- Home
- »
- Medical Devices
- »
-
U.S. Endobronchial Ultrasound Biopsy Market Report, 2033GVR Report cover
![U.S. Endobronchial Ultrasound Biopsy Market Size, Share & Trends Report]()
U.S. Endobronchial Ultrasound Biopsy Market (2026 - 2033) Size, Share & Trends Analysis Report By Product (Equipment, Consumables), By Procedure (Standalone EBUS Lymph Node Biopsy Procedures, Combined Procedures), By Modality, By CPT Codes, By Site, And Segment Forecasts
- Report ID: GVR-4-68040-906-5
- Number of Report Pages: 130
- Format: PDF
- Historical Range: 2021 - 2025
- Forecast Period: 2026 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
U.S. Endobronchial Ultrasound Biopsy Market Summary
The U.S. endobronchial ultrasound biopsy market size was estimated at USD 32.5 million in 2025 and is projected to reach USD 45.7 million by 2033, growing at a CAGR of 4.4% from 2026 to 2033. The market is also categorized into a procedure side which was estimated at USD 861.0 million in 2025 and is projected to grow at a CAGR of 6.5% from 2026 to 2033.
Key Market Trends & Insights
- By equipment, EBUS scopes/EBUS systems segment dominated the market with the largest revenue share of 71.9% in 2025
- By procedure, standalone EBUS lymph node biopsy procedures segment dominated the market with the largest revenue share in 2025.
- By modality, the ion-based robotic-assisted bronchoscopy segment is expected to grow at the fastest CAGR
- By site, the academic medical centers segment is expected to grow at the fastest CAGR
Market Size & Forecast
- 2025 Market Size: USD 32.5 Million
- 2033 Projected Market Size: USD 45.7 Million
- CAGR (2026-2033): 4.4%
Key factors contributing to the market growth include rising incidence of lung cancer and respiratory disorders, rapid technological evolution and increasing market competition in the U.S. endobronchial ultrasound biopsy (EBUS) market. In addition, expansion of lung cancer screening programs, shift toward advanced bronchoscopy modalities driving EBUS adoption, favorable reimbursement and clinical guidelines.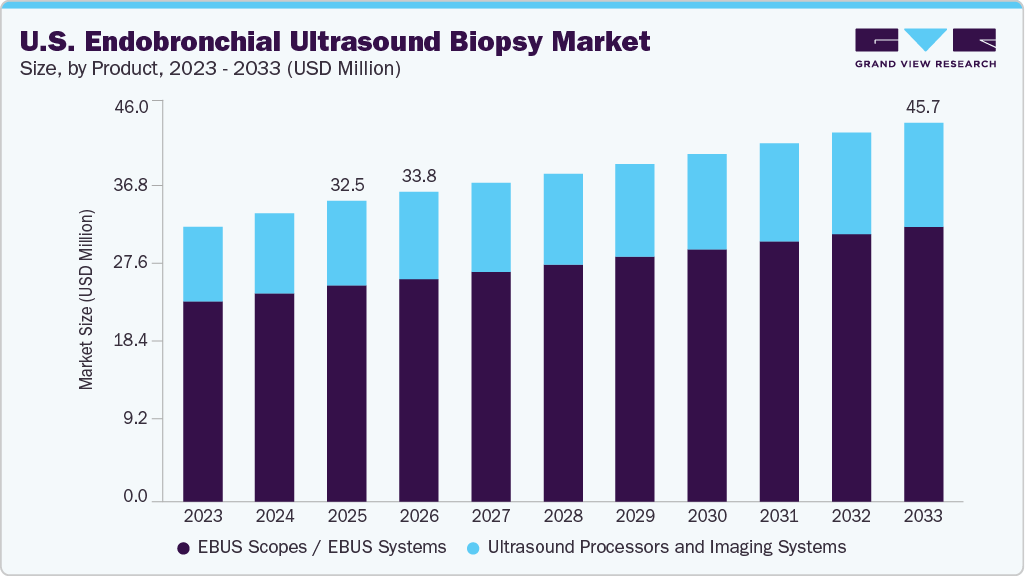
The expansion of lung cancer screening programs across U.S. states is a key driver of EBUS adoption, as the increasing use of low-dose CT (LDCT) is leading to higher detection of early-stage lung nodules and mediastinal abnormalities that require confirmatory diagnosis and staging. State-led and community-based initiatives focused on improving access, reducing disparities, and integrating advanced technologies such as AI are expanding screening coverage among high-risk and underserved populations. This is significantly increasing patient volumes entering diagnostic pathways, thereby driving demand for minimally invasive procedures such as EBUS. For instance:
-
In March 2026, the American Lung Association announced a statewide initiative offering free low-dose CT lung cancer screenings to at-risk Illinois residents through May to improve early detection. The program aims to reduce screening disparities, particularly among Black and Hispanic populations, by providing navigation support, transportation, and financial assistance to overcome barriers to care.
-
In September 2025, the Ohio State University Comprehensive Cancer Center James launched a mobile lung cancer screening unit to expand early detection and prevention efforts across Ohio, particularly targeting underserved communities. The unit travels statewide to provide low-dose CT screenings for high-risk populations, aiming to improve access, reduce geographic barriers, and enable earlier diagnosis, thereby improving treatment outcomes.
-
In April 2025, The Healthy Lungs California initiative was expanded through a five-year partnership between the University of California Lung Cancer Consortium and AstraZeneca to improve early lung cancer screening rates and reduce mortality. The program focuses on increasing access in underserved communities, launching local screening efforts, and integrating innovations such as AI-assisted detection while addressing disparities and environmental risk factors like pollution and wildfire exposure
Moreover, the growing adoption of advanced bronchoscopy modalities, including electromagnetic navigation bronchoscopy and Ion-based robotic-assisted bronchoscopy, across U.S. hospitals is directly driving the transition away from conventional diagnostic approaches such as transthoracic needle aspiration (TTNA), traditional bronchoscopy, and surgical biopsy.
These technologies enable precise navigation to small, peripheral, and hard-to-reach lung nodules using 3D imaging, navigation systems, and robotic control, significantly improving diagnostic yield while reducing complications and invasiveness. As these modalities become integral to lung nodule evaluation, they are increasingly used in combination with EBUS for comprehensive diagnosis and staging, thereby directly contributing to higher EBUS procedural volumes.
For instance, in March 2026, Mercy Hospital Fort Smith introduced robotic-assisted bronchoscopy technology to enable more precise, minimally invasive diagnosis of lung nodules and improve early detection of lung disease. The system uses advanced 3D CT mapping and robotic navigation to access small or hard-to-reach lesions, allowing same-procedure biopsy while reducing the need for invasive surgeries and hospital stays.
“Having a robotic-assisted bronchoscopy at Mercy allows patients to avoid more invasive procedures and unnecessary surgeries, this technology brings a new level of precision to lung diagnostics.”
- Dr. Paul Bean, chief medical officer at Mercy Fort Smith.
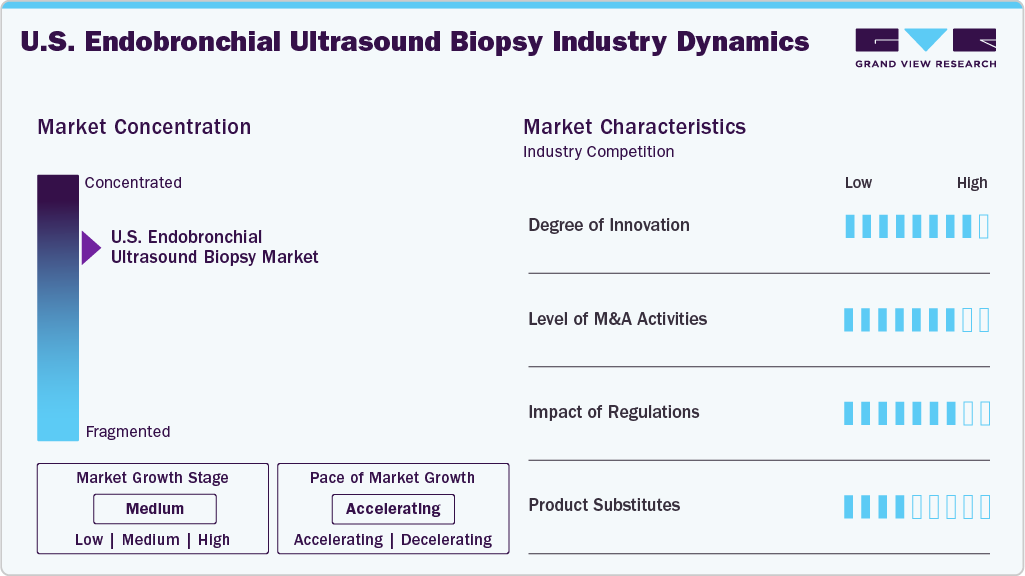
Market Concentration & Characteristics
The chart below illustrates the relationship between market concentration, industry characteristics, and industry participants. The x-axis represents the level of industry concentration, ranging from low to high, while the y-axis captures key industry characteristics, including competition intensity, regulatory impact, partnerships & collaborations, degree of innovation, and regional expansion. In a slightly concentrated market structure, the endobronchial ultrasound market is characterized by the presence of a few dominant players with a limited number of other participants. The industry exhibits high levels of innovation, moderate levels of partnership and collaboration, and significant regulatory influence, while regional expansion remains moderate.
The U.S. Endobronchial Ultrasound Biopsy (EBUS) industry is witnessing high innovation, driven by continuous advancements in devices and accessories aimed at improving procedural efficiency and patient outcomes. For instance, in October 2025, Fujifilm Healthcare Americas launched the Eluxeo 8000 imaging system in the United States, featuring 4K resolution, advanced noise reduction, and enhanced visualization for improved endoscopic diagnostics and workflow efficiency.
“We are steadfast in our commitment to advance the field of endoscopy with innovations that empower GI physicians to safely and efficiently accomplish even the most challenging procedures with the best possible outcomes,”
-Tai Fujita, vice president, Endoscopy Division, Fujifilm Healthcare Americas.
The U.S. Endobronchial Ultrasound Biopsy (EBUS) market is witnessing moderate merger and acquisition activities, with companies pursuing selective acquisitions to strengthen their geographic presence and expand distribution capabilities. For instance, in January 2025, Olympus America acquired the distribution business from Sur Medical SpA in Chile, enabling Olympus Latin America to directly sell, distribute, and service its full medical technology portfolio through its newly established local subsidiary.
The impact of regulations is high in the U.S. Endobronchial Ultrasound Biopsy (EBUS) market, as devices require stringent approval processes from the U.S. Food and Drug Administration, including 510(k) clearance for most systems and accessories. These requirements ensure strong standards of safety, efficacy, and clinical performance, supporting physician confidence and adoption. However, they also increase development timelines, compliance costs, and entry barriers, thereby restricting rapid market entry while maintaining overall market quality.
Traditional diagnostic methods such as conventional bronchoscopy, Transthoracic Needle Aspiration (TTNA), and surgical biopsy serve as potential substitutes for the endobronchial ultrasound biopsy industry. While TTNA offers direct access to peripheral lung lesions, it carries a higher risk of pneumothorax. Similarly, surgical biopsy, including mediastinoscopy, provides a definitive diagnosis but is invasive, costly, and associated with longer recovery times.
Equipment Insights
Based on equipment, EBUS scopes / EBUS systems segment dominated the market with the largest revenue share of 71.9% in 2025. The large share of the segment is attributed to the direct procedural dependence on EBUS scopes for real-time airway navigation and tissue sampling, their essential role in enabling transbronchial needle aspiration and lymph node staging, high utilization rates in both diagnostic and staging bronchoscopy procedures, and continuous upgrades in scope flexibility, image resolution, and needle compatibility that enhance procedural accuracy and efficiency.
The ultrasound processors and imaging systems segment held a significant share in the market, which is attributed to the increasing adoption of image-guided minimally invasive procedures and the growing emphasis on diagnostic accuracy in lung cancer staging and pulmonary disease evaluation, which are contributing to the growth of the ultrasound processors and imaging systems segment. These systems are widely utilized across pulmonology departments, thoracic surgery units, and interventional suites, supporting applications such as mediastinal lymph node assessment, peripheral lung nodule detection, and evaluation of complex lymphadenopathy.
Consumables Insights
Based on consumables, the EBUS-TBNA needles segment dominated the market with the largest revenue share in 2025. A large share of the segment is attributed to their essential role in enabling accurate tissue aspiration during EBUS-guided procedures, high procedural demand for minimally invasive biopsy tools in lung cancer staging and diagnosis, repeated per-procedure usage driving consistent demand, and increasing preference for advanced needle designs that improve sample quality and diagnostic yield.
However, the EBUS-related bronchoscopic procedure consumables and accessories segment is expected to grow at the fastest CAGR over the forecast period, driven by the increasing procedural volume of endobronchial ultrasound-guided interventions, recurring demand for single-use items such as biopsy needles, guide sheaths, and sampling accessories, and the growing emphasis on infection control and procedural safety in bronchoscopy suites.
Site Insights
Based on the site, the academic medical centers segment dominated the market with the largest revenue share in 2025, supported by high patient inflow for complex pulmonary cases in the U.S., early adoption of advanced endobronchial ultrasound technologies, and strong involvement in clinical research, training, and guideline-driven diagnostic practices across leading healthcare institutions. For instance, in October 2025, Marshall Health Network’s Cabell Huntington Hospital in West Virginia performed the state’s first AI-enhanced MONARCH robotic lung bronchoscopy, leveraging advanced navigation technology to improve precision in diagnosing hard-to-reach lung nodules, further highlighting the role of academic centers in driving adoption of advanced bronchoscopic innovations.
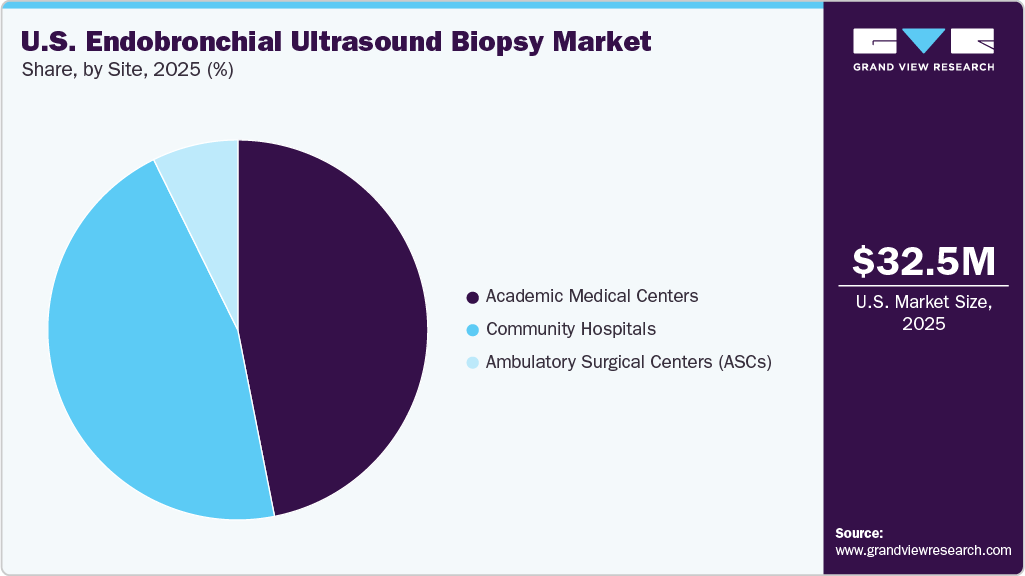
In addition, ambulatory surgical centers (ASCs) segment is expected to show significant growth over the forecast period. This growth is supported by the strong shift toward minimally invasive and outpatient diagnostic procedures, where EBUS plays a key role in early lung cancer diagnosis and lymph node staging. In addition, according to the Ambulatory Surgery Center Association, as of March 2025, there are over 6,500 Medicare-certified ASCs in the United States, highlighting the robust infrastructure supporting outpatient procedural care. The increasing preference for cost-effective, same-day procedures, along with reduced hospital stay requirements and improved procedural efficiency, is further driving the adoption of EBUS in ASC settings.
Key U.S. Endobronchial Ultrasound Biopsy Company Insights
The U.S. Endobronchial Ultrasound Biopsy (EBUS) market is shifting towards consolidation. Key players such as Olympus Corporation, FUJIFILM Holdings, Boston Scientific, Cook Group, and PENTAX Medical dominate the market with comprehensive EBUS-biopsy systems, needles, and imaging platforms. These companies are actively expanding their portfolios through product innovation, clinical collaborations, and expanded distribution networks to strengthen presence. Meanwhile, emerging firms like Praxis Medical, CONMEDACE Medical Devices Pvt. Ltd., and Medi-Globe Technologies are gaining visibility by introducing cost-effective, single-use biopsy tools and motorized needle systems tailored for precision lung diagnostics.
Key U.S. Endobronchial Ultrasound Biopsy Companies:
- PENTAX Medical (a division of HOYA Group )
- Boston Scientific Corporation
- Olympus America (Subsidiary of Olympus Corporation)
- Cook Group
- FUJIFILM Holdings Corporation (Acquired by DCC Healthcare in October 2022
- Medi-Globe Technologies GmbH
- ACE Medical Devices Pvt. Ltd.
- Praxis Medical
- CONMED
Recent Developments
-
In July 2025, Olympus Corporation of the Americas launched the EU-ME3 ultrasound processor in the United States, integrating EUS/EBUS imaging with advanced diagnostics, high-resolution visualization, and broad endoscope compatibility.
"As healthcare facilities look for ways to improve patient outcomes and save costs, we are pleased to offer our customers a versatile, state-of-the-art ultrasound platform for GI and pulmonary applications."
-Richard Reynolds, President of the Medical Systems Group at Olympus Corporation of the Americas.
-
In September 2024, FUJIFILM Holdings Corporation announced the compatibility of its ARIETTA 750 FF ENDO ultrasound system with several ultrasound endoscopes and a linear bronchoscope. This system features advanced technologies such as “eFocusing” for improved image clarity and “Carving Imaging” for enhanced visualization.
-
In October 2023, Praxis Medical received FDA clearance to the EndoCore EBUS‑TBNA biopsy device, a motorized, needle‑rotating system designed for endobronchial ultrasound-guided lung biopsies.
“We are excited by the implications of how this device could progress the standard of care. Improving EBUS-TBNA sensitivity, diagnostic yield, and ancillary testing capabilities would allow for an expedited workup of all types of adenopathy while maintaining procedural efficiency and low complication rates.”
- Dr. Joseph Romero, a Pulmonologist at Morton Plant Hospital in Clearwater, FL.
U.S. Endobronchial Ultrasound Biopsy Market Report Scope
Report Attribute
Details
Product Side Market size value in 2026
USD 33.82 million
Product Side Revenue forecast in 2033
USD 45.72 million
Procedure Side Market size value in 2026
USD 912.8 million
Procedure Side Revenue forecast in 2033
USD 1,414.3 million
Growth Rate (Product Side Market)
CAGR of 4.4% from 2026 to 2033
Actual data
2021 - 2025
Forecast period
2026 - 2033
Quantitative units
Market Value in USD million/billion, Annual Equipment Sales Volume, Procedure volume (000’), and CAGR from 2026 to 2033
Report coverage
Revenue forecast, annual equipment sales volume forecast, procedure volume (000’), forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Product side market (Equipment and Consumables), Procedure side market (Procedures, modality, CPT codes )
Key companies profiled
PENTAX Medical, Boston Scientific Corporation, Olympus America, Cook Group, FUJIFILM Holdings Corporation, Medi-Globe Technologies GmbH, ACE Medical Devices Pvt. Ltd., Praxis Medical, CONMED Corporation
Customization scope
Free report customization (equivalent to 8 analysts working days) with purchase. Addition or alteration to country, regional & segment scope.
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
U.S. Endobronchial Ultrasound Biopsy Market Report Segmentation
This report forecasts revenue growth at U.S. levels and provides an analysis of the latest industry trends in each of the sub-segments from 2021 to 2033. For this study, Grand View Research has segmented the U.S. Endobronchial Ultrasound Biopsy market report based on the product side market (Equipment and Consumables), the procedure side market (Procedures, modality, CPT codes, end use).
-
Product Side Market
-
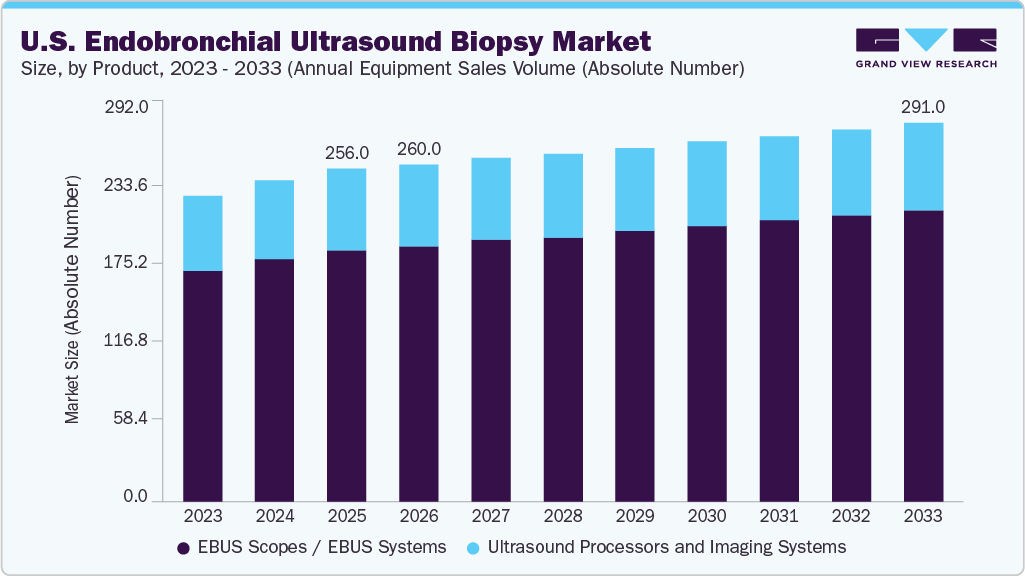
Equipment Outlook (Annual Equipment Sales Volume (Absolute Number), Revenue, USD Million 2021 - 2033)
-
EBUS Scopes / EBUS Systems
-
Linear EBUS (CP-EBUS)
-
Radial Probe EBUS (RP-EBUS)
-
-
Ultrasound Processors and Imaging Systems
-
-
Consumables Outlook (Annual Equipment Sales Volume (Absolute Number), Revenue, USD Million 2021 - 2033)
-
EBUS-TBNA Needles
-
EBUS-Related Bronchoscopic Procedure Consumables and Accessories
-
Biopsy Forceps
-
Cytology Brushes
-
Cryoprobes
-
Guide Sheaths
-
Accessories
-
-
-
-
Procedure Side Market Outlook
-
Procedure Outlook (Procedure Volume, in ‘000 Procedures, Revenue, USD Million 2021 - 2033)
-
Standalone EBUS Lymph Node Biopsy Procedures
-
Combined Procedures
-
-
CPT Codes Outlook (Procedure Volume, in ‘000, Revenue, USD Million 2021 - 2033)
-
31652
-
31653
-
31654
-
-
Site Outlook (Procedure Volume, in ‘000, Revenue, USD Million 2021 - 2033)
-
Academic Medical Centers
-
Community Hospitals
-
Ambulatory Surgical Centers (ASCs)
-
-
Modality Outlook (Procedure Volume, in ‘000, Revenue, USD Million 2021 - 2033)
-
Transthoracic Needle Aspiration (TTNA)
-
Traditional Bronchoscopy
-
Surgical Biopsy
-
Electromagnetic Navigation Bronchoscopy
-
Robotic-Assisted Bronchoscopy
-
-
Procedure Vs Site Outlook (Procedure Volume, in ‘000 Procedures, 2021 - 2033)
-
Standalone EBUS Lymph Node Biopsy Procedures
-
Academic Medical Centers
-
Community Hospitals
-
Ambulatory Surgical Centers (ASCs)
-
-
Combined Procedures
-
Academic Medical Centers
-
Community Hospitals
-
Ambulatory Surgical Centers (ASCs)
-
-
-
Frequently Asked Questions About This Report
b. The U.S. endobronchial ultrasound biopsy market size was estimated at USD 32.5 million in 2025 and is expected to reach USD 33.8 million in 2026.
b. Based on equipment the EBUS scopes / EBUS systems segment dominated the market with the largest revenue share of 71.9% in 2025. The large share of the segment is attributed to the direct procedural dependence on EBUS scopes for real-time airway navigation and tissue sampling
b. Some key players operating in the U.S. endobronchial ultrasound biopsy (EBUS) market include PENTAX Medical, Boston Scientific Corporation, Olympus America, Cook Group, FUJIFILM Holdings Corporation, Medi-Globe Technologies GmbH, ACE Medical Devices Pvt. Ltd., Praxis Medical, CONMED Corporation
b. Key factors that are driving the market growth include rising incidence of lung cancer and respiratory disorders, rapid technological evolution and increasing market competition in the U.S. Endobronchial Ultrasound Biopsy market, , expansion of lung cancer screening programs, and shift toward advanced bronchoscopy modalities.
b. The U.S. endobronchial ultrasound biopsy market is expected to grow at a compound annual growth rate of 4.4% from 2026 to 2033 to reach USD 45.7 million by 2033.
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










