- Home
- »
- Biotechnology
- »
-
Induced Pluripotent Stem Cells Production Market Report, 2033GVR Report cover
![Induced Pluripotent Stem Cells Production Market Size, Share & Trends Report]()
Induced Pluripotent Stem Cells Production Market (2025 - 2033) Size, Share & Trends Analysis Report By Process, By Workflow (Reprogramming, Cell Culture), By Product, By Application, By End Use, By Region, And Segment Forecasts
- Report ID: GVR-4-68039-549-7
- Number of Report Pages: 120
- Format: PDF
- Historical Range: 2021 - 2023
- Forecast Period: 2025 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
Induced Pluripotent Stem Cells Production Market Summary
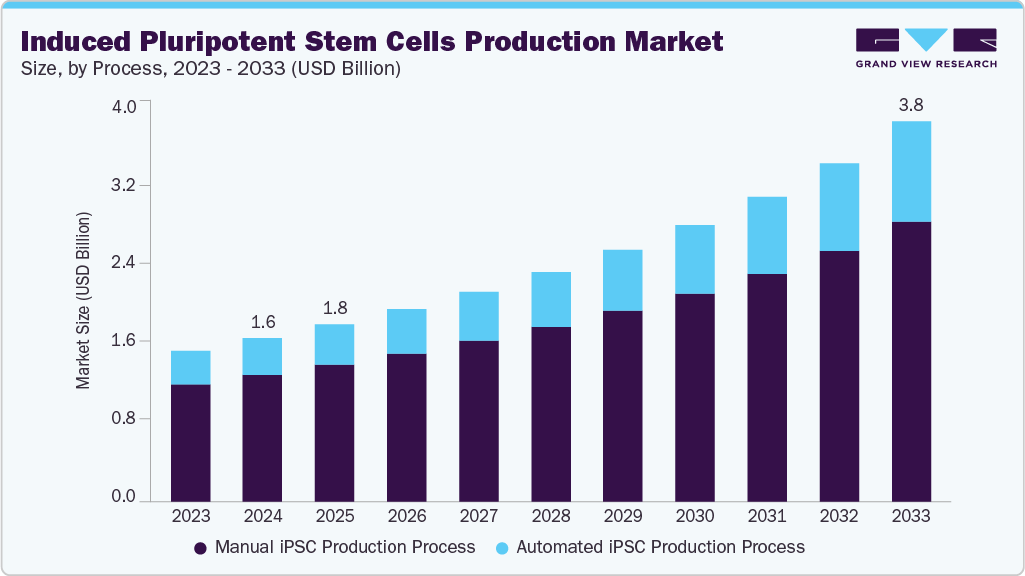
The global induced pluripotent stem cells production market size was estimated at USD 1.62 billion in 2024 and is projected to reach USD 3.78 billion by 2033, growing at a CAGR of 10.01% from 2025 to 2033. The potential of stem cell-based therapies, the growing incidence of cancer, capital investments for stem cell-based research, and multiple advantages of induced pluripotent stem cells (iPSCs) over embryonic stem cells (ESCs) are driving the market growth significantly.
Key Market Trends & Insights
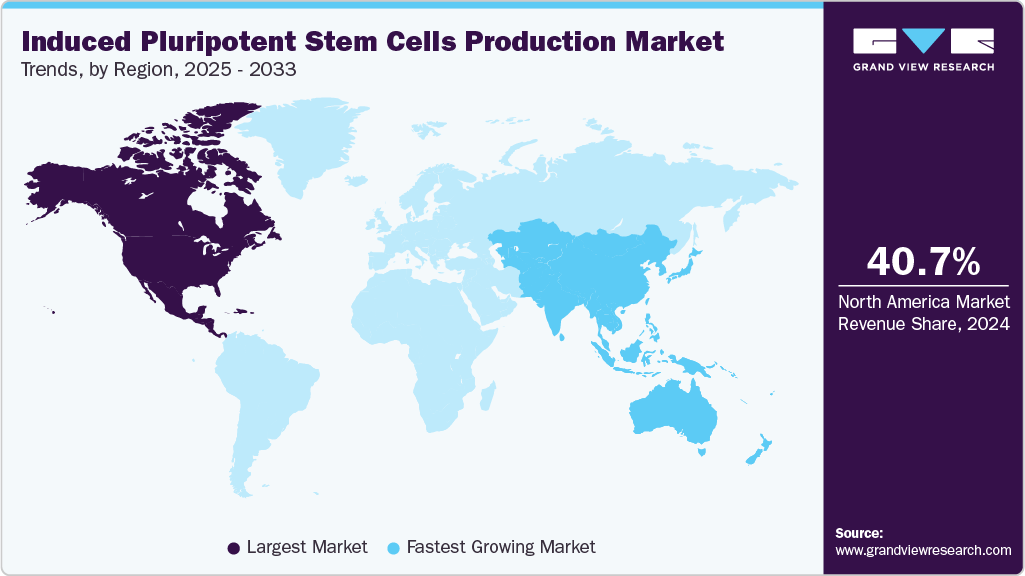
- The North America induced pluripotent stem cells production market held the largest global revenue share of 40.66% in 2024.
- The induced pluripotent stem cells (iPSC) production market in the U.S. is expected to grow from 2025 to 2033.
- Based on process, the manual iPSC production process segment held the highest market share of 77.55% in 2024.
- By workflow, the cell culture segment held the highest market share in 2024.
- By product, the consumables & kits segment held the highest market share in 2024.
Market Size & Forecast
- 2024 Market Size: USD 1.62 Billion
- 2033 Projected Market Size: USD 3.78 Billion
- CAGR (2025-2033): 10.01%
- North America: Largest Market in 2024
- Asia Pacific: Fastest-growing Market from 2025 to 2033
Expanding Role of iPSC-Derived Cells in Drug Discovery and Toxicology Screening
The use of induced pluripotent stem cell (iPSC)-derived cells in drug discovery and toxicology screening is emerging as a major growth driver for the iPSC production market. Pharma and biotech companies are increasingly adopting iPSC-derived cardiomyocytes, hepatocytes, and neuronal cells to create physiologically relevant in vitro models that better mimic human biology than conventional cell lines or animal models. These models enable high-throughput screening, improve early-stage safety and efficacy assessments, and align with the global push to reduce animal testing, meeting ethical and regulatory expectations.
Ongoing Clinical Trials Related to iPSC
NCT Number
Conditions
Sponsor
Completion Time
NCT05886205
Refractory Focal Epilepsy
Peking Union Medical College Hospital
2025
NCT07065409
Moyamoya Disease
Huaqiu Zhang
2026
NCT06367673
AML, Adult
Zhejiang University
2026
NCT05445063
Macular Degeneration
Beijing Tongren Hospital
2026
NCT04339764
Dry Age-Related Macular Degeneration
National Eye Institute (NEI)
2029
NCT05647213
Univentricular Heart
HeartWorks, Inc.
2029
NCT06342986
Gynecologic Cancer
Masonic Cancer Center, University of Minnesota
2027
NCT06027853
AML, Adult
Zhejiang University
2026
NCT05643638
Graft Versus Host Disease, Acute
Cynata Therapeutics Limited
2027
NCT06692426
Duchenne Muscular Dystrophy
Masonic Cancer Center, University of Minnesota
2027
NCT06255028
Systemic Lupus Erythematosus
Century Therapeutics, Inc.
2028
NCT06482268
PD - Parkinson's Disease
University of California, San Diego
2028
NCT06138210
Acute Ischemic Stroke
Xuanwu Hospital, Beijing
2025
NCT06951074
Stem Cells
Ain Shams University
2026
NCT05774509
Heart Failure With Reduced Ejection Fraction
Assistance Publique - Hôpitaux de Paris
2026
NCT07111325
Lateral Epicondylitis
Shanghai 6th People's Hospital
2025
NCT06687837
Parkinson Disease
Jeffrey S. Schweitzer, MD, PhD
2028
NCT04396899
Heart Failure
University Medical Center Goettingen
2027
NCT06789445
Primary Photoreceptor Disease
BlueRock Therapeutics
2030
NCT06394232
Retinal Disease
Eyestem Research Pvt. Ltd.
2030
NCT06299033
Chronic Ischemic Stroke
Hopstem Biotechnology Inc.
2025
NCT06978920
Parkinson Disease
Nuwacell Biotechnologies Co., Ltd.
2028
Source: Clinical trial, Secondary Research, Grand View Research
Regulatory bodies such as the FDA and EMA actively encourage human cell-based testing to improve translational predictability and reduce costly late-stage clinical failures. iPSC-based platforms support precision medicine by enabling patient-specific testing for differential drug responses across genetic profiles. As demand for safer, more effective therapeutics rises, iPSC-derived models' scalability, reproducibility, and predictive accuracy drive sustained demand for advanced production and differentiation platforms, solidifying their role in modern drug development pipelines.
Growing Adoption of iPSC in Personalized and Precision Medicine
The increasing emphasis on personalized and precision medicine is a key factor fueling the demand for induced pluripotent stem cell (iPSC) production. iPSCs can be generated from a patient’s somatic cells, such as skin fibroblasts or blood cells, and subsequently differentiate into virtually any cell type. This unique capability significantly reduces the risk of immune rejection in therapeutic applications, enabling the development of patient-specific treatments for a broad range of chronic and degenerative diseases, including cardiovascular disorders, neurodegenerative conditions, and metabolic syndromes.
Moreover, iPSC technology supports creating disease-specific cell models that reflect the exact genetic and molecular profile of individual patients. These models are instrumental in understanding disease mechanisms, identifying novel drug targets, and testing treatment responses in a controlled, patient-matched environment. As healthcare systems worldwide shift towards precision therapies aimed at maximizing efficacy while minimizing side effects, demand for scalable, GMP-compliant iPSC production platforms is expected to rise.
Market Concentration & Characteristics
The induced pluripotent stem cells production industry has seen significant innovation in recent years, with the introduction of advanced technologies that have improved the efficiency and accuracy of data analysis. The development of 3D culture systems and organoid technology has further allowed for more accurate modeling of human tissues and organs, improving the utility of iPSCs in drug testing and disease modeling. This has led to broader application and more intricate analyses.
The induced pluripotent stem cells production industry demonstrates moderate partnerships and collaborative activities, indicating medium engagement among stakeholders. For instance, in April 2023, Axol Biosciences entered into an exclusive agreement with the Francis Crick Institute, securing exclusive rights to a proprietary methodology for generating human pluripotent stem cells (hPSC) for drug screening and cardiotoxicity assays.

The regulatory environment significantly impacts the induced pluripotent stem cell production industry. Regulatory authorities are focusing on establishing stringent guidelines for biosafety, contamination control, cell line authentication, and other factors. Complying with regulatory standards represents significant challenges that may limit the market's growth.
The global iPSC production industry is witnessing steady product expansion as companies introduce advanced reprogramming kits, feeder-free culture media, and scalable differentiation systems to address growing demand across research, drug discovery, and clinical applications. Manufacturers increasingly focus on GMP-compliant, xeno-free solutions, integrated bioreactor platforms, and ready-to-use iPSC-derived cell types to streamline workflows and ensure regulatory compliance. For instance, in June 2022, Axol BioScience Ltd introduced Comprehensive In Vitro Proarrhythmia Assay (CiPA)- validated, hiPSC-derived ventricular cardiomyocytes to help enhance drug discovery. This diversification enhances product portfolios and broadens application potential, strengthening competitive positioning in a rapidly evolving market.
The induced pluripotent stem cells production market is experiencing a high level of regional expansion, indicating rapid growth and increasing market presence across different geographic regions. This expansion is driven by several factors, including increased research and development focused on iPSCs for regenerative medicine, disease modeling, and drug discovery, which drives demand and expansion. For instance, in August 2022, Thermo Fisher Scientific, Inc. expanded its cell culture media manufacturing site in the U.S., and such expansions can help the company increase its iPSC generation capacity.
Process Insights
The manual iPSC production process segment dominated the induced pluripotent stem cells production market and accounted for the largest revenue share in 2024. The manual iPSC production process begins with the introduction of genes/transcription factors into rising somatic cells, a reprogramming step & selection of iPSC colonies, expansion, and passaging of iPSCs. Companies with expertise in tissue procurement, gene introduction, RNA reprogramming, and cell culture and expansion drive revenue generation in this segment.
The automated iPSC production process segment is estimated to witness the fastest CAGR during the forecast period. The increasing demand for stem cell-based therapies requires scaling up. This demand for generating large-scale induced pluripotent stem cells (iPSCs) in a reproducible manner with minimum alterations is propelling the adoption of automated production platforms. Most companies working on iPSC-based therapies rely on lines of allogenic cells that are taken from multiple donors. These cells are then created into off-the-shelf products on a large scale, which can be used for the treatment of patients affected with the same diseases.
Workflow Insights
The cell culture segment dominated the market with the largest revenue share of 37.26% in 2024. Cell culture workflow includes revenues generated from iPSC harvest products, expansion products, and differentiation products, thus contributing to the market relevance of this segment. Cell culture plays a pivotal role in various stages of induced pluripotent stem cells (iPSCs) generation, and continuous investments by key players operating in this market globally have made an important contribution to the segment’s revenue generation. For example, in August 2021, HebeCell Corp. announced that it had raised USD 53 million in financing to advance its R&D program, clinical operations, and commercialization for off-the-shelf pluripotent stem cell-CAR-NK products. Moreover, costs associated with cell culture matrices, media, and regular maintenance of these cells add up to the revenue generated by this segment.
The cell characterization/analysis segment is expected to grow at the fastest CAGR over the forecast period, owing to factors such as the growing use of assays based on cells in drug discovery due to their consistency in producing tissue-specific results. Moreover, the segment is anticipated to grow due to the rising incidence of chronic diseases like cancer, which has seen an increase in the use of cell-based assays for detection and treatment.
Product Insights
The consumables and kits segment dominated the induced pluripotent stem cells production industry with the largest revenue share of 39.97% in 2024. The never-ceasing research and development activities in the iPSCs space are driving the demand for various consumables & kits. Multiple induced pluripotent stem cells (iPSCs) kits, such as reprogramming kits, differentiation kits, and generation kits, along with a selection of media & other consumables by key players, are allowing end users to utilize these products as per the intended application. Moreover, the utility of kits and media during toxicology testing is further propelling the growth of this product segment.
The automated platforms segment is expected to grow at the fastest CAGR during the forecast period, due to the demand for stem cell products, the scaling up of clinical phase iPSC therapies, and regulatory-level quality control. The automated platforms provide reproducible & reliable results, reduce labor dependence, and provide well-maintained manufacturing conditions and standardization of protocols while eliminating human biases. For instance, Hitachi Ltd. offers automated equipment for iPSC culture to spread regenerative medicines. Many other companies also provide such automated platforms to increase throughput, decrease operational costs, and increase adaptability for iPSC production.
Application Insights
The drug development & discovery segment accounted for the largest revenue share of 43.26% in 2024, driven by the increasing use of iPSC-derived cell models for advanced screening, disease modeling, and toxicity testing, which offer superior physiological relevance and predictive accuracy compared to conventional immortalized cell lines or animal models. Moreover, regulatory encouragement from agencies such as the FDA and EMA for human cell-based testing methods is accelerating adoption. The growing integration of iPSC-derived systems into preclinical pipelines and advancements in automated culture platforms and GMP-compliant manufacturing are expected to sustain the segment’s dominance over the forecast period.
The regenerative medicine segment is expected to grow at the fastest CAGR from 2025 to 2033, owing to companies' vast applications and robust product pipelines. Moreover, iPSCs can be used for disease-specific therapies and provide greater and safer drug development platforms. Also, iPSC-derived organoids and organs have recently been analyzed for disease modeling and regenerative therapy. In disease modeling, induced pluripotent stem cells enable drug discovery for innovative therapeutics, and in regenerative treatment, iPSCs are proceeding with clinical trials. For example, in April 2022, Editas Medicine, Inc. announced its preclinical data (both in vitro & in vivo) for the tumor-killing capability of its NK cell therapy derived from induced pluripotent stem cells (iPSCs).
End Use Insights
The biotechnology & pharmaceutical companies segment dominated the induced pluripotent stem cells production market with the largest revenue share of 59.41% in 2024. The availability and adoption of various kits, consumables, and instruments, along with iPSC products, are driving the revenue generation in this segment. To utilize the market opportunity of the stem cell-based therapy market, biotechnology and pharmaceutical companies are boosting new product developments via intensive R&D efforts. For example, in October 2022, Fate Therapeutics announced that it would present its preclinical and clinical data of multiple iPSC product platforms at the Society for Immunotherapy of Cancer (SITC) meeting in November 2022. This demonstrates the robust induced pluripotent stem cells (iPSCs) product pipeline, further fueling the growth.
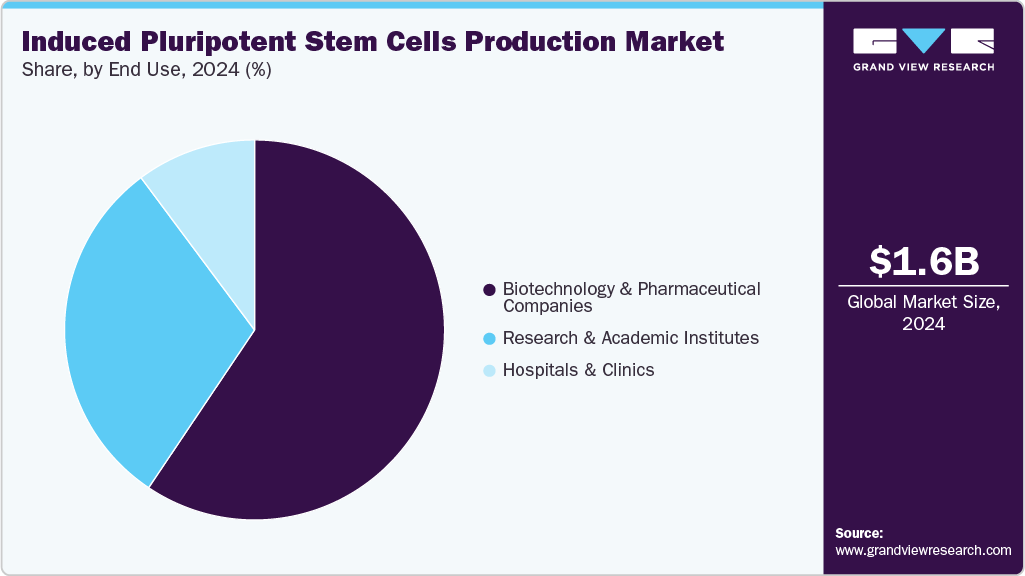
The research & academic institutes segment is expected to grow at the fastest CAGR over the forecast period. This is due to iPSCs having wide applications and thus being utilized in many research and clinical studies. The increasing utilization of iPSCs in regenerative studies provides favorable prospects for translating this technology into clinical use. For instance, in May 2023, UK-based Axol Bioscience secured exclusive rights from The Francis Crick Institute to commercialize advanced hiPSC-derived ventricular cardiomyocyte technology for global drug screening and cardiotoxicity applications.
Regional Insights
North America led the induced pluripotent stem cells production market with the largest revenue share of 40.66% in 2024. The growing incidence of chronic diseases in this region, developed healthcare infrastructure, funds from private and government initiatives, and strategic business models drive revenue generation in the market space.
U.S Induced Pluripotent Stem Cells Production Market Trends
The induced pluripotent stem cells (iPSC) production industry in the U.S. is expected to grow over the forecast period due to a growing availability of funds from government organizations. Moreover, stem cell-based companies received significant support during the COVID-19 pandemic to develop therapies.
Europe Induced Pluripotent Stem Cells Production Market Trends
The induced pluripotent stem cells production industry in Europe was identified as lucrative. This can be attributed to the increasing demand for advanced therapy products, and several companies and clinics are entering the region.
The UK induced pluripotent stem cells production market held a significant revenue share in 2024. This is attributed to major private players entering the market through strategic initiatives, such as mergers and acquisitions.
The induced pluripotent stem cells production market in Germany is anticipated to grow significantly over the forecast period. The presence of a substantial number of providers offering iPSC products and initiatives undertaken by them to expand their businesses is propelling the growth of the market in Germany. For instance, in May 2023, Evotec SE introduced new High-Throughput Screening (HTS) facilities at its headquarters in Hamburg, Germany. These facilities would be utilized for research related to human iPSCs. Such launches of facilities for iPSC-based research are anticipated to boost iPSC production.
Asia Pacific Induced Pluripotent Stem Cells Production Market Trends
The induced pluripotent stem cells (iPSC) production industry in Asia Pacific is expected to grow at the fastest CAGR of 11.20% over the forecast period. Key factors such as intensive low-cost driven stem cell-based research, growing economies, better healthcare plans, and the rising awareness of the potential of personalized treatments will drive revenue generation during the forecasted period. Moreover, the presence of multiple start-ups, academic universities, collaborative efforts by international companies, and new product/service launches in this region will contribute to APAC’s growth.
The China induced pluripotent stem cells production market is expected to grow over the forecast period. This market is driven by several initiatives undertaken by companies and research institutes. For instance, in June 2023, Merck KGaA expanded reagent manufacturing in China, investing USD 74.2 million for this expansion. Such investments in reagent production can fuel iPSC production in the country.
The induced pluripotent stem cells production market in Japan is witnessing rapid growth, supported by the country’s pioneering role in iPSC technology, strong government funding for regenerative medicine, and a favorable regulatory framework that accelerates clinical translation. For instance, in April 2025, Japan’s Kyoto University Hospital completed a clinical trial transplanting insulin-producing iPSC-derived pancreatic islet cells into a type 1 diabetes patient, marking a significant step toward future therapeutic applications. Moreover, increasing investments by domestic biopharma companies, rising demand for patient-specific therapies, and the commercialization of iPSC-based products drive market expansion, positioning Japan as one of the most influential contributors to global iPSC innovation.
MEA Induced Pluripotent Stem Cells Production Market Trends
The induced pluripotent stem cells production industry in MEA is expected to grow exponentially over the forecast period. This is attributed to considerable support received from the local government and the healthcare transformation mission undertaken for the next few years. With the rapid expansion of cell banks and increased awareness about stem cell preservation, the market for induced pluripotent stem cell production is expected to grow rapidly in the coming years. For instance, in April 2025, the UAE’s Abu Dhabi Stem Cells Center developed virus-free, clinical-grade iPSCs using proprietary GMP-compliant protocols, the first such achievement in the Middle East, advancing safe, personalized regenerative therapies for complex diseases.
The Kuwait-induced pluripotent stem cell (iPSC) production market is at an early stage but shows growth potential. It is driven by rising interest in regenerative medicine, increasing biomedical research investment, and emerging collaborations with global biotech firms. While limited infrastructure and expertise remain challenges, government initiatives to advance life sciences are expected to support market expansion.
Key Induced Pluripotent Stem Cells Production Company Insights
The global induced pluripotent stem cells (iPSC) production market is defined by a blend of established life sciences leaders, specialized biotech innovators, and emerging technology providers. Collectively, these players shape a rapidly evolving landscape driven by regenerative medicine breakthroughs, personalized therapy demand, and expanding applications in drug discovery. Industry leaders such as Lonza, Thermo Fisher Scientific, Inc., and Merck KGaA have built strong market positions through comprehensive product portfolios, GMP-compliant manufacturing facilities, and continued investment in automation, advanced reprogramming methods, and scalable differentiation platforms.
Specialist companies, including Axol Bioscience Ltd.; REPROCELL Inc.; Applied StemCells, Inc.; and Fate Therapeutics, are expanding their presence through proprietary stem cell lines, disease-specific modeling tools, and targeted therapeutic pipelines. Global players like Evotec and Hitachi, Ltd., leverage strategic collaborations, contract development services, and regional manufacturing strengths to accelerate clinical translation of iPSC-derived therapies. Meanwhile, niche contributors such as StemCellsFactory III and other emerging innovators provide specialized technologies in cell reprogramming, process optimization, and custom assay development, supporting both large-scale production and research-focused needs.

Strategic partnerships between iPSC producers, contract development and manufacturing organizations (CDMOs), and pharmaceutical companies underscore the industry’s collaborative nature, bridging early-stage innovation with commercial-scale manufacturing. With rising demand for patient-specific therapies, predictive preclinical models, and scalable cell production systems, market success will favor companies that integrate technological innovation with flexible, regulatory-ready solutions, positioning themselves at the forefront of the next wave in iPSC commercialization.
Key Induced Pluripotent Stem Cells Production Companies:
The following are the leading companies in the induced pluripotent stem cells production market. These companies collectively hold the largest market share and dictate industry trends.
- Lonza
- Axol Bioscience Ltd.
- Evotec
- Hitachi, Ltd.
- REPROCELL Inc.
- Merck KGaA
- Fate Therapeutics
- Thermo Fisher Scientific, Inc.
- StemCellsFactory III
- Applied StemCells, Inc.
Recent Developments
-
In August 2025, REPROCELL launched StemRNA Clinical iPSC Seed Stock Clones, offering ethically sourced, regulatory-compliant, virus-free, clinical-grade iPSC lines from U.S. and Japan facilities to accelerate early-stage cell therapy development.
-
In April 2024, Singapore-based SCG Cell Therapy and A*STAR launched joint labs with nearly USD 30 million funding to advance scalable, GMP-grade iPSC manufacturing for novel cellular immunotherapies and talent development under the RIE2025 plan.
Induced Pluripotent Stem Cells Production Market Report Scope
Report Attribute
Details
Market size value in 2025
USD 1.76 billion
Revenue forecast in 2033
USD 3.78 billion
Growth rate
CAGR of 10.01% from 2025 to 2033
Base year for estimation
2024
Historical data
2021 - 2023
Forecast period
2025 - 2033
Quantitative units
Revenue in USD million/billion and CAGR from 2025 to 2033
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Process, workflow, product, application, end use, region
Regional scope
North America; Europe; Asia Pacific; Latin America; MEA
Country scope
U.S.; Canada; Mexico; UK; Germany; France; Italy; Spain; Denmark; Sweden; Norway; India; China; Japan; Australia; South Korea; Thailand; Brazil; Argentina; Saudi Arabia; UAE; South Africa; Kuwait
Key companies profiled
Lonza; Axol Bioscience Ltd.; Evotec; Hitachi, Ltd.; REPROCELL Inc.; Merck KGaA; Fate Therapeutics; Thermo Fisher Scientific, Inc.; StemCellsFactory III; Applied StemCells, Inc.
Customization scope
Free report customization (equivalent up to 8 analyst’s working days) with purchase. Addition or alteration to country, regional & segment scope.
Global Induced Pluripotent Stem Cells Production Market Report Segmentation
This report forecasts revenue growth and provides an analysis on the latest trends in each of the sub-segments from 2021 to 2033. For this report, Grand View Research has segmented the global induced pluripotent stem cells production market report based on process, workflow, product, application, end use, and region:
-
Process Outlook (Revenue, USD Million, 2021 - 2033)
-
Manual iPSC Production Process
-
Automated iPSC Production Process
-
-
Workflow Outlook (Revenue, USD Million, 2021 - 2033)
-
Reprogramming
-
Cell Culture
-
Cell Characterization/Analysis
-
Engineering
-
Others
-
-
Product Outlook (Revenue, USD Million, 2021 - 2033)
-
Instruments/ Devices
-
Automated Platforms
-
Consumables & Kits
-
Media
-
Kits
-
Others
-
-
Services
-
-
Application Outlook (Revenue, USD Million, 2021 - 2033)
-
Drug Development & Discovery
-
Regenerative Medicine
-
Toxicology Studies
-
Others
-
-
End Use Outlook (Revenue, USD Million, 2021 - 2033)
-
Research & Academic Institutes
-
Biotechnology & Pharmaceutical Companies
-
Hospitals & Clinics
-
-
Regional Outlook (Revenue, USD Million, 2021 - 2033)
-
North America
-
U.S.
-
Canada
-
Mexico
-
-
Europe
-
UK
-
Germany
-
France
-
Italy
-
Spain
-
Denmark
-
Sweden
-
Norway
-
-
Asia Pacific
-
Japan
-
China
-
India
-
Australia
-
Thailand
-
South Korea
-
-
Latin America
-
Brazil
-
Argentina
-
-
Middle East and Africa (MEA)
-
South Africa
-
Saudi Arabia
-
UAE
-
Kuwait
-
-
Frequently Asked Questions About This Report
b. The global induced pluripotent stem cells production market size was estimated at USD 1.62 billion in 2024 and is expected to reach USD 1.76 billion in 2025.
b. The global induced pluripotent stem cells production market is expected to grow at a compound annual growth rate of 10.01% from 2025 to 2033 to reach USD 3.78 billion by 2033.
b. The manual iPSCs production process segment dominated the global iPSCs production market and held the largest revenue share in 2024. It serves as a convenient method for parallel production of various iPSC lines, which has led to the segment’s dominance
b. Some key players operating in the iPSCs generation market include Lonza; Axol Biosciences Ltd.; Evotec; Hitachi Ltd.; Merck KGaA; REPROCELL Inc.; Fate Therapeutics; Thermo Fisher Scientific, Inc.; StemCellFactory III; and Applied StemCell Inc.
b. Key factors that are driving the iPSCs production market growth include a robust pipeline for induced pluripotent stem cell-derived cell therapeutics coupled with increasing demand for iPSCs.
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










