- Home
- »
- Medical Devices
- »
-
U.S. Chemical Indicators Market Size, Industry Report, 2033GVR Report cover
![U.S. Chemical Indicators Market Size, Share & Trends Report]()
U.S. Chemical Indicators Market (2026 - 2033) Size, Share & Trends Analysis Report By Product (Strips, Indicator Tapes, Cards), By Method (Steam Sterilization, Hydrogen Peroxide Sterilization, Ethylene Oxide Sterilization), By End-use, And Segment Forecasts
- Report ID: GVR-4-68040-860-1
- Number of Report Pages: 110
- Format: PDF
- Historical Range: 2026 - 2033
- Forecast Period: 2026 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
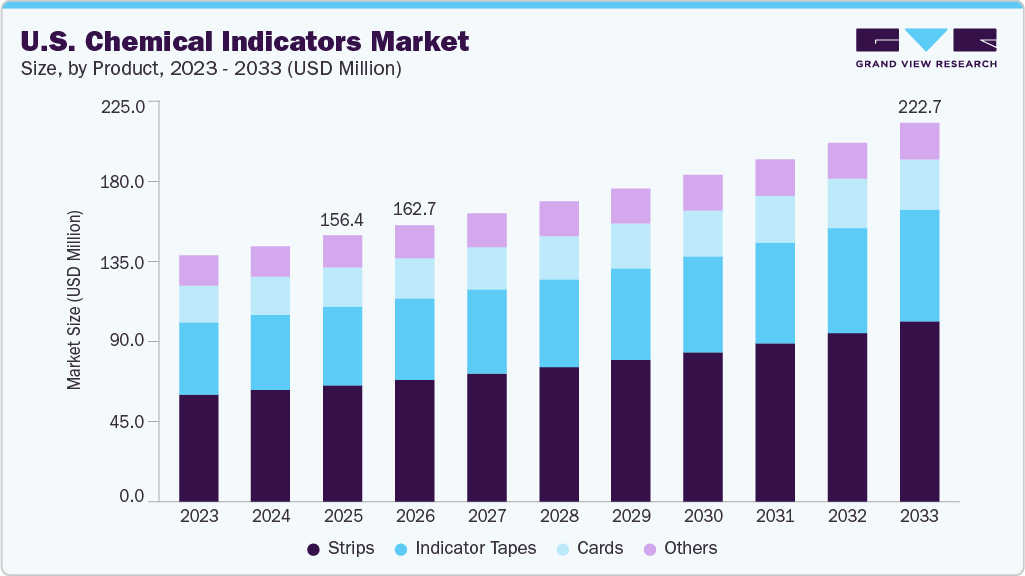
U.S. Chemical Indicators Market Summary
The U.S. chemical indicators market size was estimated at USD 156.43 million in 2025 and is projected to reach USD 222.73 million by 2033, growing at a CAGR of 4.59% from 2026 to 2033. The market is driven by stringent sterilization regulations and compliance requirements across healthcare facilities.
Key Market Trends & Insights
- By product, the strips segment led the market with the largest revenue share in 2025.
- By method, the steam sterilization segment led the market with the largest revenue share in 2025.
- By end-use, the hospitals and clinics segment led the market with the largest revenue share in 2025.
Market Size & Forecast
- 2025 Market Size: USD 156.43 Million
- 2033 Projected Market Size: USD 222.73 Million
- CAGR (2026-2033): 4.59%
Rising surgical procedure volumes and the growing number of outpatient and dental centers increase the demand for routine sterilization monitoring. Increased focus on infection prevention and patient safety further supports consistent use of chemical indicators. In addition, the adoption of advanced sterilization technologies is creating demand for specialized indicator products.The increasing awareness of infection prevention continues to drive the U.S. market for chemical indicators, as healthcare facilities strengthen sterilization monitoring to reduce hospital-acquired infections. Greater emphasis on patient safety and regulatory compliance has led to routine validation of sterilization cycles using chemical indicators across hospitals, laboratories, and surgical centers. For instance, in September 2025, Infection Control Today underscored the critical role of medical device sterility in patient safety, highlighting steam sterilization as the most widely used method and emphasizing the importance of chemical indicators in verifying process effectiveness. The article also referenced ISO 11140-1 standards and compared Type 4 and Type 5 indicators, outlining their performance characteristics and role in ensuring compliant sterilization practices.
According to ISO 11140-1, Type 4 chemical indicators are designed to react to two or more critical sterilization process variables. In comparison, Type 5 chemical indicators respond to all critical variables and are formulated to more closely mimic the performance of a biological indicator (BI). For moist heat sterilization, these variables generally include exposure time and temperature in saturated steam, such as 132°C for 4 minutes. The standard also specifies the required performance criteria and pass/fail conditions for both Type 4 and Type 5 indicators.
According to the U.S. Centers for Disease Control and Prevention in November 2025, chemical indicators are recommended inside every sterilization package to verify that items have been exposed to appropriate sterilizing conditions. The CDC further recommends using chemical indicators in combination with mechanical and biological monitoring methods to enhance assurance of sterilization and support effective infection prevention protocols. As a result, increasing awareness of infection prevention is driving wider adoption of chemical indicators across healthcare and allied sectors. Greater emphasis on sterilization validation and patient safety is reinforcing the routine use of these monitoring products. This sustained focus on compliance and quality assurance continues to contribute to steady growth in the chemical indicators market.
Rising healthcare infrastructure significantly drives the U.S. market for chemical indicators by increasing the number of hospitals, ambulatory surgical centers, specialty clinics, and dental facilities requiring sterilization monitoring. Expansion of surgical departments, diagnostic laboratories, and outpatient care centers leads to a higher volume of sterilization cycles performed daily. Each sterilization cycle requires routine validation using chemical indicators, thereby increasing product consumption. As healthcare systems modernize and expand capacity, demand for reliable sterilization assurance solutions continues to grow. For instance, in June 2025, UC Davis Health highlighted the 48X Complex as part of a major expansion initiative that will add nearly 1 million square feet of new space in the year. The C Street Clinic expansion in East Sacramento opened on May 8, and additional facilities in Elk Grove, Rocklin, and Folsom are scheduled to open in August. By 2030, more than 3.5 million square feet of new facilities, including the 14-story California Tower replacing older structures to meet state seismic requirements, are expected to be completed. The program represents one of the largest health system capital improvement initiatives in the U.S.
“We are passionate about meeting the needs of our patients and their families. We know the demand for outpatient surgery is going to grow dramatically in the coming years,” said UC Davis Health Interim Vice Chancellor of Human Health Sciences and Chief Clinical Officer Bruce Hall. “The future of care delivery is to meet patients where they need us, when they need us, and how they need us, which might mean outpatient, in their home, or even virtual; and enabled by new medical technologies such as wearable devices. We are trying to put patients at the center of what we do, ensuring they feel listened to, heard, respected, and valued. These are all key elements in healing and creating long-term health partnerships with the patients and families we will serve in this new center.”
Such a large-scale expansion of healthcare infrastructure directly supports growth in the U.S. market for chemical indicators. The addition of new hospitals, surgical units, outpatient clinics, and specialty centers increases the daily volume of sterilization cycles. Each new operating room, procedure suite, and diagnostic facility requires routine sterilization monitoring in compliance with regulatory standards. As a result, expanding healthcare capacity drives higher and recurring demand for chemical indicators across the U.S.
Market Concentration & Characteristics
The U.S. market for chemical indicators exhibits a high degree of innovation, driven by the development of advanced, rapid-response indicator technologies that improve safety and compliance across healthcare, food, and industrial applications. Manufacturers are integrating smart and digital sensing capabilities to enhance accuracy, traceability, and real-time monitoring. There is also growing innovation in environmentally friendly and biodegradable indicator formats to meet sustainability demands. Continuous R&D efforts are expanding product functionality and performance, strengthening market competitiveness.
The U.S. market for chemical indicators is witnessing a moderate level of mergers and acquisitions as companies seek to broaden their product portfolios and strengthen technological capabilities. Strategic transactions often focus on acquiring niche players with specialized indicator technologies or complementary sensor solutions. Larger firms are also consolidating to expand their geographic reach and distribution networks. This M&A activity supports competitive positioning and accelerates innovation in the market.
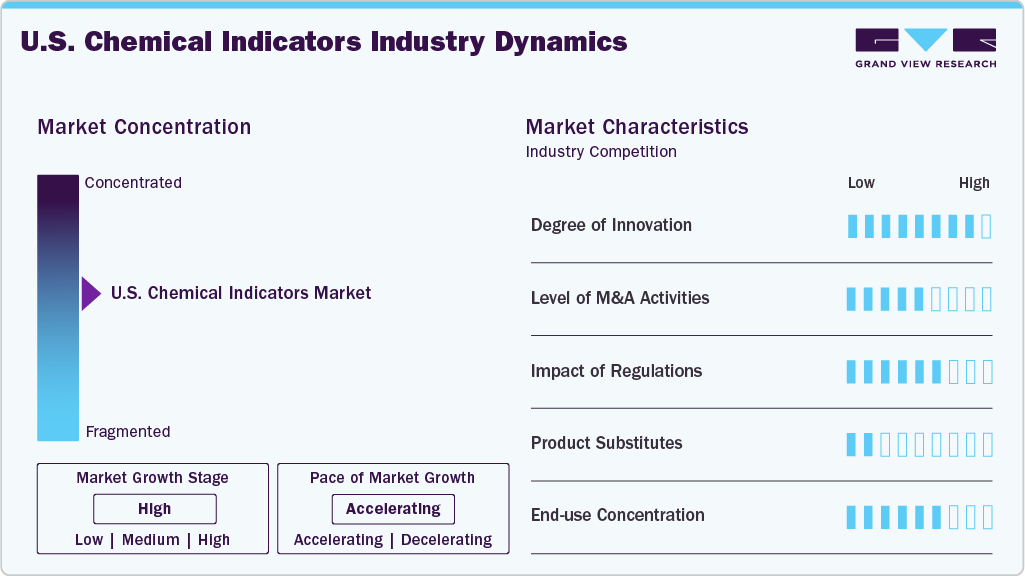
Regulations have a significant impact on the U.S. chemical indicators industry, particularly in healthcare sterilization and infection control applications. Compliance with FDA guidelines, CDC standards, and ANSI/AAMI sterilization requirements increases product validation and quality assurance costs for manufacturers. However, strict regulatory oversight also drives demand for high-performance, reliable chemical indicators to ensure patient safety and compliance with accreditation standards. Evolving regulatory updates continue to shape product development and market entry strategies.
In the U.S. chemical indicators industry, alternatives such as biological indicators, electronic monitoring systems, and physical process monitors act as key substitutes for conventional chemical indicator strips and tapes. Advanced digital tracking and sensor-based sterilization-monitoring solutions provide real-time data and greater accuracy, reducing reliance on traditional indicators in certain settings. These substitute technologies are adopted in hospitals and industrial facilities seeking enhanced compliance and automation. As a result, manufacturers are focusing on product innovation to remain competitive.
Product Insights
The strips segment dominated the U.S. market in 2025 and is expected to grow fastest over the forecast period, driven by widespread use in routine sterilization monitoring across healthcare facilities. Strips offer clear visual results, ease of application inside instrument packs, and cost-effectiveness, making them preferred for daily sterilization assurance. Their compatibility with various sterilization methods, especially steam and ethylene oxide, further supports strong adoption among hospitals, clinics, and central sterile processing departments.
Indicator tapes are witnessing significant growth in the U.S. market due to their simplicity and ease of use in sterilization processes. They are widely applied on the exterior of packs and trays to quickly differentiate processes from unprocessed items. Their cost-effectiveness and suitability for steam and ethylene oxide sterilization make them a preferred choice in hospitals and clinics. The increased focus on routine sterilization validation is further supporting the growth of the indicator tape segment.
Method Insights
The steam sterilization segment dominated the U.S. market in 2025 due to the widespread use of autoclaves in hospitals, ambulatory surgical centers, and dental clinics. Steam sterilization is the most adopted and cost-effective method for sterilizing surgical instruments and medical devices. As a result, demand for steam-specific chemical indicator strips, tapes, and integrators remains consistently high. Strict compliance requirements for routine steam cycle monitoring further support the segment’s leading position.
The hydrogen peroxide sterilization segment is expected to grow at the fastest rate during the forecast period. Increasing adoption of low-temperature sterilization systems, particularly for heat- and moisture-sensitive medical devices, is driving demand for compatible chemical indicators. Healthcare facilities are using vaporized hydrogen peroxide (VHP) systems for faster cycle times and greater material compatibility. This shift is accelerating the need for specialized hydrogen peroxide chemical indicators.
End-use Insights
Hospitals and clinics dominated the U.S. market in 2025, due to the high volume of surgical procedures and routine instrument reprocessing conducted in these settings. These facilities require continuous monitoring of sterilization to comply with infection control guidelines and patient safety standards. The presence of central sterile services departments (CSSDs) in large hospitals further drives consistent use of chemical indicators. In addition, strict regulatory oversight in healthcare institutions strengthens demand within this segment.
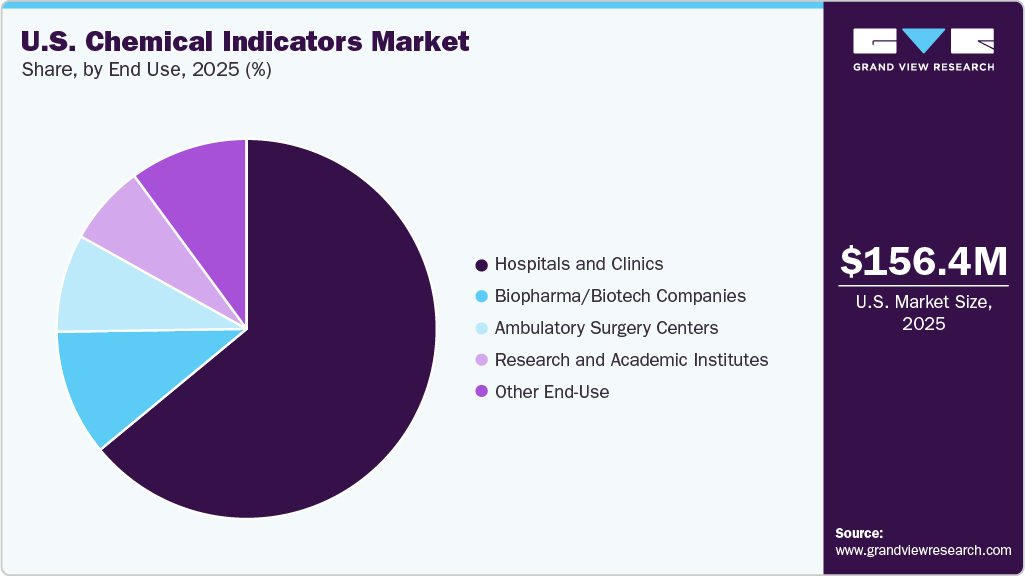
Biopharma and biotech companies are expected to grow at the fastest rate during the forecast period. Increasing production of sterile drugs, biologics, and advanced therapies is driving the need for stringent sterilization validation processes. These companies require reliable monitoring solutions to meet regulatory compliance and quality assurance standards. Expanding research activities and manufacturing capacity worldwide are further supporting rapid segment growth.
Key U.S. Chemical Indicators Company Insights
The U.S. chemical indicators industry is led by established manufacturers such as STERIS, ASP, Mesa Laboratories, and Propper Manufacturing Co., supported by their strong product portfolios and nationwide distribution presence. Market share is largely concentrated among these key players due to strict regulatory compliance requirements and consistent demand from hospitals, surgical centers, and dental facilities.
Key U.S. Chemical Indicators Companies:
- Propper Manufacturing Co.
- STERIS
- ASP
- Solventum
- Getinge
- Tuttnauer
- Andersen Sterilizers
- Mesa Laboratories, Inc.
- Hu-Friedy Mfg. Co.
- True Indicating
Recent Developments
- In January 2025, True Indicating, LLC, a company of sterilization monitoring technologies, announced FDA 510(k) clearance for its CSPN-15 Type 5 Integrating Indicator for Steam. The clearance confirmed the product’s substantial equivalence to legally marketed predicate devices, enabling the company to launch this sterilization assurance solution to market.
"We are proud to receive FDA clearance for our CSPN-15, which represents years of dedicated research and development," said Bob Napierala, Co-Founder & CEO. "This approval reinforces our mission to support healthcare providers with products that meet the highest standards."
"Our CSPN-15 indicator is engineered for accuracy and reliability," said Thomas Riha, Chief Scientific Officer. "It offers confidence during every sterilization cycle-especially in high-stakes clinical environments."In December 2025, LifeNet Health, a global provider of regenerative medicine, announced the acquisition of Tissue Testing Technologies LLC (T3), a biotechnology company specializing in biopreservation. T3’s proprietary technologies and services are designed to preserve living cells and tissues for clinical and research applications, supporting the quality and effectiveness of life-saving therapies.
-
In September 2024, Propper Manufacturing Company announced that its EO Chex Ethylene Oxide (EO) Indicator Tape received FDA clearance, making it the only FDA-cleared ethylene oxide indicator tape available for sale in the U.S.
U.S. Chemical Indicators Market Report Scope
Report Attribute
Details
Market size value in 2026
USD 162.69 million
Revenue forecast in 2033
USD 222.73 million
Growth rate
CAGR of 4.59% from 2026 to 2033
Actual data
2021 - 2025
Forecast period
2026 - 2033
Quantitative units
Revenue in USD million, and CAGR from 2026 to 2033
Segments covered
Product, method, end-use
Regional scope
U.S.
Report coverage
Revenue, competitive landscape, growth factors, and trends
Key companies profiled
Propper Manufacturing Co.; STERIS; ASP; Solventum; Getinge; Tuttnauer; Andersen Sterilizers; Mesa Laboratories, Inc.; Hu-Friedy Mfg. Co.; True Indicating
Customization scope
Free report customization (equivalent to 8 analysts working days) with purchase. Addition or alteration to country, regional & segment scope
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
U.S. Chemical Indicators Market Report Segmentation
This report forecasts revenue growth at the country levels and provides an analysis of the latest industry trends in each of the sub-segments from 2021 to 2033. For this study, Grand View Research has segmented the U.S. chemical indicators market report based on product, method, and end-use:
-
Product Outlook (Revenue, USD Million, 2021 - 2033)
-
Strips
-
Indicator Tapes
-
Cards
-
Others
-
-
Method Outlook (Revenue, USD Million, 2021 - 2033)
-
Steam Sterilization
-
Ethylene Oxide Sterilization
-
Hydrogen Peroxide Sterilization
-
Others
-
-
End-use Outlook (Revenue, USD Million, 2021 - 2033)
-
Hospitals and Clinics
-
Biopharma/Biotech Companies
-
Ambulatory Surgery Centers
-
Research and Academic Institutes
-
Other End Use
-
Frequently Asked Questions About This Report
b. The U.S. chemical indicators market size was estimated at USD 156.43 million in 2025 and is expected to reach USD 162.69 million in 2026.
b. The U.S. chemical indicators market is expected to grow at a compound annual growth rate of 4.59% from 2026 to 2033 to reach USD 222.73 million by 2033.
b. Strips segment dominated the U.S. chemical indicators market with a share of 43.88% in 2025. This is attributable to widespread use in routine sterilization monitoring across healthcare facilities. Strips offer clear visual results, ease of application inside instrument packs, and cost-effectiveness, making them preferred for daily sterilization assurance.
b. Some key players operating in the U.S. chemical indicators market include Propper Manufacturing Co., STERIS, ASP, Solventum, Getinge, Tuttnauer, Andersen Sterilizers, Mesa Laboratories, Inc., Hu-Friedy Mfg. Co., True Indicating
b. Key factors that are driving the market growth include stringent sterilization regulations and compliance requirements across healthcare facilities. Rising surgical procedure volumes and the growing number of outpatient and dental centers increase the demand for routine sterilization monitoring. Increased focus on infection prevention and patient safety further supports consistent use of chemical indicators.
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










