- Home
- »
- Medical Devices
- »
-
U.S. Peripheral Vascular Devices And Accessories Market Report 2033GVR Report cover
![U.S. Peripheral Vascular Devices And Accessories Market Size, Share & Trends Report]()
U.S. Peripheral Vascular Devices And Accessories Market (2026 - 2033 ) Size, Share & Trends Analysis Report By Product (Peripheral Stents, PTA Balloons), By Application (Peripheral Arterial Disease, Aneurysms), By End Use (Hospitals, Ambulatory Surgical Centers), And Segment Forecasts
- Report ID: GVR-4-68039-243-5
- Number of Report Pages: 120
- Format: PDF
- Historical Range: 2021 - 2024
- Forecast Period: 2026 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
U.S. Peripheral Vascular Devices And Accessories Market Summary
The U.S. peripheral vascular devices and accessories market size was estimated at USD 4.1 billion in 2025 and is projected to reach USD 6.2 billion by 2033, growing at a CAGR of 5.1% from 2026 to 2033. The industry is driven by the rising prevalence of peripheral arterial disease (PAD), fueled by aging and lifestyle factors.
Key Market Trends & Insights
- By product, the peripheral stents segment led the market with the largest revenue share of 22.4% in 2025.
- By indication, the PAD accounted for the largest market revenue share in 2025.
- By end use, the hospital segment accounted for the largest market revenue share in 2025.
Market Size & Forecast
- 2025 Market Size: USD 4.1 Billion
- 2033 Projected Market Size: USD 6.2 Billion
- CAGR (2026-2033): 5.1%
Technological advancements, such as minimally invasive procedures and innovative devices, are enhancing treatment effectiveness. In addition, the growing geriatric population, more prone to vascular conditions, is increasing the demand for vascular interventions and devices.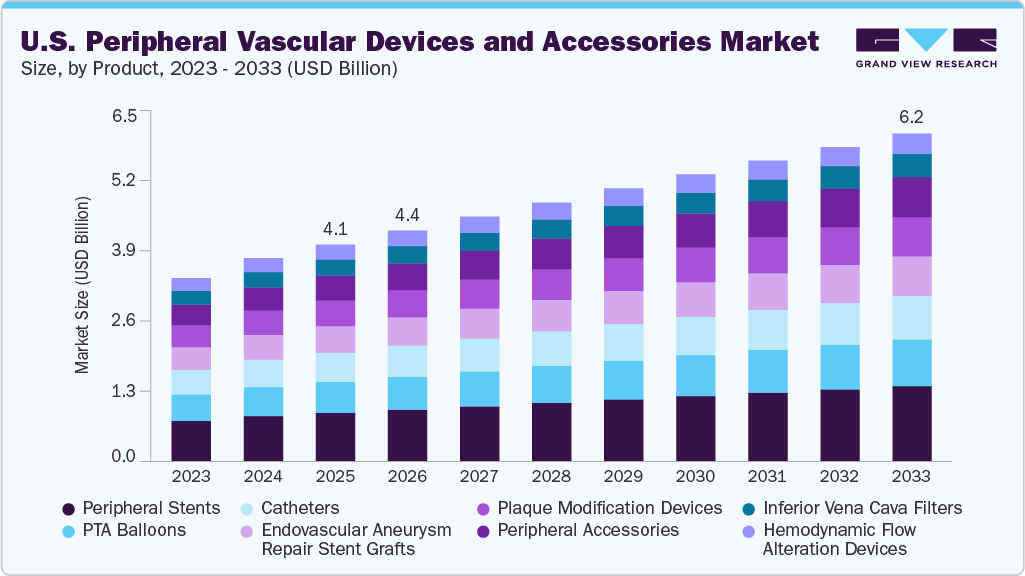
The market is undergoing a significant transformation driven by rising clinical demand, technological advancements, and evolving care models. The shift toward minimally invasive procedures are also driving the market growth.
Market Drivers
Rising Prevalence & Increasing Diagnosis of Peripheral Artery Disease (PAD)
The growing burden of peripheral artery disease (PAD) is a key driver of the market. PAD affects blood circulation in the lower extremities due to arterial narrowing caused by plaque buildup. According to a CDC article published in July 2025, approximately 6.5 million individuals in the U.S. were living with PAD in 2024, highlighting the significant patient population requiring diagnosis and treatment. While the disease progresses, many patients require interventional procedures such as angioplasty, stenting, and atherectomy, which rely on specialized peripheral vascular devices and accessories. The increasing prevalence of PAD, therefore, directly contributes to the rising demand for vascular intervention technologies across hospitals and cardiovascular centers in the U.S.
Growing Burden of Diabetes and Diabetes Related PAD
The increasing prevalence of diabetes in the U.S. is a key market driver, as it raises the risk of vascular complications, particularly in the lower limbs. Persistent high blood sugar damages blood vessels and accelerates atherosclerosis, leading to reduced circulation and peripheral artery disease (PAD). While the condition worsens, patients often require diagnostic imaging and minimally invasive procedures such as angioplasty, thrombectomy, and stenting to restore blood flow.
According to CDC article published in March 2026, approximately 40.1 million people of all ages in the U.S. were living with diabetes in 2023, representing around 12.0% of the total population. Among adults aged 18 years and older, about 39.8 million individuals (14.9% of U.S. adults) had diabetes, including 11.0 million adults with undiagnosed diabetes, which accounts for 27.6% of all diabetes cases. Moreover, diabetes prevalence rises steadily with age, affecting nearly three in ten adults aged 65 and above. The presence of a large undiagnosed population indicates that many individuals remain at risk of developing severe vascular complications before receiving appropriate medical intervention.
Technologically Advanced Product Launches in Peripheral Vascular Devices
Technological innovation and the launch of advanced endovascular devices are significantly driving the market growth. The introduction of advanced thrombectomy technology for vascular blockages. In February 2026, Avantec Vascular, a Nipro company, received U.S. FDA 510(k) clearance for its thrombectomy system designed to remove fresh emboli and thrombi from peripheral venous vessels measuring 7 mm or larger in diameter. This device supports minimally invasive treatment of conditions such as deep vein thrombosis and other peripheral vascular obstructions.
The integration of artificial intelligence (AI)-driven predictive analytics in peripheral endovascular procedures presents a strong growth opportunity for the market by enabling clinicians to forecast patient outcomes and potential complications such as major limb events or mortality. These advanced models support better clinical decision-making by helping physicians customize treatment plans, select appropriate devices, and refine procedural planning, particularly for high-risk patients. This enhances both clinical efficiency and resource utilization. According to the NCBI article published in March 2024, using data from over 235,000 U.S. patients in the Vascular Quality Initiative (VQI) registry demonstrated that a machine learning model (XGBoost) could accurately predict one-year adverse limb events or death, achieving a high AUROC of 0.94 and outperforming traditional statistical methods.
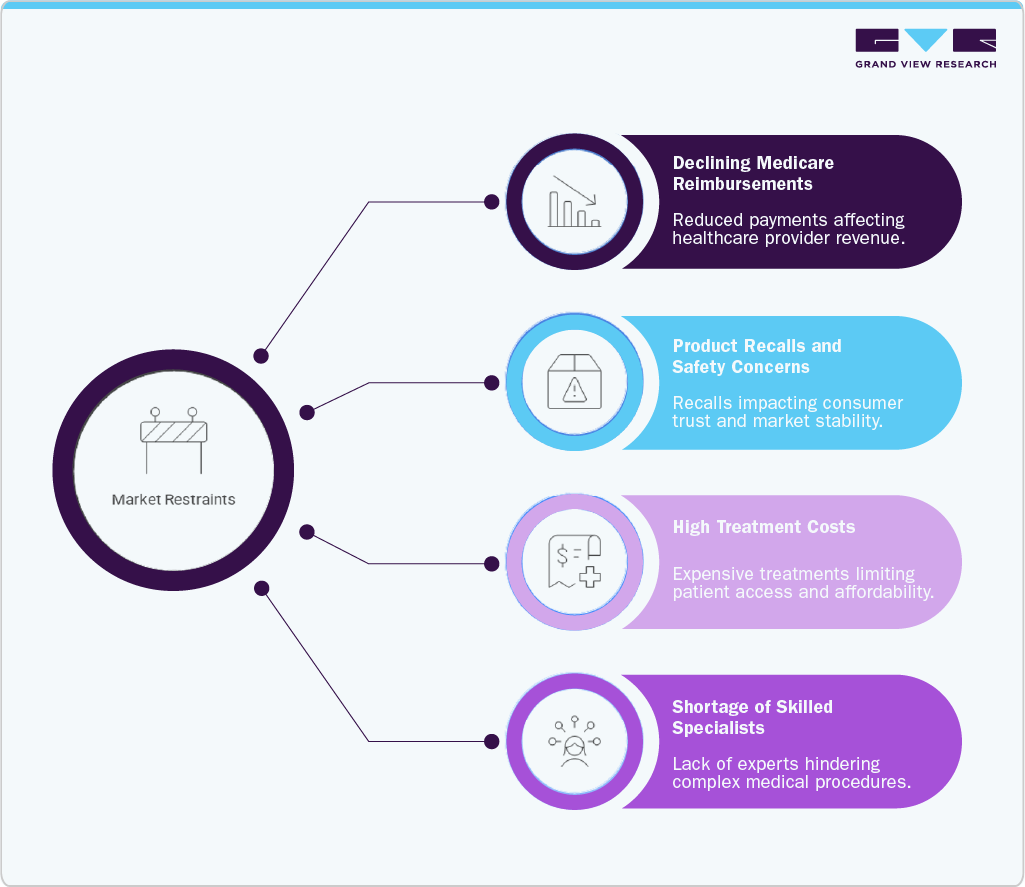
Market Restraints And Challenges
The U.S. peripheral vascular devices industry faces several key challenges, including declining reimbursement rates for procedures, which discourage the adoption of advanced and often higher-cost interventional devices among healthcare providers. In addition, product recalls and safety concerns related to devices such as stents, catheters, guidewires, and stent grafts pose risks in complex procedures where reliability is critical. High treatment costs for conditions like peripheral arterial disease (PAD) and aneurysms further limit accessibility, as these require advanced technologies, specialized infrastructure, and ongoing clinical monitoring. Moreover, a shortage of skilled specialists trained in complex endovascular interventions continues to restrict procedural volumes and the adoption of innovative treatment approaches.
Market Opportunity Analysis
Growing Office-Based Laboratories for Peripheral Vascular Interventions
The rapid growth of office-based laboratories (OBLs) and outpatient vascular treatment centers in the U.S. represents a key opportunity for the market. Traditionally, many peripheral vascular interventions, such as angioplasty, atherectomy, and stent placement, were performed in hospital catheterization laboratories. However, advancements in minimally invasive endovascular technologies, improved catheter systems, and compact imaging equipment have enabled physicians to safely perform many of these procedures in office-based or outpatient settings. This transition is creating new demand for peripheral vascular devices and accessories that are optimized for outpatient workflows, including low-profile catheters, guidewires, vascular access devices, and atherectomy systems.
AI‑Based Predictive Modeling to Optimize Endovascular Intervention Outcomes in PAD
The use of artificial intelligence (AI)based predictive analytics to forecast patient outcomes after peripheral endovascular procedures offers a meaningful growth opportunity for the U.S. peripheral vascular devices industry. Predictive models that accurately anticipate major adverse eventssuch as limb events or deathafter interventions identical angioplasty, stenting, atherectomy, or thrombectomy, help clinicians tailor treatment strategies, select appropriate devices, and optimize procedural planning. These tools support peripheral vascular specialists in identifying high-risk patients who benefit most from specific therapeutic devices (For instance, guidewires, balloons, stents, or plaque modification tools) and procedural accessories, thereby enhancing clinical efficiency and resource utilization.
Market Concentration & Characteristics
The degree of innovation in the U.S. peripheral vascular devices industry is high, driven by continuous advancements in medical technology. Companies focus on developing minimally invasive procedures, improved stent designs, and drug-eluting devices to enhance patient outcomes. In November 2024, Koninklijke Philips N.V. revealed the launch of the U.S. THOR IDE clinical trial, marking the enrollment of its first patient. The trial is focused on evaluating a groundbreaking catheter that combines laser atherectomy with intravascular lithotripsy to treat peripheral artery disease (PAD).
The level of mergers and acquisitions (M&A) activities in the U.S. peripheral vascular devices industry is moderate. Companies often pursue acquisitions to expand their product portfolios, integrate new technologies, and enter new markets. In March 2026, Medtronic announced an agreement to acquire Scientia Vascular for USD 550 million to strengthen vascular access and intervention capabilities. Scientia’s portfolio includes advanced guidewires, microcatheters, and access products, enhancing Medtronic’s reach in neurovascular and vascular procedural workflows, a strategic bolt-on that complements existing systems used in peripheral and neurovascular interventions.
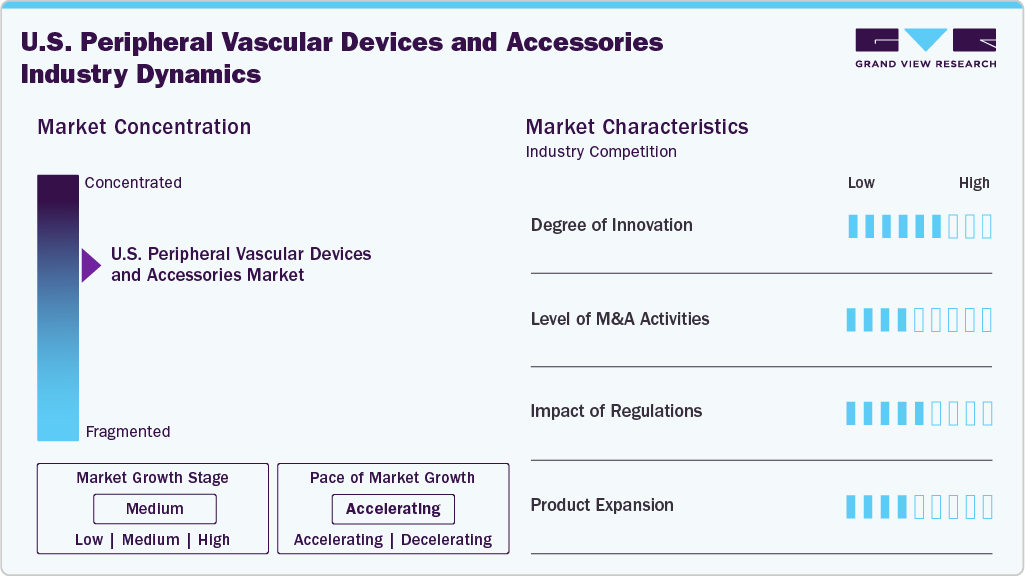
The impact of regulations on the U.S. peripheral vascular devices industry is significant, as the industry is subject to strict oversight by agencies such as the FDA. Regulatory approval processes for new devices, clinical trials, and ongoing post-market surveillance ensure that only safe and effective products reach the U.S. peripheral vascular devices industry. While regulations are essential for patient safety, they also lengthen the time to market and can increase costs for companies, particularly in terms of compliance and approvals.
The product and service expansion in the U.S. peripheral vascular devices industry is moderate, as companies are continually improving existing devices and introducing new products to meet evolving patient needs. Expansions typically focus on offering more advanced, tailored solutions such as drug-coated balloons, stents, and endovenous laser treatment systems. In addition, many companies are enhancing services by offering comprehensive vascular intervention solutions that include diagnostics, devices, and post-procedure care.
Product Insights
The peripheral stents segment led the market with the largest revenue share of 22.4% in 2025 and is anticipated to grow at the fastest CAGR over the forecast period. This high share is attributable to technological advancements in stent technology and the increasing demand for minimally invasive procedures. The peripheral stent segment is primarily driven by a growing patient pool requiring immediate stent placement, technological innovations in peripheral vascular interventions, and an increasing number of government initiatives. In April 2024, Abbott announced FDA approval of the Esprit BTK Everolimus Eluting Resorbable Scaffold System, a significant advancement for patients with chronic limb-threatening ischemia below the knee. This innovative dissolvable stent releases Everolimus to promote healing before dissolving over three years. It aims to improve upon traditional balloon angioplasty, which often leads to recurring blockages. The Esprit BTK System offers a minimally invasive approach to maintaining open arteries.
The PTA balloons segment is expected to grow at a significant CAGR during the forecast period. Increasing CVD incidence and the technologically advanced product launch drive the growth of the market. For instance, in June 2024, the Sugacoated PTA Balloon Dilatation catheter, created by Kossel Medtech, received approval from the National Medical Products Administration (NMPA) for commercial use. This device, designed the expand narrowed peripheral vessels, features a special Niti wire constraint to ensure even expansion, facilitating plaque restructuring, decreasing dissection risk, and reducing the need for additional stent placement and overall healthcare costs.
Application Insights
The peripheral arterial disease (PAD) segment led the market with the largest revenue share of 47.2% in 2025, driven by the rising prevalence of diabetes in the U.S., as it increases the risk of lower-limb vascular complications. Prolonged high blood sugar damages blood vessels and accelerates atherosclerosis, leading to poor circulation and peripheral artery disease (PAD). While the disease progresses, patients often require diagnostic imaging and minimally invasive treatments such as angioplasty, thrombectomy, and stent placement to restore blood flow. According to a CDC article published in March 2026, approximately 40.1 million people of all ages in the U.S. were living with diabetes in 2023, representing around 12.0% of the total population. Among adults aged 18 years and older, about 39.8 million individuals (14.9% of U.S. adults) had diabetes, including 11.0 million adults with undiagnosed diabetes, which accounts for 27.6% of all diabetes cases. Moreover, diabetes prevalence rises steadily with age, affecting nearly three in ten adults aged 65 and above.
The aneurysms segment is expected to grow at the fastest CAGR during the forecast period. Aneurysms, particularly abdominal aortic aneurysms (AAA) and peripheral aneurysms, also significantly drive the demand for peripheral vascular devices. Aneurysms are often treated surgically, such as with endovascular aneurysm repair (EVAR), which requires specialized devices, including stent grafts and catheters. In May 2024, the CDC reported that abdominal aortic aneurysms are more prevalent in men and individuals aged 65 and older. In addition, they occur more frequently in White individuals compared to Black individuals. On the other hand, thoracic aortic aneurysms, which occur in the chest, affect both men and women equally, and their incidence increases with age.
End Use Insights
The hospital segment led the market with the largest revenue share of 41.0% in 2025. Hospitals are increasingly investing in catheterization labs, hybrid operating rooms, and specialized vascular units to manage the growing volume of complex cases. These upgrades enable more precise and efficient endovascular procedures such as angioplasty, stenting, and atherectomy, while improving patient safety and procedural outcomes, ultimately driving higher adoption of peripheral vascular devices and accessories. For instance, in May 2025, UC Davis Medical Center became the first hospital in the Sacramento region to perform a percutaneous transmural arterial bypass (PTAB) procedure using the newly approved DETOUR System, offering patients with complex PAD an effective minimally invasive alternative to open bypass surgery.
“This next-generation device allows us to provide an alternative treatment option with comparable outcomes to surgical bypass while avoiding potential bypass complications.”
-Melissa R. Keller, assistant professor of vascular surgery, UC Davis Health.
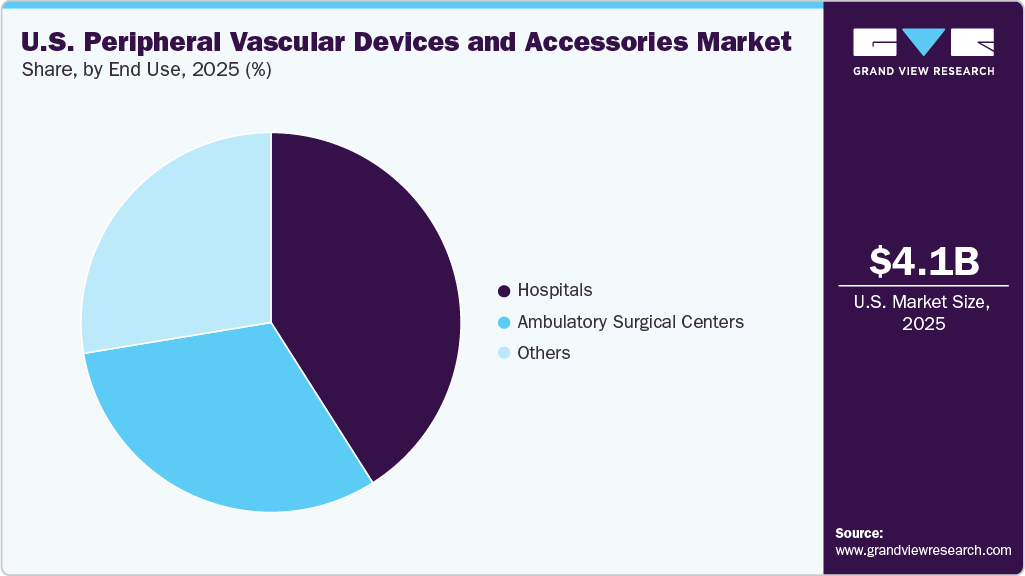
The ambulatory surgical centers segment is expected to grow at the fastest CAGR during the forecast period, driven by the shift toward minimally invasive therapies, cost containment, and patient preference for shorter hospital stays. ASCs facilitate efficient patient flow while maintaining high standards of procedural safety and recovery. In February 2026, an article published in Ambulatory Surgery Center News highlighted that select ambulatory surgery centers (ASCs) performing percutaneous coronary interventions reported complication rates comparable to hospitals at about 1%, while achieving cost savings of roughly USD 4,000 per procedure, emphasizing the potential for safe and more efficient cardiac care in the ASC setting.
“At the national level, most states allow the performance of PCIs in ASCs with approximately 20 of these states reporting detailed data which indicate that the complication rate in ASCs essentially matches those performed in hospitals at just [1%].”
-New York State Public Health and Health Planning Council
Key U.S. Peripheral Vascular Devices And Accessories Company Insights
Some of the key players operating in the U.S. peripheral vascular device and accessories industry include Boston Scientific Corporation, Abbott, Koninklijke Philips N.V., Medtronic, BIOTRONIK SE & Co. KG, and BD. These companies are making significant infrastructure investments, enabling them to develop, manufacture, and commercialize many devices in the country.
In addition, companies enter into several strategic partnerships with distributors and other companies to expand their presence.
Key U.S. Peripheral Vascular Devices And Accessories Companies:
- Abbott
- Terumo Corporation
- Nipro
- Medtronic
- Boston Scientific Corporation
- Cordis
- Koninklijke Philips N.V.,
- AngioDynamics.
- ASAHI INTECC CO., LTD.
- BD
- Teleflex Incorporated. (Biotronik)
- Edward Lifesciences Corporation
- B. Braun SE
- W. L. Gore & Associates, Inc.
- Merit Medical Systems, Inc.
- Cook
Recent Developments
-
In February 2026, Nipro’s U.S.-based subsidiary Avantec Vascular (a Nipro company) received U.S. FDA 510(k) clearance for a new thrombectomy system designed to treat peripheral venous thrombus (For instance DVT). The device combines rotational and aspiration mechanisms to improve clot removal efficiency and reduce procedure time, strengthening Nipro’s position in minimally invasive peripheral vascular interventions.
-
In February 2026, AngioDynamics partnered with the PERT Consortium to launch the ALPHA-PE Research Fund, supporting physician-led research in pulmonary embolism and vascular thrombectomy care.
-
In November 2025, AngioDynamics received FDA IDE approval for the APEX-Return study, evaluating this system in combination with AlphaVac for vascular procedures.
-
In August 2025, Abbott revealed a key expansion by building a new cardiovascular device manufacturing facility in Georgia (U.S.), expected to be completed by 2028. This investment supports growing demand across vascular and structural heart devices and strengthens domestic production capacity.
U.S. Peripheral Vascular Devices And Accessories Market Report Scope
Report Attribute
Details
Market size value in 2026
USD 4.4 billion
Revenue forecast in 2033
USD 6.2 billion
Growth rate
CAGR of 5.1% from 2026 to 2033
Base year for estimation
2025
Historical data
2021 - 2024
Forecast period
2026 - 2033
Quantitative units
Revenue in USD million/billion, Unit Volume, In Thousand Units, and CAGR from 2026 to 2033
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Product, application, end use
Country scope
U.S.
Key companies profiled
Abbott; Terumo Corporation; Nipro; Medtronic; Boston Scientific Corporation; Cordis; Koninklijke Philips N.V.; AngioDynamics.; ASAHI INTECC CO.; LTD.; BD; Teleflex Incorporated. (Biotronik); Edward Lifesciences Corporation; B. Braun SE; W. L. Gore & Associates, Inc.; Merit Medical Systems, Inc.; Cook.
Customization scope
Free report customization (equivalent up to 8 analyst’s working days) with purchase. Addition or alteration to country, regional & segment scope.
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
U.S. Peripheral Vascular Devices And Accessories Market Report Segmentation
This report forecasts revenue growth at the country level and provides an analysis of the latest industry trends in each of the sub-segments from 2021 to 2033. For this study, Grand View Research has segmented the U.S. peripheral vascular devices and accessories market report based on product, application, and end use:
-
Product Outlook (Revenue, USD Million, Unit Volume, In Thousand Units, 2021 - 2033)
-
Peripheral Stents
-
Iliac Artery Stents
-
Femoral Artery Stents
-
Carotid Artery Stents
-
Renal Artery Stents
-
Others
-
-
PTA Balloons
-
Catheters
-
Angiography Catheters
-
Guiding Catheters
-
IVUS/OCT Catheters
-
-
Endovascular Aneurysm Repair Stent Grafts
-
Thoracic Endovascular Aneurysm Stent Grafts
-
Abdominal Endovascular Aneurysm Stent Grafts
-
-
Plaque Modification Devices
-
Atherectomy Devices
-
Thrombectomy Devices
-
-
Peripheral Accessories
-
Guidewires
-
Workhorse Guidewires
-
Specialty Guidewires
-
Extra Support Guidewires
-
Frontline Finesse Guidewires
-
-
Peripheral Vascular Closure Devices
-
Balloon Inflation Devices
-
Introducer Sheaths
-
-
Inferior Vena Cava Filters
-
Permanent Filters
-
Retrievable Filters
-
-
Hemodynamic Flow Alteration Devices
-
Chronic Total Occlusion Devices
-
Embolic Protection Devices
-
-
-
Application Outlook (Revenue, USD Million, Unit Volume, In Thousand Units, 2021 - 2033)
-
Peripheral Arterial Disease (PAD)
-
Aneurysms
-
Venous Diseases
-
Others
-
-
End Use Outlook (Revenue, USD Million, Unit Volume, In Thousand Units, 2021 - 2033)
-
Hospitals
-
Ambulatory Surgical Centers
-
Others
-
Frequently Asked Questions About This Report
b. Some key players operating in the U.S. peripheral vascular devices & accessories market include Abbott; Terumo Medical Corporation; Medtronic; Boston Scientific Corporation; Cordis; Koninklijke Philips N.V.; AngioDynamics, Inc.; Asahi Intecc Co. Ltd.; BD; BIOTRONIK SE & Co. KG; Biosensors International Group Ltd.; B. Braun SE; iVascular; Merit Medical Systems, Inc.; Cook Group Inc.
b. Key factors driving the U.S. peripheral vascular devices & accessories market growth include higher adoption rates of minimally invasive surgeries coupled with the presence of high patient awareness and healthcare expenditure levels.
b. The U.S. peripheral vascular devices & accessories market size was estimated at USD 4.1 billion in 2025 and is expected to reach USD 4.4 billion in 2026.
b. The U.S. peripheral vascular devices & accessories market is expected to grow at a compound annual growth rate of 5.1% from 2026 to 2033 to reach USD 6.2 billion by 2033.
b. The peripheral stents segment, categorized by product, held the largest market share of 22.4% in 2025 and is anticipated to grow at the fastest CAGR over the forecast period. This is attributable to the growing patient pool requiring immediate stent placement, technological innovations in peripheral vascular interventions, and growing government initiatives.
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










