced experimental reproducibility. They drive advancements in personalized medicine and biologics manufacturing by forming strategic partnerships, upgrading technologies, and expanding into specialized cell culture systems. As the need for cell-based assays, gene therapies, and 3D culture models grows, the U.S. cell culture media & cell lines market is increasingly focused on delivering integrated, end-to-end solutions. Continued emphasis on collaboration, innovation, and regulatory compliance shapes a competitive landscape aimed at streamlining workflows, accelerating drug development, and improving patient outcomes across various therapeutic areas.
Key U.S. Cell Culture Media And Cell Lines Companies:
- Sartorius AG
- Danaher
- Merck KGaA
- Thermo Fisher Scientific, Inc.
- FUJIFILM Corporation
- Lonza
- STEMCELL Technologies
- PromoCell GmbH
- ATCC
- AllCells
Recent Developments
-
In April 2025, Thermo Fisher Scientific introduced a new CHO K-1 cell line and platform technology that cut IND filing timelines from 13 to 9 months. With protein expression up to 8 g/L and enhanced stability, the cell line boosts productivity from pre-clinical to commercial stages. The integrated solution streamlines development and simplifies procurement for biotech and pharma companies.
-
In January 2025, Danaher revealed a USD 1.5 billion investment to boost the manufacturing capabilities of Cytiva and Pall Corporation. This expansion targets rising global demand for biotech products such as cell culture media, chromatography resins, and single-use technologies.
-
In May 2023, Lonza launched TheraPEAK T-VIVO, a GMP-grade, serum-free, chemically defined medium for CAR T-cell manufacturing. Free of animal components enhances consistency, lowers contamination risk, and supports scalable production across platforms like bioreactors and the Cocoon Platform.
U.S. Cell Culture Media And Cell Lines Market Report Scope
Attribute
Details
Market size value in 2025
USD 2.12 billion
Revenue forecast in 2033
USD 4.36 billion
Growth rate
CAGR of 9.45% from 2025 to 2033
Actual data
2021 - 2024
Forecast period
2025 - 2033
Quantitative units
Revenue in USD million/billion and CAGR from 2025 to 2033
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Product, application, end use
Key companies profiled
Sartorius AG; Danaher; Merck KGaA; Thermo Fisher Scientific, Inc.; FUJIFILM Corporation; Lonza; STEMCELL Technologies; PromoCell GmbH; ATCC; AllCells
Customization scope
Free report customization (equivalent up to 8 analyst’s working days) with purchase. Addition or alteration to country, regional & segment scope.
U.S. Cell Culture Media And Cell Lines Market Report Segmentation
This report forecasts revenue growth at the country level and provides an analysis of the latest industry trends in each of the sub-segments from 2021 to 2033. For this study, Grand View Research has segmented the U.S. cell culture media & cell lines market report on the basis of product, application, and end use.
-
Product Outlook (Revenue, USD Million, 2021 - 2033)
-
Traditional Cell Lines
-
Classical Media
-
Stem Cells & Blood-derived Cells
-
Specialty Media Products
-
-
Application Outlook (Revenue, USD Million, 2021 - 2033)
-
Biopharmaceutical Production
-
Diagnostics
-
Drug Screening and Development
-
Tissue Engineering and Regenerative Medicine
-
Other Applications
-
-
End Use Outlook (Revenue, USD Million, 2021 - 2033)
-
Large Biopharmaceutical Companies
-
Small and Mid-sized Biotechnology Companies
-
Contract Research and Manufacturing Organizations (CROs/CMOs)
-
Research and Academic Institutes
-
Hospitals and Diagnostic Laboratories
-
Other End Uses
-
Frequently Asked Questions About This Report
b. The U.S. cell culture media & cell lines market size was estimated at USD 1.97 billion in 2024 and is expected to reach USD 2.12 billion in 2025.
b. The U.S. cell culture media & cell lines market is expected to grow at a compound annual growth rate of 9.45% from 2025 to 2033 to reach USD 4.36 billion by 2033.
b. On the basis of product, specialty media products held the largest revenue share of 43.08% in 2024 and are anticipated to grow at the fastest CAGR over the forecast period for the U.S. cell culture media & cell lines market. This growth is driven by their enhanced formulation, supporting high-yield cell growth and complex bioproduction needs. Rising demand for targeted biologics and cell-based therapies further fuels segment expansion.
b. Some of the key players operating in the market include Sartorius AG; Danaher; Merck KGaA; Thermo Fisher Scientific, Inc.; FUJIFILM Corporation; Lonza; STEMCELL Technologies; PromoCell GmbH; ATCC; and AllCells
b. The major factors driving the market growth include increasing demand for biopharmaceuticals, rising adoption of cell-based therapies, and ongoing advancements in media formulations to support high-efficiency cell growth and productivity. Additionally, strong investments in regenerative medicine and cancer research are accelerating market expansion.
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










