- Home
- »
- Pharmaceuticals
- »
-
Hypertrophic Cardiomyopathy Therapeutics Market Report 2033GVR Report cover
![Hypertrophic Cardiomyopathy Therapeutics Market Size, Share & Trends Report]()
Hypertrophic Cardiomyopathy Therapeutics Market (2026 - 2033) Size, Share & Trends Analysis Report By Drug Class, (Beta Blockers, Calcium Channel Blockers), By Disease Type, By Route Of Administration, By Distribution Channel, By Region, And Segment Forecasts
- Report ID: GVR-4-68040-873-2
- Number of Report Pages: 150
- Format: PDF
- Historical Range: 2021 - 2024
- Forecast Period: 2026 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
Hypertrophic Cardiomyopathy Therapeutics Market Summary
The global hypertrophic cardiomyopathy therapeutics market size was estimated at USD 1.43 billion in 2025 and is projected to reach USD 2.16 billion by 2033, growing at a CAGR of 5.5% from 2026 to 2033. The industry is expanding due to the increasing adoption of targeted pharmacological therapies that address underlying disease mechanisms.
Key Market Trends & Insights
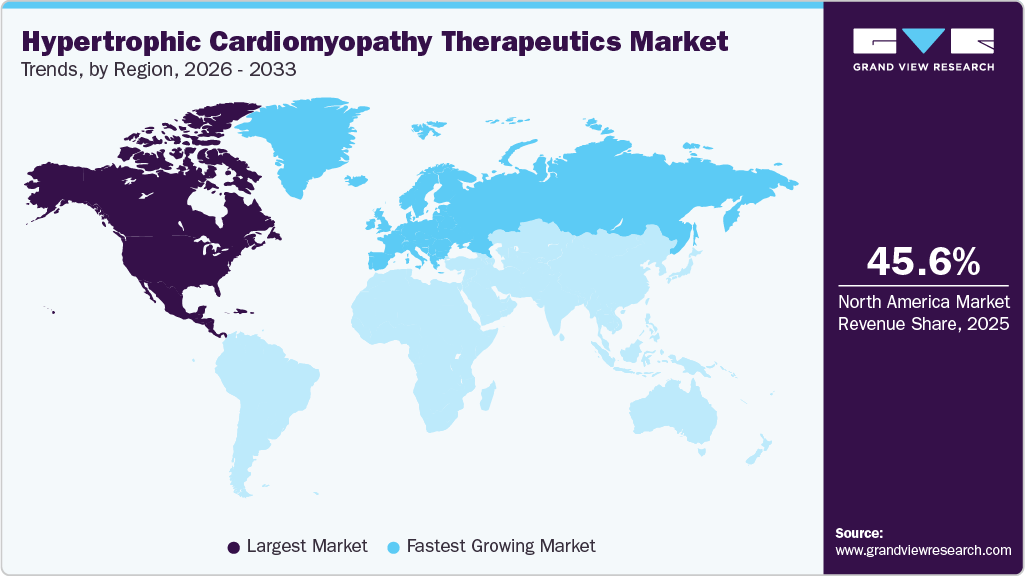
- The North America hypertrophic cardiomyopathy therapeutics market held the largest share of 45.66% of the global market in 2025.
- The hypertrophic cardiomyopathy therapeutics industry in the U.S. is expected to grow significantly over the forecast period.
- By drug class, the beta blockers segment held the largest market share of 36.89% in 2025.
- By disease type, the obstructive hypertrophic cardiomyopathy (oHCM) segment held the largest market share in 2025.
- By route of administration, the oral segment held the largest market share in 2025.
Market Size & Forecast
- 2025 Market Size: USD 1.43 Billion
- 2033 Projected Market Size: USD 2.16 Billion
- CAGR (2026-2033): 5.5%
- North America: Largest market in 2025
- Europe: Fastest growing market
The introduction of cardiac myosin inhibitors has shifted treatment strategies toward disease modification rather than symptom control, supporting long-term clinical management. Rising diagnosis rates driven by advanced imaging technologies and genetic testing have broadened the pool of identifiable patients across age groups. Greater clinical awareness among cardiologists has encouraged earlier intervention. For instance, in January 2025, The Guardian reported that hypertrophic cardiomyopathy, affecting one in 500 people, had been underdiagnosed in women due to a uniform 15 mm wall thickness threshold used for 50 years. Two in three diagnosed patients were men, although women were equally likely to have the condition. A study of 1,600 patients improved female identification by 20 percentage points, and analysis of over 43,000 individuals showed women represented 44 percent under personalized criteria. Improved monitoring and specialty distribution networks further support sustained market growth globally.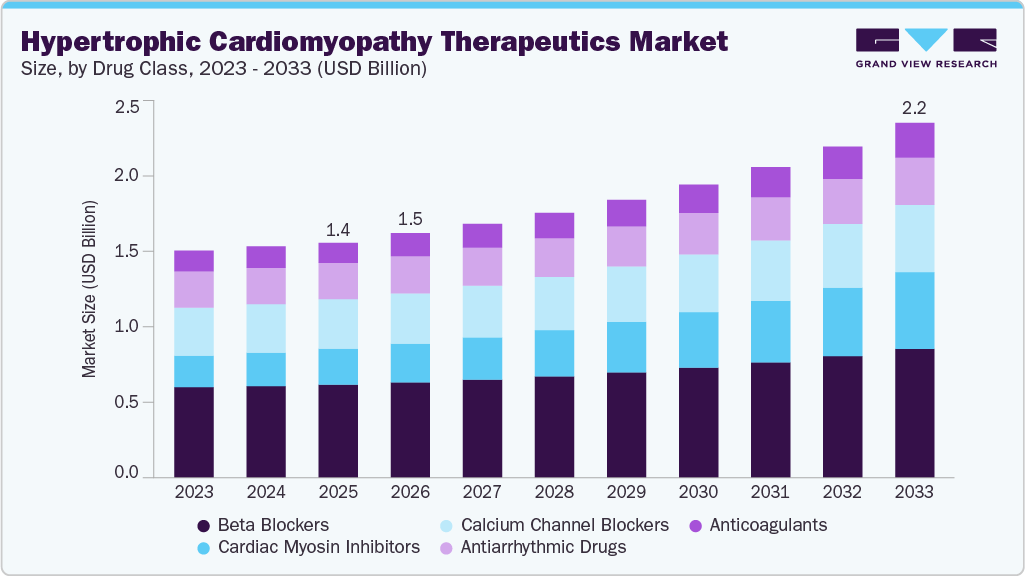
Another major growth driver is the continuous advancement of clinical research and product innovation within the cardiovascular therapeutics pipeline. Pharmaceutical companies are investing in next-generation sarcomere modulators, novel small molecules, and precision medicine approaches designed to improve efficacy and safety profiles. For instance, in February 2026, Elsevier published “Mavacamten and Aficamten in Hypertrophic Cardiomyopathy” on ScienceDirect, stating that hypertrophic cardiomyopathy was defined by left ventricular hypertrophy and myocyte disarray. The article reviewed the roles of mavacamten and aficamten in modifying disease processes rather than only addressing symptoms, and discussed phase 2 and phase 3 studies involving adults with obstructive and nonobstructive phenotypes with sample sizes in the thousands. It referenced reductions in left ventricular outflow tract gradients and improvements in exercise capacity and quality of life compared with placebo benchmarks, highlighting numerical endpoints that supported therapeutic shifts. Expanding clinical validation and regulatory progress continue to accelerate commercial uptake across major pharmaceutical markets.
The market is further supported by the growing burden of cardiovascular disorders and increased recognition of genetic cardiomyopathies in aging populations worldwide. Lifestyle related risk factors, including hypertension and metabolic conditions, have contributed to more frequent cardiac assessments and incidental diagnosis of hypertrophic cardiomyopathy. For instance, in July 2024, Medicom Medical Publishers reported on the phase 3 SEQUOIA-HCM trial evaluating aficamten in obstructive hypertrophic cardiomyopathy. The study enrolled 282 participants randomized 1:1 to aficamten plus standard of care or placebo. Eligibility criteria included a left ventricular outflow tract gradient ≥30 mmHg and Valsalva ≥50 mmHg, New York Heart Association class II or III symptoms, and predicted peak oxygen uptake ≤90% for age and sex. At week 24, peak oxygen uptake improved by +1.8 mL/kg/min versus 0.0 mL/kg/min, with a least-squares mean difference of 1.74 mL/kg/min, 95% CI 1.0 to 2.4, P=0.000002, supporting sustained therapeutic adoption globally.
Market Concentration & Characteristics
The hypertrophic cardiomyopathy therapeutics industry demonstrates a high degree of innovation driven by advances in targeted molecular therapies and precision cardiology. Cardiac myosin inhibitors represent a significant scientific breakthrough that directly modifies sarcomere function and disease progression. Ongoing research focuses on next-generation compounds with improved safety profiles and broader patient eligibility. Biotechnology companies are exploring gene-based therapies and RNA-targeted approaches to address genetic mutations associated with the condition. Clinical data generation and biomarker development continue to refine treatment selection and monitoring strategies. This innovation intensity supports competitive differentiation and premium pricing across advanced therapeutic segments.
Barriers to market entry in this sector are substantial due to complex clinical development requirements and specialized scientific expertise. Drug development for hypertrophic cardiomyopathy involves long trial durations, stringent endpoints, and significant financial investment. Limited patient populations create recruitment challenges and increase research costs for new entrants. Intellectual property protection around novel molecular targets further restricts competition. Established pharmaceutical companies benefit from strong cardiology networks and commercialization infrastructure. These factors collectively limit the number of competitors entering the market.

Regulatory frameworks play a critical role in shaping the market through strict approval and safety monitoring requirements. Therapies that influence cardiac contractility require comprehensive clinical evidence to demonstrate efficacy and cardiovascular safety. Post-marketing surveillance programs and risk evaluation protocols influence prescribing patterns and patient eligibility criteria. Approval timelines vary across regions, affecting global launch strategies and revenue realization. Label expansion approvals based on new clinical evidence create additional growth opportunities for manufacturers. Regulatory oversight also contributes to high development standards and product credibility within the medical community.
Product substitutes in this market include conventional pharmacological therapies and invasive treatment procedures used to manage symptoms. Beta blockers, calcium channel blockers, and antiarrhythmic medications remain widely used first-line treatment options in many patients. Surgical septal myectomy and alcohol septal ablation provide alternatives for patients with severe obstructive disease who do not respond to medication. Device-based interventions such as implantable cardioverter defibrillators address arrhythmia risk rather than structural disease mechanisms. Lifestyle modification and long-term monitoring strategies also complement pharmacological management. Despite these substitutes, novel targeted therapies offer differentiated clinical value through disease-specific mechanisms.
Geographical expansion in the industry is influenced by improvements in cardiovascular diagnostics and specialty care infrastructure across regions. North America and Europe currently represent major revenue contributors due to early adoption of innovative therapies and strong specialist networks. Asia Pacific is emerging as a high-growth region with increasing awareness, urban healthcare development, and expanding cardiology services. Pharmaceutical companies are strengthening regional presence through partnerships, distribution agreements, and clinical collaborations. Market entry strategies often prioritize countries with advanced reimbursement systems and high diagnostic capacity. Broader geographic penetration is expected to support long-term market growth and patient access to novel treatments.
Drug Class Insights
The beta blockers segment dominated the market with the largest revenue share of 36.89% in 2025, driven by their long-standing use as first-line therapy for symptom management in hypertrophic cardiomyopathy patients. Physicians frequently prescribed these medications to control heart rate, reduce myocardial oxygen demand, and improve exercise tolerance. Broad clinical familiarity and established treatment guidelines supported consistent utilization across healthcare settings. For instance, in October 2025, Open Heart published a systematic review and meta-analysis including 21 studies with 775 adults, comprising 262 females, 301 males, and 212 with unreported sex, covering publications from 1964 to 2022. The analysis showed a pooled standardized mean difference of –1.57, 95 percent confidence interval –2.07 to –1.07, in reducing left ventricular outflow tract gradient, and –1.19, 95 percent confidence interval –2.24 to –0.14, in lowering heart rate. Across nine studies with 189 patients, improvements in New York Heart Association class were observed, reinforcing sustained segment dominance.
The cardiac myosin inhibitors segment is projected to grow at a CAGR of 10.2% over the forecast period, fueled by increasing adoption of targeted therapies that address underlying sarcomere dysfunction. These therapies represent a shift toward disease-modifying treatment approaches rather than symptomatic management. Expanding clinical evidence demonstrating improvement in cardiac function and patient outcomes supports physician confidence and broader prescribing trends. For instance, in January 2026, Bristol Myers Squibb announced positive topline results from the phase 3 SCOUT-HCM trial evaluating Camzyos, mavacamten, in adolescents with symptomatic obstructive hypertrophic cardiomyopathy. The randomized, double blind, placebo-controlled study met its primary endpoint with a statistically significant reduction in Valsalva left ventricular outflow tract gradient at week 28 versus placebo in patients aged 12 to <18 years. Secondary endpoints were achieved, and no new safety signals were observed compared with established adult data. The first phase 3 evaluation in this population reinforced disease-modifying potential, supporting premium positioning and accelerating segment expansion globally.
Disease Type Insights
The obstructive hypertrophic cardiomyopathy (oHCM) segment dominated the market, accounting for the largest revenue share of 63.38% in 2025, driven by higher diagnosis rates and well-established treatment pathways for symptomatic patients. Obstructive disease presentation frequently led to earlier clinical evaluation and timely intervention, particularly in individuals experiencing exertional dyspnea, chest pain, or syncope. Availability of approved targeted pharmacological therapies contributed to greater treatment adoption and sustained prescription volumes. For instance, in August 2025, the International Journal of Applied and Basic Medical Research published “Hypertrophic Obstructive Cardiomyopathy: A Brief Account,” reporting that hypertrophic cardiomyopathy affected between 0.2 percent and 0.5 percent of the global population, equivalent to one in 500 to one in 200 individuals, translating to an estimated 2 million to 7 million cases in India. A 2023 study identified 34 MYBPC3 variants among 115 Indian patients, including 19 novel variants and 2.6 percent compound heterozygous mutations, with a 4 percent allele frequency for a 25-base pair intron 32 deletion, while myocardial fibrosis was observed in over 90 percent of symptomatic patients.
The non-obstructive hypertrophic cardiomyopathy (nHCM) segment is projected to grow at a CAGR of 6.1% over the forecast period over the forecast period owing to rising research focus and emerging therapeutic options targeting unmet clinical needs. Historically, limited treatment choices have created demand for innovative pharmacological solutions. For instance, in November 2025, Elsevier published a state-of-the-art review in JACC: Heart Failure reporting that hypertrophic cardiomyopathy affected more than 1 in 500 individuals globally, with non-obstructive forms accounting for approximately 30% to 70% of cases. The review noted substantial heterogeneity and morbidity, highlighting daily life limitations, ventricular tachyarrhythmias, progression to heart failure, and atrial fibrillation despite the absence of left ventricular outflow tract obstruction. It emphasized that no pharmaceutical therapies had been approved specifically for non-obstructive HCM and that several clinical trials are ongoing, underscoring unmet need. Advancements in diagnostics, increasing physician awareness, and the expansion of precision medicine approaches are expected to accelerate segment growth.
Route of Administration Insights
The oral segment dominated the market with the largest revenue share of 75.68% in 2025, due to convenience, ease of administration, and suitability for chronic disease management. Most conventional therapies for hypertrophic cardiomyopathy are available in oral formulations, supporting widespread utilization. Patients generally prefer oral medications for long-term treatment adherence. Lower administration complexity reduces the need for clinical supervision. Oral targeted therapies entering the market further strengthen this segment. Cost advantages compared with injectable options support broader accessibility. These factors collectively maintain the dominance of oral administration.
The injectable / parenteral segment is projected to grow at a CAGR of 6.4% over the forecast period, owing to advancements in biologics, gene therapies, and specialized treatment approaches. Emerging pipeline therapies are exploring injectable delivery methods to achieve sustained therapeutic effects. Complex cases requiring intensive management may benefit from parenteral administration. Clinical innovation in gene-based treatments contributes to long-term growth potential. Increasing investment in advanced cardiovascular therapeutics supports segment expansion. Healthcare providers are adopting specialized treatment modalities for patients with severe conditions. These developments are expected to accelerate growth in this segment.
Distribution Channel Insights
The hospital pharmacies segment dominated the market, accounting for the largest revenue share of 52.53% in 2025, driven by treatment initiation primarily in specialized cardiology care settings. Patients often receive diagnosis and early therapy management in hospitals or specialty clinics. Physician supervision and monitoring requirements support hospital-based dispensing. Availability of advanced diagnostic and treatment facilities strengthens this distribution channel. Complex cases requiring multidisciplinary management are frequently handled in hospital environments. Institutional purchasing agreements contribute to higher dispensing volumes. These factors collectively support the segment’s leading position.
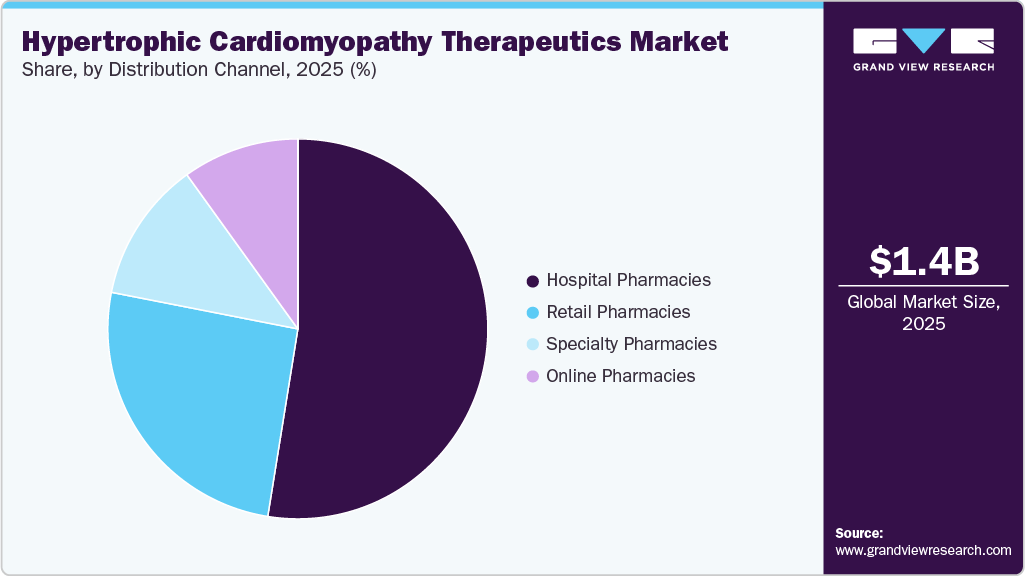
The online pharmacies segment is projected to grow at a CAGR of 9.7% over the forecast period, due to increasing patient preference for convenient medication access and home delivery services for chronic disease management. Rising adoption of digital health platforms and e-commerce integration within healthcare systems is supporting prescription fulfillment through online channels. Patients receiving long-term therapies for hypertrophic cardiomyopathy benefit from automated refill programs and remote ordering options that improve adherence. The expansion of teleconsultation services has further enabled electronic prescriptions, facilitating the use of online pharmacies. The availability of specialty medications through certified digital pharmacies is improving patient access in geographically distant areas. Competitive pricing strategies and discounts offered by online platforms are also contributing to increased adoption across diverse patient populations.
Regional Insights
The North America hypertrophic cardiomyopathy therapeutics market held the largest market share of 45.66% in 2025, due to advanced healthcare infrastructure and high adoption of innovative cardiovascular drugs. A strong presence of leading pharmaceutical companies supports the rapid commercialization of newly approved therapies across the region. Early diagnosis through widespread use of genetic testing and cardiac imaging contributes to a larger treated patient population. High healthcare spending enables access to premium specialty medications and long-term disease management programs. Established centers of excellence for cardiomyopathy care improve treatment outcomes and physician familiarity with novel therapies. These factors collectively sustain the region’s leading market position and revenue contribution.
U.S. Hypertrophic Cardiomyopathy Therapeutics Market Trends
The hypertrophic cardiomyopathy therapeutics industry in the U.S. accounts for the largest share in North America, driven by high disease awareness and strong clinical research activity. The availability of advanced diagnostic tools supports early detection and prompt treatment initiation across diverse patient groups. Favorable reimbursement coverage for specialty cardiovascular therapies increases patient access to innovative medications. The presence of major pharmaceutical manufacturers accelerates product launches and physician education initiatives. Robust clinical trial participation strengthens confidence in emerging treatment options. The country continues to lead global revenue generation through consistent adoption of advanced therapeutics.
Europe Hypertrophic Cardiomyopathy Therapeutics Market Trends
The hypertrophic cardiomyopathy therapeutics industry in Europe is expected to register the fastest CAGR of 6.8% over the forecast period, due to increasing adoption of targeted cardiovascular therapies and expanding access to advanced diagnostic technologies across the region. Growing awareness among healthcare professionals of inherited cardiac disorders is contributing to earlier detection and treatment initiation. The presence of established cardiology centers and multidisciplinary care programs is improving the management of complex hypertrophic cardiomyopathy cases. Rising clinical research activity and participation in multinational trials are supporting the availability of innovative treatment options. Expanding use of genetic testing and cardiac imaging is enhancing patient identification and risk stratification. These factors collectively are expected to accelerate therapeutic uptake and market growth throughout the forecast period.
The UK hypertrophic cardiomyopathy therapeutics industry is driven by strong clinical expertise in cardiology and increasing recognition of inherited cardiac disorders. The availability of specialized diagnostic programs improves the identification of hypertrophic cardiomyopathy across different age groups. Growing adoption of advanced pharmacological therapies supports improved disease management outcomes. Collaborative research initiatives between academic institutions and industry enhance treatment innovation. Access to multidisciplinary cardiac care centers improves patient monitoring and therapy optimization. These factors contribute to gradual market expansion within the country.
The hypertrophic cardiomyopathy therapeutics industry in Germany represents a major European market due to its advanced healthcare facilities and high cardiovascular disease management standards. Strong physician awareness and diagnostic capabilities contribute to consistent patient identification and treatment rates. Adoption of innovative cardiovascular medications is supported by established clinical guidelines and specialist networks. The presence of pharmaceutical manufacturing and research activities enhances the availability of advanced therapies. Increasing focus on early intervention strategies improves long-term treatment demand. The country maintains a stable growth trajectory within the regional market.
The France hypertrophic cardiomyopathy therapeutics industry shows steady growth in the market, supported by improvements in cardiology diagnostics and patient management programs. Increasing awareness of hereditary cardiac conditions helps detect and monitor them earlier. Availability of specialized cardiac centers enhances access to advanced treatment options for symptomatic patients. Adoption of innovative therapies is gradually expanding across tertiary care facilities. Clinical research participation strengthens a physician’s familiarity with emerging treatment approaches. These factors collectively support moderate market growth across the country.
Asia Pacific Hypertrophic Cardiomyopathy Therapeutics Market Trends
The hypertrophic cardiomyopathy therapeutics industry in the Asia Pacific is experiencing growth due to expanding healthcare access and improving diagnostic capabilities across developing and developed economies in the region. The rising prevalence of cardiovascular disorders is increasing the number of patients undergoing cardiac evaluation and specialist consultation. Growing healthcare expenditure among urban populations is supporting the adoption of advanced pharmacological therapies and innovative treatment options. Expansion of private healthcare providers and specialty cardiac centers is improving access to cardiology expertise and long-term disease management services. Increasing awareness among healthcare professionals is contributing to earlier diagnosis and timely therapeutic intervention. The region offers substantial untapped potential for pharmaceutical companies seeking broader patient reach and future commercial expansion.
The hypertrophic cardiomyopathy therapeutics industry in Japan represents a key market in the Asia Pacific, driven by advanced medical technology adoption and strong cardiology expertise. High availability of diagnostic imaging and genetic screening supports accurate disease identification. Aging population trends contribute to increased clinical evaluation for cardiac conditions. Adoption of innovative cardiovascular therapies is supported by physician familiarity with precision medicine approaches. The presence of domestic pharmaceutical research enhances therapeutic development and commercialization. The country maintains a consistent demand for advanced treatment options within the region.
The China hypertrophic cardiomyopathy therapeutics industry is experiencing rapid growth due to expanding healthcare infrastructure and increasing cardiovascular disease awareness. Large population size contributes to a substantial undiagnosed patient pool with future treatment potential. Improvements in hospital capabilities and diagnostic technologies support rising detection rates. Growing urban healthcare spending enables middle-income populations to access innovative specialty medications. Expansion of pharmaceutical partnerships strengthens distribution and market penetration. These dynamics position China as a major growth contributor in the Asia Pacific.
Latin America Hypertrophic Cardiomyopathy Therapeutics Market Trends
The hypertrophic cardiomyopathy therapeutics industry in Latin America demonstrates emerging growth potential supported by improving healthcare access and increasing awareness of cardiovascular diseases. The expansion of private healthcare services enhances the availability of advanced diagnostic tools in major urban centers. Rising physician education initiatives contribute to improved recognition of hypertrophic cardiomyopathy symptoms. Adoption of innovative pharmacological therapies remains concentrated in higher-income populations. Increasing collaborations with international pharmaceutical companies support treatment availability. The region shows gradual expansion with opportunities for future market development.
The Brazil hypertrophic cardiomyopathy therapeutics industry represents the largest market in Latin America, driven by growing cardiovascular disease burden and expanding healthcare services. Increasing availability of cardiac imaging and specialist consultations improves disease detection rates. The adoption of innovative therapies is gradually increasing in private healthcare institutions. Urban population growth increases treatment demand in metropolitan areas. Pharmaceutical company presence supports product distribution and physician training initiatives. These factors collectively support steady market development within the country.
Middle East & Africa Hypertrophic Cardiomyopathy Therapeutics Market Trends
The hypertrophic cardiomyopathy therapeutics industry in the Middle East and Africa shows moderate growth, supported by improving healthcare infrastructure in selected countries. Increasing investment in advanced medical facilities enhances access to specialized cardiology services. Rising awareness of genetic disorders contributes to improved diagnosis in urban healthcare centers. Adoption of innovative therapies remains limited to higher-income segments of the population. Expansion of private hospitals supports the availability of advanced treatment options. The region presents long-term growth potential as healthcare capabilities continue to evolve.
The Saudi Arabia hypertrophic cardiomyopathy therapeutics industry is a leading market within the Middle East, driven by expanding healthcare modernization and strong demand for advanced medical treatments. The increasing prevalence of cardiovascular risk factors contributes to higher diagnostic screening rates. The availability of specialized cardiac centers improves patients' access to advanced therapeutic options. Growing physician awareness supports the adoption of innovative pharmacological treatments for hypertrophic cardiomyopathy. Pharmaceutical partnerships enhance supply and distribution efficiency within the country. These factors collectively support steady market growth in Saudi Arabia.
Key Hypertrophic Cardiomyopathy Therapeutics Company Insights
Bristol Myers Squibb and Cytokinetics, Inc. are expanding their presence in the hypertrophic cardiomyopathy therapeutics market through the development and commercialization of cardiac myosin inhibitors that target core disease mechanisms. Novartis AG and Bayer AG are advancing cardiovascular research programs through strategic collaborations and investments in precision medicine. Sanofi is participating in specialty therapeutic segments through ongoing research initiatives and portfolio diversification within cardiovascular care. Tenaya Therapeutics, Rocket Pharmaceuticals, and Lexeo Therapeutics are focusing on gene therapy platforms to address the genetic causes of hypertrophic cardiomyopathy. Imbria Pharmaceuticals and Edgewise Therapeutics are advancing novel metabolic and sarcomere-modulating therapies through their respective clinical development pipelines. The market landscape reflects increased innovation, partnerships, and an emphasis on targeted treatment approaches to improve patient outcomes and expand therapeutic options.
Key Hypertrophic Cardiomyopathy Therapeutics Companies:
The following key companies have been profiled for this study on the hypertrophic cardiomyopathy therapeutics market.
- Bristol Myers Squibb
- Cytokinetics, Inc.
- Novartis AG
- Sanofi
- Bayer AG
- Tenaya Therapeutics
- Imbria Pharmaceuticals
- Rocket Pharmaceuticals
- Lexeo Therapeutics
- Edgewise Therapeutics
Recent Developments
-
In January 2026, Bristol Myers Squibb announced positive topline results from the phase 3 SCOUT-HCM trial of Camzyos, mavacamten, in adolescents aged 12 to <18 years with symptomatic obstructive hypertrophic cardiomyopathy. The randomized, double blind, placebo controlled study met its primary endpoint, showing a statistically significant reduction in Valsalva left ventricular outflow tract gradient at week 28 versus placebo, with no new safety signals observed.
-
In January 2026, Cytokinetics announced that MYQORZO, aficamten, was commercially available in the U.S. for adults with symptomatic obstructive hypertrophic cardiomyopathy following FDA approval on December 19, 2025. The therapy was launched in 5 mg, 10 mg, 15 mg, and 20 mg tablets and distributed under a Risk Evaluation and Mitigation Strategy program. Treatment required echocardiographic monitoring, with avoidance recommended in patients with a left ventricular ejection fraction below 55 percent.
-
In January 2026, Sanofi announced that the National Medical Products Administration in China approved two of its innovative medicines, Myqorzo (aficamten) for the treatment of obstructive hypertrophic cardiomyopathy and Redemplo (plozasiran) for adult patients with familial chylomicronemia syndrome. The approval included Myqorzo for oHCM and Redemplo for FCS based on pivotal phase 3 studies, expanding treatment options for these serious conditions. The press release underscored Sanofi’s commitment to addressing unmet medical needs and enhancing patient access in Greater China.
Hypertrophic Cardiomyopathy Therapeutics Market Report Scope
Report Attribute
Details
Market size value in 2026
USD 1.48 billion
Revenue forecast in 2033
USD 2.16 billion
Growth rate
CAGR of 5.5% from 2026 to 2033
Base year for estimation
2025
Historical data
2021 - 2024
Forecast period
2026 - 2033
Quantitative units
Revenue in USD million/billion and CAGR from 2026 to 2033
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, trends
Segments covered
Drug class, disease type, route of administration, distribution channel, region
Regional scope
North America; Europe; Asia Pacific; Latin America; Middle East & Africa
Country scope
U.S.; Canada; Mexico; UK; Germany; France; Italy; Spain; Denmark; Sweden; Norway; China; Japan; India; Australia; South Korea; Thailand; Brazil; Argentina; South Africa; Saudi Arabia; UAE; Kuwait
Key company profiled
Bristol Myers Squibb, Cytokinetics, Inc.; Novartis AG; Sanofi; Bayer AG; Tenaya Therapeutics; Imbria Pharmaceuticals; Rocket Pharmaceuticals; Lexeo Therapeutics; Edgewise Therapeutics
Customization scope
Free report customization (equivalent up to 8 analysts working days) with purchase. Addition or alteration to country, regional & segment scope.
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
Global Hypertrophic Cardiomyopathy Therapeutics Market Report Segmentation
This report forecasts revenue growth at global, regional, and country levels and provides an analysis of the latest industry trends in each of the sub-segments from 2021 to 2033. For this study, Grand View Research has segmented the global hypertrophic cardiomyopathy therapeutics market report based on drug class, disease type, route of administration, distribution channel, and region:
-
Drug Class Outlook (Revenue, USD Million, 2021 - 2033)
-
Beta Blockers
-
Calcium Channel Blockers
-
Antiarrhythmic Drugs
-
Cardiac Myosin Inhibitors
-
Anticoagulants
-
Others
-
-
Disease Type Outlook (Revenue, USD Million, 2021 - 2033)
-
Obstructive Hypertrophic Cardiomyopathy (oHCM)
-
Non-Obstructive Hypertrophic Cardiomyopathy (nHCM)
-
-
Route of Administration Outlook (Revenue, USD Million, 2021 - 2033)
-
Oral
-
Injectable / Parenteral
-
Others
-
-
Distribution Channel Outlook (Revenue, USD Million, 2021 - 2033)
-
Hospital Pharmacies
-
Retail Pharmacies
-
Online Pharmacies
-
Specialty Pharmacies
-
-
Regional Outlook (Revenue, USD Million, 2021 - 2033)
-
North America
-
U.S.
-
Canada
-
Mexico
-
-
Europe
-
UK
-
Germany
-
France
-
Italy
-
Spain
-
Denmark
-
Sweden
-
Norway
-
-
Asia Pacific
-
Japan
-
China
-
India
-
Australia
-
South Korea
-
Thailand
-
-
Latin America
-
Brazil
-
Argentina
-
-
Middle East & Africa
-
South Africa
-
Saudi Arabia
-
UAE
-
Kuwait
-
-
Frequently Asked Questions About This Report
b. The global hypertrophic cardiomyopathy therapeutics market size was estimated at USD 1.43 billion in 2025 and is projected to reach USD 1.48 billion in 2026.
b. The global hypertrophic cardiomyopathy therapeutics market is expected to grow at a compound annual growth rate of 5.5% from 2026 to 2033, reaching approximately USD 2.16 billion by 2033.
b. Based on drug class, the beta blockers segment dominated the hypertrophic cardiomyopathy therapeutics market with the largest revenue share of 36.89% in 2025, driven by their long standing use as first line therapy for symptom management. These medications help control heart rate, reduce myocardial oxygen demand, and improve exercise tolerance in patients with hypertrophic cardiomyopathy. Their broad clinical familiarity, established treatment guidelines, and wide availability across healthcare settings continue to support strong adoption among physicians.
b. Key players operating in the hypertrophic cardiomyopathy therapeutics market include Bristol Myers Squibb, Cytokinetics, Inc., Novartis AG, Sanofi, Bayer AG, Tenaya Therapeutics, Imbria Pharmaceuticals, Rocket Pharmaceuticals, Lexeo Therapeutics, and Edgewise Therapeutics. These companies are actively engaged in developing innovative cardiovascular therapies, including cardiac myosin inhibitors, gene therapies, and targeted small molecules aimed at improving disease management and patient outcomes.
b. Key factors driving market growth include increasing prevalence and diagnosis of hypertrophic cardiomyopathy, advancements in cardiac imaging and genetic testing, and growing adoption of targeted pharmacological therapies such as cardiac myosin inhibitors. Rising clinical research activity and investments in precision cardiology are also supporting the development of innovative therapies designed to modify disease progression rather than only manage symptoms.
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










