- Home
- »
- Medical Devices
- »
-
Asia Pacific Bioregenerative Aesthetic Injectable Market, 2033GVR Report cover
![Asia Pacific Bioregenerative Aesthetic Injectable Market Size, Share & Trends Report]()
Asia Pacific Bioregenerative Aesthetic Injectable Market (2026 - 2033) Size, Share & Trends Analysis Report By Product (PN, Peptide based Injectables, PRP Injectables), By Application, By End-use (MedSpas, Aesthetic Surgery Centers), And Segment Forecasts
- Report ID: GVR-4-68040-861-1
- Number of Report Pages: 180
- Format: PDF
- Historical Range: 2021 - 2025
- Forecast Period: 2026 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
Asia Pacific Bioregenerative Aesthetic Injectable Market Summary
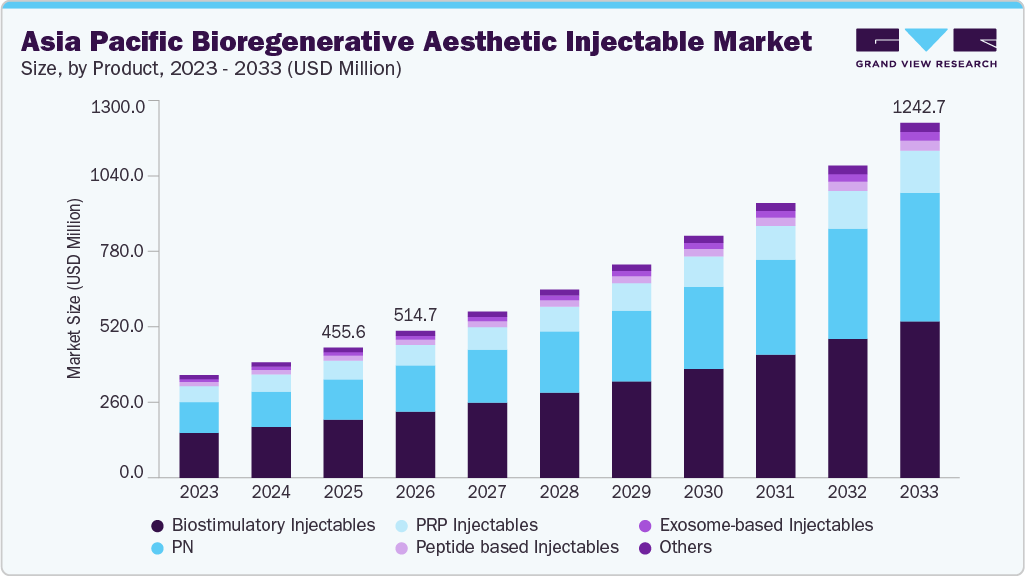
The Asia Pacific bioregenerative aesthetic injectable market size was estimated at USD 455.6 million in 2025 and is projected to reach USD 1,242.7 million by 2033, growing at a CAGR of 13.4% from 2026 to 2033. This growth is driven by rising consumer demand for natural-looking, minimally invasive treatments that promote long-term skin health.
Key Market Trends & Insights
- China bioregenerative aesthetic injectable industry held the largest share of 48.0% in 2025.
- By product, the biostimulatory injectables segment held the highest market share in 2025.
- By application, the skin hydration & moisturization (For Face) segment held the largest market share in 2025.
- By end use, the dermatology centers segment accounted for the largest revenue share in 2025.
Market Size & Forecast
- 2025 Market Size: USD 455.6 Million
- 2033 Projected Market Size: USD 1,242.7 Million
- CAGR (2026-2033): 13.4%
Advances in regenerative technologies such as platelet-rich plasma, polynucleotides, and exosomes are boosting product effectiveness. Increasing awareness, higher disposable incomes, and expanding medical aesthetics infrastructure worldwide are further driving adoption across key markets.The Asia Pacific bioregenerative aesthetic injectables industry is progressing into a more mature, value-driven stage, influenced by distinct consumer behaviors across China, Japan, and South Korea.
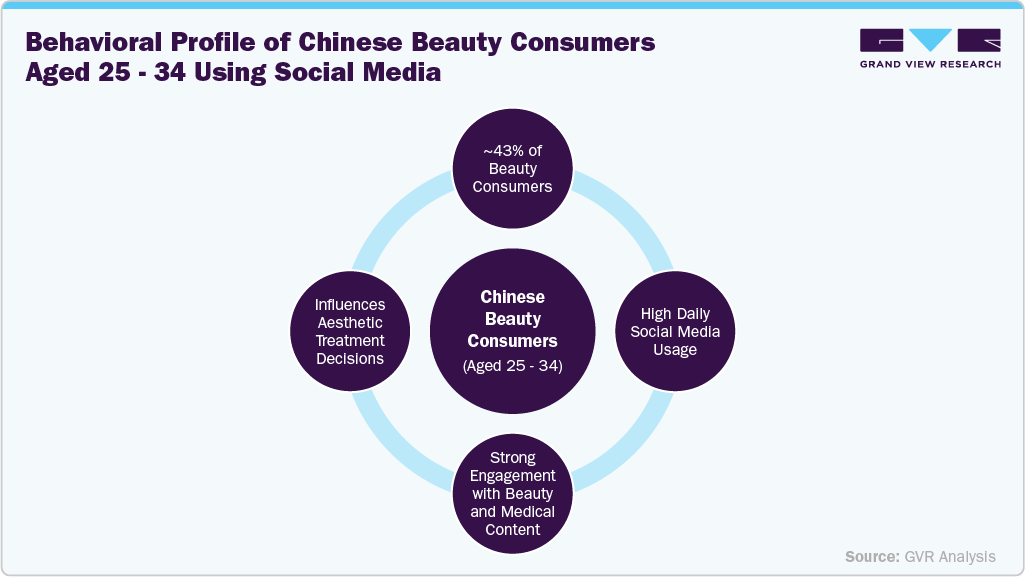
In China, demand is fueled by strong beauty awareness, digital engagement, and openness to medical innovation. Younger urban professionals increasingly favor regenerative procedures centered on skin repair, collagen stimulation, and preventive anti-aging rather than dramatic cosmetic change. PRP and biostimulatory injectables serve as common entry treatments, while peptide- and exosome-based solutions are gaining traction through physician-led education and digital beauty platforms.
“In China’s beauty space, social platforms such as Xiaohongshu and Douyin don’t just advertise, they educate. Our users treat clinician-led drops, before-and-after reel stories, and peer reviews as primary decision inputs, not just inspiration.”
-Medical Aesthetic Campaign Lead
Source: GVR Analysis
Consumers aged 25-34 play a central role in shaping demand, relying heavily on online medical content and physician credibility. However, while Chinese patients are open to innovation, they expect visible results and science-backed explanations, creating a high-expectation, fast-adoption environment.
In Japan, adoption is more cautious and precision-oriented, with strong emphasis on safety and subtle, long-term improvement.
“Surveys in Japan suggest many patients approach aesthetic treatments with a healthy skepticism and often require high-quality clinical education and clear outcome expectations before they engage.”
-Japanese Consumer Behavior Summary
Japanese patients favor dermatology-led care and gradual, predictable outcomes, resulting in slower but highly loyal adoption.
South Korea represents one of the most advanced aesthetic markets globally, where consumers prioritize skin quality and regeneration over volumization. PRP, polynucleotides, and biostimulators are commonly integrated into comprehensive treatment plans.
“South Korea’s blend of high-tech skincare, personalized clinic experiences, and rapid aesthetic innovation shapes consumer behavior that prioritizes nuanced results and ongoing skin health over one-time cosmetic fixes.”
-Vogue
Market Concentration & Characteristics
The Asia Pacific bioregenerative aesthetic injectables industry is moderately concentrated, with a mix of global pharmaceutical leaders and strong regional players competing across premium and mid-tier segments. Multinational companies dominate high-value, clinically validated biostimulatory products, while domestic manufacturers drive volume growth through localized innovation and competitive pricing. The market is characterized by rapid product advancements, rising demand for minimally invasive regenerative treatments, and increasing physician-led education. Digital influence, medical tourism, and expanding dermatology clinic networks further shape industry dynamics. Although regulatory standards vary by country, growing clinical validation and consumer awareness continue to strengthen market maturity and structured competition across the region.
The Asia Pacific bioregenerative aesthetic injectables industry exhibits strong innovation, supported by Sweden’s growing influence in Asia Pacific aesthetics. In January 2025, Nordberg Medical introduced Juläine, a Swedish-developed PLLA biostimulator, across Central Asia Pacific. Developed using proprietary LASYNPRO technology, Juläine utilizes microsphere-based collagen stimulation with controlled inflammatory response to gradually activate fibroblasts. Positioned as a minimally invasive alternative to traditional dermal fillers, the product highlights Sweden’s commitment to science-led innovation, natural aesthetic outcomes, and sustained tissue regeneration through long-term collagen remodeling.
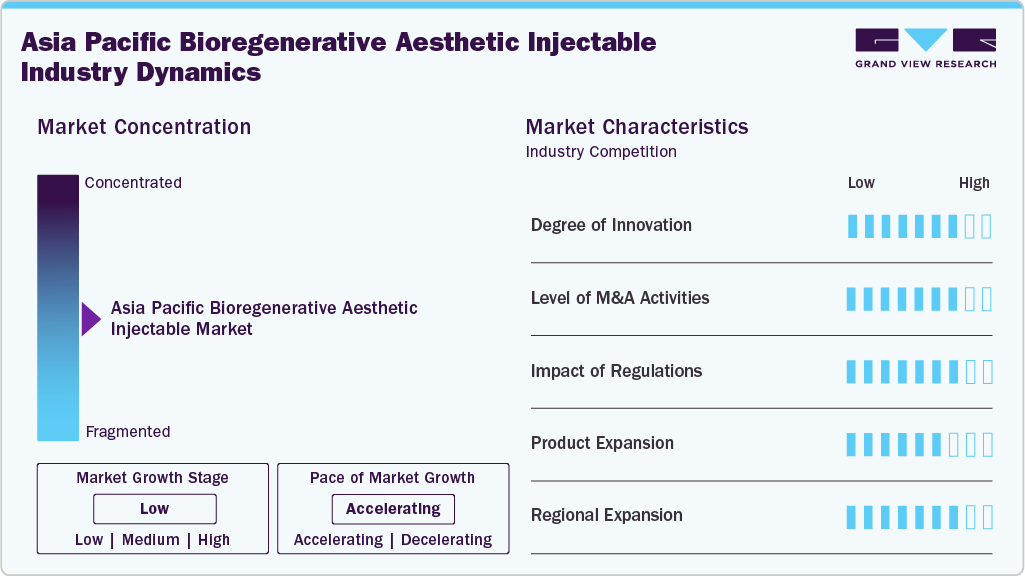
The Asia Pacific bioregenerative aesthetic injectables industry is witnessing a moderate to high level of M&A activity, driven by strategic expansion and portfolio diversification. Global aesthetic and pharmaceutical companies are acquiring regional brands and technology developers to strengthen their presence and access innovative regenerative formulations. Domestic players are also forming joint ventures and distribution partnerships to expand into new markets across the region. These transactions often focus on securing proprietary technologies such as polynucleotides, exosome platforms, and advanced biostimulators. Overall, M&A activity reflects growing competition, increasing investor interest, and the strategic importance of regenerative aesthetics within broader aesthetic medicine portfolios.
Regulation plays a critical role in shaping the Asia Pacific bioregenerative aesthetic injectables industry, influencing product approvals, labeling, clinical validation, and marketing claims. Key regulatory authorities such as China’s National Medical Products Administration (NMPA), Japan’s Pharmaceuticals and Medical Devices Agency (PMDA), South Korea’s Ministry of Food and Drug Safety (MFDS), and Australia’s Therapeutic Goods Administration (TGA) enforce strict safety and efficacy standards. These agencies require robust clinical data, particularly advanced regenerative products like exosomes and polynucleotide injectables. While stringent oversight can lengthen approval timelines, it enhances patient safety, strengthens consumer confidence, and supports long-term market credibility and sustainable industry growth.
Product substitutes in the Asia Pacific bioregenerative aesthetic injectables industry includes traditional dermal fillers, neuromodulators (such as botulinum toxin), energy-based devices, topical cosmeceuticals, and minimally invasive skin treatments. Hyaluronic acid fillers and Botox-type products remain strong alternatives for patients seeking immediate volumization or wrinkle reduction rather than regenerative outcomes. Laser therapies, radiofrequency, ultrasound-based tightening, and microneedling procedures also compete by stimulating collagen through non-injectable approaches. In addition, medical-grade skincare products and at-home aesthetic devices appeal to cost-sensitive consumers. While bioregenerative injectables emphasize long-term tissue repair and collagen induction, these substitutes attract patients prioritizing quicker results, lower cost, or non-invasive solutions.
Growth across the Asia Pacific bioregenerative aesthetic injectables industry is being reinforced by strategic capacity expansion initiatives. For example, Galderma is evaluating the establishment of a fifth, fully automated manufacturing facility in Singapore to strengthen its regional presence. Backed by the Singapore Economic Development Board, the proposed plant is expected to increase annual production capacity by approximately 350 million units. This expansion would help meet the growing demand for premium dermo-cosmetic and regenerative injectable products while supporting the company’s long-term expansion strategy and supply chain resilience within the Asia Pacific region.
Product Insights
The biostimulatory injectables segment accounted for the largest revenue share of 44.6% in 2025, primarily due to its ability to deliver long-lasting, natural-looking aesthetic results by stimulating the body’s own collagen production. Unlike traditional fillers that provide immediate volume, biostimulatory injectables such as poly-L-lactic acid and calcium hydroxylapatite promote gradual tissue regeneration and structural improvement. Growing patient preference for minimally invasive treatments with durable outcomes, reduced maintenance frequency, and improved skin quality has driven adoption. Strong physician confidence, expanding clinical indications, and increasing use in combination therapies further support the segment’s dominant market position.
The PN (polynucleotide) injectables segment is projected to record the fastest growth from 2026 to 2033 in the market, driven by rising demand for advanced skin regeneration therapies. PN injectables are increasingly favored for their ability to stimulate collagen production, improve skin elasticity, and support tissue repair with natural-looking results. Growing consumer preference for minimally invasive treatments, combined with strong clinical adoption in South Korea, Japan, and China, is accelerating uptake. In addition, expanding practitioner awareness, increasing product launches, and positive patient outcomes are strengthening confidence in PN-based treatments, positioning the segment for robust expansion across the region.
Application Insights
The skin hydration & moisturization (face) segment accounted for the largest revenue share of 19.39% in 2025. This dominance is primarily driven by growing consumer emphasis on skin quality, glow, and long-term dermal health rather than structural alteration. Increasing demand for minimally invasive procedures that enhance hydration, elasticity, and overall skin texture has supported strong adoption across urban markets. Products positioned around collagen stimulation and deep dermal hydration appeal to younger consumers seeking preventive care as well as mature patients targeting early aging signs. Expanding clinic networks and practitioner recommendations further reinforce segment leadership across the region.
The under eye-area (face) segment is projected to register the fastest CAGR during the forecast period. Growth is driven by rising concerns related to dark circles, fine lines, and skin thinning, particularly among urban consumers exposed to high screen time and stress-related fatigue. Patients increasingly prefer regenerative injectables that improve skin quality and collagen density without excessive volumization. Polynucleotides, PRP, and low intensity biostimulators are gaining traction for delicate periorbital rejuvenation. Strong demand in South Korea, China, and Japan, combined with growing practitioner expertise, is accelerating adoption in this high-precision treatment segment.
End Use Insights
The dermatology centers segment accounted for the largest revenue share of 38.5% in 2025. This dominance is driven by strong patient trust in dermatologist-led procedures, higher treatment volumes, and access to advanced regenerative technologies. Dermatology clinics are often preferred for PRP, polynucleotide, and biostimulatory injectables due to their clinical expertise, safety standards, and ability to deliver personalized skin-focused treatment protocols.
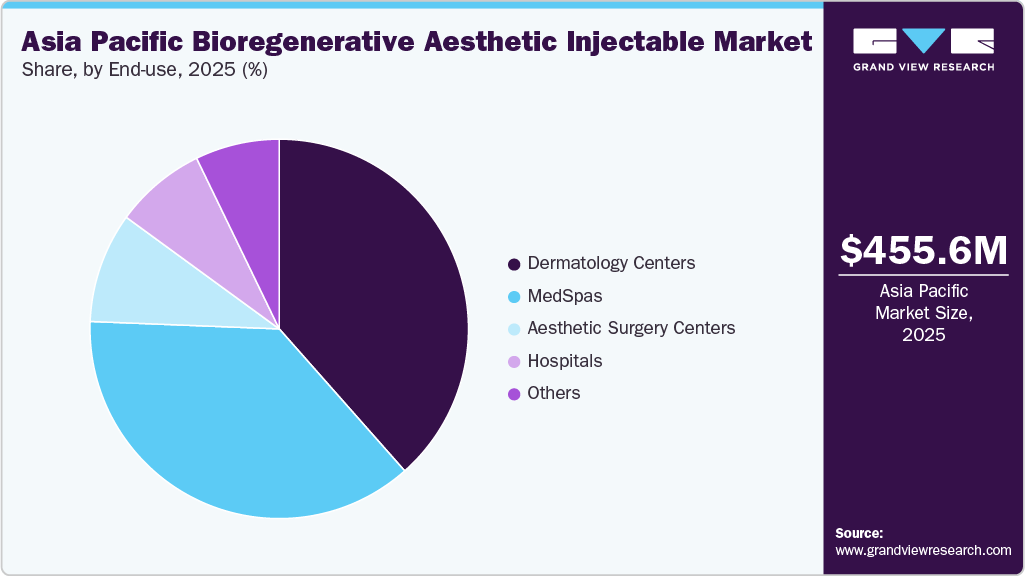
The MedSpas segment is expected to witness the fastest CAGR during the forecast period, driven by rising consumer demand for minimally invasive, quick-recovery treatments in non-hospital settings. Growing preference for preventive aesthetics, combination therapies, and personalized skin rejuvenation programs is accelerating adoption. MedSpas offer convenient access, competitive pricing, and strong digital marketing outreach, particularly among younger demographics.
Country Insights
Asia Pacific bioregenerative aesthetic injectable industry held the largest share of the global market, exceeding 30.3% in 2025, and is expected to witness sustained growth in the coming years. The rapid expansion of digital platforms, medical tourism, and e-commerce has further enhanced the accessibility and market presence of these injectables and associated treatment protocols across the region. Both international and regional manufacturers are actively introducing a broad range of bioregenerative injectable products, including hyaluronic acid- and vitamin-based formulations as well as advanced solutions containing polynucleotides, peptides, and growth-enhancing complexes. Market entry is largely supported through collaborations with dermatology and aesthetic clinics, established distribution networks, and formalized practitioner training and certification programs, facilitating broader clinical adoption across major Asia-Pacific countries.
Medical tourism remains a significant contributor to market growth. Asia Pacific has emerged as a preferred destination for international patients seeking advanced aesthetic procedures at competitive costs, leading to increased treatment volumes and greater visibility of bioregenerative injectables. Leading aesthetic centers in the region continue to report rising inflows of overseas patients pursuing cosmetic and combination treatments, supported by advanced infrastructure and growing global demand for high-quality aesthetic services.
For example, in September 2025, Contour Clinic Thailand reported that international patients represented approximately 25.4% of its total aesthetic procedure volumes substantially above the global average of around 10% underscoring strong cross-border demand. In addition, global aesthetic procedure volumes reached nearly 34.9 million cases in 2023, reflecting year-over-year growth across both surgical and non-surgical categories. Improved accessibility, shifting beauty standards, and the strong appeal of the region’s medical tourism ecosystem have collectively accelerated demand for bioregenerative aesthetic injectables across Asia Pacific.
China Bioregenerative Aesthetic Injectable Market Trends
China’s bioregenerative aesthetic injectable industry is expanding rapidly, driven by growing consumer demand and continuous product innovation. The competitive landscape includes multinational companies such as PharmaResearch Products, Galderma, Merz Aesthetics, Mastelli, Ipsen, and BR Pharma, particularly within premium and biostimulatory segments. These players are strengthening their presence through localized product strategies, partnerships with domestic aesthetic clinics, clinician training initiatives, and the launch of globally recognized regenerative injectables. Competition is shaped by pricing strategies, deeper penetration into lower-tier cities, and diversified portfolios that address both traditional and regenerative aesthetic needs.
Market growth is supported by the rising popularity of minimally invasive procedures and increasing acceptance of aesthetic enhancements across age groups. An aging population seeking youthful appearance solutions remains a key demand driver, while younger consumers especially millennials and Gen Z are influenced by social media trends and urban lifestyles, favoring natural-looking, subtle results. The transition from conventional cosmetic treatments to bioregenerative injectables reflects a stronger emphasis on safety, collagen stimulation, and long-term skin quality improvement.
India Bioregenerative Aesthetic Injectable Market Trends
India’s bioregenerative aesthetic injectables industry is witnessing leading companies actively implementing strategic initiatives to strengthen their position. Global players such as Mastelli s.r.l., Merz Aesthetics, Caregen Co. Ltd., Galderma, and Fillmed are emphasizing product innovation, including next-generation injectables with enhanced biostimulatory effects and improved delivery technologies designed to enhance safety, effectiveness, and durability. In addition to innovation, companies are expanding regionally by moving beyond major metropolitan areas through collaborations with local dermatology and aesthetic clinics, while also strengthening distribution networks to widen market reach.
In India, bioregenerative injectables are increasingly offered by private dermatology and aesthetic centers. Market growth is being driven by rising interest in minimally invasive procedures that stimulate the body’s natural repair mechanisms rather than simply providing cosmetic correction. Treatments such as platelet-rich therapies, exosome-based solutions, and other regenerative formulations are gaining popularity among both clinicians and consumers seeking collagen stimulation, tissue regeneration, and longer-lasting improvements. This trend reflects a broader shift toward natural rejuvenation and regenerative outcomes, in line with global aesthetic preferences.
South Korea Bioregenerative Aesthetic Injectable Market Trends
South Korea’s bioregenerative aesthetic injectable industry is witnessing robust growth, supported by both global and domestic players. International brands such as Allergan (AbbVie) and Galderma compete alongside local leaders like Medytox, which leverage strong clinical networks and localized strategies. Demand is rising due to consumer preference for natural, regenerative, and minimally invasive treatments. The country’s rapidly aging population further strengthens market potential, with over 21% aged 65 and above as of 2026. Advanced formulations, including regenerative skin boosters and collagen-stimulating hybrid injectables, align with evolving beauty standards, supporting sustained expansion of the market.
Australia Bioregenerative Aesthetic Injectable Market Trends
Australia’s bioregenerative aesthetic injectable industry is growing steadily, supported by strategic efforts from key players such as Mastelli, Merz Aesthetics, Galderma, and Allergan. These companies are focusing on next-generation biostimulatory injectables and regenerative skin boosters that offer gradual, natural-looking results with strong safety profiles well aligned with Australian preferences for subtle enhancement and preventive skincare. Market growth is fueled by rising interest in anti-aging solutions, higher disposable income, and increased awareness of regenerative technologies like PRP and collagen stimulators. Product launches, clinician training, and marketing initiatives, including Rejuran’s recent expansion, are further boosting consumer awareness and demand nationwide.
Key Asia Pacific Bioregenerative Aesthetic Injectable Company Insights
Some of the key companies in the Asia Pacific bioregenerative aesthetic injectable industry include Galderma, Merz Pharma and among others. Most companies focus on R&D activities to develop technologically advanced products to gain a competitive edge. Companies are also adopting various strategies, such as mergers and acquisitions, joint ventures, and developing low-cost equipment, especially for developing economies.
Key Asia Pacific Bioregenerative Aesthetic Injectable Companies:
- PharmaResearch Products Co., Ltd.
- MD Skin Solutions
- Mastelli s.r.l.
- Merz Aesthetics
- Caregen Co. Ltd
- Croma Pharma
- PROMOITALIA
- Galderma
- Fillmed
- IBSA
- Bio-Formula
- BR Pharma
- Allergan (AbbVie)
- Merz
- Fidia
- VAIM
Recent Developments
-
In November 2025, Germany’s Merz Aesthetics launched two flagship injectables, Xeomin and Radiesse, in China to address rising demand for regenerative aesthetics. The move strengthens Merz’s long-term China strategy amid tighter regulation and growing consumer interest in biostimulators.
-
In November 2024, Galderma celebrated 25 years of Sculptra, the first regenerative biostimulator, with new clinical data and recent approval in China, expanding its global reach. The PLLA-SCA formulation remodels collagen, elastin, and adipose tissue, restoring volume, skin quality, and firmness. Sculptra remains a cornerstone in regenerative aesthetic treatments worldwide.
Asia Pacific Bioregenerative Aesthetic Injectable Market Report Scope
Report Attribute
Details
Market size value in 2026
USD 514.7 million
Revenue forecast in 2033
USD 1,242.7 million
Growth rate
CAGR of 13.4% from 2026 to 2033
Historical data
2021 - 2025
Forecast period
2026 - 2033
Quantitative units
Revenue in USD billion/million and CAGR from 2026 to 2033
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Product, application, end use, country
Regional scope
Asia Pacific
Country scope
China; South Korea; Japan; India; Thailand; Singapore; Malaysia; Indonesia; Vietnam; Philippines; Taiwan; Pakistan; Hong Kong; Australia; New Zealand
Key companies profiled
MD Skin Solutions; Mastelli s.r.l.; Merz Aesthetics; Caregen Co. Ltd; Croma Pharma; PROMOITALIA; Galderma; Fillmed; IBSA; Bio-Formula; BR Pharma; Allergan (AbbVie); Merz; Fidia; VAIM
Customization scope
Free report customization (equivalent to 8 analyst’s working days) with purchase. Addition or alteration to country, regional & segment scope
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
Asia Pacific Bioregenerative Aesthetic Injectable Market Report Segmentation
This report forecasts revenue growth at regional and country levels and provides an analysis of the latest industry trends in each of the sub-segments from 2021 to 2033. For this study, Grand View Research has segmented the Asia Pacific bioregenerative aesthetic injectable market report based on product, application, end use, and country:
-
Product Outlook (Revenue, USD Million, 2021 - 2033)
-
PN
-
PDRN
-
PN Injectables
-
-
Peptide based Injectables
-
Exosome-based Injectables
-
Biostimulatory Injectables
-
PLLA
-
CaHA
-
-
PRP Injectables
-
Others
-
-
Application Outlook (Revenue, USD Million, 2021 - 2033)
-
Skin Hydration & Moisturization (For Face)
-
Elasticity & Firmness (For Face)
-
Under eye-areas (For Face)
-
Fine Lines (For Face)
-
Acne Scar (For Face)
-
Wrinkle Correction (For Face)
-
Body Application-
-
Arms & Thighs
-
Décolletage
-
Intimate Areas
-
Knees
-
Scalp & Hair Rejuvenation
-
-
-
End Use Outlook (Revenue, USD Million, 2021 - 2033)
-
MedSpas
-
Aesthetic Surgery Centers
-
Hospitals
-
Dermatology Centers
-
Others
-
-
Country Outlook (Revenue, USD Million, 2021 - 2033)
-
Asia Pacific
-
China
-
South Korea
-
Japan
-
India
-
Thailand
-
Singapore
-
Malaysia
-
Indonesia
-
Vietnam
-
Philippines
-
Taiwan
-
Pakistan
-
Hong Kong
-
Australia
-
New Zealand
-
-
Frequently Asked Questions About This Report
b. The Asia Pacific bioregenerative aesthetic injectable market is expected to grow at a compound annual growth rate of 13.4% from 2026 to 2033 to reach USD 1,242.7 million by 2033.
b. The Asia Pacific bioregenerative aesthetic injectable market was estimated at USD 455.6 million in 2025 and is expected to reach USD 514.7 million in 2026.
b. The biostimulatory injectables segment of the product category dominated the Asia Pacific bioregenerative aesthetic injectable market with a share of 44.6% in 2025, primarily due to its ability to deliver long-lasting, natural-looking aesthetic results by stimulating the body’s own collagen production.
b. Some key players operating in the Asia Pacific bioregenerative aesthetic injectable market include MD Skin Solutions, Mastelli s.r.l., Merz Aesthetics, Caregen Co. Ltd, Croma Pharma, PROMOITALIA, Galderma, Fillmed, IBSA, Bio-Formula, BR Pharma, Allergan (AbbVie), Merz, Fidia, and VAIM
b. Key factors that are driving the Asia Pacific bioregenerative aesthetic injectable market growth include the rising consumer demand for natural-looking, minimally invasive treatments that promote long-term skin health. Advances in regenerative technologies like platelet-rich plasma, polynucleotides, and exosomes are boosting product effectiveness. Increasing awareness, higher disposable incomes, and expanding medical aesthetics infrastructure worldwide are further driving adoption across key markets.
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










