- Home
- »
- Medical Devices
- »
-
Healthcare Contract Research Organization Market, 2033GVR Report cover
![Healthcare Contract Research Organization Market Size, Share & Trends Report]()
Healthcare Contract Research Organization Market (2025 - 2033) Size, Share & Trends Analysis Report By Type (Drug Discovery, Pre-clinical, Clinical), By Service (Data Management, Regulatory/Medical Affairs, Medical Writing), By Therapeutic Area (Oncology, CNS Disorders), By Molecule, By Region, And Segment Forecasts
- Report ID: 978-1-68038-688-2
- Number of Report Pages: 120
- Format: PDF
- Historical Range: 2021 - 2024
- Forecast Period: 2025 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
Healthcare Contract Research Organization Market Summary
The global healthcare contract research organization market size was estimated at USD 55.84 billion in 2024 and is projected to reach USD 105.73 billion by 2033, growing at a CAGR of 7.42% from 2025 to 2033. The market growth is driven by increasing investments in drug development, growing R&D activities, expanding regulatory requirements, globalization of trials, and the increasing complexity of clinical trials.
Key Market Trends & Insights
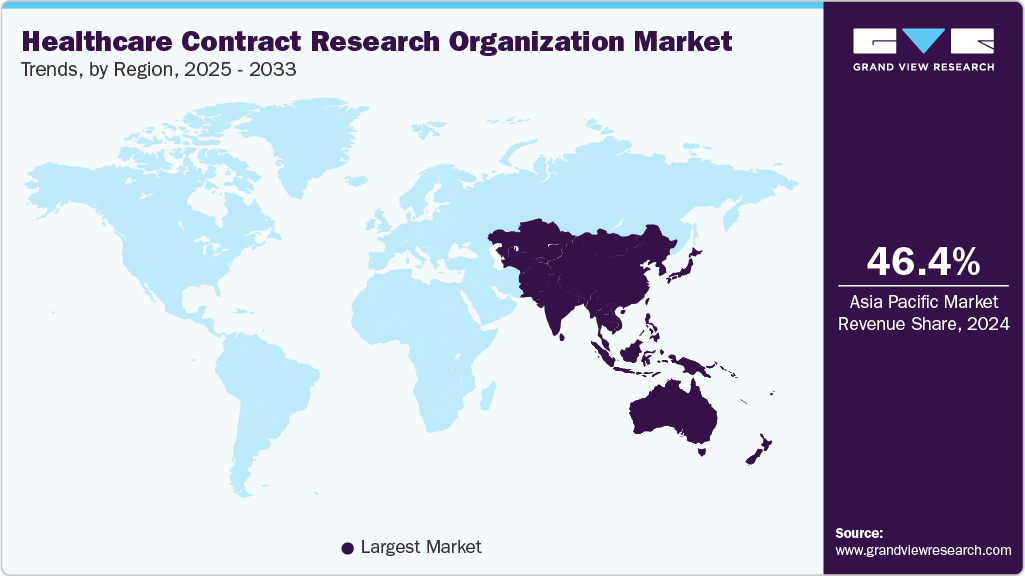
- The Asia Pacific healthcare contract research organization market held the largest share of 46.40% of the global market in 2024.
- The healthcare contract research organization (CRO) in India is expected to grow significantly over the forecast period.
- Based on type, the clinical segment held the largest market share of 75.67% in 2024.
- Based on service, the clinical monitoring segment captured the largest market share in 2024.
Market Size & Forecast
- 2024 Market Size: USD 55.84 Billion
- 2033 Projected Market Size: USD 105.73 Billion
- CAGR (2025-2033): 7.42%
- Asia Pacific: Largest market in 2024
Contract research organizations offer a range of expertise in various services compared with pharmaceutical companies in conducting clinical trials across multiple geographies and developing drugs in specific therapeutic areas, which is one of the major factors driving the market growth in the pharmaceutical industry. Moreover, the reliance on CROs has increased due to rising pressure on drug developers regarding clinical data management, evolving regulatory requirements, and stringent safety standards, further creating rising requirements for services. In addition, most pharmaceutical and medical device companies are outsourcing drug/device production, leading to increased clinical trials with trial privatization fueling increasing engagement in the developed countries.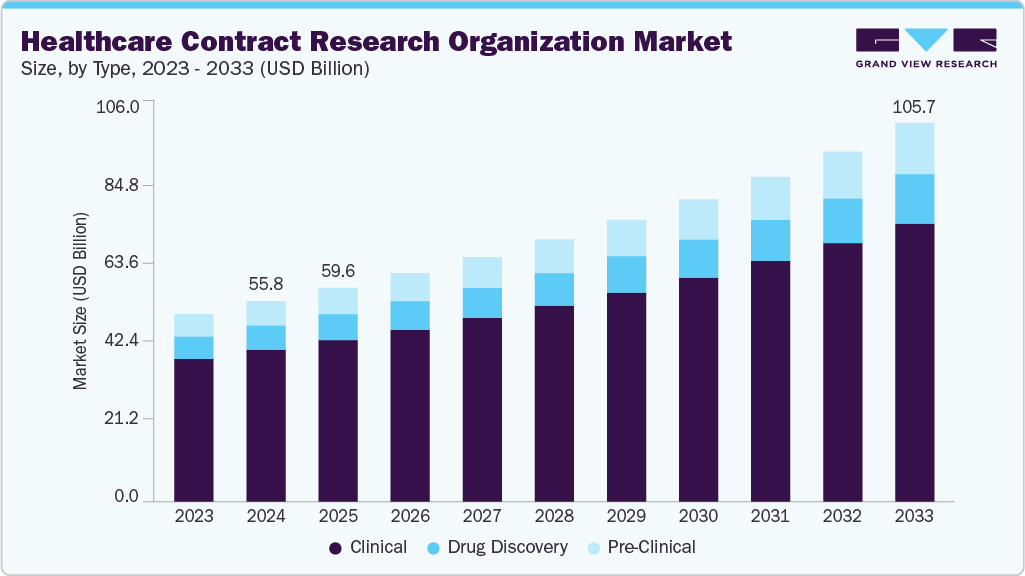
Furthermore, an increasing burden of non-communicable diseases (NCDs) and the rapidly growing aging population are also some of the factors contributing to the market growth. For instance, in October 2024, CDC mentioned that it had estimated that over 90% of adults aged 65 and above were affected by at least one chronic health condition. This increasing burden of chronic diseases is anticipated to drive the complexity of clinical trials, necessitating more specialized protocols, longer study durations, and advanced data management solutions. In addition, rising life expectancy increases morbidity, leading to more age-related diseases such as dysfunction, and dementia. This further creates a need for proper management of chronic diseases along with digital technology that transforms the entire process of drug development. The advent of mobile & wearables, Artificial Intelligence (AI), cloud technology, and associated platforms now enable the collection of frequent, precise, & multidimensional data during trials. These advanced technologies have the potential to enable innovative trial designs that lead to easy recruitment & retention, an improved patient experience, and established novel endpoints in clinical studies.
Furthermore, as the globalization of clinical trials increases, companies navigate diverse regulatory frameworks across multiple geographies. The rising stringency of regulatory bodies such as the FDA, EMA, and local health authorities has propelled the need for robust compliance systems. CROs offer professional regulatory knowledge and standardized processes to ensure trials meet global standards. They also provide continuous support for documentation and ethics submissions. CROs allow sponsors to achieve faster market access by mitigating compliance risks and accelerating approvals. As regulatory environments become more complex, reliance on CROs has intensified, driving sustained growth in the healthcare CRO market.
Opportunity Analysis
The healthcare CRO market is witnessing significant opportunities driven by decentralized and virtual trials, increasing outsourcing trends, and the growing complexity of clinical trials across therapeutic areas. Pharmaceutical and biotechnology companies are increasingly leveraging CRO expertise to optimize costs, accelerate timelines, and access advanced technologies such as AI-driven trial designs, real-world evidence integration, and adaptive clinical trial models. The shift toward patient-centric, remote, and digital trial models creates opportunities for CROs to offer virtual monitoring, telemedicine integration, and wearables-based data collection. The need for specialized CRO services is strengthened due to the expanding demand for personalized medicine, biologics, and rare disease treatments. Moreover, the globalization of clinical trials, particularly in emerging markets with large patient pools, offers CROs substantial scope to expand networks and enhance service capabilities.
Impact of U.S. Tariffs on the Global Healthcare Contract Research Organization Market
The imposition of 100% tariffs on branded and patented pharmaceutical imports by the U.S. has had a major impact across the healthcare ecosystem, with significant implications for the healthcare contract research organization industry. This tariff policy threatens to escalate drug costs, disrupt supply chains, and delay clinical trials requiring steady access to pharmaceuticals. Several medical communities, including the report of the American Hospital Association (AHA), have cautioned that shortages in critical drugs for cancer, heart disease, and organ failure could place patients’ lives at risk, highlighting the severity of the issue. CROs, which depend on continuous access to investigational drugs and ingredients, face rising operational complexity and pressure to diversify sourcing while maintaining trial continuity.
Technological Advancements
Technological advancements are driving significant growth and operational efficiency. Decentralized clinical trials have been increasingly adopted, strengthening telemedicine, wearables, and virtual monitoring to enhance patient recruitment, retention, and real-world data capture. Artificial intelligence and machine learning have been applied to optimize protocol design, site selection, and early safety signal detection.

Cloud-based platforms and integrated data ecosystems have enabled collaboration, transparency, and faster reporting across global trials. Real-world evidence and big data analytics have strengthened regulatory submissions and market access strategies, as well as digital biomarkers and wearables have improved monitoring, patient engagement, and adherence, transforming clinical development workflows.
Pricing Model Analysis
In the Healthcare CRO ecosystem, four primary pricing models are employed to align services with sponsor needs. Milestone-based pricing links payments to project achievements, incentivizing timely delivery and quality. Value-based pricing ties fees to measurable outcomes, rewarding CROs for improving efficiency, regulatory success, or time-to-market.
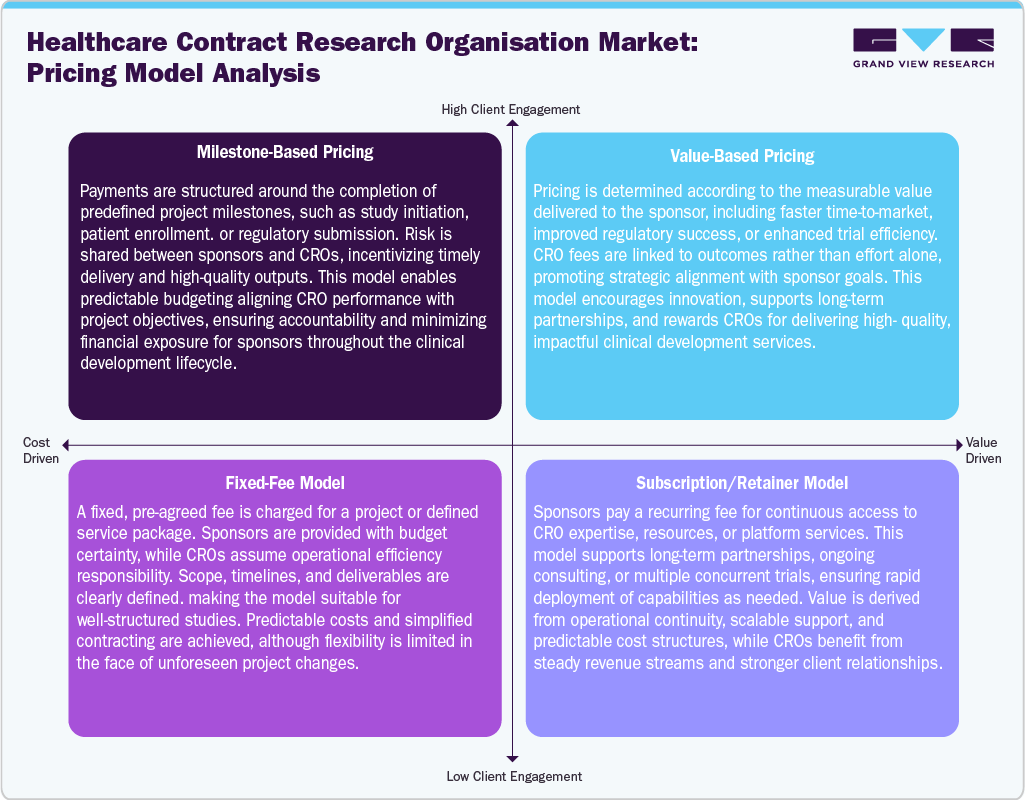
Fixed-fee models provide predictable costs for well-defined projects, with CROs assuming operational efficiency responsibility. Subscription or retainer models offer recurring access to expertise and resources, supporting multiple trials or ongoing consulting. Each model balances risk, flexibility, and financial predictability, allowing sponsors to select the approach that best aligns with project complexity and strategic objectives.
Market Concentration & Characteristics
The industry growth stage is moderate, and growth is accelerating. The market is characterized by the degree of innovation, level of M&A activities, regulatory impact, service expansion, and regional expansion.
The adoption of innovative technologies, including AI, decentralized trials, digital biomarkers, and real-world evidence analytics, has enhanced operational efficiency and data quality. CROs investing in innovative solutions can offer differentiated services, shorten development timelines, and attract clients seeking advanced capabilities for complex or novel therapeutic programs.

Mergers and acquisitions are transforming the market, enabling firms to expand service portfolios, geographic reach, and therapeutic expertise. Consolidation enhances operational scale, integration of niche capabilities, and access to new client segments, while strategic acquisitions allow CROs to increase market competitiveness and revenue growth potential.
Stringent global regulatory frameworks for pharmaceuticals and medical devices drive demand for CRO expertise. Compliance with evolving guidelines, data integrity requirements, and patient safety standards necessitates sophisticated operational and regulatory capabilities.
CROs are broadening their offerings beyond core clinical trials to include early-phase research, real-world evidence, regulatory consulting, analytics, and post-market surveillance. This expansion allows clients to access integrated solutions, simplify outsourcing, enhance project continuity, and position CROs as strategic partners capable of addressing diverse development and commercialization needs.
Geographic diversification is increasingly pursued to access cost efficiencies, diverse patient populations, and emerging markets. Expansion into Asia, Latin America, and Eastern Europe enables faster recruitment, regulatory advantages, and localized expertise. CROs benefit from scalable operations, increased client engagement, and competitive positioning in the global clinical development ecosystem.
Type Insights
On the basis of type, the clinical segment held the largest market share in 2024, accounting for a revenue share of 75.67%. As research advances, pharmaceutical companies are increasingly focusing on strict budgets and timelines, prompting a preference for outsourcing to CROs or setting up dedicated clinical trial labs. Besides, rising complexity of trials due to orphan diseases and cancer, requiring innovative drug development approaches, is also supporting the segment’s market growth.
The pre-clinical segment is emerging as the fastest-growing area in the market. This growth is driven by increasing demand for early-stage drug development services, including in vitro and in vivo testing, toxicology studies, and pharmacokinetic evaluations. Pharmaceutical and biotechnology companies are outsourcing pre-clinical activities to CROs to optimize costs, adhere to strict timelines, and leverage specialized expertise and advanced laboratory infrastructure. In addition, rising investments in drug discovery, the emergence of complex biologics, and the focus on accelerating time-to-market for novel therapies are further fueling the segment expansion.
Service Insights
In terms of service, the clinical monitoring segment held the largest revenue share in 2024 due to the growing number of clinical trials and the rising demand for monitoring services. Outsourcing to CROs has increased over the past decade, driven by cost efficiency and specialized expertise. Advancements such as smart analytics and real-time data acquisition are enhancing monitoring accuracy, enabling early detection of drug safety or toxicity issues and timely corrective actions like trial redesign or termination. In addition, IT and consulting companies are supporting the innovation in clinical monitoring platforms and further contributing to segment growth. For instance, in February 2025, Paradigm Biopharmaceuticals selected Advanced Clinical as its CRO for a pivotal Phase III osteoarthritis trial of iPPS, following strong Phase II results, due to its expertise, capabilities, and strategic alignment.
Regulatory and medical affairs are expected to witness the fastest growth driven by rising R&D, clinical trial applications, product registrations, and expanding drug pipelines. The growing need for faster product approvals, strict compliance requirements, and efficiency in resource utilization is fueling outsourcing requirements. In addition, CROs are also pursuing expansion strategies to strengthen their global presence. For example, in September 2022, PharmaLex GmbH, a leading regulatory services provider, opened a new office in Beijing, offering clients access to regional experts specializing in pharmaceutical and biopharmaceutical regulatory support.
Therapeutic Area Insights
On the basis of source, oncology represented the largest segment in 2024, driven by the rising global prevalence of cancer and the urgent need for innovative therapies. Cancer research requires highly complex, large-scale clinical trials with advanced designs, extensive patient recruitment, and strict regulatory compliance, making outsourcing to CROs increasingly essential. CROs provide specialized expertise, global networks, and advanced technologies to manage oncology trials efficiently, supporting drug development pipelines for targeted therapies, immuno-oncology, and precision medicine.
The CNS disorders segment is projected to witness notable growth during the forecast period, as these conditions represent a significant share of the global health burden and affect diverse population groups. Approximately 50 million Americans live with neurological disorders, with a similar prevalence seen worldwide. The rising incidence of conditions such as Alzheimer’s, stroke, and Parkinson’s is closely tied to the rapidly aging population. According to the WHO, the global population aged 60 and above is expected to increase from 1.4 billion in 2020 to 2.1 billion by 2050, driving demand for advanced CNS-focused research and treatments.
Molecule Insights
On the basis of molecules, the pharmaceutical segment accounted for the largest share in 2024. The growth of this segment is driven by the rising number of promising small and large molecules in the global market and the increasing use of specialty medicines. Small molecules, including APIs and finished drugs, contribute significantly to pharmaceutical sales, fueling demand for CRO outsourcing. These services help reduce costs, ensure faster drug production, and optimize operational efficiency. Outsourcing also allows companies to focus on R&D and facility management. Notably, according to the U.S. FDA, small molecules accounted for the largest share in new drug approvals, highlighting the clinical monitoring requirement in the market. Similarly, large molecules are expected to grow at the fastest CAGR over the estimated period. Large molecules provide specialized biologics services, including monoclonal antibodies and vaccines, through advanced bioanalytical testing, process development, and clinical support. Their expertise accelerates complex drug development, ensuring regulatory compliance, data accuracy, and faster time-to-market for biopharmaceutical innovations. Such factors are expected to drive the market.
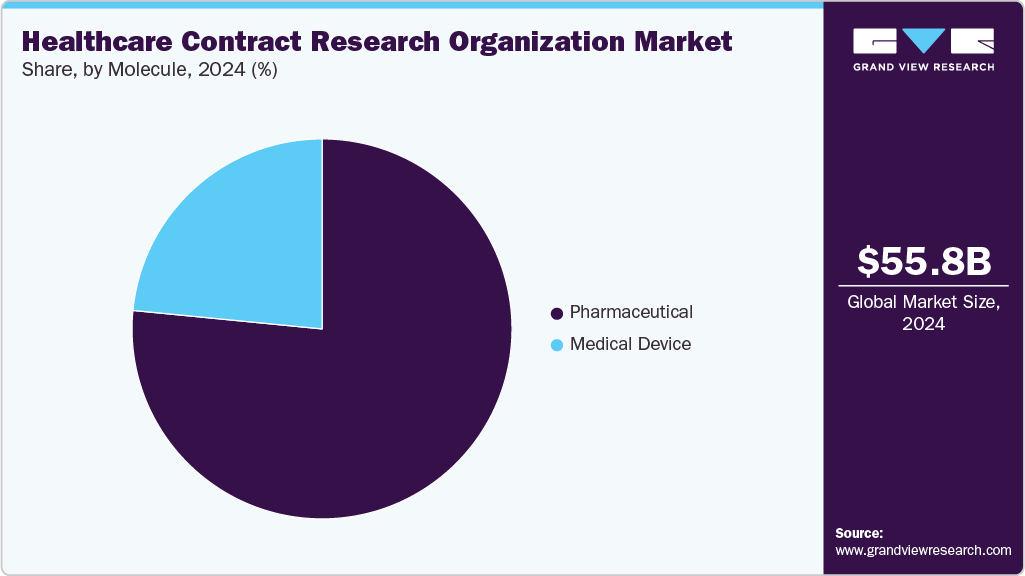
The medical device segment is projected to grow at a notable CAGR over the forecast period. The segment growth is driven by diverse materials, complex product designs, and the rising number of small to mid-sized manufacturers. Stringent regulatory norms and supportive government initiatives further drive the CRO demand across the globe. Besides, CROs offer specialized expertise, therapeutic insights, and strategic guidance, enabling faster product development and market entry. By streamlining compliance and accelerating innovation, CROs play a vital role in advancing medical device research and development. These factors are expected to significantly enhance the reliance on CROs for medical device studies and clinical support worldwide.
Regional Insights
Asia Pacific healthcare contract research organization industry held the largest revenue share of 46.40% in 2024, driven by a large patient population and increased outsourcing in the biopharmaceutical industry, particularly during clinical R&D stages, boosting regional clinical studies. India’s minimal import-export regulations accelerate processes, while China’s vast patient pool and regulatory adjustments support its largest market share. Access to treatment-naive patients in these countries promotes advanced outsourcing of clinical trials and related services, enhancing efficiency and supporting the region’s rapid expansion in the global CRO landscape.
The healthcare contract research organization industry in Japan accounted for a notable share (in the Asia Pacific) in 2024. Market growth is driven by the expanding pharmaceutical sector, increasing healthcare expenditure, and heightened awareness, along with initiatives from government bodies and institutions supporting clinical research and development activities across the country.
The India healthcare contract research organization industry is projected to register an impressive CAGR during the forecast period, driven by its large patient base, cost advantages, and growing clinical trial expertise. The country is emerging as a preferred destination for Phase II and III studies, especially in oncology, cardiovascular, and infectious diseases. Rising healthcare infrastructure investment, government research support, and a favorable regulatory framework further propel CRO market growth.
The healthcare contract research organizations industry in China is expected to register substantial growth during the forecast period. Growth is driven by strong government support for medical research, expansion of the biotechnology sector, and rising demand for innovative therapies. The country is emerging as a key hub for early-phase trials, particularly in oncology, neurology, and rare diseases. Local CROs capitalize on China’s large patient population, digital health technologies, and favorable regulatory environment to support clinical development and market expansion.
North America Healthcare Contract Research Organization Market Trends
North America healthcare contract research organization industry dominated the market in 2024,attributed to rising chronic diseases, complex clinical trials, and increasing outsourcing by biopharma sponsors. Advanced technologies such as AI, decentralized trials, and real-world evidence analytics are being adopted to improve efficiency and reduce timelines. Key players, including IQVIA, Parexel, PPD, and ICON, are expanding capabilities through M&A and service diversification. Strong regulatory expertise, integrated offerings, and innovation in digital platforms continue to differentiate market leaders and attract global clients.
U.S. Healthcare Contract Research Organization Market Trends
The healthcare contract research organization industry in the U.S.accounted for the largest share in the North America, owing to growing biopharma R&D investment and expedited regulatory pathways. In a notable development of September 2025, ACL Digital acquired U.S.-based Symbiance, strengthening its clinical research footprint and integrating AI-driven data management, pharmacovigilance, and end-to-end CRO capabilities. Such strategic acquisitions, coupled with digital transformation and regulatory expertise, are intensifying competition and enabling scalable, patient-centric solutions across the U.S. clinical development ecosystem.
Canada healthcare contract research organization industry is expected to grow at a significant CAGR during the forecast period, driven by government support & funding and the growing biopharma and med-tech sector. Technological advancements, including AI, virtual reality diagnostics, and digital health platforms, are enhancing trial efficiency and patient engagement. In August 2025, the Government of Canada expanded healthcare to Indigenous communities through the CAN Health Network and Virtual Health Hub is enabling remote trial access, supporting homegrown innovation, and strengthening healthcare delivery, positioning CROs to leverage both advanced technologies & national healthcare initiatives for growth and broader patient inclusion.
Europe Healthcare Contract Research Organization Market Trends
The Europe healthcare contract research organizations industry is projected to experience a substantial CAGR during the forecast period, driven by rising clinical trial activity, precision medicine adoption, and innovations in drug development. The region is supported by a skilled workforce, robust regulatory frameworks, and an established pharmaceutical ecosystem. Some of the key trends driving this market include decentralized trials and digital technologies, such as AI and blockchain, improving efficiency and patient engagement. Demand for specialized services, including oncology and rare disease studies, is increasing and reflecting the growing complexity and sophistication of clinical research across Europe.
The healthcare contract research organization industry in Germany held the largest share in 2024, fueled by rising R&D investment, increasing healthcare awareness, and technological innovations. A focus on precision medicine and advanced therapies strengthens innovation in clinical trial approaches. Enhanced collaboration between CROs and academic institutions further accelerates research capabilities and strengthens Germany’s position in the global clinical development aspect.
The UK healthcare contract research organization industry accounted for a considerable share in 2024. Regional growth is driven by rising clinical trial activity, increasing outsourcing demand, and advancements in biotechnology. The market progresses due to decentralized trials, AI, and big data integration for analytics, and emphasis on rare disease studies. Stringent regulations and Brexit-related changes have influenced the market, prompting CROs to expand services and geographic presence to meet evolving client requirements and maintain compliance.
Latin America Healthcare Contract Research Organization Market Trends
The healthcare Contract Research Organizations industry in Latin Americais projected to register a significant CAGR during the forecast period, driven by growing demand for clinical trials across therapeutic areas such as oncology and infectious diseases. The region’s diverse patient base, cost advantages, and developing healthcare infrastructure attract global pharmaceutical sponsors. Regulatory reforms and collaborations between local and international CROs enhance research capabilities, while adoption of digital technologies and data analytics further strengthens Latin America’s role in global clinical research and development.
Brazil healthcare contract research organization industry is experiencing robust growth as its large, diverse patient population offers unique opportunities for clinical trials, especially in oncology, cardiovascular, and infectious disease areas. Improvements in the regulatory environment, coupled with increased focus on research and development, further enhance Brazil’s appeal as a destination for global pharmaceutical companies.
Middle East & Africa Healthcare Contract Research Organization Market Trends
Middle East healthcare contract research organization industry is growing steadily due to strategic geopolitical positioning and strategic investments in biotechnology infrastructure. Countries like Saudi Arabia and the UAE are emerging as regional hubs, attracting global CDMOs through incentives, infrastructure, and government visionary initiatives. Major players such as Lonza, WuXi AppTec, Catalent, Thermo Fisher Scientific, and Piramal Pharma Solutions are actively expanding operations in the region. For instance, in June 2023, Saudi Arabia’s Public Investment Fund launched Lifera, a commercial-scale CDMO, to strengthen local biomanufacturing, enhance skills, enable technology transfer, and propel the national pharmaceutical capabilities and resilience.
South Africa healthcare contract research organization industryis expected to depict a robust CAGR during the forecast period, as the country emerges as a key destination for clinical trials in sub-Saharan Africa. Its well-developed infrastructure, diverse patient population, and cost advantages attract global pharmaceutical sponsors. Strong regulatory frameworks, skilled workforce, and growing focus on research in infectious diseases, oncology, and chronic conditions further drive CRO demand, strengthening South Africa’s position in the global healthcare CRO market.
Key Healthcare Contract Research Organization Company Insights
Major players in the global market are pursuing various strategic initiatives, including acquisitions, mergers, service launches, partnerships, expansions, and collaborations, to expand geographic presence and strengthen competitive positioning. For example, in June 2025, Fortrea and Emery Pharma have partnered to provide rapid MNP testing of rifampin for safe, FDA-compliant DDI studies, enabling sponsors to accelerate trials, ensure participant safety, and generate high-quality clinical data.
Key Healthcare Contract Research Organization Companies:
The following are the leading companies in the healthcare contract research organization market. These companies collectively hold the largest market share and dictate industry trends.
- ICON Plc
- Charles River Laboratories
- Syneos Health
- IQVIA Inc.
- GVK Biosciences Private Limited (Aragen)
- LabCorp
- Parexel International Corporation
- Thermo Fisher Scientific
- CTI Clinical Trial & Consulting
- PSI
- Medpace
- Ergomed
- WuXi AppTec
- Worldwide Clinical Trials
- Medidata Solutions, Inc
- Pharmaron GMBH
- SGS SA
- KCR S.A.
- Advanced Clinical Research Services, LLC
- Pharm-Olam, LLC (Allucent)
Recent Developments
-
In September 2025, ACL Digital acquired Symbiance, a technology-driven CRO, to enhance AI-powered clinical research, pharmacovigilance, and data services. This acquisition strengthens ACL Digital’s end-to-end global clinical research and life sciences capabilities.
-
In September 2025, StarTrials reportedly announced its expansion into key Asian markets, establishing offices and local partnerships to enhance clinical trial operations, patient recruitment, and data quality, accelerating drug development timelines and regional CRO capabilities.
-
In November 2024, Novotech formed a strategic partnership with Biostar to support clinical development using Novotech’s platforms, following Biostar’s HKEX listing. The collaboration reinforces CRO support for innovative oncology drug development globally.
Healthcare Contract Research Organization Market Report Scope
Report Attribute
Details
Market size value in 2025
USD 59.62 billion
Revenue forecast in 2033
USD 105.73 billion
Growth rate
CAGR of 7.42% from 2025 to 2033
Actual data
2021 - 2024
Forecast period
2025 - 2033
Quantitative units
Revenue in USD million/billion and CAGR from 2025 to 2033
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Type, service, therapeutic area, molecule, region
Regional scope
North America; Europe; Asia Pacific; Latin America; MEA
Country scope
U.S.; Canada; Mexico; UK; Germany; France; Italy; Spain; Denmark; Sweden; Norway; Japan; China; India; Thailand; South Korea; Australia; Brazil; Argentina; South Africa; UAE; Saudi Arabia; Kuwait; Qatar; Oman
Key companies profiled
ICON Plc; Charles River Laboratories; Syneos Health; IQVIA Inc.; GVK Biosciences Private Limited (Aragen); LabCorp; Parexel International Corporation; Thermo Fisher Scientific; CTI Clinical Trial & Consulting; PSI; Medpace; Ergomed; WuXi AppTec; Worldwide Clinical Trials; Medidata Solutions, Inc; Pharmaron GMBH; SGS SA; KCR S.A.; Advanced Clinical Research Services, LLC; Pharm-Olam, LLC (Allucent)
Customization scope
Free report customization (equivalent up to 8 analysts working days) with purchase. Addition or alteration to country, regional & segment scope.
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
Global Healthcare Contract Research Organization Market Report Segmentation
This report forecasts revenue growth at global, regional, and country levels and provides an analysis of the latest industry trends in each of the sub-segments from 2021 to 2033. For this study, Grand View Research has segmented the global healthcare contract research organization market report based on type, service, therapeutic area, molecule and region:
-
Type Outlook (Revenue, USD Million, 2021 - 2033)
-
Drug Discovery
-
Target Validation
-
Lead Identification
-
Lead Optimization
-
-
Pre-Clinical
-
Clinical
-
Phase I Trial Services
-
Phase II Trial Services
-
Phase III Trial Services
-
Phase IV Trial Services
-
-
-
Service Outlook (Revenue, USD Million, 2021 - 2033)
-
Project Management/Clinical Supply Management
-
Data Management
-
Regulatory/Medical Affairs
-
Medical Writing
-
Clinical Monitoring
-
Quality Management/ Assurance
-
Bio-statistics
-
Investigator Payments
-
Laboratory
-
Sterility Testing
-
Container/Closure Testing
-
Extractables and Leachable Testing
-
Environmental Monitoring (Including Microbiology Testing)
-
Disinfectant Efficacy Studies
-
Others
-
-
Patient And Site Recruitment
-
Technology
-
Others
-
-
Therapeutic Area Outlook (Revenue, USD Million, 2021 - 2033)
-
Oncology
-
CNS Disorders
-
Infectious Diseases
-
Immunological Disorders
-
Cardiovascular Diseases
-
Respiratory Diseases
-
Diabetes
-
Ophthalmology
-
Pain Management
-
Others
-
-
Molecule Outlook (Revenue, USD Million, 2021 - 2033)
-
Pharmaceutical
-
Small Molecules
-
Biologics
-
-
Medical Device
-
-
Regional Outlook (Revenue, USD Million, 2021 - 2033)
-
North America
-
U.S.
-
Canada
-
Mexico
-
-
Europe
-
UK
-
Germany
-
France
-
Italy
-
Spain
-
Denmark
-
Sweden
-
Norway
-
-
Asia Pacific
-
Japan
-
China
-
India
-
Australia
-
Thailand
-
South Korea
-
-
Latin America
-
Brazil
-
Argentina
-
-
Middle East & Africa
-
South Africa
-
UAE
-
Saudi Arabia
-
Kuwait
-
Qatar
-
Oman
-
-
Frequently Asked Questions About This Report
b. The global healthcare contract research organization market size was estimated at USD 55.84 billion in 2024 and is expected to reach USD 59.62 billion in 2025.
b. The global healthcare contract research organization market is expected to grow at a compound annual growth rate of 7.42% from 2025 to 2033 to reach USD 105.73 billion in 2033.
Which type segment accounted for the highest healthcare contract research organization market share?b. The clinical segment dominated the healthcare contract research organization market with a share of 75.67% in 2024. The market growth is attributed to growing research advances and pharmaceutical companies' rising focus on strict budgets and timelines. This prompts a preference for outsourcing to CROs or setting up dedicated clinical trial labs, which further contributes to market growth.
b. Some key players operating in the healthcare CRO market include ICON Plc, Charles River Laboratories, Syneos Health, IQVIA Inc., GVK Biosciences Private Limited (Aragen), LabCorp, Parexel International Corporation, Thermo Fisher Scientific, CTI Clinical Trial & Consulting, PSI, Medpace, Ergomed, WuXi AppTec, Worldwide Clinical Trials, Medidata Solutions, Inc, Pharmaron GMBH, SGS SA, KCR S.A., Advanced Clinical Research Services, LLC, and Pharm-Olam, LLC (Allucent), among others.
b. The market growth is driven by increasing investments in drug development, growing R&D activities, expanding regulatory requirements, and the globalization of trials. In addition, the increasing complexity of clinical trials and growing reliance on CROs is due to rising pressure on drug developers regarding clinical data management, evolving regulatory requirements, and stringent safety standards. Moreover, the rising requirement for services is expected to drive the market.
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










