- Home
- »
- Healthcare IT
- »
-
Drug Reference Apps Market Size, Industry Report, 2033GVR Report cover
![Drug Reference Apps Market Size, Share & Trends Report]()
Drug Reference Apps Market (2025 - 2033) Size, Share & Trends Analysis Report By Application, By Pricing Model (Freemium (Free), Paid (Subscription)), By Device (Smartphones, Tablets), By End Use (Patients, Pharmacists), By Region, And Segment Forecasts
- Report ID: GVR-4-68040-770-0
- Number of Report Pages: 150
- Format: PDF
- Historical Range: 2021 - 2024
- Forecast Period: 2025 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
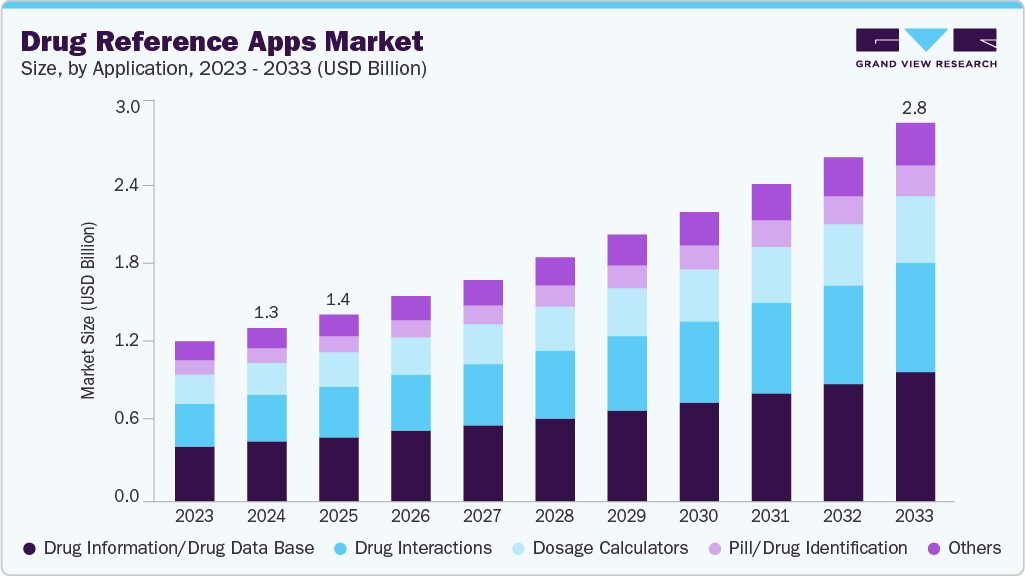
Drug Reference Apps Market Summary
The global drug reference apps market size was estimated at USD 1.30 billion in 2024 and is projected to reach USD 2.84 billion by 2033, growing at a CAGR of 9.17% from 2025 to 2033. Increasing adoption of reminders & adherence features among chronic disease patients is anticipated to boost market growth.
Key Market Trends & Insights
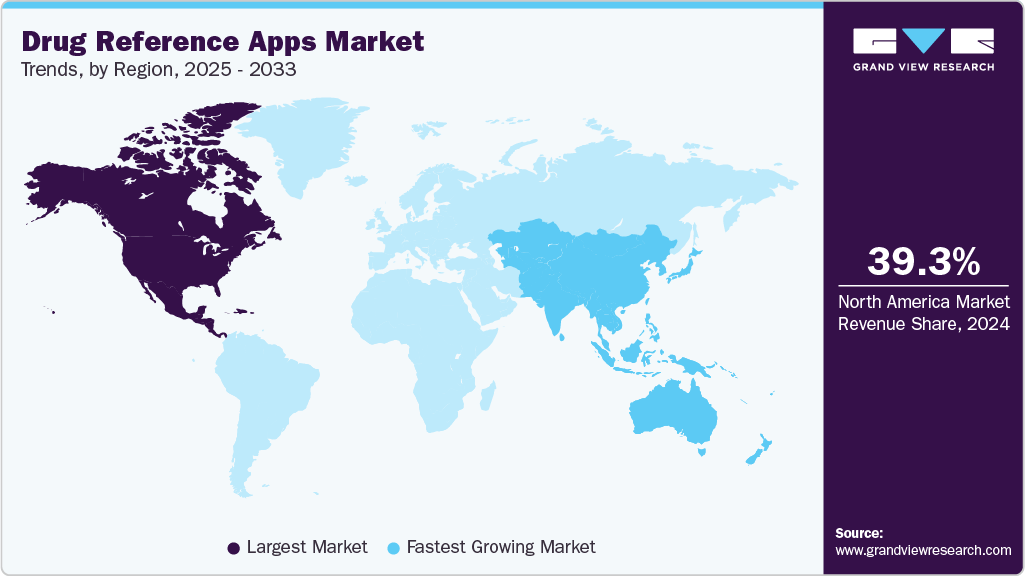
- The North America drug reference apps market dominated globally in 2024, accounting for the largest revenue share of 39.33%.
- The Canada drug reference apps industry is anticipated to register the fastest growth rate during the forecast period.
- In terms of application, the drug information/drug database segment held the largest revenue share in 2024.
- In terms of the pricing model, the freemium (free) segment held the largest revenue share in 2024.
- In terms of the device, the smartphones segment held the largest revenue share in 2024.
- In terms of the end use, the healthcare professionals segment held the largest revenue share in 2024.
Market Size & Forecast
- 2024 Market Size: USD 1.30 Billion
- 2033 Projected Market Size: USD 2.84 Billion
- CAGR (2025-2033): 9.17%
- North America: Largest market in 2024
- Asia Pacific: Fastest growing market
According to a JMIR Publications article published in April 2025, a systematic review analyzed nine studies involving 1,023 patients with chronic kidney disease (CKD) and found that 5 out of the nine mobile applications assessed demonstrated a statistically significant positive impact on medication adherence.These apps employed features such as automated reminders, patient education modules, gamification elements, and medication monitoring tools. This evidence underscores the growing influence of (DRA) in enhancing adherence, supporting safer medication management, and strengthening its role as an essential tool in digital healthcare for chronic disease patients.
Rising clinical use of smartphones and apps for drug reference drives the growth of the market. According to an NCBI article published in March 2023, a review of smartphone and mobile app use among physicians found that in many settings, doctors routinely use their phones not just for communication or medical education but also to access drug compendia and clinical decision tools.
Healthcare is undergoing a rapid digital transformation, shifting from paper-based systems to fully integrated digital platforms such as Electronic Health Records (EHRs), Electronic Medical Records (EMRs), and Clinical Decision Support Systems (CDSS). This transformation enables healthcare providers to access critical patient information in real time. Drug reference apps are increasingly being embedded within these digital systems, allowing clinicians to instantly access up-to-date information on drug interactions, dosing guidelines, contraindications, and side effects. For instance, hospitals integrating apps such as Micromedex or Lexicomp into their EHR systems enable physicians and pharmacists to access drug data directly while reviewing a patient’s medical record, significantly improving workflow efficiency and reducing errors. This trend is impactful in large hospitals and multi-specialty clinics, where rapid access to accurate drug information is crucial for patient safety and care quality.
Furthermore, patient safety is a factor driving the adoption of drug reference apps. Medication errors remain one of the leading causes of preventable hospital admissions and adverse drug events worldwide. Drug reference apps provide real-time, evidence-based information to help clinicians avoid prescribing errors, such as incorrect dosing, contraindicated medications, or dangerous drug interactions. They offer alerts for potential adverse effects and provide detailed information on drug administration and patient-specific considerations. For instance, a physician prescribing multiple medications for a cardiac patient can use a reference app to check for drug-drug interactions and adjust prescriptions accordingly. By minimizing risks associated with medication errors, these apps play a critical role in improving clinical outcomes and enhancing overall patient safety.
The rise of telemedicine and remote healthcare has driven demand for mobile drug reference apps, enabling physicians to access comprehensive drug databases during virtual consultations for safe and accurate prescribing. These apps allow clinicians to verify medications, check interactions, and suggest alternatives without being physically present, which is particularly valuable in rural or underserved areas. Incorporating artificial intelligence (AI) and machine learning further enhances functionality by analyzing patient-specific data such as age, comorbidities, and concurrent medications to provide personalized recommendations and predict adverse reactions. AI-driven alerts for uncommon drug interactions ensure up-to-date, evidence-based guidance, reducing errors, improving outcomes, and making these apps a trusted tool in both clinical practice and medical education.
Moreover, according to a Taylor & Francis Online article published in December 2024, patients were surveyed about how they perceived their physicians’ smartphone use in consultations. Its findings indicated that when doctors used smartphones in a clearly professional way (e.g., looking up references or data), patient satisfaction was less negatively affected than when use seemed casual or unrelated. This reflects how smartphones are increasingly embedded in clinical workflow, including for lookup, rather than being seen as distractions. These insights illustrate how the normalization of smartphones in clinical settings has created a natural pathway for adopting (DRA). Since most physicians already carry and rely on these devices, integrating specialized applications that provide immediate access to reliable information represents a seamless evolution in clinical workflow, supporting safer prescribing practices and more informed patient care.
Case Study: Implementing the "Kidney Health" Mobile App to Improve Medication Adherence in Chronic Kidney Disease Patients.
Case: Chronic Kidney Disease (CKD) patients often face challenges in adhering to prescribed medication regimens, which can lead to disease progression and increased healthcare costs. A 2025 randomized controlled trial aimed to evaluate the effectiveness of a pharmacist-led mobile application, "Kidney Health," designed to enhance medication adherence among CKD patients in May 2025.
Study Design:
The study was conducted at a single-center facility and involved 86 CKD patients. Participants were randomly assigned to two groups:
-
Control Group: Received standard care without the mobile app.
-
Intervention Group: Utilized the "Kidney Health" app, which provided medication reminders, educational content, and direct communication with pharmacists.
Findings: The intervention group demonstrated a significant improvement in medication adherence compared to the control group. Patients using the app showed better understanding and management of their medication schedules, leading to more consistent adherence. Additionally, the app facilitated timely interventions by pharmacists, addressing potential issues before they became critical.
Conclusion: The "Kidney Health" mobile application proved to be an effective tool for improving CKD patients' medication adherence. This case underscores the potential of digital health solutions in managing chronic conditions and highlights the importance of integrating technology into patient care strategies.
Key Funding & Investment Activities in the Drug Reference Apps Market
Instance
Funding & Investment
In October 2024
Suki, a healthcare startup focusing on AI assistants, secured USD 70 million in a Series D funding round led by Hedosophia, with participation from Venrock and March Capital. The company aims to alleviate administrative burdens for healthcare providers through its AI-powered tools, potentially integrating drug reference functionalities to streamline clinical workflows.
In October 2024
Glooko, a digital platform for managing chronic conditions like diabetes, raised USD 100 million in Series F funding. The company plans to expand its product offerings, including data insights and device integrations, which could encompass drug reference functionalities to support medication management.
In September 2024
ACTO, an AI-powered Intelligent Field Excellence platform, announced a USD 10 million investment in Generative AI to enhance healthcare provider engagement in the life sciences industry. This initiative aims to improve the "human touch" in omnichannel engagement strategies.
Technological advancements drive the growth of the market. For instance, in May 2025, Pathway, a medical AI platform, recently introduced a drug reference tool that checks for drug-drug interactions. It gives healthcare professionals access to detailed information on more than 2,000 medications and lets them review potential interactions without leaving their usual clinical interface. This feature helps clinicians make safer prescribing decisions and supports more efficient patient care by providing real-time, evidence-based insights.

Moreover, in March 2024, DrugGPT, developed by Oxford University, was developed as an AI tool designed to assist healthcare professionals in prescribing medications accurately. DrugGPT recommends drugs, flags potential adverse effects, and provides explanations with supporting research and guidelines by evaluating patient conditions entered into its chatbot. This tool aims to reduce medication errors and improve patient adherence by offering instant, evidence-based second opinions during prescribing.
Market Concentration & Characteristics
The drug reference app industry is experiencing significant innovation by integrating AI-powered features that enhance clinical decision-making. In September 2025, Epocrates introduced a conversational AI assistant within its app, enabling clinicians to obtain concise, conversational answers about prescription medications. This feature aims to streamline the prescribing process by providing quick, evidence-based information, thereby improving efficiency and reducing the risk of medication errors.
The DRA industry is characterized by the leading players' high merger and acquisition (M&A) activity. This is due to several factors, including the desire to expand the business to cater to the growing demand for solutions. In December 2024, Bayer announced its acquisition of HiDoc Technologies, a digital health company specializing in gastrointestinal disorders. As part of the deal, Bayer plans to commercialize HiDoc's Cara Care app in Spring 2025. The Cara Care app offers personalized, evidence-based support for individuals managing irritable bowel syndrome (IBS), providing users with tools to track symptoms, receive dietary recommendations, and access educational content.
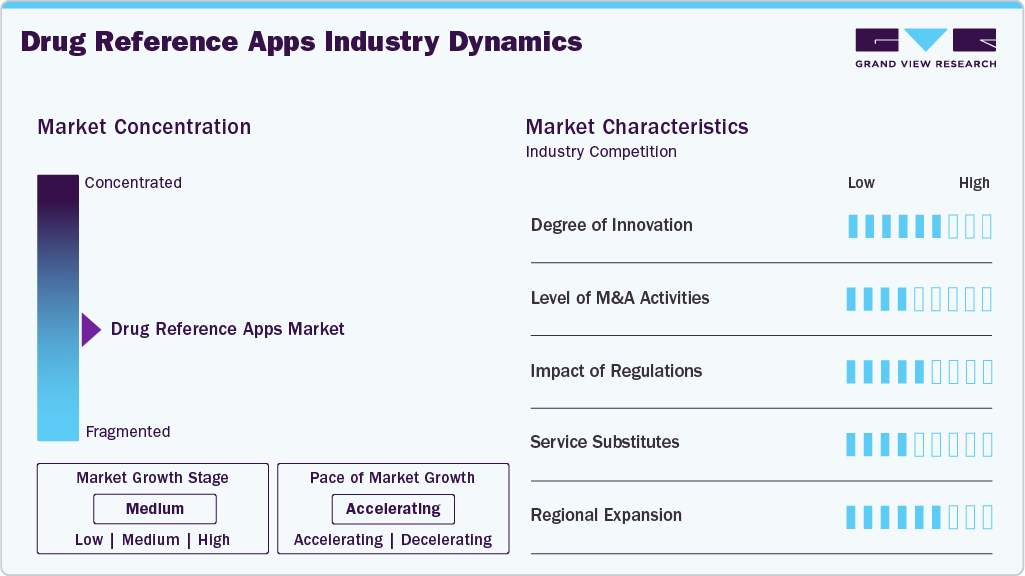
Regulatory compliance is critical in the drug reference app (DRA) industry to ensure patient safety, data security, and adherence to clinical standards. For instance, in February 2025, the U.S. Food and Drug Administration (FDA) issued draft guidance titled “Considerations for the Use of Artificial Intelligence to Support Regulatory Decision Making for Drug and Biological Products.” This guidance provides recommendations on using AI in drug development, emphasizing the need for rigorous validation and secure data handling. Such regulations encourage drug reference app developers to invest in high-quality technologies and continuous monitoring, ultimately supporting improved patient outcomes and wider adoption in healthcare systems.
In the (DRA) industry, service substitutes refer to alternative approaches or technologies that complement or replace traditional printed drug guides. For example, mobile units equipped with AI-driven diagnostic apps or remote consultations can, in specific scenarios, reduce reliance on hospital-based interventions. These substitutes offer faster assessment, timely treatment, and enhanced accessibility for patients in rural or underserved areas, while driving innovation and adoption of drug reference solutions across healthcare systems.
Several industry players are expanding their business by entering new geographical regions to strengthen their market position and service portfolio. Rising service launches create more opportunities for market players to enter new areas.
Application Insights
By application, the drug information/drug database segment dominated the market in 2024 and accounted for the largest revenue share of 34.37%. The growing trend of centralized, evidence-based digital resources in healthcare, enabling clinicians to access reliable medication information quickly and improving patient safety, drives the market growth. For instance, in May 2025, Purdue University's Center for Medication Safety launched the first comprehensive open-access database of all clinically tested drugs. This innovative platform aims to provide healthcare professionals with a centralized resource for evidence-based information on medications, enhancing clinical decision-making and patient safety. The database is designed to be user-friendly and accessible, promoting transparency and collaboration in the medical community.
The drug interactions segment in the market is anticipated to witness the fastest CAGR over the forecast period. Integrating drug interaction checkers into medication management platforms enhances patient safety by providing real-time alerts for potential drug interactions. For instance, in August 2025, mySeniorCareHub introduced a Drug Interaction Checker feature within its platform tailored for seniors and caregivers. This tool assists users in identifying potential interactions between prescription and over-the-counter medications, thereby improving medication safety and adherence. Designed with user-friendliness, it enables easy checking of interactions, contributing to safer medication management for the elderly population.
Pricing Model Insights
By pricing model, the freemium (free) segment dominated the market in 2024 and accounted for the largest revenue share of 64.88%. The freemium model is increasingly popular in the market, offering basic medication information for free while providing optional premium features for advanced clinical support. For instance, several leading DRA continue to leverage the freemium model. Epocrates offers free access to essential drug information, dosing, interactions, and contraindications, with premium upgrades for additional clinical tools. Medscape also provides a freemium model, giving free access to drug monographs, medical news, and treatment guidelines, while advanced analytics and CME resources are part of the paid tier. Similarly, Drugs.com allows users free access to a comprehensive drug database, interaction checker, and pill identifier, with subscription options for enhanced decision-support features.
The paid (subscription) segment in the market is anticipated to witness the fastest CAGR over the forecast period. Subscription-based pricing models are prevalent in the DRA market, offering healthcare professionals comprehensive, evidence-based drug information and clinical decision support tools. Several key DRA adopted subscription-based pricing models to provide healthcare professionals with in-depth, evidence-based drug information. For instance, UpToDate offers Lexidrug, a comprehensive drug reference tool that provides clinicians with evidence-based information to support medication decisions. This platform is available through individual and institutional subscriptions, catering to various healthcare settings. Moreover, Wolters Kluwer's Lexicomp provides a subscription-based service offering detailed drug monographs, clinical decision support, and patient education materials. These platforms enhance clinical decision-making by providing reliable and up-to-date drug information.
Device Insights
By device, the smartphones segment dominated the market in 2024 and accounted for the largest revenue share of 67.28%. Smartphone-based (DRA) systems increasingly integrate advanced features like AI-driven clinical decision support and real-time medication tracking to enhance healthcare professionals' efficiency and patient safety. For instance, in September 2025, True Rx Health Strategists launched the True Rx+ mobile app, which helps patients manage pharmacy benefits, compare medication prices, and live chat with a Health Strategist. The app offers a personalized dashboard, real-time medication prices across local pharmacies, prior authorization status tracking, and one-on-one health guidance, providing patients with comprehensive support for their medication management needs.
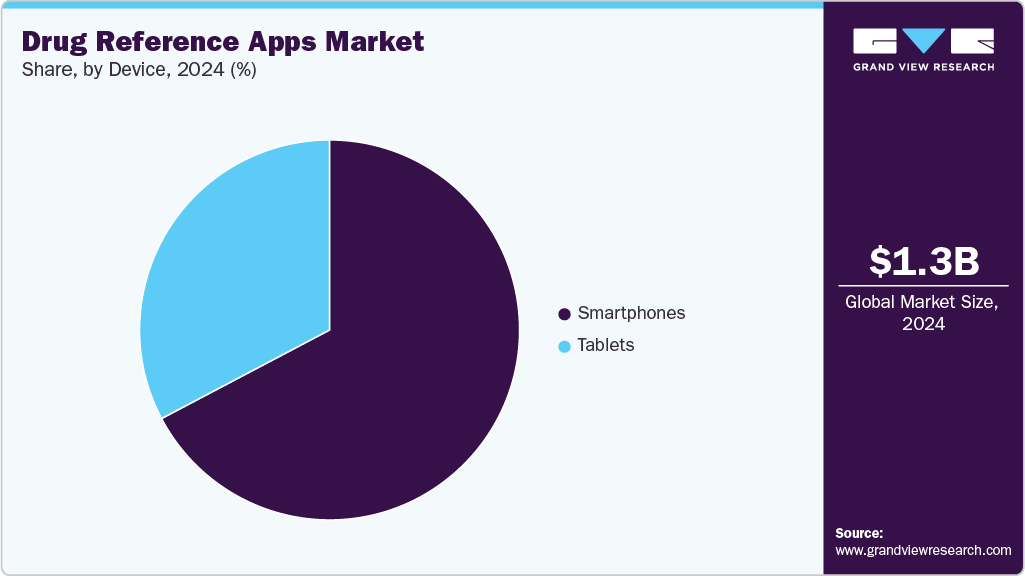
The tablets segment in the DRA market is anticipated to witness the fastest CAGR over the forecast period. Tablet-based (DRA) systems increasingly integrate advanced features like AI-driven clinical decision support and real-time medication tracking to enhance healthcare professionals' efficiency and patient safety. For instance, in August 2025, the Pill Identifier & Med Scanner app for iOS launched a significant update (v2.3), introducing key features such as camera-based pill identification, a barcode scanner for medication packaging, and detailed drug information retrieval, including dosage, side effects, and interactions. Optimized for tablet use, this update provides a more user-friendly interface, enabling faster and easier access to pill data, thereby enhancing medication safety and management.
End Use Insights
The healthcare professionals segment dominated the market in 2024 with a revenue share of 40.54%. Healthcare professionals are increasingly adopting advanced (DRA) that integrate AI-driven clinical decision support, real-time medication tracking, and personalized treatment recommendations to enhance patient safety and streamline clinical workflows. For instance, in September 2025, Ram Manohar Lohia Institute of Medical Sciences (RMLIMS) in Lucknow introduced a QR code system to simplify the reporting of Adverse Drug Reactions (ADRs). This initiative allows patients and healthcare professionals to instantly access an online platform for ADR reporting via a smartphone scan, simplifying the previously cumbersome process involving complex websites and paper forms. The system is part of a broader national effort to modernize drug safety protocols and enhance the speed and efficiency of ADR data collection.
The patient segment of the market is anticipated to register the fastest growth rate over the forecast period. Patient-focused (DRA) is evolving to offer real-time medication tracking, personalized health insights, and AI-driven support, empowering individuals to manage their health more effectively. For instance, in March 2025, the Utsarjan app was introduced to assist children with nephrotic syndrome manage their condition. This app enables real-time communication with healthcare providers, tracks vital health parameters like blood pressure and urine protein levels, and provides medication reminders. By offering a graphical overview of a patient's health, Utsarjan aims to improve medication adherence and prevent relapses, especially for patients in rural areas with limited access to specialists.
Regional Insights
North America drug reference apps industry dominated the global DRA market in 2024 and accounted for the largest revenue share of 39.33% owing to a well-established healthcare infrastructure and high healthcare expenditure. In North America, DRA increasingly integrates AI-driven clinical decision support, real-time medication tracking, and personalized treatment recommendations to enhance patient safety and streamline clinical workflows. For instance, in January 2025, CVS Health launched a new health app that replaces its previous CVS Pharmacy app. The new platform allows users to manage subscriptions across all CVS brands, access health spending and benefit details, and schedule immunizations. It also features an AI-powered search and personalized health recommendations.
U.S. Drug Reference Apps Market Trends
The drug reference apps industry in the U.S. held the largest share, 82.21%, in the North American region in 2024. In the U.S., DRA is increasingly integrating AI-driven clinical decision support, real-time medication tracking, and personalized treatment recommendations to enhance patient safety and streamline clinical workflows. For instance, in August 2025, Blue Shield of California launched a first-of-its-kind digital experience that lets members know when their doctor initiates an e-prescription order. The mobile push notifications prompt patients to view drug prices and compare alternatives, including lower-cost options. This initiative is part of Blue Shield's Pharmacy Care Reimagined program, aimed at improving medication choice and transparency.
Canada drug reference apps market is anticipated to register the fastest growth rate during the forecast period, driven by the growing reliance on mobile platforms for quick and accurate drug information, which enhances medication safety, reduces errors, and improves patient care. Integrating artificial intelligence (AI) technologies is also pivotal in this transformation, offering personalized treatment recommendations and real-time medication tracking. These advancements are particularly beneficial in rural and underserved areas, where access to healthcare professionals may be limited.
Europe Drug Reference Apps Market Trends
Europe drug reference apps industry is anticipated to register a significant growth rate during the forecast period. Increasing digital health solutions, emphasizing integrating artificial intelligence (AI) and machine learning (ML) to enhance clinical decision-making and medication safety. For instance, in June 2025, in collaboration with Click Therapeutics, Otsuka Pharmaceutical launched Rejoyn, a prescription-only smartphone app in Great Britain to treat depressive disorder. Unlike traditional cognitive behavioral therapy-based technologies, Rejoyn employs an interactive cognitive-emotional and behavioral therapeutic approach, designed to increase cognitive control over emotions. The app has demonstrated efficacy in reducing depressive symptoms within a six-week treatment program, with continued improvements observed at week 10 in the MIRAI clinical study.
Germany drug reference apps market is anticipated to grow considerably during the forecast period. In Germany, the digital health landscape is evolving, with an increasing number of digital health applications (DiGAs) receiving reimbursement approval. For instance, in August 2025, 58 DiGAs were eligible for reimbursement, reflecting the country's commitment to integrating digital health solutions into its healthcare system.
Drug reference apps market in Italy is anticipated to grow considerably during the forecast period. Increasing emphasis on mobile applications that provide comprehensive drug information, aiming to enhance medication safety and accessibility for both healthcare professionals and patients. For instance, in December 2022, the Italian Medicines Agency (AIFA) launched the "AIFA Medicinali" mobile app to provide citizens and caregivers with easy access to medicinal product information and updates. The app offers features such as notifications on drug shortages, expiry date tracking, and the ability to store health cards for quick access. This initiative reflects Italy's commitment to integrating digital tools into healthcare to improve accessibility and patient safety.
The UK drug reference apps market is anticipated to register a significant growth rate during the forecast period, driven by the increasing adoption of digital health technologies and the government's efforts to integrate innovative solutions into the healthcare system. For instance, in August 2025, the NHS App introduced an 'Amazon-style' prescription tracking feature, allowing patients to check the status of their medications. This feature has been used nearly 400,000 times in the first 10 weeks since its launch, with over 1,650 pharmacies, including all Boots locations in England, participating in the service. The implementation of this feature aims to enhance medication adherence and improve patient satisfaction by providing real-time updates on prescription statuses.
Asia Pacific Drug Reference Apps Market Trends
Asia Pacific drug reference apps industry is anticipated to be the fastest-growing market over the forecast period, fueled by increasing awareness of medication safety, the importance of timely access to drug information, and the rising use of digital health tools. Healthcare providers and patients in urban and rural areas increasingly rely on mobile and tablet-based applications to access accurate drug data, check interactions, and manage prescriptions efficiently. The integration of AI-driven features, cloud-based platforms, and real-time updates is enhancing the usability and reliability of these apps, supporting better clinical decision-making and patient outcomes.
Japan drug reference apps market is anticipated to register a significant growth rate during the forecast period. Growing with a focus on integrating digital therapeutics and enhancing accessibility to medication information through mobile platforms. For instance, in February 2025, CureApp, a Tokyo-based health tech company, received approval from Japan's Ministry of Health, Labour and Welfare (MHLW) for its digital therapeutic app to treat alcoholism. The app aims to reduce alcohol consumption in alcohol-dependent patients and is now available for prescription through medical institutions across Japan. This approval marks a significant advancement in integrating digital health solutions into Japan's healthcare system.
Drug reference apps market in China is anticipated to register a significant growth rate during the forecast period, driven by integrating advanced technologies and government initiatives to enhance healthcare accessibility and efficiency. For instance, in August 2025, China introduced the Commercial Insurance Innovative Drug Catalogue, expanding access to high-value drugs beyond the Basic Insurance Catalogue. This move allows commercial insurers to include innovative drugs in their coverage, thereby increasing the availability of advanced treatments to patients and promoting the development of the pharmaceutical industry in China.
Latin America Drug Reference Apps Market Trends
Latin America drug reference apps industry is growing over the forecast period, driven by increasing awareness of medication safety and the benefits of timely access to reliable drug information. Healthcare professionals and patients in urban and rural areas increasingly rely on mobile and tablet-based applications to quickly access drug data, check interactions, and manage prescriptions effectively. The adoption of AI-driven features, cloud-based platforms, and real-time updates is enhancing the accuracy and usability of these apps, helping improve clinical decision-making and patient outcomes across the region.
Brazil's drug reference apps market has grown in recent years, driven by advancements in digital health technologies and regulatory initiatives to enhance medication safety and accessibility. For instance, in July 2024, Brazil's National Health Regulatory Agency (ANVISA) approved a pilot project for digital medication leaflets. This initiative allows patients to access up-to-date medication information via QR codes or links on their smartphones or other digital devices, replacing traditional paper leaflets. The move aims to improve patient access to accurate medication information and promote environmental sustainability.
MEA Drug Reference Apps Market Trends
MEA drug reference apps industry is growing over the forecast period, fueled by expanding internet connectivity, rising smartphone adoption, and government efforts to strengthen healthcare infrastructure. Healthcare providers and patients increasingly use mobile applications to access reliable drug information, check interactions, and manage prescriptions efficiently. Countries like the United Arab Emirates are leading the digital healthcare transformation, driving innovation and adoption of drug reference apps across the region.
South Africa drug reference apps market is growing over the forecast period, driven by advancements in digital health technologies and regulatory initiatives to enhance medication safety and accessibility. For instance, in August 2025, the South African Health Products Regulatory Authority (SAHPRA) launched the Med Safety App to engage patients and healthcare providers on medicine safety issues. The app simplifies and promotes the reporting of suspected adverse drug reactions (ADRs), including adverse events following immunisation (AEFIs), by both the public and healthcare providers. It also allows users to access safety news from SAHPRA, creating awareness of medicines, their potential adverse effects, and pharmacovigilance.
Key Drug Reference Apps Company Insights
Key participants in the drug reference apps industry are focusing on devising innovative business growth strategies in the form of product portfolio expansions, partnerships & collaborations, mergers & acquisitions, and business footprint expansions.
Key Drug Reference Apps Companies:
The following are the leading companies in the drug reference apps market. These companies collectively hold the largest market share and dictate industry trends.
- Epocrates
- Medscape
- UpToDate Lexidrug (formerly Lexicomp)
- Micromedex (Merative US L.P.)
- PillPack
- Pill Identifier & Med Scanner
- Mango Health
- Drugs.com
- MDCalc
- Medisafe
- MyTherapy
Recent Developments
-
In September 2025, Glooko plans to extend its diabetes management solutions to hospitals by acquiring Monarch Medical Technologies and its EndoTool software, which helps manage inpatient insulin therapy. Approximately 30-40% of hospitalized patients need insulin to control glucose levels and prevent complications during other treatments.
-
In March 2025, Wolters Kluwer integrated its UpToDate drug information into Suki’s AI assistant platform, enhancing clinical decision support with trusted content.
-
In December 2024, the S-Check app, launched by researchers at the National Centre for Clinical Research on Emerging Drugs (NCCRED), is a self-guided smartphone tool designed to help people who use methamphetamine monitor their drug use and its impact on their health and wellbeing. Developed based on the Stimulant Check-Up service model at St Vincent’s Hospital in Sydney, the app targets individuals who have not previously accessed traditional treatment.
Drug Reference Apps Market Report Scope
Report Attribute
Details
Market size value in 2025
USD 1.41 billion
Revenue forecast in 2033
USD 2.84 billion
Growth rate
CAGR of 9.17% from 2025 to 2033
Actual data
2021 - 2024
Forecast data
2025 - 2033
Quantitative units
Revenue in USD million/billion and CAGR from 2025 to 2033
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Application, pricing model, device, end use, region
Regional scope
North America; Europe; Asia Pacific; Latin America; MEA
Country scope
U.S.; Canada; Mexico; Germany; UK; Spain; Italy; France; Norway; Denmark; Sweden; Japan; China; India; Australia; Thailand; South Korea; Brazil; Argentina; South Africa; Saudi Arabia; UAE; Kuwait
Key companies profiled
Epocrates; Medscape; UpToDate Lexidrug (formerly Lexicomp); Micromedex (Merative US L.P.); PillPack; Pill Identifier & Med Scanner; Mango Health; Drugs.com; MDCalc; Medisafe; MyTherapy
Customization scope
Free report customization (equivalent up to 8 analysts working days) with purchase. Addition or alteration to country, regional & segment scope.
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
Global Drug Reference Apps Market Report Segmentation
This report forecasts revenue growth at global, regional, and country levels and analyze industry trends in each sub-segments from 2021 to 2033. For this study, Grand View Research, Inc. has segmented the drug reference apps market report based on application, pricing model, device, end use, and region:
-
Application Outlook (Revenue, USD Million, 2021 - 2033)
-
Drug Information/Drug Data Base
-
Drug Interactions
-
Dosage Calculators
-
Pill/Drug Identification
-
Others
-
-
Pricing Model Outlook (Revenue, USD Million, 2021 - 2033)
-
Freemium (Free)
-
Paid (Subscription)
-
-
Device Outlook (Revenue, USD Million, 2021 - 2033)
-
Smartphones
-
Tablets
-
-
End Use Outlook (Revenue, USD Million, 2021 - 2033)
-
Healthcare Professionals
-
Patients
-
Pharmacists
-
Researchers and Educators
-
Others
-
-
Regional Outlook (Revenue, USD Million, 2021 - 2033)
-
North America
-
U.S.
-
Canada
-
Mexico
-
-
Europe
-
Germany
-
UK
-
Spain
-
Italy
-
France
-
Denmark
-
Norway
-
Sweden
-
-
Asia Pacific
-
Japan
-
China
-
India
-
Australia
-
Thailand
-
South Korea
-
-
Latin America
-
Brazil
-
Argentina
-
-
MEA
-
South Africa
-
Saudi Arabia
-
UAE
-
Kuwait
-
-
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










