- Home
- »
- Medical Devices
- »
-
Antimicrobial Catheters Market Size, Industry Report, 2033GVR Report cover
![Antimicrobial Catheters Market Size, Share & Trends Report]()
Antimicrobial Catheters Market (2026 - 2033) Size, Share & Trends Analysis Report By Product Type (Urological, Vascular Access, Specialty & Surgical), By Technology (Silver-Based Technology, Antibiotic-Impregnated), By Material, By End Use, By Region, And Segment Forecasts
- Report ID: GVR-4-68040-863-9
- Number of Report Pages: 150
- Format: PDF
- Historical Range: 2021 - 2025
- Forecast Period: 2026 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
Antimicrobial Catheters Market Summary
The global antimicrobial catheters market size was estimated at USD 1.43 billion in 2025 and is projected to reach USD 2.86 billion by 2033, growing at a CAGR of 9.0% from 2026 to 2033. The growth of this market is primarily driven by the increasing incidence of hospital-acquired infections, particularly catheter-associated urinary tract infections, which pose significant clinical and economic burdens on healthcare systems worldwide.
Key Market Trends & Insights
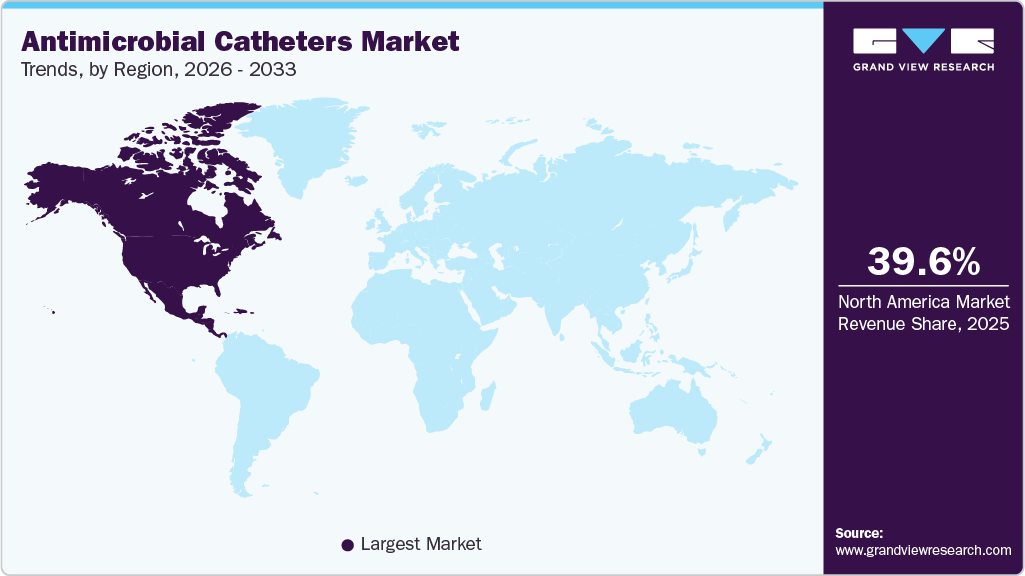
- North America dominated the antimicrobial catheters market with the largest revenue share 39.62% in 2025.
- The antimicrobial catheters market in the U.S. accounted for the largest market revenue share in North America in 2025.
- By product type, the vascular access catheters segment led the market with the largest revenue share in 2025.
- By technology, silver-based technology segment led the market with the largest revenue share in 2025.
- By end use, the hospitals segment led the market with the largest revenue share in 2025.
Market Size & Forecast
- 2025 Market Size: USD 1.43 Billion
- 2033 Projected Market Size: USD 2.86 Billion
- CAGR (2026-2033): 9.0%
- North America: Largest market in 2025
Rising awareness among healthcare providers and patients about infection prevention and the benefits of antimicrobial catheters is further fueling market adoption. In addition, ongoing technological advancements, such as catheters coated with silver alloy, antibiotics, or novel antimicrobial polymers, are enhancing catheter efficacy and reducing infection rates. The expanding geriatric population, who are more prone to urinary complications and prolonged catheter use, also contributes to higher demand.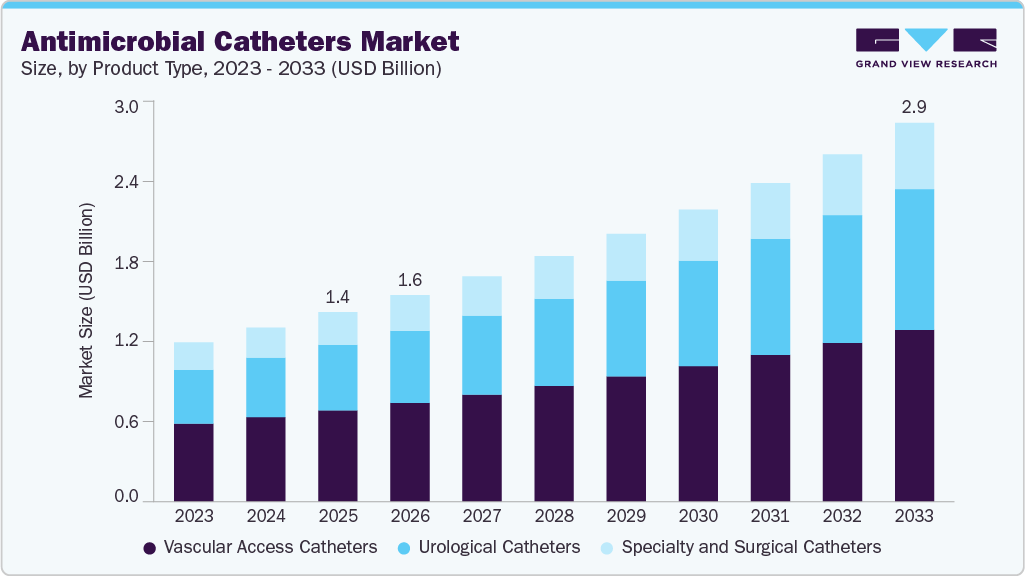
Ventilator-associated events (VAE), catheter-associated urinary tract infections (CAUTIs), and central line-associated bloodstream infections (CLABSI) are key healthcare-associated infections that significantly drive the growth of the antimicrobial catheters market. These infections are common complications in hospitalized and critically ill patients, leading to increased morbidity, extended hospital stays, and higher healthcare costs. Antimicrobial catheters, designed to resist microbial colonization and biofilm formation, reduce the incidence of these infections. The rising prevalence of VAEs, CAUTIs, and CLABSIs has created strong demand for catheter solutions with advanced antimicrobial technologies, as hospitals and healthcare providers prioritize patient safety, infection prevention, and regulatory compliance. Accordingly, the need to minimize these infections fuels market adoption and innovation in antimicrobial catheter products.
“UTIs are increasingly common at home and in hospitals. More serious in older patients, especially those with catheters, but can occur at any age. Not often related to poor hygiene. Managing UTIs through prevention, education, and access to healthcare-including diagnostics and treatment-is vital.”- Professor Matt Inada-Kim, National Clinical Director for Infections Management and AMR at NHS England
U.S. Hospital Infection Statistics - CLABSI, CAUTI, and VAE by State and Location, 2024
2024
Central line-associated bloodstream infections (CLABSI)
Catheter-associated urinary tract infections (CAUTI)
Ventilator-associated events (VAE)
Inpatient Locations
Inpatient Locations
Inpatient Locations
State
Total
ICU
Wards
NICU
Total
ICU
Wards
Total
ICU
Wards
Alabama
545
146
375
24
531
146
385
86
69
17
Alaska
59
10
43
6
56
10
46
9
8
1
Arizona
484
100
362
22
468
100
368
43
36
7
Arkansas
347
65
245
37
325
65
260
37
34
3
California
2,895
566
2,069
260
2,667
566
2,101
405
288
117
Colorado
433
80
318
35
408
80
328
73
59
14
Connecticut
Upgrade report license to gain access to the complete analysis.
D.C.
Delaware
Florida
Georgia
Guam
Hawaii
Idaho
Illinois
Indiana
Iowa
Kansas
Kentucky
Louisiana
Maine
Maryland
Massachusetts
Michigan
Minnesota
Mississippi
Missouri
Montana
Nebraska
Nevada
New Hampshire
New Jersey
New Mexico
New York
North Carolina
North Dakota
Ohio
Oklahoma
Oregon
Pennsylvania
Puerto Rico
Rhode Island
South Carolina
South Dakota
Tennessee
Texas
Utah
Vermont
Virgin Islands
Virginia
Washington
West Virginia
Wisconsin
Wyoming
All US
29,837
5,726
22,182
1,929
28,427
5,721
22,706
4,512
3,066
1,446
Source: CDC, 2026
The expansion of healthcare infrastructure in emerging markets significantly drives the antimicrobial catheters market by increasing access to hospitals, clinics, and specialized care units where catheter use is essential. As countries in Asia, Latin America, and the Middle East invest in modernizing healthcare facilities, there is a growing demand for advanced medical devices that ensure patient safety and reduce hospital-acquired infections. This development leads to higher adoption of antimicrobial catheters, particularly in critical care units, surgical centers, and outpatient settings. In addition, improved healthcare infrastructure often includes enhanced training for medical personnel, improved sterilization practices, and stricter infection control protocols, further boosting the market for devices that prevent catheter-related infections.
Key National Health Initiatives
Initiative / Country
Year / Target
Objective / Focus Area
Impact on Antimicrobial Catheters Market
Healthy China 2030
2030
Improve overall healthcare access, strengthen hospital infrastructure, enhance infection prevention and control
Expansion of hospitals, ICUs, and surgical units increases demand for advanced catheter technologies, including antimicrobial catheters
Saudi Vision 2030 - Health Sector Transformation Program
2030
Transform healthcare system to improve quality, efficiency, and patient safety; digitalization and modernization of hospitals
Modernized healthcare infrastructure and stricter infection control measures drive adoption of antimicrobial catheters in hospitals and ICUs
UAE Vision 2031 - Health Sector Transformation
2031
Enhance healthcare quality, access, and innovation; reduce healthcare-associated infections (HAIs)
Increased focus on infection prevention and patient safety boosts demand for antimicrobial catheters and related vascular access devices
Source: GVR Analysis
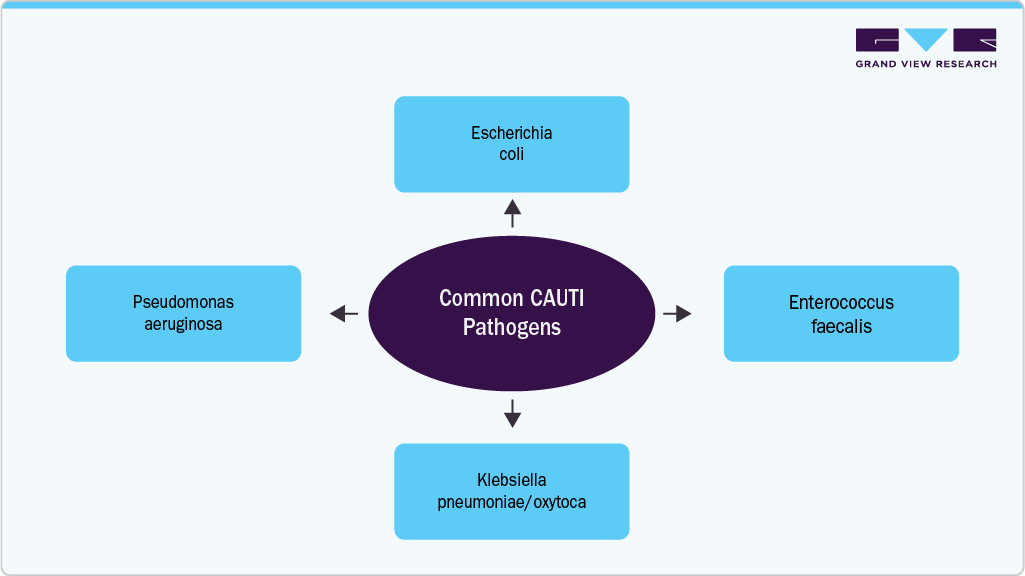
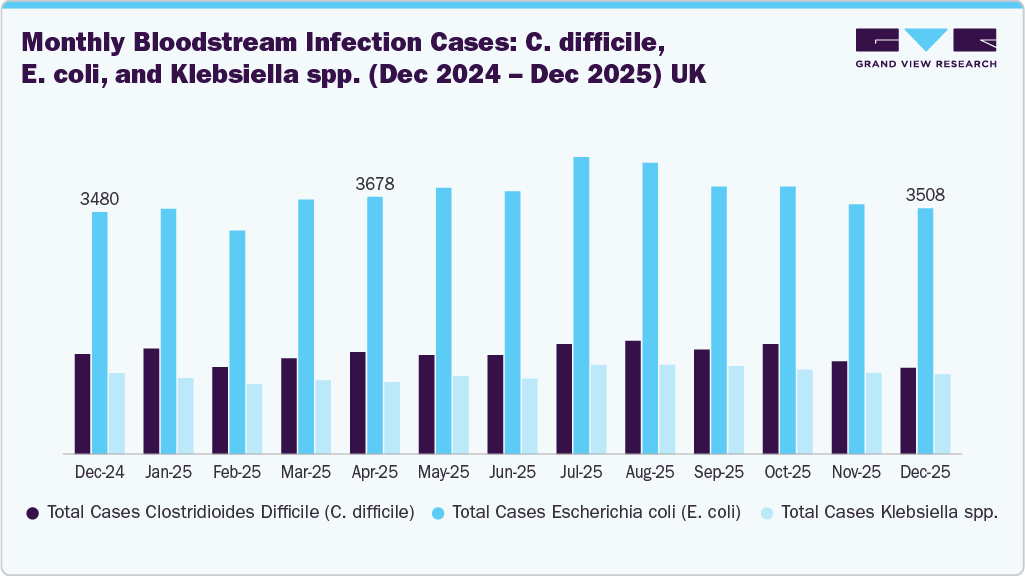
The increasing number of Clostridioides difficile (C. difficile), Escherichia coli (E. coli), and Klebsiella spp. Infections can indirectly drive the antimicrobial catheters market by highlighting the critical need for better infection‑prevention strategies in healthcare settings. These pathogens are common causes of healthcare‑associated infections (HAIs) that often occur in patients with indwelling medical devices such as urinary and vascular catheters. A higher incidence of such infections raises awareness among clinicians and hospital administrators about the risks associated with catheter use, encouraging adoption of antimicrobial‑coated catheters designed to reduce microbial colonization and prevent biofilm formation on device surfaces. As hospitals and healthcare systems prioritize infection control to improve patient outcomes and reduce treatment costs tied to these bacterial infections, the demand for advanced antimicrobial catheter technologies increases, supporting overall market growth.
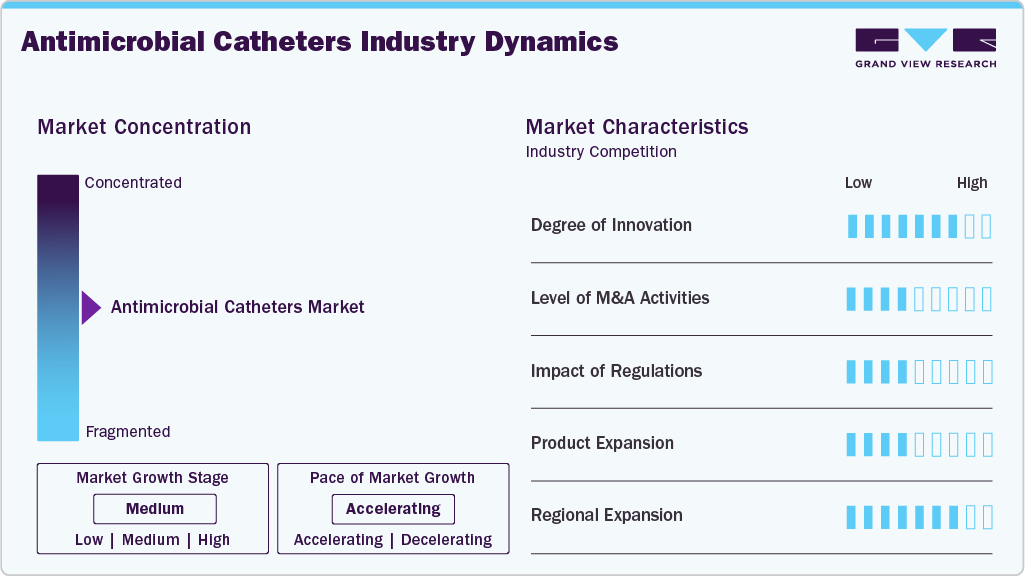
Market Concentration & Characteristics
The antimicrobial catheters market is a rapidly growing segment within medical devices, driven by the increasing prevalence of hospital-acquired infections, rising awareness of infection prevention, and adoption of advanced antimicrobial technologies in vascular and urinary catheters. Hospitals currently dominate the market, while home care and outpatient settings are emerging as fast-growing segments. The market is also supported by innovations such as silver-based and antibiotic-impregnated catheters, along with regional expansions and strategic partnerships by major players.
The antimicrobial catheters market is highly innovative, driven by the need to reduce healthcare-associated infections, improve patient outcomes, and extend device dwell time. Manufacturers are continuously developing advanced coating technologies, such as silver-based, antibiotic-impregnated, and organic/polymer-based antimicrobial surfaces, to enhance infection prevention while minimizing the risk of antimicrobial resistance. In addition, research is focused on next-generation materials that provide sustained antimicrobial release, improved biocompatibility, and reduced biofilm formation.
- In July 2024, Teleflex Incorporated announced the launch of the Teleflex Center for Antimicrobial Protection (TCAP), a strategic initiative under its Vascular Access business unit. Located in the Research Triangle, North Carolina, the center focuses on reducing infections caused by multidrug-resistant organisms by supporting education, innovation, and collaboration with infection prevention professionals. The initiative aims to reduce complications, such as central line-associated bloodstream infections (CLABSI), which can increase hospital stays and healthcare burden. Through TCAP, Teleflex is advancing antimicrobial catheter technology and developing innovative vascular access solutions.
Regulations play a critical role in shaping the antimicrobial catheters market by influencing product development timelines, approval costs, and commercialization strategies. Globally, stringent regulatory frameworks such as the EU Medical Device Regulation (EU MDR 2017/745) have strengthened requirements for clinical evidence, post-market surveillance, and safety monitoring, thereby improving patient safety but increasing compliance costs and time-to-market for manufacturers. The MDR replaced older directives to enhance oversight, strengthen market surveillance, and address safety gaps identified in previous regulatory systems.
- In June 2025, ASTM released ASTM F623-25, updating the Foley catheter standard and replacing ASTM F623-19. The revision clarifies the scope to single-use balloon retention catheters for short-term use (24 hours to 30 days) and introduces updated physical test methods. Chemically treated catheters, including those with antimicrobial or hydrophilic coatings, are not fully covered and require additional chemical or biocompatibility testing. Manufacturers should adjust regulatory submissions and testing strategies accordingly.
The level of mergers and acquisitions (M&A) activities in the antimicrobial catheters market is moderate to high, driven by the need for technology advancement, portfolio expansion, and geographic market penetration. Large medical device companies are actively acquiring smaller firms with specialized antimicrobial coating technologies, infection prevention solutions, or niche catheter product portfolios to strengthen their competitive positioning. M&A activities are also being used to enhance manufacturing capabilities, expand distribution networks, and accelerate entry into high-growth emerging markets.
- In March 2022, Vygon announced the acquisition of Macatt Medica, a Peru-based distributor of single-use medical devices, to strengthen its presence in South America, particularly in the enteral nutrition segment. With this acquisition, Macatt Medica became a wholly owned subsidiary of Vygon, supporting the company’s regional expansion strategy.
Regional expansion in the antimicrobial catheters market is being driven by increasing healthcare infrastructure development, rising awareness about infection prevention, and growing demand for advanced medical devices across emerging economies. Market players are actively expanding their distribution networks, establishing regional offices, and forming partnerships with local healthcare providers to strengthen their market presence. Regions such as Asia Pacific, Latin America, and the Middle East & Africa are witnessing increased investments in hospitals, diagnostic centers, and long-term care facilities, which are supporting the adoption of antimicrobial catheter technologies.
- In January 2026, Medline announced the opening of a new office in Riyadh, Saudi Arabia, to strengthen its presence and enhance service capabilities across the Middle East. The new facility supports Medline’s expansion strategy in the region and aims to improve customer engagement, supply chain coordination, and localized support for healthcare providers. The Riyadh office houses teams across management, business development, product management, regulatory affairs, and human resources, offering services in both Arabic and English to ensure faster response and communication.
“Establishing our Saudi Arabia office marks a pivotal step in Medline’s global expansion strategy, firmly embedding us within the Middle East's healthcare landscape and serving as a strategic cornerstone for the region. This central hub will enable us to streamline regional coordination and connect customers to our organisation, thereby enhancing speed, consistency, and tailored support across the Kingdom." - Hervé Million, President of Medline Europe, Middle East and Africa
Product Type Insights
The vascular access catheters segment dominates the antimicrobial catheters market in 2025, due to their extensive use in hospitals for drug delivery, fluid management, parenteral nutrition, chemotherapy, and critical care treatments. The high volume of inpatient procedures, intensive care unit admissions, and long-term intravenous therapies significantly drives demand for antimicrobial vascular access catheters. In addition, strong clinical focus on preventing catheter-related bloodstream infections (CRBSIs), along with strict infection control guidelines from healthcare authorities, supports the widespread adoption of antimicrobial coatings in vascular access devices. The availability of multiple product types, such as central venous catheters, PICCs, and peripheral IV catheters, further strengthens segment dominance.
The urological catheters segment is expected to grow at the fastest CAGR due to the rising prevalence of urinary incontinence, benign prostatic hyperplasia, and neurogenic bladder disorders, and the increasing number of urological surgeries globally. The growing elderly population, which is more prone to chronic urinary conditions requiring long-term catheterization, is also driving demand. Moreover, the growing shift toward home-based care and self-catheterization, combined with rising awareness of catheter-associated urinary tract infections, is accelerating the adoption of antimicrobial urological catheters, particularly in long-term and home care settings.
Size Insights
Silver-based technology dominates the antimicrobial catheters market in 2025, due to its broad-spectrum antimicrobial activity, long clinical history, and strong acceptance among healthcare providers. Silver coatings are effective against a wide range of bacteria and some fungi, helping reduce the risk of catheter-associated infections without relying on antibiotics, which supports antimicrobial stewardship efforts. In addition, silver-based technologies are widely available across multiple catheter types, including urological, vascular access, and specialty catheters. Established clinical evidence, regulatory approvals, and strong manufacturer adoption further contribute to the dominant market position of silver-based antimicrobial catheters.
Organic or polymer-based antimicrobial technologies segment is expected to grow at the fastest CAGR over the forecast period, driven by increasing concerns about antimicrobial resistance and the need for non-antibiotic infection-prevention solutions. These technologies use antiseptic agents or antimicrobial polymers that prevent microbial adhesion and biofilm formation on catheter surfaces. Growing research investments, development of next-generation surface modification technologies, and rising regulatory and clinical focus on reducing antibiotic usage are supporting the rapid adoption of polymer-based antimicrobial catheters. In addition, these technologies are gaining traction in long-term catheterization and home care applications due to their safety profile and sustained antimicrobial performance.
Material Insights
The Polyurethane (PU) segment dominates the antimicrobial catheters market in 2025, due to its excellent balance of strength, flexibility, and biocompatibility, making it highly suitable for a wide range of catheter applications, particularly vascular access catheters. PU materials enable thinner catheter walls while maintaining high tensile strength, improving flow rates, and overall device performance. In addition, polyurethane is highly compatible with various antimicrobial coating technologies, including silver, antiseptic, and antibiotic-based coatings, further supporting its widespread adoption. The material’s durability, resistance to kinking, and stability during more extended indwelling periods make it a preferred choice among healthcare providers, especially in hospital and critical care settings.
The silicone segment is expected to grow at the fastest CAGR from 2026 to 2033, due to its superior biocompatibility, softness, and reduced risk of tissue irritation, making it ideal for long-term catheterization, particularly in urological applications such as Foley catheters. Silicone is associated with lower encrustation risk and improved patient comfort during extended use, which is driving its growing preference in long-term care and home care settings. In addition, the growing focus on patient safety, the rising demand for long-duration catheterization, and the increasing adoption of premium catheter materials are driving the rapid global growth of silicone-based antimicrobial catheters.
End Use Insights
Hospitals segment dominate the antimicrobial catheters market in 2025 due to the high volume of surgical procedures, critical care admissions, and long-term patient management requiring catheterization. Hospitals are primary centers for infection prevention initiatives and adopt advanced antimicrobial technologies to reduce the risk of catheter-associated infections. In addition, the presence of skilled healthcare professionals, improved reimbursement structures, and strict infection control protocols further support greater use of antimicrobial catheters in hospital settings compared to other healthcare facilities, such as ambulatory surgical centers or home care environments.
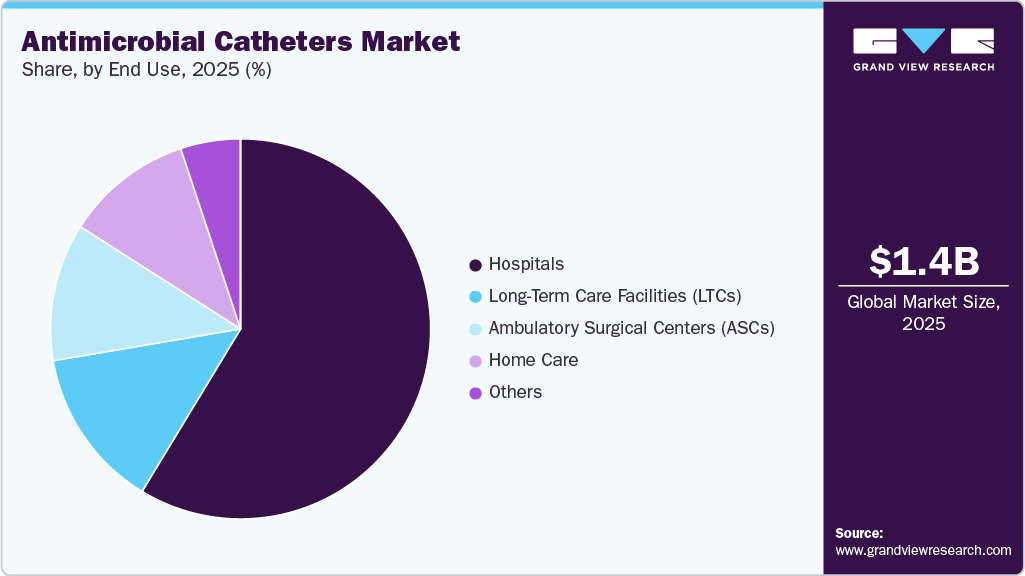
The home care segment is expected to grow at the fastest CAGR over the forecast period, driven by the rapid shift toward decentralized healthcare delivery and long-term disease management outside hospital settings. The increasing global burden of chronic conditions such as urinary incontinence, neurological disorders, diabetes, and cancer is driving the need for long-term catheterization, which is increasingly being managed at home. In addition, the rising geriatric population, which is more prone to mobility limitations and chronic illnesses, is significantly contributing to demand for home-based catheter care. Healthcare systems across developed and emerging countries are actively promoting early discharge programs and home treatment models to reduce hospital overcrowding and lower treatment costs, further accelerating demand for antimicrobial catheters in home settings. Technological advancements such as improved antimicrobial coatings, pre-lubricated and easy-to-use catheter designs, and patient education programs are also supporting safe catheter use at home.
Regional Insights
The North America antimicrobial catheters market is growing due to strong infection prevention mandates, high hospital adoption of antimicrobial-coated devices, and the region’s advanced healthcare infrastructure. Healthcare systems in the U.S. and Canada actively prioritize reducing healthcare-associated infections, which are strongly linked to urinary and central venous catheter use, driving consistent demand for antimicrobial solutions. High ICU utilization, large chronic disease patient pools, and aging populations further increase long-term catheterization needs, supporting sustained product adoption across hospitals and long-term care facilities. In addition, regulatory pressure and hospital safety programs are accelerating the shift toward antimicrobial technologies, with many hospitals integrating antimicrobial catheters into standard infection-control protocols to improve patient outcomes and reduce catheter-associated infections.
U.S. Antimicrobial Catheters Market Trends
Antimicrobial catheters market in the U.S. is growing, supported in part by strong infection-prevention frameworks and clinical practice standards issued by organizations such as the CDC. CDC guidelines for prevention of catheter-associated infections emphasize appropriate catheter selection, strict aseptic insertion, maintenance protocols, and early removal when clinically unnecessary. These guidelines drive healthcare facilities to adopt advanced infection-control solutions, including antimicrobial-coated or impregnated catheters, especially in high-risk patient populations or settings with elevated infection risk.
In addition, the CDC framework highlights the need for continuous quality improvement programs, surveillance of catheter-associated infections, and the adoption of newer technologies to reduce infection burden across acute and non-acute care settings. This encourages hospitals and integrated delivery networks to standardize procurement toward infection-prevention-focused devices, indirectly supporting the adoption of antimicrobial catheter technologies across urology, vascular access, and critical care applications.

Healthcare facilities should follow the 2009 CDC Guideline for Prevention of Catheter-associated Urinary Tract Infections. The guideline emphasizes the proper use, insertion, and maintenance of urinary catheters in different healthcare settings. It also presents effective quality improvement programs that healthcare facilities can use to prevent CAUTIs.
Europe Antimicrobial Catheters Market Trends
Antimicrobial catheters market in Europe is growing due to a combination of infection control pressure, an aging population, healthcare quality regulations, and rising clinical adoption of advanced-coated catheters across hospitals and long-term care settings. European healthcare systems are increasingly focused on reducing hospital-acquired infections, especially catheter-associated urinary tract infections and bloodstream infections, thereby accelerating the adoption of antimicrobial-coated devices. Many hospitals are integrating infection-prevention protocols and antimicrobial technologies into routine clinical practice, supporting steady demand across urology, ICU, and oncology care. In addition, Europe’s aging population is increasing the use of long-term catheterization procedures, particularly in elderly care facilities, driving higher use of antimicrobial urinary and vascular catheters. Strong regulatory frameworks, patient safety mandates, and continuous product innovation, including silver, antibiotic, and combination coatings, are further supporting market expansion across major countries such as Germany, the UK, and France.
Antimicrobial catheters market in the UK is growing due to the rising burden of urinary incontinence, which increases the need for intermittent and long-term urinary catheterization across hospitals, long-term care facilities, and home care settings. Patients with moderate to severe incontinence require catheter-based bladder management, particularly among elderly populations, patients with neurological disorders, and those in post-surgical or chronic care settings. This sustained catheter utilization increases the risk of catheter-associated urinary tract infections (CAUTIs), prompting healthcare providers to adopt antimicrobial or antiseptic-coated catheters as part of infection prevention and patient safety strategies. In addition, infection control remains a major priority across the UK healthcare system, encouraging the use of advanced catheter technologies when standard infection prevention protocols are insufficient. Furthermore, the growing focus on reducing hospital-acquired infections, improving patient outcomes, and lowering treatment-related complications is supporting the transition toward antimicrobial catheter adoption, thereby contributing to market growth.
Prevalence and Impact of Urinary Incontinence in UK (2025)
Category
Key Statistic / Impact (UK)
Prevalence of bladder problems
1 in 5 people experience bladder problems (~14 million people)
Women affected
Around 1 in 3 women (34%) live with urinary incontinence
Men affected
Over 2 million men experience urinary incontinence
Hospital-acquired UTIs
Incontinence increases risk of hospital-acquired UTIs
Impact on hospital stay
Hospital-acquired UTIs extend length of stay by ~6 days
Care home residents
Over 50% of care home residents have urinary incontinence
Source: The Urology Foundation
The Germany antimicrobial catheters market is experiencing growth driven by strong infection-control practices, advanced healthcare infrastructure, and an increasing focus on preventing catheter-associated infections in hospitals and long-term care settings. Germany maintains high clinical standards through strict, evidence-based catheter management protocols and active infection surveillance systems, which encourage the adoption of antimicrobial-coated catheters to reduce hospital-acquired infections. In addition, the country’s rapid digital health adoption, including electronic monitoring systems for CAUTIs, is improving early detection and infection tracking, further supporting demand for advanced antimicrobial catheter technologies. The rising elderly population and the greater need for long-term catheterization and catheter management solutions in elder care facilities are also key drivers of market expansion.
Furthermore, in Germany, CAUTI prevention is primarily driven by the KISS program (Krankenhaus-Infektions-Surveillance-System), coordinated by the Robert Koch Institute and implemented via Charité Berlin. This national surveillance system monitors device-associated infections, using standardized protocols, benchmarking, and evidence-based interventions to reduce rates, particularly in ICUs.
Asia Pacific Antimicrobial Catheters Market Trends
Antimicrobial catheters market in APAC is driven by rising healthcare infrastructure development, increasing infection awareness, and growing demand for advanced infection-prevention medical devices across hospitals and specialty care centers. Rapid expansion of hospital networks, particularly in countries such as China, India, and Southeast Asian nations, is increasing the volume of surgical procedures and catheter-based treatments, thereby raising the need for antimicrobial catheter technologies to reduce infection risk. In addition, the region is experiencing a growing elderly population and rising prevalence of chronic diseases such as diabetes, kidney disorders, and cardiovascular conditions, all of which increase long-term catheter utilization. Improving healthcare access, expansion of private hospital chains, and increasing adoption of international infection-control protocols are also encouraging the transition toward antimicrobial-coated urinary and vascular catheters. Furthermore, government investments in healthcare modernization, the growth of medical tourism, and increased clinician awareness of catheter-associated infections are supporting steady market expansion across the Asia Pacific region.
Diabetes Burden Comparison in South-East Asia and Western Pacific Regions, 2024 and 2050
Metric
South-East Asia (SEA) - 2024
Western Pacific (WP) - 2024
South-East Asia (SEA) - 2050 (Projected)
Western Pacific (WP) - 2050 (Projected)
People with Diabetes (in 1,000s)
106,870.20
215,440.70
184,509.40
253,810.00
Age-Standardized Prevalence (%)
10.80%
11.10%
13.00%
12.80%
Proportion of Undiagnosed Diabetes (%)
42.70%
50.00%
-
-
People with Undiagnosed Diabetes (in 1,000s)
45,607.50
107,619.20
-
-
Source: IDF, 2025
The India antimicrobial catheters market is driven by the rapid expansion of healthcare infrastructure, rising burden of chronic diseases, and increasing awareness of hospital-acquired infection prevention across healthcare settings. India is witnessing strong growth in hospital capacity, private healthcare investments, and the expansion of tertiary care centers, which is increasing the volume of surgical procedures and catheter-based treatments. In addition, the rising prevalence of diabetes, kidney disease, cardiovascular disorders, and neurological conditions is increasing long-term catheter utilization, particularly for urinary management and dialysis procedures. Growing awareness among clinicians regarding catheter-associated infections and the importance of infection control protocols is also encouraging the adoption of antimicrobial-coated catheters in hospitals and critical care units. Furthermore, government healthcare initiatives, the expansion of health insurance coverage, and an increased focus on improving patient safety and reducing treatment complications are supporting the transition toward advanced infection-prevention medical devices, thereby contributing to market growth in India.
As of March 2025, the Government of India has been actively strengthening the country’s healthcare ecosystem through multiple national programs aimed at expanding infrastructure, improving rural healthcare access, and enhancing critical care capacity. Initiatives such as PM-ABHIM, National Health Mission (NHM), PMSSY, and Finance Commission health grants are supporting the development of health and wellness centers, the addition of critical care beds, the establishment of district public health laboratories, and the expansion of tertiary healthcare facilities and medical education infrastructure. These initiatives are collectively improving healthcare accessibility, strengthening public health systems, and increasing the availability of trained healthcare professionals nationwide.
Antimicrobial catheters market in China is growing due to rising focus on hospital infection control, increased healthcare utilization, expanding hospital infrastructure, and strong demand for minimally invasive procedures. China accounts for a significant share of antimicrobial catheter consumption in Asia-Pacific, driven by the widespread adoption of coated central lines and increasing use in ICUs and surgical settings. A major driver is the growing burden of chronic diseases, neurological disorders, and cardiovascular conditions, which increases the need for catheter-based procedures and long-term vascular access devices. Expansion of national health insurance coverage, hospital modernization, and government healthcare investments are improving patient access to advanced medical technologies, indirectly supporting the adoption of antimicrobial catheters.
In 2024, according to the China Neurological Disorders Report compiled by the National Center for Neurological Disorders (China), neurological disease burden data showed that Alzheimer’s disease and related dementias affected about 16.99 million people in China, while Parkinson’s disease affected around 3.62 million people, highlighting the significant and growing clinical burden associated with aging populations. These findings were based on analysis of more than 15,000 scientific publications and national epidemiology data sources used to characterize disease prevalence, incidence, and demographic distribution across China. In addition, cerebrovascular disease was identified as one of the leading causes of death in Chinese adults, with ischemic stroke accounting for about 72.8% of stroke cases, reflecting a major neurological disease driver linked to vascular risk factors and aging demographics.
Latin America Antimicrobial Catheters Market Trends
Antimicrobial catheters market in Latin America is growing due to improving infection-control awareness, expanding healthcare infrastructure, and rising adoption of advanced medical devices across major countries such as Brazil and Argentina. The growing focus on preventing catheter-associated infections is driving hospitals to shift toward antimicrobial-coated catheters to improve patient safety and clinical outcomes. Rising healthcare expenditure, hospital modernization, and the strengthening of private healthcare systems are supporting technology adoption across the region. However, pricing pressure, import duties, and uneven access to advanced technologies continue to influence the pace of adoption in some countries.
MEA Antimicrobial Catheters Market Trends
The Middle East & Africa (MEA) antimicrobial catheters market is growing due to an infection-control needs, healthcare infrastructure expansion, and increasing critical-care capacity across key countries. Rising hospital-acquired infection burden is a major driver; for instance, ICU infection rates in some Middle Eastern countries have been reported at notable levels, pushing hospitals to adopt antimicrobial and coated catheters to reduce complications and treatment costs. In addition, large-scale hospital modernization projects and the expansion of medical tourism in GCC countries such as Saudi Arabia and the UAE are accelerating the adoption of advanced infection-prevention devices.
Key Antimicrobial Catheters Company insights
The antimicrobial catheters market is competitive, with key players holding significant positions. The major companies are undertaking various organic as well as inorganic strategies such as new product development, collaborations, acquisitions, mergers, and regional expansion for serving the unmet needs of their customers.
Key Antimicrobial Catheters Companies:
The following key companies have been profiled for this study on the antimicrobial catheters market.
- B. Braun SE
- Teleflex Incorporated
- SilvoGuard
- BD
- MEDLINE
- ConvaTec Group PLC
- Silq Technologies
- AvantGuard (Halomine Inc.)
- Medtronic
- Cook
- Integra LifeSciences Corporation
- Vygon
- Camstent
Recent Developments
-
In November 2025, AvantGuard, Inc., a biotechnology company developing next-generation antimicrobial technologies inspired by the human immune system, announced that it had received three National Institutes of Health (NIH) research grants totaling USD2.65 million.
-
In September 2025, B. Braun SE announced the complete acquisition of True Digital Surgery (TDS), a Goleta, California-based company specializing in digital robotic-assisted 3D surgical microscopy. The acquisition strengthens B. Braun’s position in digital microsurgery and supports continued advancement.
-
In January 2025, BD announced expanded investments to strengthen its U.S. manufacturing network for essential medical devices to support healthcare system demand. Following more than USD 10 million invested in 2024, BD installed new syringe and needle production lines in Connecticut and Nebraska, significantly increasing capacity for safety-engineered injection devices and conventional syringes. The company also plans to invest over USD 30 million in 2025 to expand IV line manufacturing in Utah, building on earlier investments that already boosted IV catheter production.
Antimicrobial Catheters Market Report Scope
Report Attribute
Details
Market size value in 2026
USD 1.56 billion
Revenue forecast in 2033
USD 2.86 billion
Growth rate
CAGR of 9.0% from 2026 to 2033
Actual data
2021 - 2025
Forecast period
2026 - 2033
Quantitative units
Revenue in USD million/billion and CAGR from 2026 to 2033
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Product type, technology, material, end use, region
Regional scope
North America; Europe; Asia Pacific; Latin America; MEA
Country scope
U.S.; Canada; Mexico; UK; Germany; France; Italy; Spain; Denmark; Sweden; Norway; Japan; China; India; Australia; Thailand; South Korea; Brazil; Argentina; South Africa; Saudi Arabia; UAE; Kuwait.
Key companies profiled
B. Braun SE; Teleflex Incorporated; SilvoGuard; BD; Medline; ConvaTec Group PLC; Silq Technologies; AvantGuard (Halomine Inc.); Medtronic; Cook; Integra LifeSciences Corporation; Vygon; Camstent
Customization scope
Free report customization (equivalent up to 8 analysts working days) with purchase. Addition or alteration to country, regional & segment scope.
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
Global Antimicrobial Catheters Market Report Segmentation
This report forecasts revenue growth at country levels and provides an analysis on the latest industry trends and opportunities in each of the sub-segments from 2021 to 2033. For the purpose of this study, Grand View Research has segmented the antimicrobial catheters market report on the basis of product type, technology, material, end use, and region:
-
Product Type Outlook (Revenue, USD Million, 2021 - 2033)
-
Urological Catheters
-
Vascular Access Catheters
-
Specialty and Surgical Catheters
-
-
Technology Outlook (Revenue, USD Million, 2021 - 2033)
-
Silver-Based Technology
-
Antibiotic-Impregnated
-
Organic/Polymer-Based (Non-Metal/Non-Antibiotic)
-
Combination Technologies
-
-
Material Outlook (Revenue, USD Million, 2021 - 2033)
-
Silicone
-
Polyurethane (PU)
-
Latex
-
Others
-
-
End Use Outlook (Revenue, USD Million, 2021 - 2033)
-
Hospitals
-
Ambulatory Surgical Centers (ASCs)
-
Long-Term Care Facilities (LTCs)
-
Home Care
-
Others
-
-
Region Outlook (Revenue, USD Million, 2021 - 2033)
-
North America
-
U.S.
-
Canada
-
Mexico
-
-
Europe
-
UK
-
Germany
-
France
-
Italy
-
Spain
-
Denmark
-
Sweden
-
Norway
-
-
Asia Pacific
-
Japan
-
China
-
India
-
Australia
-
South Korea
-
Thailand
-
-
Latin America
-
Brazil
-
Argentina
-
-
Middle East & Africa
-
South Africa
-
Saudi Arabia
-
UAE
-
Kuwait
-
-
Frequently Asked Questions About This Report
North America dominated the antimicrobial catheters market in 2025.
Key companies operating in the antimicrobial catheters market include B. Braun SE, Teleflex Incorporated, SilvoGuard, BD, Medline Industries, ConvaTec Group PLC, Silq Technologies, AvantGuard (Halomine Inc.), Medtronic, Cook Medical, Integra LifeSciences Corporation, Vygon, and Camstent.
The antimicrobial catheters market is driven by the rising prevalence of hospital-acquired infections (HAIs) and increasing emphasis on infection prevention in healthcare settings. Growing surgical procedures, aging populations, and stricter regulatory guidelines promoting antimicrobial-coated devices further support market expansion.
The global antimicrobial catheters market size was estimated at USD 1.43 billion in 2025 and is expected to reach USD 1.56 billion in 2026.
The global antimicrobial catheters market is expected to grow at a compound annual growth rate of 9.0% from 2026 to 2033 to reach USD 2.86 billion by 2033.
About the authors:
Author: GVR Medical Devices Research Team | Last Updated:
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.









