- Home
- »
- Clinical Diagnostics
- »
-
HIV Self-testing Market Size & Trends, Industry Report, 2033GVR Report cover
![HIV Self-testing Market Size, Share & Trends Report]()
HIV Self-testing Market (2025 - 2033) Size, Share & Trends Analysis Report By Type (Blood-based Kits, Saliva-based Kits), By Technology (Immunofiltration Assays, Lateral Flow Assays (LFA)), By Distribution Channel, By Region, And Segment Forecasts
- Report ID: GVR-4-68040-710-7
- Number of Report Pages: 150
- Format: PDF
- Historical Range: 2021 - 2023
- Forecast Period: 2025 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
HIV Self-testing Market Summary
The global HIV Self-testing market size was estimated at USD 211.55 million in 2024 and is expected to reach USD 643.41 million by 2033, growing at a CAGR of 13.55% from 2025 to 2033. This growth is fueled by an increasing emphasis on early HIV detection, rising HIV prevalence, and a global shift toward personal, confidential health solutions.
Key Market Trends & Insights
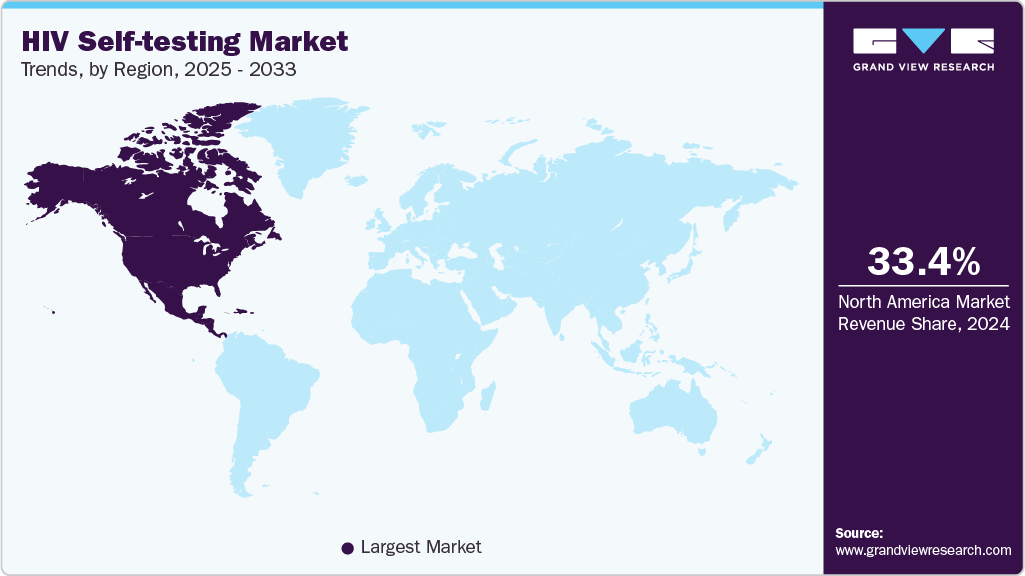
- North America HIV self-testing market dominated the global market and accounted for the largest revenue share of 33.40% in 2024.
- The U.S. led the North American market and held the largest revenue share in 2024
- Based on type, the blood-based kits segment dominated the global market and accounted for the largest revenue share of 54.65% in 2024.
- Based on technology, the Lateral flow assays (LFA) segment held the largest revenue share of 71.99% in 2024.
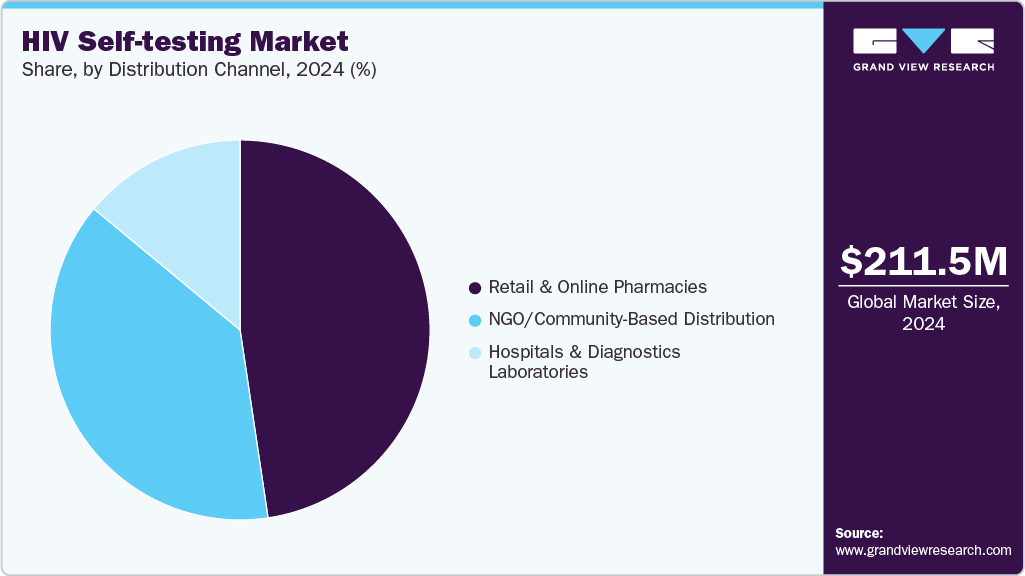
- Based on distribution channel, the retail and online pharmacies segment held the largest revenue share of 34.08% in 2024.
Market Size & Forecast
- 2024 Market Size: USD 211.55 Million
- 2033 Projected Market Size: USD 643.41 Million
- CAGR (2025-2033): 13.55%
- North America: Largest market in 2024
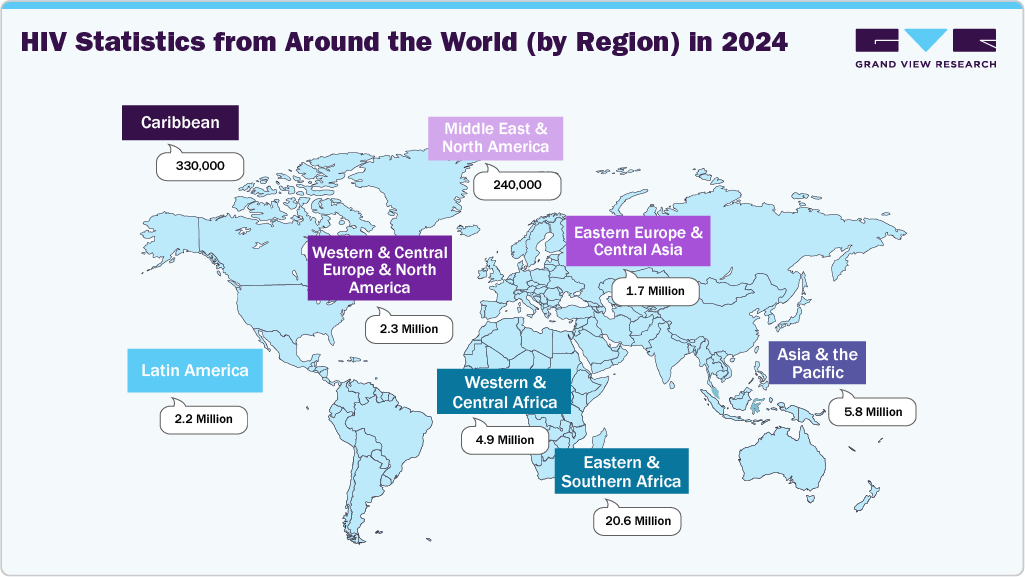
Over 100 countries have adopted HIVST policies, and more than 20 million kits were distributed globally in 2023 alone. The affordability and scalability of rapid diagnostic kits have made them integral in both developed and developing health systems. As of the most recent estimates, approximately 40.8 million people worldwide are living with HIV, including 1.4 million children between the ages of 0 and 14. In 2024 alone, there were about 1.3 million new HIV infections, with 120,000 cases occurring in children. That same year, an estimated 630,000 people died from HIV-related causes, including 75,000 children. Since the beginning of the HIV epidemic, it is estimated that 44.1 million people have lost their lives due to HIV-related illnesses. Despite the ongoing impact, global progress has been substantial over the past decade and a half. Between 2010 and 2024, the number of new HIV infections decreased by approximately 40%, dropping from 2.2 million to 1.3 million annually. During the same period, HIV-related deaths fell by 54%, declining from 1.4 million to 630,000. These reductions reflect the success of widespread awareness, prevention efforts, antiretroviral therapy scale-up, and increasing adoption of strategies such as HIV self-testing, which have contributed to earlier diagnosis and treatment initiation.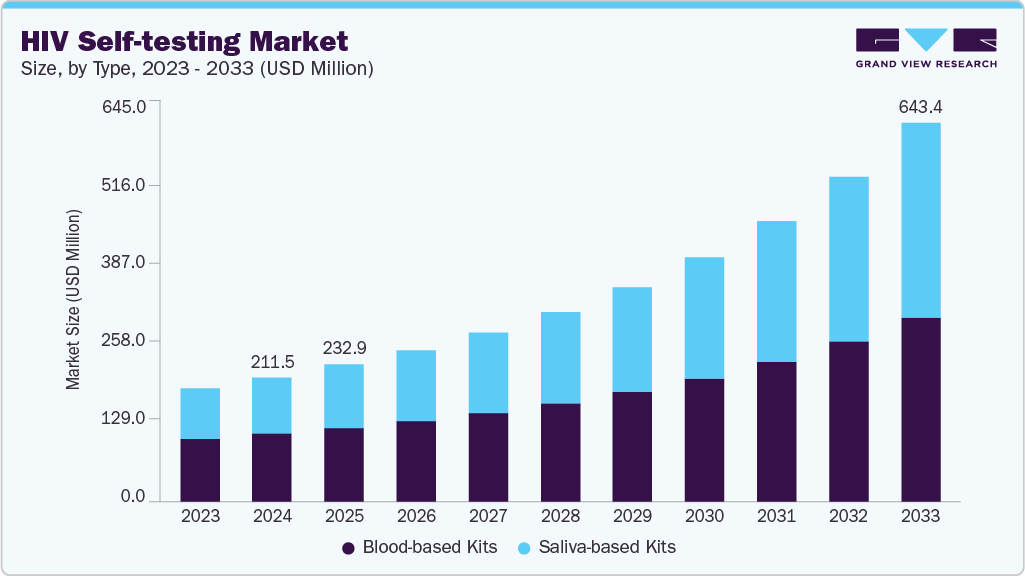
One of the strongest drivers of the HIV self-testing market is the rising demand for discreet, accessible, and user-friendly diagnostic options, particularly in populations facing stigma or barriers to clinic-based testing. According to UNAIDS, nearly 40% of people living with HIV globally remain undiagnosed, a gap largely attributed to fear of discrimination, social stigma, and inconvenient access to health facilities. HIV self-tests provide a private, at-home alternative that empowers individuals to know their status without fear. Saliva-based kits such as OraQuick In-Home HIV Test have become popular for their non-invasive nature, simplicity, and 20-minute result window. In regions such as Sub-Saharan Africa, where HIV prevalence is highest, self-testing has been pivotal: countries like Kenya, Malawi, and South Africa have scaled up national HIVST programs through community and NGO distribution. In high-income countries, rising e-commerce penetration has enabled widespread access to HIV self-tests via online pharmacies. This combination of anonymity, convenience, and technological simplicity is not only improving diagnosis rates but also fundamentally shifting how public health systems approach HIV prevention and treatment linkage.
Technological advancements are transforming HIV self-testing into a more accurate, efficient, and user-centric process. The most common format-lateral flow immunoassays (LFA)-uses a simple strip that detects HIV-1/2 antibodies through a saliva or finger-prick blood sample. These tests deliver results in 15-20 minutes, and newer devices incorporate all-in-one integrated systems, such as Atomo’s HIV Self-Test, which combines lancet, buffer, and strip in a single device. In addition, smartphone-linked digital self-tests are being piloted, allowing users to scan results, receive guided instructions, and access telehealth services-enhancing test reliability and post-test linkage to care. In clinical trials, smartphone-based readers have shown >98% concordance with trained visual interpretation. Furthermore, innovations in microfluidics and nano-based detection promise to make future HIVST kits even more sensitive and early-stage infection-aware. The industry is also shifting toward combination kits (e.g., HIV + syphilis), especially in areas with co-infection burdens. These technological improvements not only reduce human error but also make self-testing viable in low-literacy populations, further expanding its global reach.

Market Concentration & Characteristics
Innovation in HIV self-testing is focused on user-centric design, digital result integration, and combination tests. Devices now feature all-in-one kits, app connectivity, and minimal sample handling. Startups and health tech firms are also exploring AI-based test interpretation and blockchain-linked anonymity. Innovation is not only improving accuracy but also enhancing the testing experience and follow-up ecosystem.
Mergers and acquisitions are accelerating as major diagnostic companies seek to enter or strengthen their positions in the self-testing segment. Notable examples include OraSure’s acquisitions in mobile diagnostics and partnerships between Atomo Diagnostics and global NGOs. Larger players aim to acquire smaller, agile tech firms to capitalize on digital testing trends and expand global reach.
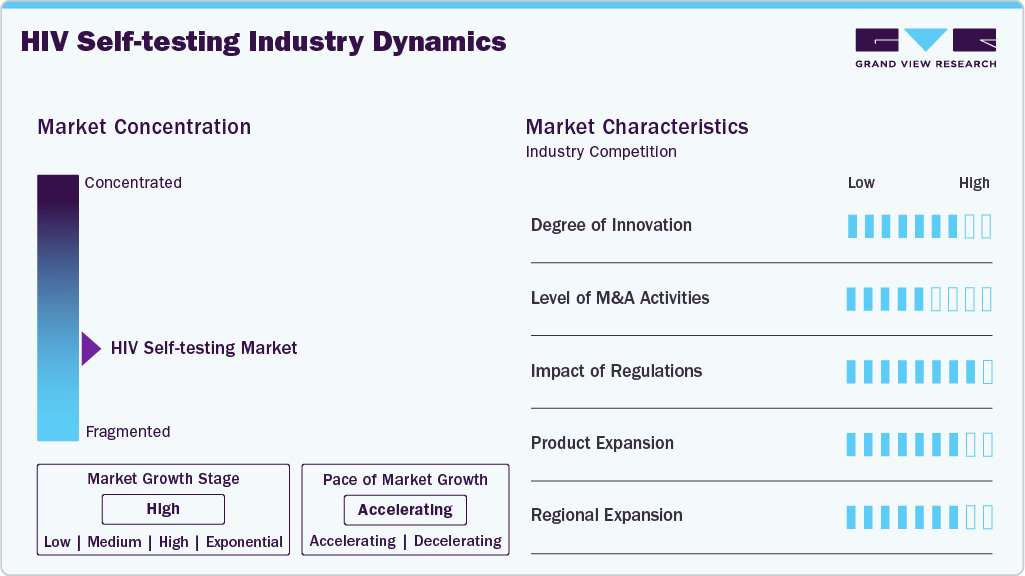
Regulatory bodies such as the FDA, CE, and WHO Prequalification Program are streamlining approvals, ensuring quality and safety. Regulatory harmonization has accelerated market entry, especially in Africa and Southeast Asia. However, varied regional standards, lack of digital health frameworks, and delays in local authorization still pose barriers to rapid, widespread distribution and innovation scalability.
Companies are expanding from simple antibody-only HIV tests to combo kits that detect HIV + syphilis, hepatitis, or COVID-19. Enhanced product formats include digital readouts, oral swabs, and finger-prick integrations with built-in safety features. Future portfolios may include multiplexed diagnostics and reusable digital platforms, aligning with trends in home-based, consumer-directed testing.
HIV self-testing is rapidly expanding beyond traditional markets in North America and Europe into Asia-Pacific, Africa, and Latin America. Regional programs in Kenya, South Africa, India, and Brazil have driven mass uptake via NGOs and public health outreach. Multilateral funding and e-commerce penetration are unlocking growth in underserved and high-prevalence regions worldwide.
Type Insights
On the basis of type, blood-based kits market accounted for the largest market share of 54.65% in 2024, due to their higher sensitivity and accuracy. These kits detect HIV-1/2 antibodies through a finger-prick blood sample, with a strong presence in both developed and emerging markets. Products such as Atomo HIV Self-Test and BioSure HIV Self-Test are leading this segment. The Atomo test, for example, features an integrated design with built-in lancet and buffer, reducing user error and improving test efficiency. Blood-based kits are preferred in clinical-grade community outreach programs and by users seeking higher diagnostic confidence. As awareness and access expand-especially in Africa, Asia, and Latin America-this segment is expected to witness sustained growth, backed by government and NGO support for reliable, at-home HIV testing tools.
The saliva-based kits segment in the HIV self-testing market is experiencing rapid growth, driven by rising demand for non-invasive, user-friendly diagnostic options. These kits use oral fluid to detect HIV-1/2 antibodies, offering a painless alternative to finger-prick tests. A key product in this space is the OraQuick In-Home HIV Test by OraSure Technologies. It delivers results in approximately 20 minutes without needing blood or lab processing and has been FDA-approved and WHO prequalified, making it widely adopted across North America, Europe, and parts of Africa. The simplicity of use, especially for first-time testers, adolescents, and needle-averse individuals, is a key growth factor. As awareness spreads, saliva-based kits are increasingly included in e-pharmacy platforms and NGO distribution efforts globally.
Technology Insights
Lateral flow assay (LFA) held the largest market share in 2024 and anticipated to grow at the fastest rate over the forecast period. This technology enables rapid, easy-to-use diagnostic tests that produce results within 15-20 minutes, making it ideal for self-testing. LFA works by detecting HIV-1/2 antibodies via a capillary action-driven flow of a sample (blood or oral fluid) across a test strip with immobilized reagents. Products such as the OraQuick In-Home HIV Test (saliva-based) and BioSure HIV Self Test (blood-based) are prominent examples using LFA. These tests are valued for their low cost, portability, and ease of interpretation-typically providing a visual line for positive results. Innovations within LFA, such as integrated sample collection (e.g., Atomo Diagnostics), have further boosted usability and accuracy.
The Immunofiltration assay segment in the HIV self-testing market grow steady, typically used in more specialized or controlled self-testing environments. This technology involves applying a sample (usually blood) vertically onto a membrane where HIV antibodies bind to immobilized antigens, creating a visible signal. While less common in over-the-counter consumer kits, immunofiltration offers high specificity and rapid processing-often delivering results in under 10 minutes. Products such as the INSTI HIV Self-Test by bioLytical Laboratories utilize immunofiltration technology. INSTI stands out by providing instant results in 1 minute and is WHO prequalified, making it suitable for both home and field use. The segment is growing at a modest rate, driven by demand in clinical outreach programs and NGO-led campaigns that prioritize speed and accuracy. Although it trails behind LFA in market size, the Immunofiltration Assay segment is expected to maintain a niche but reliable role, especially in point-of-care self-testing initiatives.
Distribution Channel Insights
Retail and online pharmacies dominated the market in 2024, accounting for a 47.65% share and is expected to grow at the fastest CAGR over the forecast period. This growth is driven by increasing consumer demand for convenient, private access to self-test kits. Retail pharmacies provide over-the-counter availability in many countries, while online platforms have expanded reach, especially post-pandemic. Products such as the OraQuick In-Home HIV Test are widely available through Amazon, Walgreens, CVS, and Boots, offering discreet delivery and clear user instructions. Online pharmacies enable access in remote areas, enhance privacy for stigmatized groups, and often include digital support such as video guides and telehealth referrals. In high-income countries such as the U.S., UK, and Germany, online and retail sales dominate, while emerging markets such as India and South Africa are rapidly adopting this channel due to rising e-commerce and pharmacy digitization.
The hospitals and diagnostics laboratories segment is expected to grow significantly over the forecast period. While traditionally central to HIV diagnostics, this channel plays a supportive role in the self-testing landscape by offering confirmatory testing, distributing kits during clinical visits, or integrating them into broader health screening programs. Some hospitals also use self-testing kits such as the INSTI HIV Self Test or BioSure HIV Self Test in pre-counseling or discharge packages, especially in public health facilities. However, growth in this segment is relatively slower, as users increasingly prefer home-based and pharmacy-based testing for privacy and convenience. In countries with universal healthcare (e.g., UK, Canada), hospitals may still distribute self-test kits under government programs. Despite this, the channel is gradually shifting from being a primary distributor to acting more as a referral and follow-up point, supporting post-test care rather than being a core sales driver.
Regional Insights
The North America HIV self-testing market is experiencing strong growth, driven by high public awareness, supportive FDA regulations, and increasing consumer preference for private, at-home testing options. Self-testing is particularly popular among key populations including MSM (men who have sex with men) and younger adults aged 18-35, who are statistically more likely to use online health tools. Over 75% of pharmacies in the U.S. and Canada now offer HIV self-tests, while growing e-commerce sales and digital health campaigns are pushing penetration further. According to the CDC, nearly 40% of high-risk individuals in the U.S. prefer self-testing over traditional clinic visits. The pandemic accelerated adoption of at-home diagnostics, and HIVST benefited from this shift. Government-funded pilot programs and NGO-supported community outreach campaigns are expanding access, especially among underserved communities. Strong internet penetration and insurance coverage for preventive health continue to support market expansion across the region.
U.S. HIV Self-testing Market Trends
The U.S. HIV self-testing market is growing due to a combination of policy support, consumer behavior shifts, and telehealth integration. In 2024, over 60% of HIV self-tests in the U.S. were purchased online, reflecting a preference for privacy and convenience. The CDC’s "Let’s Stop HIV Together" campaign and distribution partnerships with dating apps like Grindr and Tinder have improved awareness among high-risk groups. The FDA’s early approval of OraQuick gave the U.S. a head start in commercialization. Ongoing at-home test reimbursement policies and the inclusion of self-tests in PrEP programs have further fueled adoption. University health centers and community clinics across states like California, New York, and Illinois actively promote self-testing as part of routine sexual health screening. Digital tracking systems and partnerships with telehealth services allow users to self-test and quickly access confirmatory care, reducing barriers to diagnosis and treatment linkage. This digital-health alignment continues to drive adoption.
Europe HIV Self-testing Market Trends
Europe is experiencing strong growth in the HIV self-testing market, fueled by EU-wide public health strategies, regulatory harmonization, and increasing self-care adoption. Several countries have legalized over-the-counter HIV self-test sales, and CE-marked kits such as BioSure and INSTI are widely available. The European Centre for Disease Prevention and Control (ECDC) encourages self-testing to increase testing coverage among key populations like MSM, migrants, and sex workers. Over 30 European countries have adopted HIV self-testing in national strategies, and eHealth tools support distribution in regions with high stigma or rural limitations. Digital outreach and NGO campaigns, particularly during European Testing Week, have significantly increased uptake. University and youth programs in France, Spain, and Italy incorporate self-testing as part of preventive health education. Continued investment in online pharmacy networks and public-private collaborations are expected to enhance accessibility and normalization of HIV self-testing across both Western and Eastern Europe.
The UK HIV self-testing market is growing rapidly, propelled by strong government support, NHS integration, and rising use of digital health services. Initiatives such as HIV Testing Week and the THT (Terrence Higgins Trust) online self-test distribution platform have proven highly effective-over 1 million self-tests have been distributed across the UK through government-backed channels since 2020. A national shift toward remote sexual health services post-COVID has further increased uptake. Studies indicate that nearly 50% of HIV testing among MSM in the UK is now self-initiated, and youth demographics are particularly engaged with discreet, mail-order services. Public health campaigns actively promote self-testing for early diagnosis and linkage to care. HIVST is also included in some regional PrEP access programs. The growing popularity of subscription health boxes and e-pharmacies, combined with favorable CE certification, continues to expand access and normalize HIV self-testing across the UK’s diverse population.
Germany’s HIV self-testing market is witnessing progressive growth, driven by strong public health advocacy, de-stigmatization efforts, and a solid legal framework that allows over-the-counter self-test sales since 2018. Public awareness campaigns supported by the German AIDS Foundation (DAH) and government health agencies have educated citizens on the importance of regular testing, particularly among MSM and migrant populations, which are key at-risk groups. The annual HIV Test Week and AIDS awareness initiatives promote self-testing as a first line of defense. Self-test kits are now available in over 80% of urban pharmacies, with increasing online distribution across platforms like Apotheke.de. Research conducted in Germany shows a 30% increase in self-test usage among men under 40 between 2020 and 2024. Additionally, regional NGOs and community outreach programs distribute free kits to vulnerable populations, expanding access. As cultural attitudes shift and digital health tools evolve, Germany’s HIV self-testing market is expected to grow steadily.
Asia Pacific HIV Self-testing Market Trends
The Asia Pacific HIV self-testing market is growing rapidly, supported by government-led pilot programs, global funding, and rising awareness in high-burden countries such as India, China, and Thailand. Organizations such as WHO, UNITAID, and the Global Fund have funded HIVST rollouts across several countries in the region. In Thailand, national campaigns boosted HIVST awareness by over 50% among MSM and youth in urban areas. In India, the National AIDS Control Organization (NACO) has launched pilot HIVST programs targeting high-risk populations. The rise of e-commerce platforms has increased accessibility in both urban and semi-urban regions. Growing smartphone usage supports digital health tools that guide testing and link users to care. In countries such as Australia and Japan, self-testing is already part of official HIV testing strategies. The combination of policy support, funding, and technological readiness positions Asia Pacific as a high-potential growth region for HIV self-testing in the coming years.
Japan is gradually embracing HIV self-testing, with growth supported by government awareness campaigns, private sector innovation, and increasing acceptance of at-home diagnostics. Historically conservative toward self-testing, Japan has made policy shifts in recent years to allow mail-order test kits, primarily distributed through certified NGOs and private online pharmacies. A rise in anonymous testing preferences among young people and MSM populations has encouraged adoption. Recent surveys indicate a 25% increase in self-test interest among Japanese adults aged 18-30 between 2020 and 2024. Although HIV prevalence in Japan is relatively low, government agencies have incorporated self-testing into broader STD awareness and prevention strategies. Private tech-health startups are also entering the market with digital self-testing platforms, offering AI-guided test instructions and follow-up services. As privacy, convenience, and technological reliability become essential in consumer diagnostics, Japan's HIV self-testing market is expected to grow gradually but steadily.
China’s HIV self-testing market is expanding rapidly, driven by government-led digital health initiatives, NGO partnerships, and strong demand from urban youth. Platforms such as WeChat Health, JD Health, and Alibaba Health now offer approved self-test kits, making access widespread. The Chinese CDC has supported multiple HIVST pilots targeting MSM and student populations, resulting in a 40% increase in voluntary self-testing from 2019 to 2024. National HIV campaigns promote self-testing as a key prevention method, particularly in regions with high incidence like Guangdong and Sichuan. NGOs also collaborate with community clinics to distribute kits to marginalized groups, including migrants and sex workers. The government's “Internet + Healthcare” model encourages telemedicine-linked testing, improving post-test care access. With a growing middle class, digital adoption, and shifting social norms around sexual health, China is poised for significant continued growth in HIV self-testing, particularly via mobile health ecosystems and online pharmacy expansion.
Latin America HIV Self-testing Market Trends
Latin America is experiencing moderate to high growth in HIV self-testing, propelled by public health campaigns, NGO outreach, and increasing government inclusion of HIVST in national strategies. Countries like Brazil, Mexico, and Argentina are leading the charge, integrating HIV self-testing into primary healthcare programs and mobile clinics. Brazil’s Ministry of Health launched a national initiative that led to a 70% increase in test distribution between 2020 and 2024. Self-test kits are now available through pharmacies and online platforms, expanding access for youth and underserved populations. Awareness campaigns focus on de-stigmatization and early diagnosis. Additionally, regional partnerships with organizations like PAHO and UNAIDS support large-scale HIVST pilot programs. Although regulatory pathways vary across countries, most governments are moving toward harmonization and inclusion of HIVST in essential health lists. With rising HIV incidence in certain key populations, the demand for private, accessible testing will continue to fuel market growth.
Middle East and Africa HIV Self-testing Market Trends
The Middle East and Africa region shows strongest potential for long-term HIV self-testing growth, primarily driven by international funding, high HIV burden in Sub-Saharan Africa, and NGO-led distribution. Countries like South Africa, Kenya, and Nigeria have rolled out large-scale HIVST initiatives supported by PEPFAR, UNITAID, and the Global Fund. In Kenya, self-testing uptake among men nearly doubled between 2019 and 2024, due to targeted outreach and availability in pharmacies and community centers. Mobile clinics and digital health pilots also support testing in remote regions. Meanwhile, Middle Eastern countries are cautiously exploring HIVST due to cultural sensitivity but are gradually adopting pilot programs in partnership with NGOs. Language-localized instructions, oral-swab kits, and discreet distribution are expanding access. Despite infrastructure and stigma challenges, ongoing awareness efforts and donor-backed procurement are expected to make HIVST a cornerstone of HIV control in Africa and a rising priority in the Middle East.
Key HIV Self-testing Company Insights
The HIV self-testing market is driven by a mix of established diagnostics companies and specialized innovators focused on expanding access and ease of testing. OraSure Technologies leads with its widely adopted OraQuick In-Home HIV Test, the first FDA-approved oral self-test. BioSURE UK offers highly accurate blood-based kits popular in Europe, while Atomo Diagnostics is known for its all-in-one integrated testing devices, minimizing user error. Chembio Diagnostic Systems develops dual HIV-syphilis kits, expanding test scope. SD Biosensor and CTK Biotech are expanding in Asia and Africa, offering affordable, scalable solutions. Trinity Biotech and Hyrex Diagnostics contribute with global distribution of rapid kits, while Abbott Laboratories supports both clinical and home settings with its combo antigen/antibody technology. Lastly, Genedrive Plc is investing in point-of-care molecular platforms with potential future relevance. Together, these players are shaping a competitive, innovation-driven market aimed at increasing accessibility, privacy, and early HIV detection across diverse global regions.
Key HIV Self-testing Companies:
The following are the leading companies in the HIV self-testing market. These companies collectively hold the largest market share and dictate industry trends.
- OraSure Technologies, Inc.
- BioSURE HIVST
- Atomo Diagnostics
- Chembio Diagnostics, Inc.
- SD Biosensor, INC.
- CTK Biotech, Inc.
- genedrive plc
- Hydrex Diagnostics Sp.
- Trinity Biotech
- Abbott
Recent Developments
-
In July 2025, MedMira Inc., in collaboration with REACH Nexus, announced the early launch of a clinical trial to assess the use of its Multiplo TP/HIV rapid test as a self-test in Canada. The trial began ahead of schedule, supported by an agreement with Health Canada, which allowed MedMira to pursue this label claim without requiring further testing by non-healthcare professionals.
-
In January 2025, OraSure Technologies, Inc., a provider of point-of-need and at-home diagnostic testing solutions, announced that the U.S. Food and Drug Administration’s Center for Biologics Evaluation and Research (CBER) has approved an update to the labeling of its OraQuick HIV Self-Test. This change expands the approved age range, allowing the test to be used by individuals aged 14 and older, compared to the previous minimum age of 17. The update is expected to significantly improve access to HIV testing among adolescents.
-
In June 2024, the New York State Department of Health marked National HIV Testing Day by urging individuals to learn their HIV status. This year’s theme, “Level up your self-love: check your status,” highlights the importance of valuing oneself and addressing personal health needs with compassion, respect, and care. In support of the initiative, the Department also launched a new free HIV self-test distribution campaign to expand access to testing.
HIV Self-testing Market Report Scope
Report Attribute
Details
Market size value in 2025
USD 232.86 million
Revenue forecast in 2033
USD 643.41 million
Growth rate
CAGR of 13.55% from 2025 to 2033
Base year for estimation
2024
Historical data
2021 - 2023
Forecast period
2025 - 2033
Quantitative units
Revenue in USD billion/million, and CAGR from 2025 to 2033
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Type, technology, distribution channel, region
Regional scope
North America; Europe; Asia Pacific; Latin America; MEA
Country scope
U.S.; Canada; Mexico; UK; Germany; France; Italy; Spain; Norway; Sweden; Denmark; Japan; China; India; Australia; South Korea; Thailand; Brazil; Argentina; South Africa; Saudi Arabia; UAE; Kuwait
Key companies profiled
OraSure Technologies, Inc.; BioSURE HIVST; Atomo Diagnostics; Chembio Diagnostics, Inc.; SD Biosensor, INC.; CTK Biotech, Inc.; genedrive plc; Hydrex Diagnostics Sp.; Trinity Biotech; Abbott
Customization scope
Free report customization (equivalent up to 8 analysts’ working days) with purchase. Addition or alteration to country, regional & segment scope
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
Global HIV Self-testing Market Report Segmentation
This report forecasts revenue growth and provides an analysis of the latest trends in each of the sub-segments from 2021 to 2033. For this study, Grand View Research has segmented the global HIV self-testing market based on type, technology, distribution channel, and region:

-
Type Outlook (Revenue, USD Million, 2021 - 2033)
-
Blood-based Kits
-
Saliva-based Kits
-
-
Technology Outlook (Revenue, USD Million, 2021 - 2033)
-
Immunofiltration Assays
-
Lateral Flow Assays (LFA)
-
Other Rapid Diagnostic Technologies
-
-
Distribution Channel Outlook (Revenue, USD Million, 2021 - 2033)
-
Retail and Online Pharmacies
-
Hospitals and Diagnostics Laboratories
-
NGO/ Community-based Distribution
-
-
Regional Outlook (Revenue, USD Million, 2021 - 2033)
-
North America
-
U.S.
-
Canada
-
Mexico
-
-
Europe
-
UK
-
Germany
-
France
-
Italy
-
Spain
-
Denmark
-
Sweden
-
Norway
-
-
Asia Pacific
-
Japan
-
China
-
India
-
Australia
-
South Korea
-
Thailand
-
-
Latin America
-
Brazil
-
Argentina
-
-
Middle East & Africa
-
South Africa
-
Saudi Arabia
-
UAE
-
Kuwait
-
-
Frequently Asked Questions About This Report
b. The global HIV self-testing market size was estimated at USD 211.55 million in 2024 and is expected to reach USD 232.86 million in 2025.
b. The global HIV self-testing market is expected to grow at a compound annual growth rate of 13.55% from 2025 to 2033 to reach USD 643.41 million by 2033.
b. The HIV self-testing market in North America is experiencing strong growth, driven by high public awareness, supportive FDA regulations, and increasing consumer preference for private, at-home testing options.
b. Some key players operating in the HIV self-testing market include OraSure Technologies, Inc., BioSURE HIVST, Atomo Diagnostics, Chembio Diagnostics, Inc., SD Biosensor, INC., CTK Biotech, Inc., genedrive plc, Hydrex Diagnostics Sp., Trinity Biotech, Abbott
b. This growth is fueled by an increasing emphasis on early HIV detection, rising HIV prevalence, and a global shift toward personal, confidential health solutions.
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










