- Home
- »
- Medical Devices
- »
-
Africa Pharmaceutical Regulatory Affairs Market Report, 2033GVR Report cover
![Africa Pharmaceutical Regulatory Affairs Market Size, Share & Trends Report]()
Africa Pharmaceutical Regulatory Affairs Market (2025 - 2033) Size, Share & Trends Analysis Report By Service By Category, By Development Stage, By Indication, By Service Provider, By Company Size, By Country, And Segment Forecasts
- Report ID: GVR-4-68040-121-3
- Number of Report Pages: 150
- Format: PDF
- Historical Range: 2021 - 2024
- Forecast Period: 2025 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
Market Size & Trends
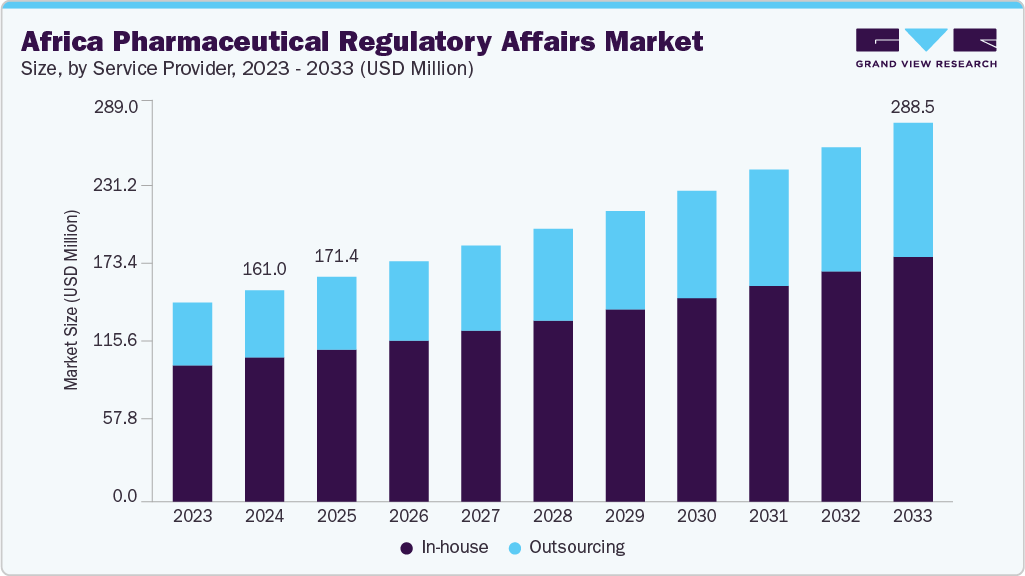
The Africa pharmaceutical regulatory affairs market size was estimated at USD 161.01 million in 2024 and is projected to reach USD 288.53 million by 2033, growing at a CAGR of 6.73% from 2025 to 2033. The market is driven by a rising elderly population and the increasing burden of chronic diseases, alongside growing healthcare expenditures and heightened demand for innovative, affordable medicines across the region. Furthermore, there is a significant shift toward strengthening local pharmaceutical manufacturing capabilities, which is drawing greater regulatory attention and driving the need for robust compliance frameworks.
The market growth is due to a significant transformation in the region’s healthcare landscape. The rise in non-communicable diseases such as diabetes, cancer, and cardiovascular conditions is pushing national health systems to adopt tighter regulatory frameworks that ensure the availability of safe and effective treatments. Moreover, expanding health coverage through national insurance schemes and global health partnerships is prompting governments to prioritize streamlined drug approvals and pharmacovigilance systems. Regulatory bodies are also under pressure to fast-track access to essential medicines, especially in underserved rural areas. This trend has heightened the demand for outsourcing firms with the expertise to navigate evolving regulatory policies, align with harmonized standards, and manage complex approval pathways across multiple countries.

Furthermore, the African governments and regional blocs are increasingly emphasizing the development of local pharmaceutical manufacturing to reduce dependency on imports and improve medicine security. This industrial shift is creating a greater need for well-defined regulatory systems, from GMP certification to dossier submissions and lifecycle management of products. Efforts led by entities such as the African Medicines Agency (AMA), regional harmonization initiatives, and cross-border cooperation are pushing countries to modernize and align their regulatory affairs processes. This transformation is not only fostering innovation but also opening opportunities for contract regulatory services, consultancy, and training programs focused on building local capacity in regulatory compliance and policy execution.
Pricing Analysis
The Africa pharmaceutical regulatory affairs market is characterized by diverse pricing models tailored to the unique needs of local and multinational clients. Fixed-fee or project-based pricing is widely used for well-defined services such as product registration and regulatory submissions, offering budget predictability to emerging local manufacturers. For projects with variable scope, such as post-approval modifications or multi-country filings, time and materials (T&M) models are preferred, allowing flexible billing based on actual resource use.
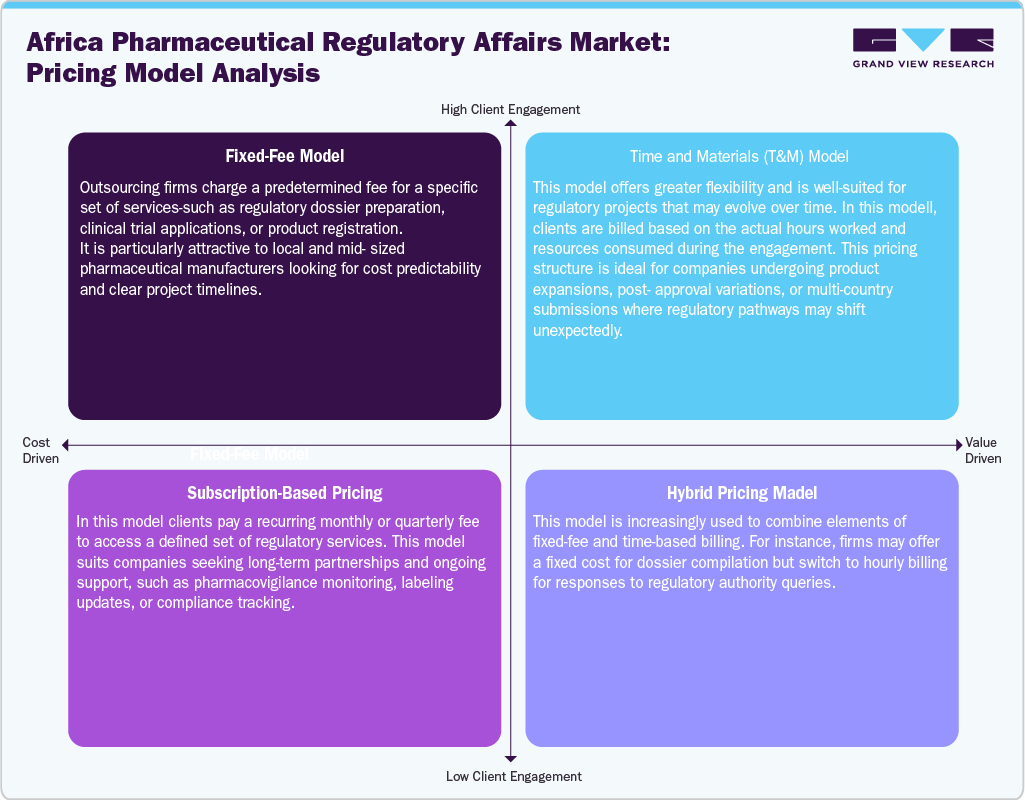
Subscription-based or retainer models are gaining traction among firms seeking continuous regulatory support, particularly for pharmacovigilance, compliance tracking, and lifecycle management. Hybrid pricing approaches are also emerging, combining fixed fees for core tasks with variable charges for unforeseen regulatory responses or country-specific adaptations. This evolving pricing landscape reflects the region’s growing demand for scalable, transparent, and adaptable regulatory outsourcing solutions.
Service Provider Insights
This segment is classified into in-house and outsourcing. The in-house segment accounted for the largest revenue share of 68.19% in the Africa pharmaceutical regulatory affairs market in 2024. The segment’s growth is due to the preference of established pharmaceutical companies to maintain greater control over regulatory compliance, intellectual property, and submission timelines. Many local and regional manufacturers continue to rely on internal regulatory teams to manage product registration, labeling, and post-market surveillance, particularly in countries where regulatory processes are still developing or vary significantly by jurisdiction.
The outsourcing segment is anticipated to grow at the fastest CAGR during the forecast period. The segmental growth is driven due to a rising need for specialized regulatory expertise and cost efficiency among pharmaceutical companies across Africa. As regulatory frameworks across the continent become more stringent and harmonized, many small to mid-sized firms lack the internal capacity to navigate complex submission processes, multi-country compliance requirements, and evolving pharmacovigilance standards.
Services Insights
This segment is segregated into regulatory consulting, legal representation, regulatory writing & publishing, product registration & clinical trial applications, and other services. The regulatory writing & publishing segment held the largest revenue share in 2024, driven by the increasing volume of drug submissions, the growing complexity of regulatory documentation, and the need for high-quality, compliant submissions aligned with evolving regional and international guidelines. As pharmaceutical companies expand their product pipelines and seek approvals across multiple African countries, the demand for specialized expertise in preparing clinical study reports, Common Technical Documents (CTDs), labeling information, and safety updates has surged.
The outsourcing segment is anticipated to grow at the fastest CAGR during the forecast period. The segment growth is driven due to a rising need for specialized regulatory expertise and cost efficiency among pharmaceutical companies across Africa. As regulatory frameworks across the continent become more stringent and harmonized, many small to mid-sized firms lack the internal capacity to navigate complex submission processes, multi-country compliance requirements, and evolving pharmacovigilance standards.
Category Insights
This segment is segregated into drugs and biologics. The drugs segment held the largest revenue share in 2024, driven by the high volume of generic and branded small-molecule drugs being developed, registered, and marketed across the continent. Many African countries continue to prioritize access to essential medicines, driving substantial regulatory activity related to drug approvals, labeling compliance, and pharmacovigilance.
The biologics segment is anticipated to grow at the fastest CAGR during the forecast period. The segment growth is driven due to the rising demand for advanced therapies such as vaccines, monoclonal antibodies, and biosimilars across African countries, particularly in response to infectious diseases, cancer, and autoimmune conditions. Increased investment in healthcare infrastructure and a growing focus on immunization programs have accelerated interest in biologic products.
Indication Insights
This segment is segregated into oncology, neurology, cardiology, immunology, and others. The oncology segment held the largest revenue share in 2024 due to the rising prevalence of cancer across the continent, driven by factors such as aging populations, lifestyle changes, and increased urbanization. The growing burden of various cancers, including breast, cervical, and prostate cancer, has intensified demand for innovative therapies and targeted treatments. This surge has led pharmaceutical companies to accelerate clinical trials, product registrations, and post-market surveillance in oncology, necessitating comprehensive regulatory support.
The immunology segment is anticipated to grow at the fastest CAGR during the forecast period. The segment growth is driven due to the increasing prevalence of autoimmune and inflammatory diseases across Africa, such as rheumatoid arthritis, psoriasis, and multiple sclerosis. Advances in biologic therapies and targeted immunomodulatory treatments are expanding treatment options, creating a higher demand for regulatory approvals and ongoing compliance management.
Development Stage Insights
This segment is segregated into preclinical, clinical studies, and PMA. The clinical studies segment held the largest revenue share in 2024, due to the increasing number of clinical trials being conducted across Africa, driven by a growing focus on developing new drugs and therapies tailored to the continent’s unique disease burden. Expanding investments in clinical research infrastructure, coupled with partnerships between local institutions and global pharmaceutical companies, have significantly boosted trial activities.
The preclinical segment is anticipated to grow at the fastest CAGR during the forecast period. The segment growth is driven due to the increasing investment in early-stage drug discovery and development activities across Africa, supported by expanding research collaborations between local institutions and global pharmaceutical companies. Enhanced focus on innovation, coupled with improvements in laboratory infrastructure and biotechnology capabilities, is fueling preclinical research.
Company Size Insights
The segment is segregated into small, medium, and large. The medium size segment held the largest revenue share in 2024, due to its balanced capacity to invest in regulatory compliance while still requiring external expertise to manage complex and evolving regulatory landscapes. Medium-sized pharmaceutical firms often face resource constraints that limit their ability to maintain large in-house regulatory teams, making outsourcing an attractive and cost-effective solution.
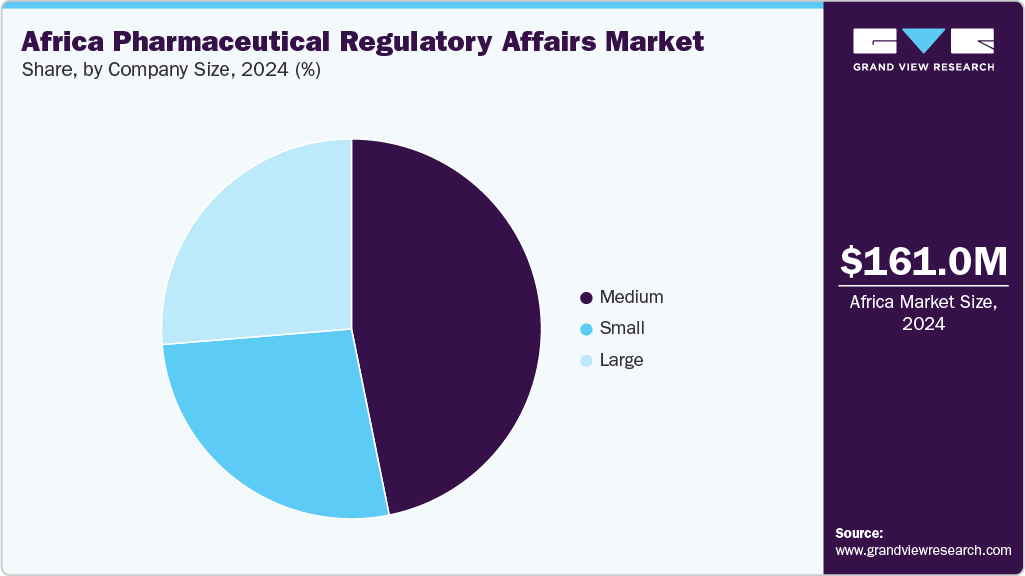
The large segment is anticipated to grow at the fastest CAGR over the forecast period. The growth of the segment is due to the increasing complexity of regulatory requirements faced by large pharmaceutical companies as they expand their operations across multiple African countries. Large firms often manage extensive and diverse product portfolios, including innovative drugs and biologics, which require sophisticated regulatory strategies and compliance management.
Country Insights
The pharmaceutical regulatory affairs market in South Africa held the largest revenue share in 2024. The country’s growth is due to increasing healthcare demands, rising chronic disease prevalence, and a commitment to strengthening local pharmaceutical manufacturing.
Botswana Pharmaceutical Regulatory Affairs Market Trends
The pharmaceutical regulatory affairs market in Botswana is expected to grow significantly due to the government initiatives aimed at strengthening local pharmaceutical manufacturing and reducing dependence on imports. The Botswana Medicines Regulatory Authority (BoMRA) has enhanced the regulatory framework by introducing electronic platforms to streamline drug approvals, improving efficiency and compliance.
Uganda Pharmaceutical Regulatory Affairs Market Trends
The pharmaceutical regulatory affairs market in Uganda held the considerable revenue share in 2024, owing to the country’s robust regulatory framework and strategic initiatives to enhance local pharmaceutical manufacturing. The National Drug Authority (NDA) has been instrumental in promoting domestic production, licensing over 45 local manufacturers and facilitating the registration of 185 conventional and 250 herbal medicines. This support includes tax exemptions on raw materials, Good Manufacturing Practice (GMP) inspections, and the implementation of the Buy Uganda Build Uganda (BUBU) policy, which offers a competitive advantage to locally produced pharmaceuticals.
Key Africa Pharmaceutical Regulatory Affairs Company Insights
Several key players are acquiring various strategic initiatives to strengthen their market position, offering diverse services to customers. The prominent strategies adopted by companies are service launches, mergers & acquisitions/joint ventures, mergers, partnerships & agreements, expansions, and others to increase market presence & revenue and gain a competitive edge drives the market growth.
Key Africa Pharmaceutical Regulatory Affairs Companies:
- Freyr
- IQVIA Inc.
- ICON plc
- WuXi AppTec
- Charles River Laboratories Inc.
- LabCorp
- Parexel International Corporation
- Pharmalex GmbH
- Pharmexon
- Genpact
Recent Developments
-
In June 2024, Pharmexon announced its expansion into the new regions, including South Africa, which aims to strengthen its regulatory affairs and pharmacovigilance services across these areas.
-
In July 2024, IQVIA was named a Leader in the Everest Group's Life Sciences Regulatory and Medical Affairs Operations PEAK Matrix Assessment 2024, highlighting its innovative solutions and global footprint.
Africa Pharmaceutical Regulatory Affairs Market Report Scope
Report Attribute
Details
Market size value in 2025
USD 171.35 million
Revenue forecast in 2033
USD 288.53 million
Growth rate
CAGR of 6.73% from 2025 to 2033
Actual data
2021 - 2024
Forecast period
2025 - 2033
Quantitative units
Revenue in USD million, and CAGR from 2025 to 2033
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Service provider, services, category, indication, development stage, company size, country
Country scope
South Africa; Botswana; Namibia; Algeria; Zimbabwe; Zambia; Tanzania; Rwanda; Ghana; Nigeria; Uganda; Mauritius; Kenya; Rest of Africa
Key companies profiled
Freyr; IQVIA Inc.; ICON plc; WuXi AppTec; Charles River Laboratories; LabCorp; Parexel International Corporation Inc.; Pharmalex GmbH; Pharmexon; Genpact
Customization scope
Free report customization (equivalent up to 8 analysts working days) with purchase. Addition or alteration to country, regional & segment scope.
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
Africa Pharmaceutical Regulatory Affairs Market Report Segmentation
This report forecasts revenue growth at regional and country levels and provides an analysis of the latest industry trends in each of the sub-segments from 2021 to 2033. For this study, Grand View Research has segmented the Africa pharmaceutical regulatory affairs market report based on service providers, services, category, indication, development stage, company size, and country:
-
Service Provider Outlook (Revenue, USD Million, 2021 - 2033)
-
In-house
-
Outsourcing
-
-
Service Outlook (Revenue, USD Million, 2021 - 2033)
-
Regulatory Consulting
-
Legal Representation
-
Regulatory Writing & Publishing
-
Writing
-
Publishing
-
-
Product Registration & Clinical Trial Applications
-
Other Services
-
-
Category Outlook (Revenue, USD Million, 2021 - 2033)
-
Drugs
-
Innovator
-
Preclinical
-
Clinical
-
Post Market Approval (PMA)
-
-
Generics
-
Preclinical
-
Clinical
-
Post Market Approval (PMA)
-
-
-
Biologics
-
Biotech
-
Preclinical
-
Clinical
-
Post Market Approval (PMA)
-
-
ATMP
-
Preclinical
-
Clinical
-
Post Market Approval (PMA)
-
-
Biosimilars
-
Preclinical
-
Clinical
-
Post Market Approval (PMA)
-
-
-
-
Indication Outlook (Revenue, USD Million, 2021 - 2033)
-
Oncology
-
Neurology
-
Cardiology
-
Immunology
-
Others
-
-
Development Stage Outlook (Revenue, USD Million, 2021 - 2033)
-
Preclinical
-
Clinical
-
Post Market Approval (PMA)
-
-
Company Size Outlook (Revenue, USD Million, 2021 - 2033)
-
Small
-
Medium
-
Large
-
-
Country Outlook (Revenue, USD Million, 2021 - 2033)
-
South Africa
-
Botswana
-
Namibia
-
Zimbabwe
-
Zambia
-
Tanzania
-
Rwanda
-
Ghana
-
Nigeria
-
Uganda
-
Mauritius
-
Kenya
-
Frequently Asked Questions About This Report
b. The Africa pharmaceutical regulatory affairs market size was estimated at USD 161.01 million in 2024 and is expected to reach USD 171.35 million in 2024
b. The Africa pharmaceutical regulatory affairs market is expected to grow at a compound annual growth rate of 6.73% from 2025 to 2033 to reach USD 288.53 million by 2033.
b. South Africa dominated the Africa pharmaceutical regulatory affairs market with a share of 57.70% in 2024. The government of South Africa has shown a strong commitment to improving healthcare access and quality. Policies such as the National Health Insurance (NHI) aim to provide universal health coverage, leading to increased demand for pharmaceutical products and regulatory compliance services.
b. Some key players operating in the Africa pharmaceutical regulatory affairs market include Freyr, IQVIA Inc, ICON plc, WuXi AppTec, Charles River Laboratories, Labcorp Drug Development, Parexel International Corporation, Pharmalex GmbH, Pharmexon, Genpact
b. Key factors that are driving the Africa pharmaceutical regulatory affairs market growth include the increasing prevalence of chronic diseases, a growing elderly population, rising healthcare expenditure, and the demand for innovative and cost-effective medicines across the continent.
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










