- Home
- »
- Clinical Diagnostics
- »
-
HSV Testing Market Size And Trends, Industry Report, 2033GVR Report cover
![HSV Testing Market Size, Share & Trends Report]()
HSV Testing Market (2025 - 2033) Size, Share & Trends Analysis By Type (HSV-1/HSV-2 Combines, HSV-1, HSV-2), By Test Type (Serological Tests, Point Of Care Tests), By Sample Type (Blood, Swabs, Cerebrospinal Fluid), By End-use, By Region, And Segment Forecasts
- Report ID: GVR-4-68040-688-1
- Number of Report Pages: 150
- Format: PDF
- Historical Range: 2021 - 2023
- Forecast Period: 2025 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
HSV Testing Market Summary
The global HSV testing market size was estimated at USD 611.97 million in 2024 and is expected to reach USD 1017.75 million by 2033, growing at a CAGR of 5.96% from 2025 to 2033. This market encompasses diagnostic solutions aimed at the timely and reliable detection of herpes simplex virus types 1 and 2.
Key Market Trends & Insights
- North America HSV testing market dominated the global industry and accounted for the largest revenue share of 38.72% in 2024.
- The U.S. led the North American market and held the largest revenue share in 2024
- Based on type, the HSV-1/HSV-2 Combines segment dominated the global market and accounted for the largest revenue share of 48.95% in 2024.
- Based on test type, the serological tests segment held the largest revenue share of 49.80% in 2024.
- Based on sample type, the blood segment dominated the HSV testing market with the largest revenue share of 62.36% in 2024.
- Based on end use, the hospitals segment held the largest revenue share of 34.08% in 2024.
Market Size & Forecast
- 2024 Market Size: USD 611.97 Million
- 2033 Projected Market Size: USD 1,017.75 Million
- CAGR (2025-2033): 5.96%
- North America: Largest market in 2024
- Asia Pacific: Fastest growing market
Testing platforms in this space range from traditional serological methods to advanced molecular assays, supporting clinical decisions across diverse healthcare environments. These solutions are primarily utilized in managing sexually transmitted infections and prenatal screening, with growing adoption in decentralized and point-of-care settings. The rising incidence of herpes simplex virus infections, along with heightened public awareness and efforts to improve sexual health screening, is supporting market expansion. Integration of HSV testing with digital platforms and remote diagnostic tools is further expanding access to care, particularly in low-resource and underserved settings, enabling earlier detection and timely clinical intervention.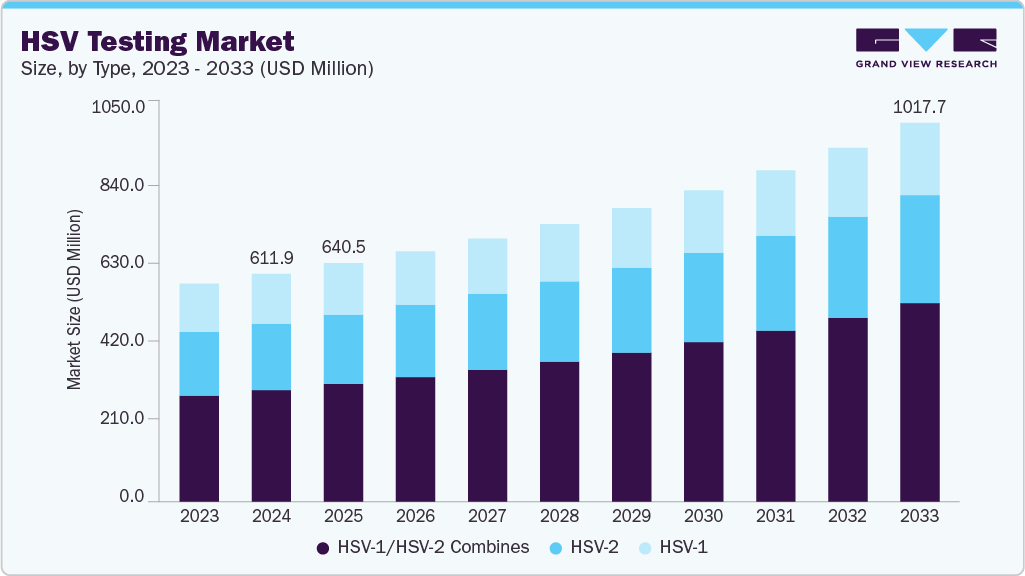
Ongoing advancements in molecular diagnostic technologies are enhancing the sensitivity and turnaround time of HSV testing. Multiplex PCR platforms capable of detecting HSV alongside other sexually transmitted pathogens are gaining clinical preference, particularly in outpatient and emergency care settings. In addition, regulatory approvals for newer test kits with improved sample-to-answer workflows support their use in primary and urgent care environments.
Increased government initiatives and funding for STI surveillance and prevention programs also contribute to market growth. Public health campaigns, especially those targeting adolescent and reproductive health, are driving demand for accessible and accurate HSV testing. Collaborations between public health agencies and private diagnostic providers enable broader test availability, helping reduce diagnostic gaps in developed and resource-limited regions.
Key trends specific to the HSV Testing Market
Growing Demand for Type-Specific Molecular Testing: There is a rising preference for HSV tests that can accurately differentiate between HSV-1 and HSV-2. Molecular diagnostic platforms, such as PCR-based assays, are increasingly used due to their high sensitivity and ability to deliver rapid, type-specific results in both symptomatic and asymptomatic patients.
Expansion of Point-of-Care HSV Testing: Point-of-care HSV testing is gaining ground in community clinics, urgent care centers, and outreach settings. The ability to perform rapid testing without central lab access is driving its adoption, particularly in regions with limited healthcare infrastructure or high STI burden.
Emphasis on Screening During Pregnancy: Clinical focus on HSV testing during prenatal care is intensifying to reduce the risk of neonatal herpes. This trend is supported by updated guidelines and increased awareness among obstetricians and primary care providers regarding the implications of undiagnosed HSV infection during pregnancy.
Integration with Digital Health and Remote Diagnostics: The HSV testing market is seeing greater alignment with digital health platforms, enabling result tracking, remote consultations, and integration with electronic medical records. This is improving access to testing and follow-up care, especially in telehealth-supported environments.
Public Sector Support and STI Control Programs: Government-funded initiatives targeting sexually transmitted infections are driving broader HSV test adoption. Partnerships between diagnostic providers and public health agencies are improving access to testing kits and services, particularly in underserved and high-incidence areas.

Overall, the HSV testing market is evolving with a strong focus on accuracy, accessibility, and integration into broader sexual health strategies. Continued innovation in diagnostic platforms, combined with expanding public health efforts and clinical awareness, is expected to support broader adoption across centralized and decentralized care environments. These factors are reinforcing the role of HSV testing in timely infection management, risk reduction, and improved patient outcomes worldwide.
Market Concentration & Characteristics
Innovation in the HSV testing market is high, with growing focus on faster, more sensitive, and user-friendly diagnostic solutions. Advancements in molecular diagnostics, such as multiplex PCR and automated sample-to-result systems, are improving detection accuracy for both HSV-1 and HSV-2. Integration with smartphone apps and cloud-based data systems is enhancing result interpretation and remote consultation capabilities. Demand for minimally invasive methods and at-home testing kits is also driving innovation.
Mergers and acquisitions are moderately active, particularly as larger diagnostic firms seek to expand their STI testing portfolios through the acquisition of specialized assay developers. These deals aim to broaden access to advanced HSV diagnostic technologies and enter new geographic markets. Collaborations between diagnostics companies and digital health firms are also rising, targeting improved test accessibility and real-time connectivity.
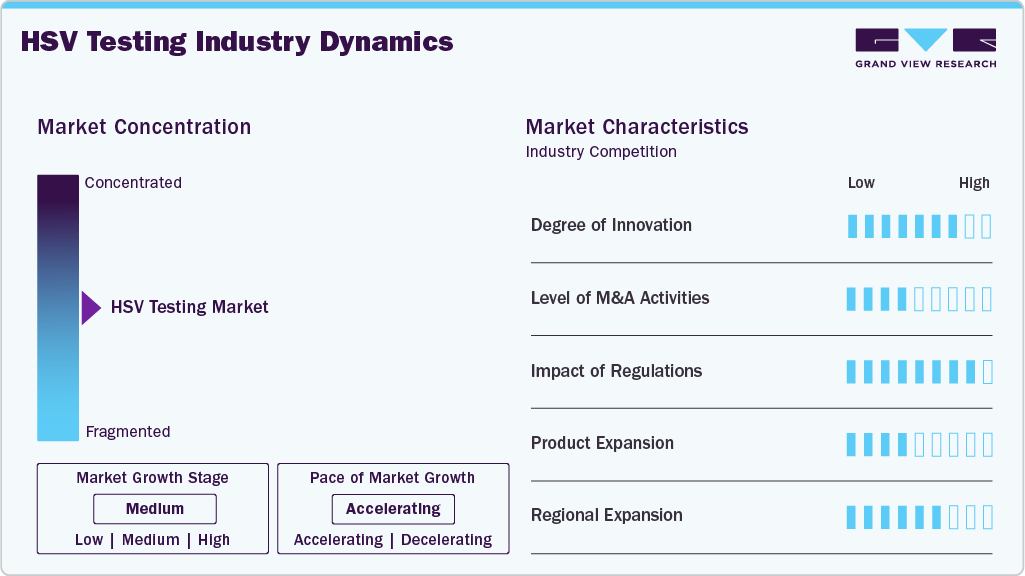
Regulatory oversight plays a crucial role in shaping the HSV testing landscape. Approvals from agencies such as the FDA and CE mark are essential for clinical adoption, especially for molecular-based and point-of-care tests. Increasing emphasis on sexual health screening in national healthcare guidelines is encouraging wider HSV test utilization. However, regulatory requirements around data privacy and home testing are becoming more stringent, particularly in the U.S. and Europe.
The market is witnessing steady product expansion, with the launch of rapid molecular HSV test kits, dual STI test panels, and sample-to-answer devices suitable for both clinical and decentralized use. Manufacturers are focusing on improving test accuracy, ease of use, and integration with electronic health systems. Direct-to-consumer test kits are also emerging, aimed at individuals seeking discreet and convenient testing options.
Regional growth is strong, particularly in Asia-Pacific, Latin America, and parts of Africa, where increasing STI prevalence and healthcare investments are creating demand for accessible HSV diagnostics. Companies are introducing cost-effective and portable solutions tailored to low-resource settings. While North America and Europe remain key markets, emerging economies are becoming vital for expanding testing coverage and volume-based growth.
Type Insights
Based on type, HSV‑1/HSV‑2 combined tests accounted for the largest revenue share of 48.95% in 2024, driven by the demand for comprehensive screening solutions capable of detecting both virus types in a single assay. These dual-target molecular tests are commonly used in clinical and public health settings due to their efficiency, diagnostic accuracy, and ability to inform tailored treatment strategies. The growing need to identify asymptomatic carriers and make informed prenatal decisions has bolstered the adoption of combined kits.
In addition, their portability and compatibility with point-of-care workflows have made them a preferred choice in sexual health clinics and decentralized healthcare environments. A notable recent development: Pune‑based Mylab Discovery Solutions received CDSCO approval in May 2023 for its PathoDetect HSV Type 1 & 2 real‑time PCR kit. This single‑tube multiplex assay delivers results in under two hours and is compatible with automated, high‑throughput systems. It exemplifies advances in rapid, type‑specific testing tailored for clinical labs and resource-limited settings.
The HSV-2 segment is expected to witness notable CAGR during the forecast period, supported by the increasing clinical importance of distinguishing HSV-2 infections due to their stronger association with genital herpes and neonatal transmission risk. Growing awareness of HSV-2-specific complications and its role in co-infections with HIV is prompting healthcare providers to adopt targeted testing strategies. The rise in sexually transmitted infection (STI) screening programs, especially in high-risk populations, is also contributing to the segment's expansion. Advances in molecular testing platforms that offer high sensitivity and specificity for HSV-2 detection are improving diagnostic accuracy in both symptomatic and asymptomatic cases. In addition, integration of HSV-2 assays into multi-pathogen test panels and their use in prenatal and reproductive health settings are further reinforcing demand for segment-specific diagnostics.
Test Type Insights
Serological tests held the largest revenue share in 2024, driven by their widespread use in routine screening, ease of implementation, and relatively lower cost than molecular methods. These tests are particularly valued for their ability to detect HSV antibodies, making them helpful in identifying past exposure and asymptomatic infections. Their application is broad, ranging from population-level surveillance to prenatal care.
A 2024 comparative study published in the Journal of Clinical Microbiology reported that Roche’s HSV-2 IgG test achieved 97.9% sensitivity and 99.5% specificity, outperforming counterparts from Bio-Rad and DiaSorin, highlighting ongoing improvements in assay performance and reliability. However, despite their dominance, concerns persist regarding sensitivity in early-stage infections and the inability to distinguish between active and latent infections. As molecular methods become more accessible, there is growing interest in transitioning to tests that offer higher accuracy and faster turnaround. Nevertheless, serological assays remain the most commonly used HSV test type globally due to their affordability, scalability, and compatibility with high-throughput lab workflows.
The point-of-care (PoC) tests segment is anticipated to grow at the fastest rate over the forecast period, driven by rising demand for rapid, accessible, and decentralized diagnostic solutions. PoC HSV tests enable immediate clinical decision-making in urgent care settings, sexual health clinics, and outreach programs, especially where laboratory infrastructure is limited. This growth is supported by shifting healthcare delivery models that prioritize early detection, minimal patient follow-up, and expanded screening in non-traditional settings. Technological advancements, such as portable molecular platforms and integrated sample-to-result systems, are enhancing test accuracy and usability. In addition, increased adoption of PoC diagnostics in prenatal care and remote screening programs is accelerating segment growth. As healthcare systems emphasize speed, convenience, and broader access, PoC HSV testing is emerging as a key tool in the market’s expansion.
Sample Type Insights
Blood-based testing dominated the market with 62.36% revenue share in 2024, due to its high accuracy, broad clinical acceptance, and ability to detect both HSV-1 and HSV-2 antibodies. Blood samples are widely used in hospital laboratories, prenatal screening programs, and routine sexual health assessments. Their reliability in identifying past exposure and asymptomatic infections has made them the preferred choice for serological testing. Advancements in assay design such as high-sensitivity ELISA and chemiluminescence-based tests-have improved result precision and reduced processing time. Additionally, blood-based tests are often integrated into comprehensive STI panels, offering efficiency in multi-pathogen detection. Despite the growth of non-invasive alternatives, the diagnostic reliability and compatibility of blood samples with high-throughput testing platforms continue to position them as the primary sample type across clinical and public health settings.
The cerebrospinal fluid (CSF) segment is expected to grow at the fastest CAGR over the forecast period, driven by the increasing clinical need for accurate and rapid diagnosis of HSV-related central nervous system (CNS) infections, such as herpes simplex encephalitis and meningitis. HSV PCR testing on CSF samples is considered the gold standard in suspected CNS cases due to its high sensitivity and specificity. The demand for CSF-based testing is rising in emergency departments and intensive care settings, where timely detection is critical for initiating antiviral therapy and reducing neurological complications.
A 2025 clinical study published in Frontiers in Microbiology demonstrated that metagenomic next-generation sequencing (mNGS) detected HSV in CSF samples more sensitively than conventional PCR and also identified unexpected co-infecting pathogens, with results available within approximately three days. Patients with lower mNGS read counts were observed to have better outcomes, reinforcing the prognostic value of early and precise diagnosis. Technological advancements in nucleic acid amplification techniques, automated sample-to-result platforms, and integration with hospital diagnostic workflows are making CSF testing more accessible and efficient. Recent regulatory developments supporting molecular classification for HSV CNS diagnostics are also expected to boost adoption in developed and emerging markets.
End-use Insights
Hospitals dominated the HSV testing market with the highest revenue in 2024, reflecting their central role in diagnosing and managing acute and asymptomatic HSV infections. These facilities rely heavily on accurate and timely diagnostic tools to inform treatment decisions, particularly in prenatal care, sexually transmitted infection (STI) management, and neurological cases involving suspected HSV encephalitis. The availability of laboratory infrastructure and trained personnel enables hospitals to adopt both serological and molecular testing methods, depending on clinical needs. Key growth drivers include the rising burden of HSV-related complications, increasing awareness around neonatal herpes prevention, and growing demand for integrated STI screening. Technological advancements-such as high-throughput analyzers, automated sample handling, and real-time PCR platforms-further improve efficiency and diagnostic confidence within hospital settings.
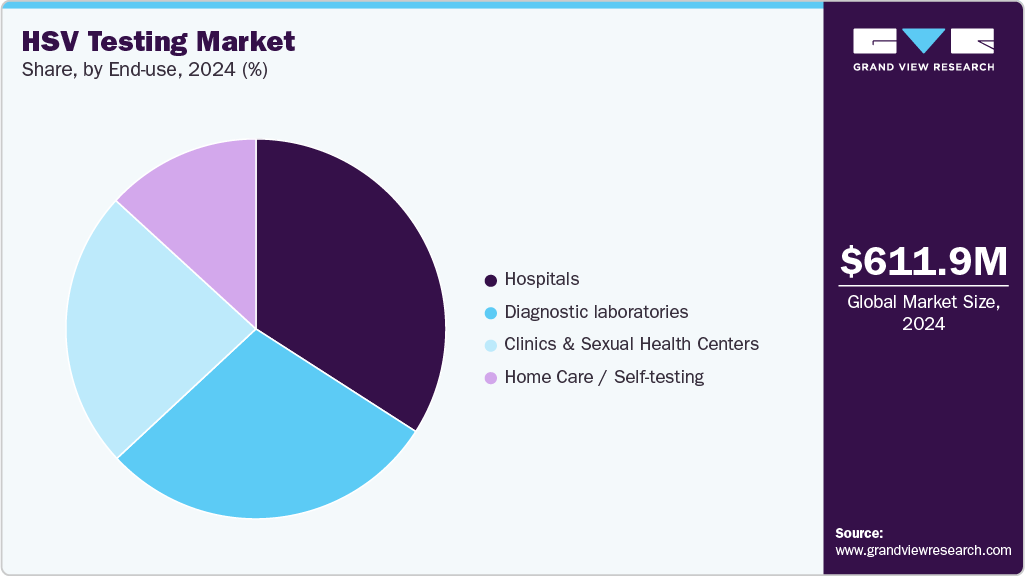
The home care and self-testing segment is expected to grow at the fastest CAGR over the forecast period, driven by increasing demand for privacy, convenience, and early detection of HSV infections. Rising awareness of sexual health and the availability of user-friendly at-home test kits are enabling individuals to screen themselves without visiting a clinic. This trend is further supported by the growing use of digital platforms that allow remote result sharing and teleconsultation. The COVID-19 pandemic accelerated the shift toward self-monitoring, reinforcing the need for decentralized diagnostic options. Newer test kits feature simplified sample collection-often using finger-prick blood or oral swabs-and are enhanced with smartphone-based instructions and cloud-enabled reporting tools. As patients seek greater control over their sexual health and healthcare systems prioritize preventive care, the home testing segment is emerging as a critical growth area within the HSV diagnostics market.
Regional Insights
The North America HSV testing market dominated the global market in 2024, supported by well-established healthcare infrastructure, high awareness of sexually transmitted infections (STIs), and broad access to both laboratory-based and point-of-care diagnostics. Routine screening protocols in sexual health clinics, prenatal care settings, and emergency departments continue to drive testing volumes across the region. Government initiatives promoting STI education and early detection have further supported demand. Moreover, clinical guidelines from organizations such as the CDC reinforce the use of type-specific serological testing, contributing to the widespread adoption of blood-based HSV diagnostics.

High investment in molecular testing technologies and the presence of major diagnostic developers continue to strengthen North America’s position in the global HSV testing market. In December 2023, QuidelOrtho received FDA 510(k) clearance for its Savanna HSV 1+2/VZV PCR assay, a rapid, sample-to-result molecular test that delivers results in approximately 25 minutes. This development reflects the growing emphasis on accessible and decentralized HSV diagnostics, supporting continued growth of the HSV testing market across North America.
U.S. HSV Testing Market Trends
The U.S. HSV testing market leads the North American market, driven by high testing volumes, strong clinical guidelines, and rising awareness around sexual and reproductive health. The country has established protocols for HSV screening in prenatal care, sexual health clinics, and emergency departments, which support widespread use of both serological and molecular diagnostics. The CDC's recommendation for type-specific HSV-2 serological testing in specific clinical scenarios continues to shape diagnostic practices. Increasing integration of HSV tests into broader STI panels and the expansion of point-of-care testing solutions further strengthen adoption. Public health campaigns, digital health integration, and recent FDA approvals for rapid molecular platforms-such as QuidelOrtho's Savanna HSV 1+2/VZV assay-underscore the U.S.'s leadership in delivering accurate, timely HSV diagnostics across care settings.
Europe HSV Testing Market Trends
Europe HSV testing market is witnessing steady growth, supported by rising awareness of sexually transmitted infections (STIs), government-led screening initiatives, and broader access to diagnostic services across the region. Countries such as Germany, the UK, and France are leading adoption, particularly within public healthcare systems that emphasize preventive care and prenatal screening. Expanded reimbursement policies and integration of HSV testing into multi-pathogen STI panels are further driving clinical uptake. In addition, the increasing availability of CE-marked molecular assays and point-of-care platforms is enhancing diagnostic accessibility across outpatient clinics, sexual health centers, and decentralized care settings. The shift toward non-invasive sample collection, combined with digital reporting tools and remote consultation services, improves patient engagement and supports continued market expansion across Western and Central Europe.
The UK HSV testing market is expanding steadily, supported by strong public health programs and NHS-led STI screening protocols. Rising awareness of HSV infections and their complications is driving increased testing in antenatal care and sexual health services. Adoption of type-specific blood tests and multiplex diagnostic panels is growing. The government’s focus on digital health and telemedicine is boosting demand for home-based HSV testing kits. Integration of HSV diagnostics with broader STI programs supports efficiency and reach. These factors position the UK as a key contributor to regional HSV testing growth.
Germany HSV testing market is showing strong growth, supported by its advanced healthcare infrastructure and structured STI screening programs. Widespread use of laboratory-based blood tests in prenatal care and sexual health services drives consistent demand. Type-specific serological testing is commonly used for early detection and clinical decision-making. The country is also witnessing growing interest in molecular diagnostics for HSV-related neurological cases. Increased integration of HSV testing into digital health records and centralized lab systems supports diagnostic efficiency. These trends are contributing to Germany’s role as a key regional market in Europe.
Asia Pacific HSV Testing Market Trends
Asia Pacific HSV testing market is the fastest-growing region in the global market, driven by rising awareness of sexually transmitted infections, growing infection burden, and expanding access to diagnostic services. Countries such as China, India, and Japan are enhancing screening efforts through public health campaigns, prenatal care programs, and digital healthcare adoption. Demand for affordable serological and molecular HSV tests is increasing across urban and semi-urban areas, supported by improvements in healthcare infrastructure. Government-led reproductive health initiatives and broader STI screening strategies are reinforcing early diagnosis and intervention. In addition, the adoption of point-of-care platforms and integration with mobile technologies are improving testing accessibility, supporting the region’s rapid market expansion.
The Japan HSV testing market is expanding gradually, supported by growing public health awareness and routine STI screening protocols. Testing is standard in prenatal care, urology, and dermatology clinics, relying strongly on lab-based blood diagnostics. Molecular tests are gaining attention for neurological HSV cases. Japan’s structured healthcare system ensures quality and consistency in diagnostics. Demand for accurate, type-specific tests continues to grow. These trends support steady market growth in clinical and preventive care settings.
China HSV testing market represents a significant growth opportunity in the global market, driven by growing public health awareness, rising STI incidence, and government-backed reproductive health initiatives. Hospitals and public clinics are increasingly adopting both serological and molecular tests, with a focus on integrating type-specific diagnostics into broader sexual health programs. Demand for accurate, scalable, and cost-effective testing solutions encourages local development and deployment of advanced diagnostic platforms.
In April 2023, Jiangsu Bioperfectus Technologies announced CE IVD certification for its Herpes Simplex Virus Type I/II Real-Time PCR Kit, designed as part of a comprehensive sexually transmitted disease testing portfolio. This reflects China’s growing role in advancing diagnostic capabilities across the region and supports continued expansion in the HSV testing market.
Latin America HSV Testing Market Trends
Latin America HSV testing market is experiencing moderate but rising demand in the HSV testing market, driven by increasing STI awareness and gradual improvements in healthcare access. Countries such as Brazil, Mexico, and Argentina are expanding diagnostic services in public hospitals and sexual health programs. Demand for affordable, easy-to-use serological tests is growing, particularly in low-resource and rural areas. Integration of HSV testing into broader STI screening initiatives supports uptake. Public health campaigns are also promoting early detection and routine screening. These factors are contributing to steady market growth across the region.
Brazil HSV testing market is expanding steadily, supported by rising STI awareness and national efforts to strengthen sexual health services. Public hospitals and community clinics are adopting serological tests for routine screening and prenatal care. Demand for accessible, low-cost diagnostics is driving uptake in underserved regions. Government programs are increasingly integrating HSV testing into broader STI initiatives. Interest in point-of-care and remote testing options is growing. These trends position Brazil as a key growth market within Latin America.
Middle East and Africa HSV Testing Market Trends
The HSV testing market in Middle East & Africa shows emerging growth, driven by rising STI awareness and efforts to expand diagnostic access. Countries such as South Africa, the UAE, and Saudi Arabia are incorporating HSV testing into sexual health and prenatal care programs. Serological testing remains the primary method due to its affordability and ease of use. Infrastructure limitations persist, but public health campaigns and NGO-led initiatives are improving outreach. Integration with broader STI screening is supporting adoption. These developments are contributing to gradual market growth across the region.
Key HSV Testing Company Insights
Key players in the HSV testing market actively focus on developing type-specific diagnostic tools and obtaining regulatory approvals to broaden their test portfolios. Companies also engage in strategic partnerships, collaborations, and acquisitions to expand their global footprint and strengthen their clinical reach. These initiatives aim to enhance the accuracy and speed of HSV detection through both serological and molecular platforms. Efforts are also underway to integrate HSV tests into broader STI panels and point-of-care solutions. Such strategies enable improved access to diagnostics across hospital, outpatient, and remote care environments.
Key HSV Testing Companies:
The following are the leading companies in the HSV testing market. These companies collectively hold the largest market share and dictate industry trends.
- F. Hoffmann-La Roche Ltd.
- Abbott
- Thermo Fisher Scientific Inc.
- BD
- Bio-Rad Laboratories, Inc.
- bioMérieux SA
- DiaSorin S.p.A.
- Hologic, Inc.
- Cepheid
- QuidelOrtho Corporation
Recent Developments
-
In June 2025, the U.S. FDA issued a Final Order classifying herpes simplex virus (HSV) nucleic acid-based assays intended for detecting central nervous system (CNS) infections-such as HSV-related meningitis and encephalitis-as Class II medical devices. This classification establishes a clear regulatory pathway with special controls for molecular diagnostic developers, particularly those working with cerebrospinal fluid (CSF) samples. The decision is expected to encourage further innovation in PCR-based HSV testing and may facilitate broader application to serum- and lesion-swab-based NAAT platforms within clinical settings.
-
In July 2025, Innovative Molecules GmbH announced the successful completion of patient enrollment for the Phase 1b portion of its ongoing Phase 1b/2a clinical trial of IM-250, a next-generation helicase-primase inhibitor targeting herpes simplex virus (HSV). This placebo-controlled study is evaluating the safety, efficacy, and pharmacokinetics of a once-weekly oral dose of IM-250 in patients with recurrent genital herpes. The completion of Phase 1b enrollment represents a significant milestone in the program, paving the way for continued development of IM-250 and its potential to transform a therapeutic landscape that has remained largely unchanged for over 40 years.
-
In October 2024, Microbix Biosystems (Canada) introduced a novel Quality Assessment Product (QAP) for HSV detection in formalin-fixed paraffin-embedded (FFPE) tissue. Co-developed with Sunnybrook Research Institute and tested in collaboration with QuidelOrtho, the QAP is designed to support both immunohistochemistry (IHC) and quantitative PCR (qPCR) workflows. This quality assurance tool enhances the reliability and consistency of molecular HSV diagnostics in tissue-based applications, supporting improved accuracy and standardization in HSV test performance across clinical and reference laboratories.
-
In December 2023, the U.S. FDA issued a safety communication warning about the potential for false-positive results in serological tests for HSV-2, particularly when index values are near the assay’s cutoff. The agency emphasized that misinterpretation of low-positive results could lead to incorrect diagnoses if not confirmed through supplemental testing. In line with CDC guidelines, the communication advised healthcare providers to use confirmatory testing-such as Western blot-before making clinical decisions. This development highlights ongoing concerns related to test specificity and reinforces the importance of diagnostic accuracy and quality control in HSV serologic testing.
-
In January 2023, Molbio Diagnostics (India) launched the Truenat HSV 1/2 real-time RT-PCR test, approved by CDSCO. This compact, battery-operated, chip-based assay delivers results within 60 minutes and integrates seamlessly with the Truelab system-enabling rapid, point-of-care HSV testing in resource-limited and decentralized healthcare settings.
HSV Testing Market Report Scope
Report Attribute
Details
Market size value in 2025
USD 640.48 million
Revenue forecast in 2033
USD 1,017.75 million
Growth rate
CAGR of 5.96% from 2025 to 2033
Base year for estimation
2024
Historical data
2021 - 2023
Forecast period
2025 - 2033
Quantitative units
Revenue in USD million, and CAGR from 2025 to 2033
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Type, test type, sample type, end-use, region
Regional scope
North America; Europe; Asia Pacific; Latin America; MEA
Country scope
U.S.; Canada; Mexico; UK; Germany; France; Italy; Spain; Norway; Sweden; Denmark; Japan; China; India; Australia; South Korea; Thailand; Brazil; Argentina; South Africa; Saudi Arabia; UAE; Kuwait
Key companies profiled
F. Hoffmann-La Roche Ltd.; Abbott; Thermo Fisher Scientific Inc.; BD; Bio-Rad Laboratories, Inc.; bioMérieux SA; DiaSorin S.p.A.; Hologic, Inc.; Cepheid; QuidelOrtho Corporation
Customization scope
Free report customization (equivalent up to 8 analysts’ working days) with purchase. Addition or alteration to country, regional & segment scope
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
Global HSV Testing Market Report Segmentation
This report forecasts revenue growth at the global, regional, and country levels and provides an analysis of the latest trends in each of the sub-segments from 2021 to 2033. For this study, Grand View Research has segmented the global HSV testing market based on type, test type, sample type, end-use, and region:
-
Type Outlook (USD Million, 2021 - 2033)
-
HSV-1/HSV-2 Combines
-
HSV-1
-
HSV-2
-
-
Test Type Outlook (USD Million, 2021 - 2033)
-
Serological tests
-
Direct detection tests
-
PCR
-
Viral culture
-
-
Point-of-care tests
-
-
Sample Type Outlook (USD Million, 2021 - 2033)
-
Blood
-
Swabs
-
Cerebrospinal fluid
-
-
End-use Outlook (USD Million, 2021 - 2033)
-
Hospitals
-
Diagnostic Laboratories
-
Clinics and Sexual Health Centers
-
Home care / Self-testing
-
-
Regional Outlook (USD Million, 2021 - 2033)
-
North America
-
U.S.
-
Canada
-
Mexico
-
-
Europe
-
UK
-
Germany
-
France
-
Italy
-
Spain
-
Denmark
-
Sweden
-
Norway
-
-
Asia Pacific
-
Japan
-
China
-
India
-
Australia
-
South Korea
-
Thailand
-
-
Latin America
-
Brazil
-
Argentina
-
-
Middle East & Africa
-
South Africa
-
Saudi Arabia
-
UAE
-
Kuwait
-
-
Frequently Asked Questions About This Report
b. The global HSV testing market size was estimated at USD 611.97 million in 2024 and is expected to reach USD 640.48 million in 2025.
b. The global HSV testing market is expected to grow at a compound annual growth rate of 5.96% from 2025 to 2033 to reach USD 1,017.75 billion by 2033.
b. North America dominated the HSV testing market in 2024, supported by well-established healthcare infrastructure, high awareness of sexually transmitted infections (STIs), and broad access to both laboratory-based and point-of-care diagnostics
b. Some key players operating in the HSV testing market includeF. Hoffmann-La Roche Ltd.; Abbott; Thermo Fisher Scientific Inc.; BD; Bio-Rad Laboratories, Inc.; bioMérieux SA; DiaSorin S.p.A.; Hologic, Inc.; Cepheid; QuidelOrtho Corporation.
b. Key factors that are driving the market growth include the rising incidence of herpes simplex virus infections, along with heightened public awareness and efforts to improve sexual health screening, which are supporting market expansion.
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










