- Home
- »
- Medical Devices
- »
-
Antibody Drug Conjugates Contract Manufacturing Market, 2030GVR Report cover
![Antibody Drug Conjugates Contract Manufacturing Market Size, Share & Trends Report]()
Antibody Drug Conjugates Contract Manufacturing Market (2025 - 2030) Size, Share & Trends Analysis Report By Linker (Cleavable, Non-cleavable), By Condition (Myeloma, Lymphoma, Breast Cancer), By Region, And Segment Forecasts
- Report ID: GVR-4-68039-623-5
- Number of Report Pages: 100
- Format: PDF
- Historical Range: 2018 - 2023
- Forecast Period: 2025 - 2030
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
Antibody Drug Conjugates Contract Manufacturing Market Summary
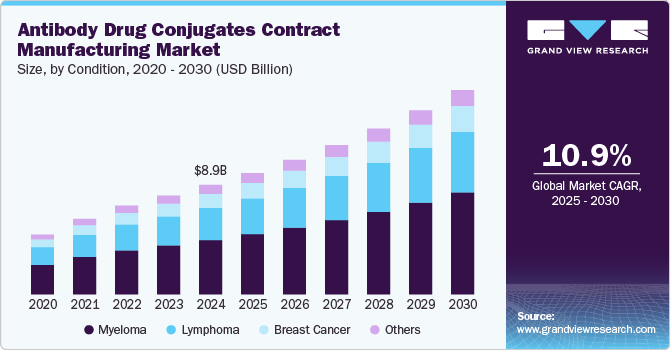
The global antibody drug conjugates contract manufacturing market size was estimated at USD 8,871.4 million in 2024 and is projected to reach USD 16,553.7 million by 2030, growing at a CAGR of 11% from 2025 to 2030. The industry is anticipated to witness significant growth over the forecast period on account of the increasing prevalence of cancer, rising demand for biologic therapy, and the challenges associated with antibody drug conjugate (ADC) manufacturing, which contribute to the demand for contract manufacturing.
Key Market Trends & Insights
- In terms of region, Asia Pacific was the largest revenue generating market in 2024.
- Country-wise, China is expected to register the highest CAGR from 2025 to 2030.
- In terms of segment, myeloma accounted for a revenue of USD 4,879.7 million in 2024.
- Lymphoma is the most lucrative condition segment registering the fastest growth during the forecast period.
Market Size & Forecast
- 2024 Market Size: USD 8,871.4 million
- 2030 Projected Market Size: USD 16,553.7 million
- CAGR (2025-2030): 11%
- Asia Pacific: Largest market in 2024
Furthermore, the growing research activities for ADC, owing to its high potential in treating cancer, are further expected to support the demand for its manufacturing in the forecasted period and, thereby is expected to support the market growth.
The rising cancer disease burden is a key driver in the antibody drug conjugate (ADC) contract manufacturing industry, as the increasing prevalence of cancer boosts demand for targeted therapies. ADCs offer high specificity and reduce systemic toxicity, making them a preferred alternative in oncology treatment. Factors such as geriatric populations, lifestyle changes, and environmental changes contribute to the growing cancer incidence, prompting pharmaceutical companies to accelerate antibody drug conjugate (ADC) development. Cancer Research UK states that over half of all cancers are diagnosed in people over the age of 70. The aging population is increasing globally. As per WHO, people aged 60 and above accounted for 1 billion in 2020, and this number is expected to rise to 2.1 billion by 2050. A rise in the geriatric population is expected to increase cancer incidence, thus contributing to the demand for ADC therapeutics, hence supporting market growth. According to WHO, high consumption of tobacco & alcohol, low fruit & vegetables intake, high body mass index, and lack of physical activity are some of the major causes of cancer. A large number of people are living sedentary lifestyles; this is expected to drive the number of cancer cases, which is likely to have a positive impact on ADC contract manufacturing industry growth.
The high cost of manufacturing antibody-drug conjugates (ADCs) is a significant driving factor in the demand for contract manufacturing in the ADC industry. The complex and resource-intensive production processes involved in ADC development, including the synthesis of highly specialized drugs and the conjugation process, require specialized equipment, skilled personnel, and stringent quality control measures. These challenges make in-house manufacturing expensive for many biopharmaceutical companies, particularly smaller players or those lacking the necessary infrastructure. As a result, many companies are increasingly turning to contract manufacturing organizations (CMOs) to reduce costs, streamline production, and leverage the expertise and scalability offered by established CMO facilities, thereby accelerating time-to-market and improving cost efficiency.
The increasing R&D activities in Antibody-Drug Conjugates (ADCs) are another driver in the ADC contract manufacturing market, as pharmaceutical companies focus on developing innovative, targeted cancer therapies. These advanced therapies combine monoclonal antibodies with cytotoxic drugs, offering precise drug delivery to tumor cells while minimizing systemic side effects. The growing prominence of ADCs, coupled with significant investments in research and clinical trials, is stimulating demand for specialized contract manufacturing services. This trend is further increased by the need for expertise in complex manufacturing processes, scalability, and compliance with regulatory standards, thereby driving industry growth and expansion.
Continuous technological advancements in antibody-drug conjugates (ADC) manufacturing, enabling enhanced efficacy, stability, and targeted drug delivery. Innovations in linker-payload technologies, site-specific conjugation, and improved bioconjugation techniques are increasing ADC potency while reducing off-target toxicity. Advancements in high-throughput analytics, single-use bioprocessing systems, and automation are streamlining manufacturing, ensuring scalability and regulatory compliance. In addition, the integration of artificial intelligence (AI) and machine learning (ML) in process optimization is improving yield consistency and cost efficiency. These technological improvements are fueling demand for specialized contract manufacturers equipped with state-of-the-art capabilities to support the growing ADC pipeline.
Market Concentration & Characteristics
The market growth stage is medium, with an accelerating pace. The market is characterized by the level of M&A activities, degree of innovation, regulatory impact, product expansions, and regional expansions.
The antibody drug conjugate (ADC) contract manufacturing industry demonstrated a high degree of innovation, driven by advancements in linker technologies, cytotoxic payloads, and bioconjugation methods. Continuous R&D efforts focus on enhancing stability, improving targeted delivery, and reducing off-target toxicity. The integration of AI and automation in bioprocessing is further optimizing manufacturing efficiency and scalability, fostering a competitive and innovation-intensive market landscape.

Regulatory oversight in the ADC contract manufacturing market is high, given the complexity of ADC production, which involves both biologics and cytotoxic small molecules. Stringent guidelines from regulatory bodies such as the FDA, EMA, and ICH to enhance product safety, efficacy, and consistency. Compliance with evolving Good Manufacturing Practices (GMP) and containment standards for handling potent compounds includes operational challenges, requiring significant investment in quality control and regulatory expertise.
The level of M&A activity in the market is moderate as companies seek to expand their technological capabilities, scale, and global footprint. With strategic consolidations aimed at expanding technological capabilities and increasing production capacity. While large CDMOs seek to acquire specialized biotech firms for expertise in high-potency APIs (HPAPIs) and conjugation technologies, the market remains fragmented, with several niche players maintaining independent operations.
The market is witnessing rapid service expansion, with CMOs investing in end-to-end solutions covering ADC development, conjugation, fill-finish, and analytical testing. Companies are enhancing their capabilities in payload-linker synthesis, bioconjugation, and formulation services to cater to rising demand. The momentum toward single-source, integrated solutions is streamlining supply chains, reducing timelines, and improving project efficiencies for biopharmaceutical clients.
The ADC contract manufacturing industry is experiencing moderate regional expansion, with North America and Europe leading in terms of production capacity and regulatory expertise. However, Asia-Pacific is emerging as a key growth region, driven by increasing investments in biopharma infrastructure, lower operational costs, and government incentives for biotech manufacturing. While expansion efforts are ongoing, regulatory harmonization and supply chain complexities pose challenges to rapid global scaling.
Condition Insights
Based on condition, the market includes myeloma, lymphoma, breast cancer and others. The myeloma segment led the market with the largest revenue share of 49.61% in 2024. As a hematologic malignancy, myeloma has spurred demand for innovative and targeted therapies, including ADCs, which offer precise drug delivery with reduced systemic toxicity. The growing adoption of ADC-based treatments, such as those targeting B-cell maturation antigen (BCMA), has driven investment in ADC manufacturing capabilities. In addition, the expanding pipeline of myeloma-focused ADCs in clinical trials has increased outsourcing demand for specialized contract manufacturing organizations (CMOs) with expertise in bioconjugation, high-potency API handling, and scalable production. This trend is further boosted by regulatory support for novel biologics and advancements in linker-payload technologies, requiring ADC contract manufacturing a critical component in addressing the rising burden of myeloma.
The lymphoma segment is anticipated to witness at the fastest CAGR during the forecast period. The segment is driven by growing demand for targeted therapies, including ADCs, which offer enhanced efficacy and reduced toxicity compared to conventional treatments. Pharmaceutical and biotechnology companies are investing heavily in ADC development to address the unmet medical needs of lymphoma patients, driving the need for specialized contract manufacturing services.
Linker Insights
Based on linker, the market is segmented into cleavable linkers and non-cleavable linkers. The cleavable linkers segment led the market with the largest revenue share of 56.1% in 2024. The segment growth is owing to its impact on drug efficacy, stability, and targeted delivery. Cleavable linkers allow precise drug release within cancer cells, enhancing ADC potency while minimizing off-target toxicity. Growing advancements in linker technology, such as acid-sensitive, glutathione-sensitive, and enzymatically cleavable linkers, are driving demand for specialized contract manufacturing services. In addition, the increasing number of ADCs in clinical pipelines and regulatory approvals accelerated the need for reliable manufacturing partners with expertise in scalable, high-purity linker production.
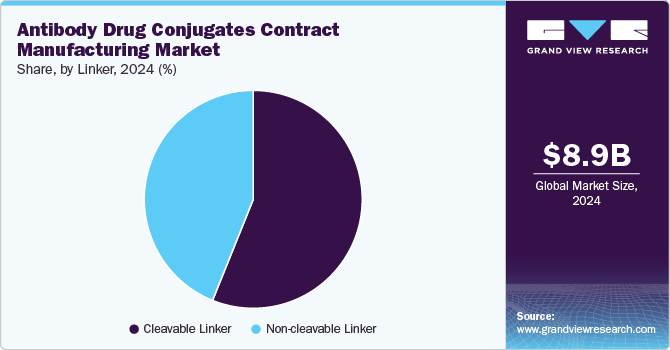
The non-cleavable linkers segment is anticipated to witness at a lucrative CAGR during the forecast period. The growing demand for next-generation ADCs with improved safety profiles and extended half-life led to increased adoption of non-cleavable linkers. In addition, advancements in linker technology, coupled with rising investments in ADC development, are driving contract manufacturers to expand their capabilities in producing high-quality, scalable linker-payload combinations.
Regional Insights
The antibody drug conjugates contract manufacturing market in North America is expected to witness at the fastest CAGR over the forecast period. Leading pharmaceutical companies in the U.S. are increasingly outsourcing ADC production to specialized CDMOs to expedite time-to-market and optimize costs. North America benefits from cutting-edge manufacturing technologies, including site-specific conjugation and high-yield production processes, enabling efficient and scalable ADC manufacturing. The presence of well-established regulatory bodies such as the U.S. FDA make sure a reliable pathway for market entry. In addition, rising collaborations and acquisitions within the industry are driving further growth.
U.S. Antibody Drug Conjugates (ADC) Contract Manufacturing Market Trends
The U.S. antibody drug conjugates contract manufacturing market is anticipated to be driven by the country's leading position in the global biotechnology and pharmaceutical sectors. The increasing adoption of ADCs for targeted cancer therapies, combined with growing demand for cost-effective manufacturing solutions, is prompting U.S.-based pharmaceutical companies to partner with CDMOs. The market benefits from high levels of innovation in bio-manufacturing techniques, such as improved drug-linker technologies and streamlined regulatory approval processes.
The antibody drug conjugates contract manufacturing market in Canada is driven by a growing hub for biopharmaceutical production, driven by strong research capabilities and favorable government initiatives supporting the biotech sector. Canadian companies are increasingly engaged in the development of ADCs for oncology, with a significant focus on expanding contract manufacturing services to meet global demand.
Asia Pacific Antibody Drug Conjugates Contract Manufacturing Market Trends
Asia Pacific antibody drug conjugates contract manufacturing market dominated the market with the largest revenue share of 40.93% in 2024. The regional growth is primarily owing to supportive regulatory reforms, especially in countries such as India & China, and low-cost labor. Furthermore, the presence of cGMP-compliant facilities and established market players in the region is also anticipated to attract more investors & fuel the market. In addition, the ease of conducting business in Asia Pacific is high due to relatively lower manufacturing costs, better tax benefits, and availability of skilled labor at lower costs than in developed economies, which supports the growth of contract manufacturing in Asia Pacific.
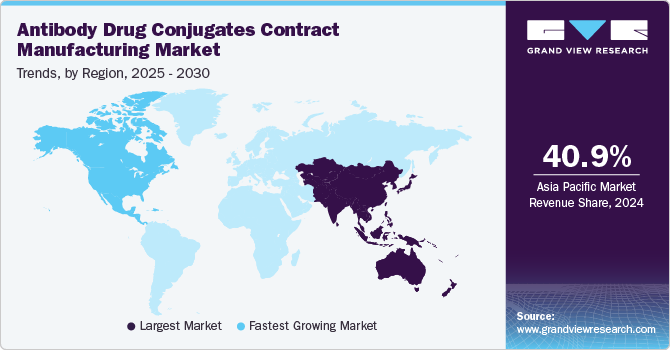
The antibody drug conjugates (ADC) contract manufacturing market in China accounted for the largest market share in the Asia Pacific in 2024, owing to increasing investment in biopharmaceutical innovation, favorable government policies, and rising demand for targeted cancer therapies. Domestic contract manufacturing organizations (CMOs) are expanding capabilities to support end-to-end ADC production, from monoclonal antibody (mAb) development to payload-linker synthesis and conjugation. Advancements in bioconjugation technologies and regulatory streamlining by the National Medical Products Administration (NMPA) are further accelerating market expansion.
The India antibody drug conjugates (ADC) contract manufacturing market is driven by increasing investments in biologics, a rising focus on oncology therapeutics, and government initiatives promoting domestic biopharmaceutical production. The demand for cost-effective and high-quality ADC manufacturing positioned India as an attractive outsourcing hub, benefiting from a well-established pharmaceutical industry and a skilled workforce. Key trends include the expansion of GMP-compliant facilities, strategic collaborations between domestic firms and global biopharma players, and advancements in conjugation technologies to enhance ADC stability and efficacy.
Europe Antibody Drug Conjugates Contract Manufacturing Market Trends:
The antibody drug conjugates (ADC) contract manufacturing market is driven by increasing demand for targeted cancer therapies and advancements in bioconjugation technologies. Pharmaceutical companies are outsourcing ADC production to contract manufacturing organizations (CMOs) to leverage their specialized expertise, state-of-the-art facilities, and regulatory compliance capabilities. The region's strong biopharmaceutical infrastructure, particularly in Germany, Switzerland, and the UK, supports this expansion. In addition, rising investments in ADC development, strategic partnerships, and a growing pipeline of clinical-stage ADCs are stimulating market growth.
The Germany antibody drug conjugates (ADC) contract manufacturing market accounted for the largest market share in Europe in 2024, owing to rising demand for targeted cancer therapies and increasing outsourcing by biopharmaceutical companies. The country’s strong pharmaceutical manufacturing infrastructure, regulatory expertise, and presence of key CMOs make it a hub for ADC production.
The antibody drug conjugates (ADC) contract manufacturing market in UK is expanding due to strong government support for biopharmaceutical innovation and a growing network of specialized CMOs. The country's leadership in advanced bioprocessing and research collaborations with academic institutions enhance its competitiveness in ADC development. Investments in high-containment manufacturing facilities and next-generation conjugation technologies are driving efficiency and scalability.
Key Antibody Drug Conjugates Contract Manufacturing Company Insights
The antibody drug conjugates (ADC) contract manufacturing industry is dominated by several key players that hold substantial market share, including Lonza Group, WuXi AppTec, Catalent, and Samsung Biologics. These companies are recognized for their advanced capabilities in biopharmaceutical manufacturing, offering comprehensive services ranging from early-stage development to large-scale production of ADCs. The key market players are undertaking several strategic initiatives, such as acquisitions, partnerships, expansion, agreements, collaborations, etc., to increase their market presence and gain a competitive edge, driving market growth.
Key Antibody Drug Conjugates Contract Manufacturing Companies:
The following are the leading companies in the antibody drug conjugates contract manufacturing market. These companies collectively hold the largest market share and dictate industry trends.
- Sterling
- Recipharm AB
- Lonza
- Catalent, Inc.
- Sartorius AG
- Wuxi Biologics
- Samsung Biologics
- Piramal Group (Piramal Pharma Solutions)
- AbbVie, Inc. (AbbVie Contract Manufacturing)
- Merck KGaA
Recent Developments
-
In May 2024, Wuxi AppTec, Inc. announced its expansion with the opening of its new R&D and manufacturing facility in Singapore. This new facility provides API R&D and manufacturing services for peptides, small molecules, complex synthetic conjugates, & oligonucleotides.
-
In December 2023, Piramal Pharma Solutions announced the expansion of its ADC manufacturing facility in Grangemouth, Scotland. The expanded capacity increases the company’s ADC production capacity by 70% to 80% and strengthens its ability to serve more customers through the drug development lifecycle.
-
In October 2023, Lonza announced a new cGMP filling line at its site in Lonza’s Stein (CH) to enhance its integrated offering for manufacturing ADCs and drug products.
Antibody Drug Conjugates Contract Manufacturing Market Report Scope
Report Attribute
Details
Market size value in 2025
USD 9.83 billion
Revenue forecast in 2030
USD 16.55 billion
Growth rate
CAGR of 10.99% from 2025 to 2030
Base year for estimation
2024
Historical data
2018 - 2023
Forecast period
2025 - 2030
Quantitative units
Revenue in USD million/billion and CAGR from 2025 to 2030
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Condition, linker, region
Regional scope
North America; Europe; Asia Pacific; Latin America; Middle East & Africa
Country scope
U.S.; Canada; Mexico; UK; Germany; France; Italy; Spain; Sweden; Denmark; Norway; Japan; China; India; Thailand; South Korea; Australia; Brazil; Argentina; South Africa; Saudi Arabia; UAE; Kuwait
Key companies profiled
Sterling; Recipharm AB; Lonza; Catalent, Inc.; Sartorius AG; Wuxi Biologics; Samsung Biologics; Piramal Group (Piramal Pharma Solutions); AbbVie, Inc. (AbbVie Contract Manufacturing); Merck KGaA
Customization scope
Free report customization (equivalent up to 8 analyst’s working days) with purchase. Addition or alteration to country, regional & segment scope.
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
Global Antibody Drug Conjugates Contract Manufacturing Market Report Segmentation
This report forecasts revenue growth at the regional and country levels and provides an analysis of the latest industry trends in each of the sub-segments from 2018 to 2030. For this study, Grand View Research has segmented the global antibody drug conjugates contract manufacturing market report based on the condition, linker, and region:
-
Condition Outlook (Revenue, USD Million, 2018 - 2030)
-
Myeloma
-
Lymphoma
-
Breast Cancer
-
Others
-
-
Linker Outlook (Revenue, USD Million, 2018 - 2030)
-
Cleavable Linker
-
Non-cleavable Linker
-
-
Regional Outlook (Revenue, USD Million, 2018 - 2030)
-
North America
-
U.S.
-
Canada
-
Mexico
-
-
Europe
-
UK
-
Germany
-
France
-
Italy
-
Spain
-
Sweden
-
Denmark
-
Norway
-
-
Asia Pacific
-
Japan
-
China
-
India
-
Thailand
-
South Korea
-
Australia
-
-
Latin America
-
Brazil
-
Argentina
-
-
Middle East & Africa
-
South Africa
-
Saudi Arabia
-
UAE
-
Kuwait
-
-
Frequently Asked Questions About This Report
b. The global antibody drug conjugates (ADC) contract manufacturing market size was estimated at USD 8.87 billion in 2024 and is expected to reach USD 9.83 billion in 2025.
b. The global antibody drug conjugates contract manufacturing market is expected to grow at a compound annual growth rate of 10.99% from 2025 to 2030 to reach USD 16.55 billion by 2030.
b. Asia Pacific dominated the ADC contract manufacturing market with a share of 40.93% in 2024. This is attributable to the availability of cheap labor coupled with cost efficiency, time-saving, and specialized expertise offered by CMOs in the region.
b. Some key players operating in the ADC contract manufacturing market include Sterling; Recipharm AB; Lonza; Catalent, Inc; Sartorius AG; Wuxi Biologics; Samsung BioLogics; Piramal Group (Piramal Pharma Solutions) ; Abbvie, Inc.(Abbvie Contract Manufacturing) and Merck KGaA
b. Key factors that are driving the antibody drug conjugates contract manufacturing market growth include the complex nature of ADCs, increasing demand for manufacturing capacity as well as an increasing number of research on antibody therapies.
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










