- Home
- »
- Biotechnology
- »
-
Bioprocess Automation Market Size, Industry Report, 2033GVR Report cover
![Bioprocess Automation Market Size, Share & Trends Report]()
Bioprocess Automation Market (2026 - 2033) Size, Share & Trends Analysis Report By Product & Services (Services & Support, Process Automation Hardware, Automation Consumables & Accessories), By Process Stage (Upstream), By End Use, By Region, And Segment Forecasts
- Report ID: GVR-4-68040-764-1
- Number of Report Pages: 150
- Format: PDF
- Historical Range: 2021 - 2025
- Forecast Period: 2026 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
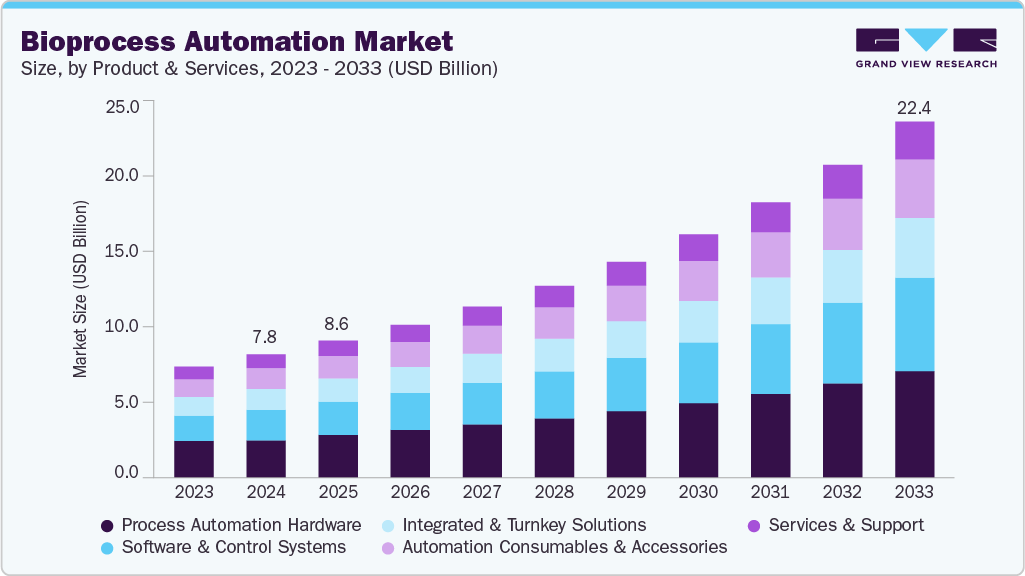
Market Size, 2025$8.6BMarket Estimate, 2026$9.5BMarket Forecast, 2033$22.2BCAGR, 2026 - 203312.8%Bioprocess Automation Market Summary
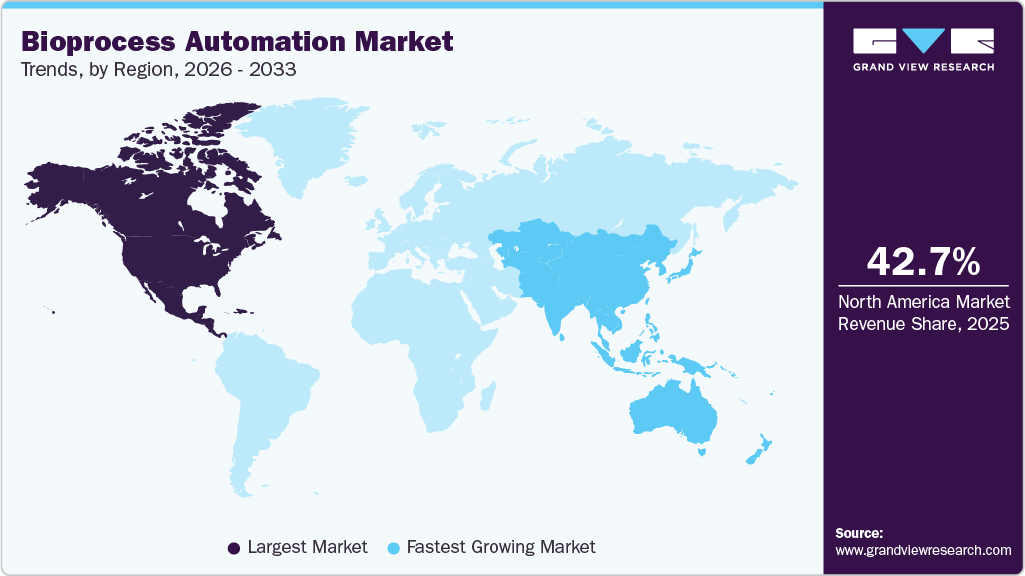
The global bioprocess automation market size was valued at USD 8.6 billion in 2025 and is projected to grow from USD 9.5 billion in 2026 to USD 22.2 billion by 2033, at a CAGR of 12.8% from 2026 to 2033. The North America market held the largest share of 42.7% of the global market in 2025, driven by increasing demand for biologics, progress in AI and robotics, and the need for regulatory compliance.

Key Market Trends & Insights
- By product & services: The process automation hardware segment held the highest market share of 31.4% in 2025.
- Based on process stage: Upstream held the highest market share in 2025.
- By end use: The pharmaceutical & biotech manufacturers segment held the highest market share in 2025.
Regional Highlights
- Largest regional market: North America (42.7% revenue share, 2025)
- Fastest-growing regional market: Asia Pacific (highest CAGR, 2026-2033)
- The bioprocess automation industry in the U.S. held the largest revenue share in 2025.
Market Size & Forecast
- Market size in 2025: USD 8.6 Billion
- Estimated market size in 2026: USD 9.5 Billion
- Projected market size by 2033: USD 22.2 Billion
- CAGR (2026-2033): 12.8%
Rising Demand for Biologics
The rising demand for biologics is among the most significant drivers of the bioprocess automation industry. Over the past decade, there has been a rapid expansion of monoclonal antibody development and vaccine production, gene therapy development, and all other biologic products because these treatments effectively address complicated medical conditions, which include cancer, autoimmune disorders, and rare genetic diseases. For these biologics to remain effective and to maintain high quality, manufacturing procedures must be extremely controlled, scalable, and precise. There is a significant need for fully automated bioprocess solutions because traditional manual or semi-automated production methods are frequently labor-intensive, prone to mistakes, and inadequate to meet the expanding market demand.
Correlation of total drug approvals vs. biologics approvals by the FDA
Year
Total Drugs Approved (Biologics and NCEs)
Biologics Approved
2023
55
17
2022
37
15
2021
50
14
2020
53
15
2019
48
12
2018
59
17
2017
46
13
2016
22
7
2015
45
13
Total
415
123
Source: Biomedicines Secondary Research, Grand View Research
Bioprocess automation is becoming essential as healthcare increasingly relies on biologics. It optimizes workflows, enhances consistency, boosts throughput, and reduces costs, variability, and production time using robotics, AI, and advanced monitoring.
Need for Efficiency & Cost Reduction
The need for cost and efficiency reduction largely drives the market for bioprocess automation. Manufacturing biopharmaceuticals is a complicated and resource-intensive process that frequently requires highly qualified staff, careful observation, and strict protocol adherence. The manual and semi-automated processes, which need extensive human effort, lead to operational errors and result in production problems, including batch failures, product defects, and increased production costs. The implementation of automated systems enables businesses to achieve higher productivity levels while reducing their need for human workers and decreasing operational mistakes, which results in more reliable production outcomes.
Automation increases productivity, drastically reduces operating costs, and improves reliability. Manufacturers can more effectively meet growing market demand thanks to automated bioprocessing systems' ability to run continuously with little downtime, optimize resource usage, and speed up production cycles. In an increasingly cost-sensitive healthcare environment, the ability to produce high-quality biologics at a lower cost also helps businesses stay competitive. As a result, the need for cost-effectiveness and operational efficiency keeps driving global investments in bioprocess automation technologies.
Market Concentration & Characteristics
The degree of innovation in the bioprocess automation industry is a key driver shaping its rapid growth. The combination of robotics, artificial intelligence (AI), machine learning, and Internet of Things (IoT) technologies creates advanced systems that can monitor and manage intricate bioprocess operations. These developments lower variability and enhance product quality by allowing adaptive control, process optimization, and predictive analytics.
The bioprocess automation industry sees strong M&A activity as companies acquire technology firms and startups to enhance AI, robotics, and digital capabilities. For instance, in September 2025, Yokogawa Corporation and Repligen launched their partnership, integrating OpreX Bio Pilot with MAVERICK to enhance automated glucose and lactate monitoring in bioprocessing. As a result of the industry's drive for integrated and end-to-end solutions, M&A is now positioned as a strategic tool to boost innovation and preserve competitiveness in the quickly changing bioprocessing market.
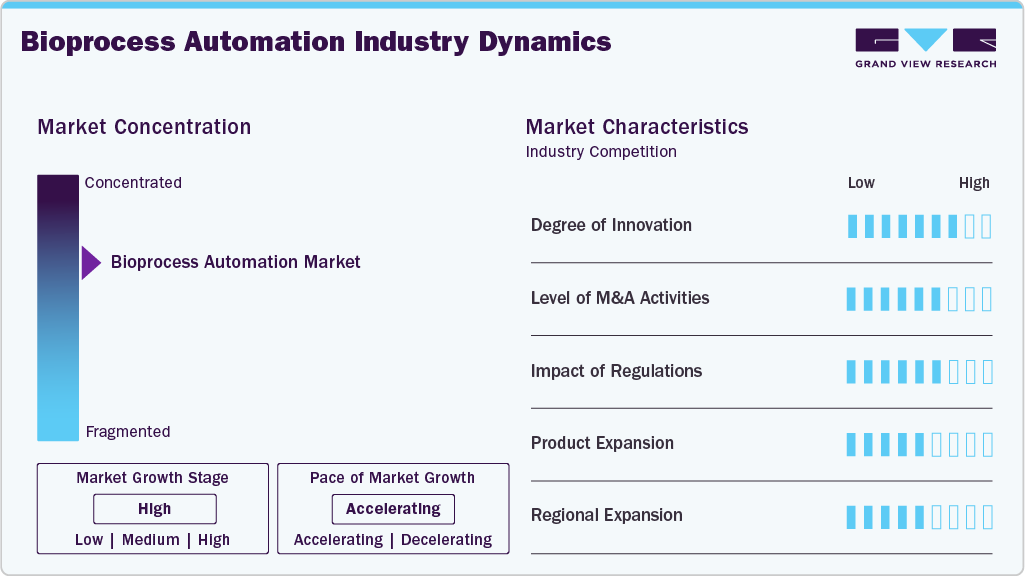
Regulations heavily influence bioprocess automation, with agencies like the FDA and EMA enforcing GMP to ensure safety and quality. Automation enhances compliance by improving traceability, reducing errors, and providing accurate, validated data for faster approvals.
Product expansion is a key driver in the bioprocess automation industry, as companies continuously develop advanced systems to meet the evolving needs of biopharmaceutical manufacturing. Numerous companies are also growing their product lines with AI-powered software, single-use devices, and end-to-end automation platforms designed for biologics, vaccines, and cell and gene therapies. Continuous product development allows manufacturers to enhance efficiency, cut costs, and speed up product launches.
Regional expansion is a key driver in bioprocess automation, with companies establishing facilities and partnerships in Asia-Pacific, Latin America, and the Middle East to access emerging markets, diversify revenue, and boost the adoption of automated solutions.
Product & Services Insights
In 2025, the process automation hardware segment led the market with 31.4% share, driven by high-performance sensors, controllers, and monitoring systems for near-real-time bioprocess control. Furthermore, the strong push of companies investing in revitalizing manufacturing infrastructure and leading to smart factories only serves to further the constant dominance across biopharmaceutical and biotechnological facilities.
The software and control systems segment is expected to grow at the fastest CAGR over the forecast period, because of the growing consumption of digital bioprocessing systems, real-time monitoring systems, and advanced analytics solutions that support process improvement and regulatory compliance. Integrating AI, machine learning, and cloud-based systems further drives demand by enabling predictive maintenance, automated decision-making, and optimization of production workflows.
Process Stage Insights
The upstream segment dominated the market in 2025 with a share of 35.0%, owing to the increasing demand for efficient cell culture, higher yield production, and advanced automation in biopharmaceutical manufacturing. The market expansion is further driven by the growing use of single-use bioreactors and real-time monitoring systems, together with the development of scalable solutions that improve process efficiency while minimizing contamination risks and enabling fast tracking of biologics and vaccine development across the globe.
The quality control and PAT segment is anticipated to experience the fastest CAGR during the forecast period due to the increasing focus on regulatory compliance, real-time product monitoring, and consistent process validation. The growing use of PAT tools is supposed to help manufacturers improve product quality, reduce batch failures, and allow for economical production at scale. In addition, the increased complexity of biologics and cell and gene therapies is further propelling the demand for quality control.
End Use Insights
The pharmaceutical & biotech manufacturers segment held the largest share of 36.3% in 2025, driven by growing demand for automated bioprocessing systems that ensure large-scale, cost-efficient, and compliant production of biologics, vaccines, and advanced therapies.
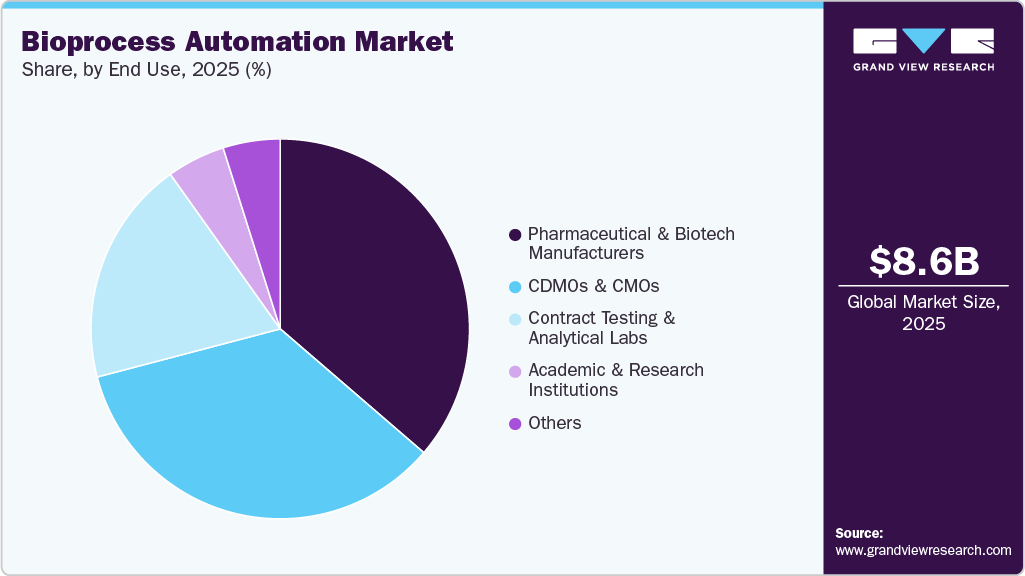
The CDMOs & CMOs segment is projected to grow at the fastest CAGR during the forecast period, driven by the recent industry trend of moving bioprocessing functions into specialized service providers to save costs, build capacities, and accelerate time-to-market. Moreover, growing demand for more flexible styles of manufacturing and increased collaborations between pharmaceutical innovators with CMOs/CDMOs will drive the market.
Regional Insights
North America bioprocess automation market dominated the global market with a share of 42.7% in 2025, supported by advanced healthcare infrastructure, strong R&D investment, and the presence of leading biopharmaceutical companies. Automation technology adoption has been accelerated by the region's established supply networks and a growing emphasis on precision medicine and the production of cell and gene therapies. Moreover, the need for effective, scalable, and compliant bioprocess solutions will support revenue growth in the forecasted period due to the growing elderly population and the rising disease prevalence.
U.S Bioprocess Automation Market Trends
The bioprocess automation market in the U.S. is highly competitive because biopharmaceutical companies invest heavily in research and development, and their regenerative medicine capabilities and their biologics pipeline continue to expand. The manufacturing process for cell and gene therapy becomes more efficient through the rapid implementation of artificial intelligence, robotic technology, and process control systems. For instance, in May 2025, Made Scientific partnered with Orchestra Life Sciences to expand its Princeton, New Jersey, facility by 12,000 square feet to meet late-phase and commercial demand.
Europe Bioprocess Automation Market Trends
The bioprocess automation market in Europe is growing steadily, driven by government funding, Horizon Europe programs, and increasing adoption of digital biomanufacturing. Strict EMA compliance and expanding portfolios from players like Sartorius, Merck KGaA, and Cytiva, along with rising cell therapy and biobanking adoption, strengthen the region’s leadership.
The UK bioprocess automation market is growing due to strong biomedical research, academic-industry collaboration, and leadership in gene therapy manufacturing. Regulatory oversight by the MHRA and investments in the UK Biobank program have accelerated automation adoption. For instance, in July 2025, Canada-UK partners launched the USD 2 million BALANCE project, an AI-powered bioreactor collaboration to enhance biologics manufacturing efficiency and scalability.
The German bioprocess automation market exists because of its strong pharmaceutical industry, along with research institutes and government innovation programs. The combination of precision medicine development and regenerative therapy research leads to digital twin technology and automated bioreactor systems. The Paul-Ehrlich-Institut regulations and the development of scalable solutions establish Germany as a European center for bioprocessing operations.
Asia Pacific Bioprocess Automation Market Trends
The bioprocess automation market in the Asia Pacific is projected to grow at the fastest CAGR of over 15.3% during the forecast period, driven by government initiatives, cost-effective labor, and rapid biologics manufacturing expansion. Countries including China, India, Japan, and South Korea are increasing investments in biopharma and automation technologies to expand capacity for producing monoclonal antibodies, vaccines, and personalized medicines.
China bioprocess automation market is rapidly emerging as a global leader, driven by “Made in China 2025,” evolving NMPA regulations, and rising adoption of GMP-compliant systems, supported by expanding cell therapy pipelines and large-scale biomanufacturing.
Japan’s bioprocess automation market remains a leader in regenerative medicine and iPSC research, driving adoption of robotics, process monitoring, and digital control platforms. Supportive AMED policies and favorable PMDA regulations further accelerate automation across academic and industrial labs.
MEA Bioprocess Automation Market Trends
The bioprocess automation market in the MEA is at an early stage but shows strong potential, supported by growing investments in biomanufacturing facilities, medical research, and healthcare infrastructure. Countries such as the UAE, Saudi Arabia, and South Africa are adopting automated platforms through government programs and public-private partnerships, with rising demand for precision therapies and advanced manufacturing driving steady bioprocess automation growth.
Kuwait bioprocess automation marketis a developing industry where automation adoption is fueled by healthcare modernization, growing biomedical research initiatives, and increasing awareness of advanced therapies. Government investments in healthcare digitization and partnerships with international bioprocess technology suppliers make GMP-compliant automation systems possible. Although still nascent, Kuwait is positioning itself as a growing contributor to the regional market.
Key Bioprocess Automation Company Insights
The market is dominated by a few established players that leverage strong product portfolios, strategic partnerships, and continuous R&D investment. Companies such as Thermo Fisher Scientific, Sartorius, Parker Hannifin, Danaher, Merck KGaA, and Eppendorf lead through advanced automation technologies, integrated platforms, and global distribution networks supporting large-scale biomanufacturing and research.
Companies such as SIA Bioreactors.net, Senieer, and Getinge are expanding their presence by offering modular, customizable automation platforms that support scalable, efficient, and compliant bioprocessing for research institutions and biopharma, cell, and gene therapy developers.
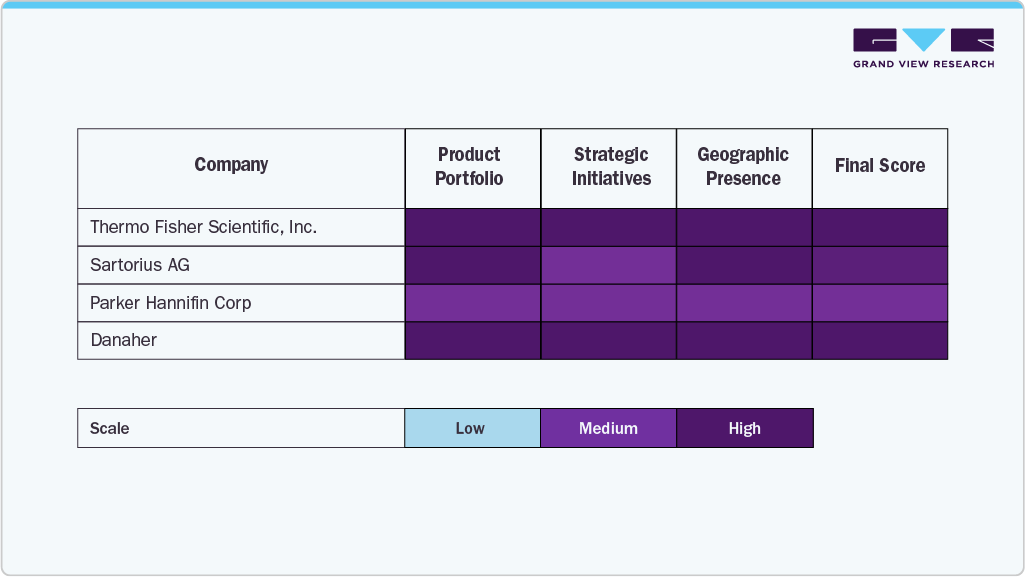
The market is growing rapidly, driven by strategic M&A, partnerships, and technological innovation. Companies that integrate advanced automation with customer-centric solutions are well-positioned to enhance global biopharmaceutical manufacturing efficiency, compliance, and scalability.
Key Bioprocess Automation Companies:
The following key companies have been profiled for this study on the bioprocess automation market.
- Thermo Fisher Scientific Inc.
- Sartorius AG
- Parker Hannifin Corp
- Danaher
- Merck KGaA
- Eppendorf SE
- SIA Bioreactors.net
- Senieer
- Getinge
- Repligen Corporation
Recent Developments
-
In January 2026, Brooks Instrument launched a non-invasive clamp-on ultrasonic flow meter with integrated EtherNet/IP, enabling sterile, real-time, and easily integrated flow measurement for upstream and downstream bioprocessing applications.
“The BCU Series redefines what’s possible for flow measurement in biopharma production. It’s the first clamp-on ultrasonic flow meter in its class with built-in EtherNet/IP, giving bioprocess engineers real-time visibility, advanced diagnostics, and plug-and-play integration with automated skid systems.”
- Steve Kannengieszer, global marketing director at Brooks Instrument
-
In June 2025, Sartorius Stedim Biotech completed a major expansion at its Aubagne, France headquarters, nearly doubling cleanroom and lab space, adding automated production and logistics systems, and enhancing R&D for innovative bioprocess solutions.
“By investing in innovation, automation, and sustainability, we are strengthening our ability to meet the evolving needs of our customers while ensuring our competitive positioning and preparing for future growth. This emphasizes our commitment to simplify and accelerate progress in bioprocessing with advanced technologies, enabling new and better therapies to be manufactured faster, more efficiently, and more sustainably.”
- René Fáber, CEO of Sartorius Stedim Biotech.
-
In December 2024, Getinge partnered with 908 Devices in Sweden to integrate MAVEN with Getinge bioreactors, enabling continuous, real-time monitoring and automated control of glucose and lactate in cell cultures.
Bioprocess Automation Market Report Scope
Report Attribute
Details
Market size in 2025
USD 8.6 billion
Estimated market size in 2026
USD 9.5 billion
Projected market size by 2033
USD 22.2 billion
Growth rate
CAGR of 12.8% from 2026 to 2033
Historical data
2021 - 2025
Forecast period
2026 - 2033
Quantitative units
Revenue in USD million/billion and CAGR from 2026 to 2033
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Product & service, process stage, end use, region
Regional scope
North America; Europe; Asia Pacific; Latin America; MEA
Country scope
U.S.; Canada; Mexico; UK; Germany; France; Italy; Spain; Denmark; Sweden; Norway; India; China; Japan; Australia; South Korea; Thailand; Brazil; Argentina; Saudi Arabia; UAE; South Africa; Kuwait
Key companies profiled
Thermo Fisher Scientific Inc.; Sartorius AG; Parker Hannifin Corp; Danaher; Merck KGaA; Eppendorf SE; SIA Bioreactors.net; Senieer; Getinge; Repligen Corporation
Customization scope
Free report customization (equivalent up to 8 analyst’s working days) with purchase. Addition or alteration to country, regional & segment scope.
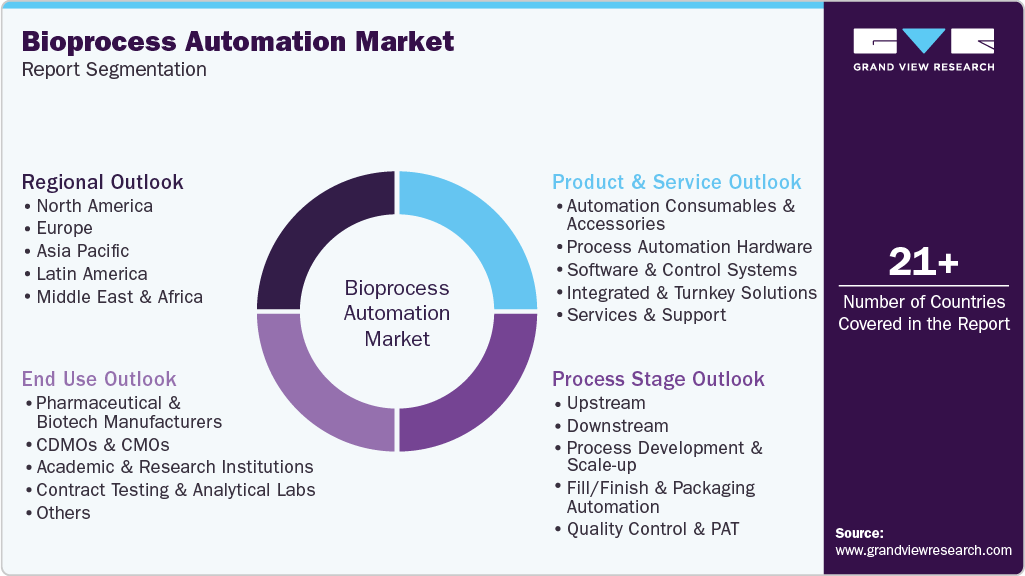
Global Bioprocess Automation Market Report Segmentation
This report forecasts revenue growth and provides an analysis of the latest trends in each of the sub-segments from 2021 to 2033. For this report, Grand View Research has segmented the global bioprocess automation market based on product & service, process stage, end use, and region:
-
Product & Service Outlook (Revenue, USD Million, 2021 - 2033)
-
Automation Consumables & Accessories
-
Process Automation Hardware
-
Software & Control Systems
-
Integrated & Turnkey Solutions
-
Services & Support
-
-
Process Stage Outlook (Revenue, USD Million, 2021 - 2033)
-
Upstream
-
Downstream
-
Process Development & Scale-up
-
Fill/Finish & Packaging automation
-
Quality Control & PAT
-
-
End Use Outlook (Revenue, USD Million, 2021 - 2033)
-
Pharmaceutical & biotech manufacturers
-
CDMOs & CMOs
-
Academic & research institutions
-
Contract testing & analytical labs
-
Others
-
-
Regional Outlook (Revenue, USD Million, 2021 - 2033)
-
North America
-
U.S.
-
Canada
-
Mexico
-
-
Europe
-
Germany
-
UK
-
France
-
Italy
-
Spain
-
Denmark
-
Sweden
-
Norway
-
-
Asia Pacific
-
China
-
India
-
Japan
-
South Korea
-
Australia
-
Thailand
-
-
Latin America
-
Brazil
-
Argentina
-
-
Middle East and Africa (MEA)
-
South Africa
-
Saudi Arabia
-
UAE
-
Kuwait
-
-
Frequently Asked Questions About This Report
North America dominated with a 42.7% revenue share in 2025.
Asia Pacific is the fastest-growing region over the forecast period.
The upstream held the largest revenue share in 2025, while software and control systems is the fastest-growing segment.
The pharmaceutical & biotech manufacturers segment held the largest share of 36.3% in 2025, while CDMOs & CMOs is the fastest-growing segment.
Some of the key players operating in the bioprocess automation market are Thermo Fisher Scientific Inc.; Sartorius AG; Parker Hannifin Corp; Danaher; Merck KGaA; Eppendorf SE; SIA Bioreactors.net; Senieer; Getinge; Repligen Corporation.
The market is driven by rising demand for biologics, advancements in AI and robotics, and the need for regulatory compliance and efficiency in the bioprocessing industry.
The global bioprocess automation market size was estimated at USD 8.6 billion in 2025 and is projected to reach USD 9.5 billion by 2026
The global bioprocess automation market size was estimated is projected to reach USD 22.2 billion by 2033, growing at a CAGR of 12.8% from 2026 to 2033,
In 2025, the process automation hardware segment was the market leader with 31.40% share, as it has witnessed extensive adoption of high-performance sensors, controllers, and monitoring systems, to achieve an accuracy of control for bioprocesses for near-real-time circumstances.
About the authors:
Author: GVR Biotechnology Research Team | Last Updated:
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.









