- Home
- »
- Medical Devices
- »
-
Injectable Drug Delivery Devices Market Size Report, 2033GVR Report cover
![Injectable Drug Delivery Devices Market Size, Share & Trends Report]()
Injectable Drug Delivery Devices Market (2026 - 2033) Size, Share & Trends Analysis Report By Product (Injectors, Pen), By Therapeutic Use (Autoimmune Disorders, Hormonal Disorders, Oncology, Obesity, Diabetes Mellitus), By End Use, By Region, And Segment Forecasts
- Report ID: 978-1-68048-GVR-8
- Number of Report Pages: 100
- Format: PDF
- Historical Range: 2021 - 2025
- Forecast Period: 2026 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Interactive Charts
- Methodology
- Download FREE Sample
-
Download Sample Report
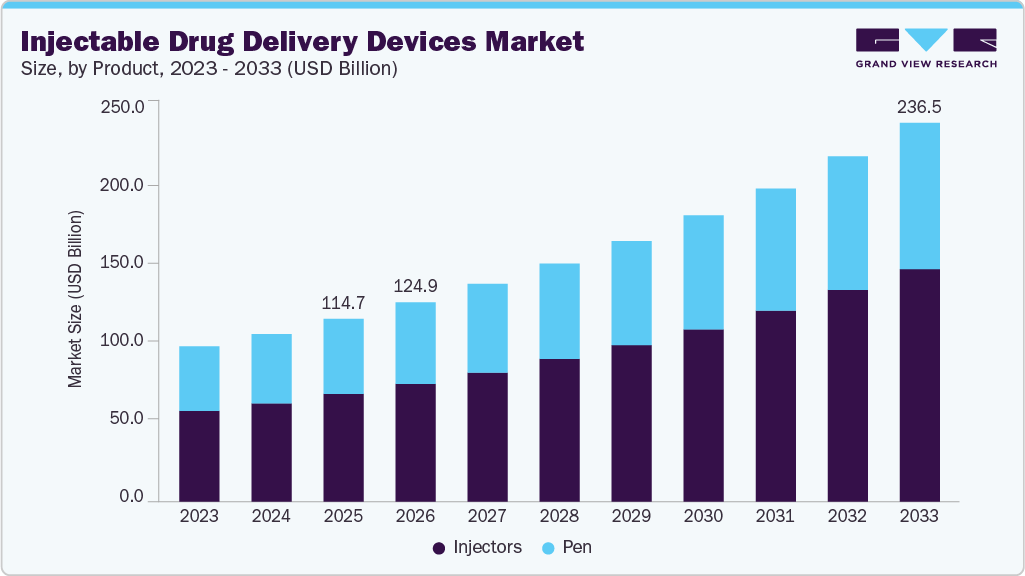
Injectable Drug Delivery Devices Market Summary
The global injectable drug delivery devices market size was estimated at USD 114.7 billion in 2025 and is projected to reach USD 236.5 billion by 2033, growing at a CAGR of 9.6% from 2026 to 2033. This significant growth is fueled by an increasing need for advanced, effective, and user-friendly drug delivery solutions in various healthcare environments.
Key Market Trends & Insights
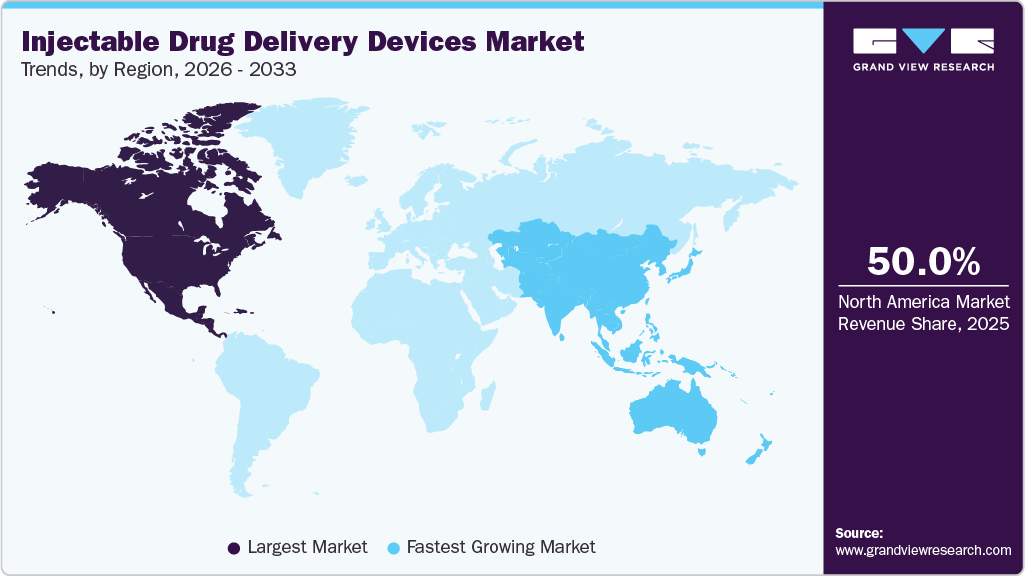
- North America injectable drug delivery devices market held the largest revenue share of 50.0% in 2025.
- U.S. dominated the North America market with a revenue share of 86.2% in 2025.
- By product, the injectors segment held the largest market share of 59.3% in 2025.
- Based on therapeutic use, the diabetes mellitus segment held the largest market share of 44.5% in 2025.
- Based on end use, homecare segment dominated the market with a revenue share of 72.5% in 2025.
Market Size & Forecast
- 2025 Market Size: USD 114.7 Billion
- 2033 Projected Market Size: USD 236.5 Billion
- CAGR (2026-2033): 9.6%
- North America: Largest market in 2025
- Asia: Fastest growing market
Key factors include the rising incidence of chronic diseases like diabetes, cancer, and autoimmune disorders that necessitate frequent injections. In addition, there is a growing preference for convenient injection instruments such as autoinjectors and pen injectors, alongside continuous technological innovations that improve safety, accuracy, and patient comfort. Moreover, the surge in biological developments, a shift toward home-based care, and an emphasis on minimizing hospital visits are all contributing to this upward trend in the market.Another factor that influences the growth is the increasing demand for advanced, effective, and user-friendly drug delivery solutions in a variety of healthcare settings. Several key factors are at play, including the rising prevalence of chronic diseases like diabetes, cancer, and autoimmune disorders that require frequent injections. There's also a growing preference for convenient injection devices such as autoinjectors and pen injectors, coupled with ongoing technological advancements that enhance safety, accuracy, and patient comfort. In addition, the rise in biopharmaceutical innovations, a shift towards home-based care, and a focus on reducing hospital visits are all contributing to this positive market trend.
According to the World Health Organization (WHO), in 2022, there were approximately 20 million new cancer cases and 9.7 million deaths. By 2050, this figure is expected to surge by 77%, reaching over 35 million cases globally. Similarly, cardiovascular diseases (CVDs), such as coronary heart disease, cerebrovascular disease, and rheumatic heart disease, remain the leading cause of death worldwide, claiming an estimated 17.9 million lives annually.
Estimated number of new cancer cases for all cancer types from 2025 to 2030, Incidence, Both sexes, age [0-85+]
Country
2025
2030
U.S.
2 535 766
2 791 752
Canada
315 177
354 384
Mexico
222 470
254 690
UK
471 780
508 505
Germany
622 649
655 540
France
499 651
532 289
Italy
444 265
470 620
Spain
292 742
317 164
Denmark
51 515
54 982
Sweden
73 587
78 777
Norway
43 203
47 841
Japan
1 042 544
1 062 688
China
5 125 778
5 717 426
India
1 532 538
1 752 347
Australia
228 202
252 173
South Korea
61 533
79 356
Thailand
208 546
232 143
Brazil
659 557
756 337
Argentina
141 457
154 919
South Africa
118 364
135 916
Saudi Arabia
31 209
39 900
UAE
5 835
7 328
Kuwait
5 957
8 111
Source: International Agency for Research on Cancer (IARC), GVR analysis
Chronic diseases often require long-term treatment, making it challenging for many patients to stick to complex injectable regimens. Advanced drug delivery devices for injections help overcome these obstacles by making administration easier, reducing the frequency of doses, and minimizing errors. Autoinjectors and pen injectors streamline the injection process, promoting consistent dosing of biologics and specialty medications. Many next-generation devices also feature built-in dose reminders, connectivity for real-time monitoring, and digital tracking, all of which enhance adherence and enable patients to manage their treatment schedules more effectively. When adherence improves, it not only leads to better health outcomes but also decreases disease flare-ups, emergency visits, and overall healthcare costs.
Chronic diseases remain the leading and fastest-growing global health issue, responsible for nearly 75% of all deaths worldwide, as reported by the World Health Organization (WHO, 2024). Noncommunicable diseases (NCDs) like cardiovascular diseases, cancer, chronic respiratory illnesses, and diabetes cause approximately 41 million deaths annually. Low- and middle-income countries account for more than 77% of this mortality. The Global Burden of Disease (GBD) 2024 data indicate that over 1.3 billion people worldwide live with at least one chronic condition requiring continuous management, many of whom depend on injectable treatments such as insulin, monoclonal antibodies, and biologics. The increasing rates of obesity, sedentary lifestyles, and aging populations continue to intensify this issue.
Estimated global prevalence and mortality of major chronic diseases, 2024
Disease Category
Estimated Global Prevalence
Annual Deaths (in Millions)
Cardiovascular Diseases (CVDs)
523 million people
20.5 million
Cancer (all types)
~20 million new cases annually
9.7 million
Chronic Respiratory Diseases (COPD, Asthma)
550 million people
4.2 million
Diabetes (Type 1 & 2)
589 million adults (20-79 years)
6.7 million
Chronic Kidney Disease (CKD)
850 million people
4.3 million
Rheumatoid Arthritis & Autoimmune Disorders
150-200 million people
-
Obesity (as a risk factor)
1.0 billion adults
-
Source: WHO, World Heart Federation. IDF Diabetes Atlas, Global Burden of Disease
Market Concentration & Characteristics
The global market for injectable drug delivery devices is dominated by a few major multinational companies, which control a considerable share thanks to their well-established brand portfolios, robust R&D capabilities, and widespread geographic outreach. Competition in this sector is driven by constant innovation, especially in areas like self-injectors, autoinjectors, prefilled syringes, and wearable injectors. There's a noticeable trend toward developing devices focused on patient needs that allow for easy self-administration. Top companies are pursuing both organic growth and strategic partnerships, taking advantage of advancements in technology, such as smart connected injectors and AI-powered delivery systems, to strengthen their market presence. For new entrants, breaking into this market can be quite difficult due to stringent regulations and the necessity for advanced manufacturing capabilities. The overall landscape is marked by moderate to high concentration, rapid innovation cycles, and significant investments in global supply chains as well as digital health solutions.
Innovation in the injectable drug delivery devices industry is very high. New technologies and patient-focused designs are driving this growth. Smart connected injectors with Bluetooth and AI help track doses and improve treatment adherence. Wearable injectors, ultra-thin needle autoinjectors, and reusable devices make treatment easier and more comfortable. For instance, in October 2024, Nemera launched a reusable autoinjector at CPHI Milan. The device features durable electronics and a long-life design to improve patient comfort, support sustainable self-administration, and reduce costs. Advances in drug formulations, such as sustained-release and needle-free systems, further increase safety and patient outcomes. Innovation remains a key force shaping this market.
Partnerships and collaborations play a critical role in the injectable drug delivery devices industry, enabling companies to combine expertise in pharmaceuticals, device engineering, and digital health to develop advanced, patient-centric delivery solutions such as autoinjectors, wearable injectors, and prefilled syringe systems. For instance, in January 2026, Halozyme Therapeutics, Inc. and Skye Bioscience, Inc. announced a global collaboration and licensing agreement to utilize Halozyme’s ENHANZE drug delivery technology for developing a subcutaneous formulation of nimacimab for obesity treatment. The partnership aims to enable higher-dose injectable delivery and support the commercialization of more patient-friendly therapies.
Regulatory approval plays a critical role in shaping the injectable drug delivery devices industry by ensuring that drug-device combination products meet stringent safety, efficacy, and performance standards before commercialization. Authorities such as the U.S. Food and Drug Administration, European Medicines Agency (EMA), and other global regulators classify many injectable systems, including autoinjectors, prefilled syringes, and wearable injectors, as combination products, requiring rigorous clinical validation, biocompatibility testing, and device reliability assessments. Compliance with these regulations enhances trust among healthcare providers and patients, supports reimbursement pathways, and accelerates global market access, while also driving innovation in safer, user-friendly, and self-administration-focused delivery systems.
Product substitutes in the injectable drug delivery devices industry include oral medications, transdermal patches, microneedle patches, inhalation systems, intravenous (IV) infusions, and emerging needle-free technologies such as jet injectors and robotic pills. For patients requiring long-term therapy, oral drugs and transdermal patches are often preferred due to ease of administration and improved convenience, while IV infusions are commonly used in hospital settings for controlled dosing. Advanced alternatives such as microneedle patches and needle-free injectors are gaining traction as minimally invasive solutions that reduce pain and improve patient compliance, particularly in chronic disease management. These substitutes can limit the adoption of traditional injectable devices, especially in cases where non-invasive delivery is feasible; however, injectables remain essential for biologics, vaccines, and therapies requiring rapid or precise systemic delivery.
Regional expansion in the injectable drug delivery devices industry is gaining strong momentum across major global regions, driven by the rising prevalence of chronic diseases, increasing adoption of biologics, and growing demand for self-administration and home-based care. Companies are actively expanding their geographical footprint through strategic partnerships, licensing agreements, and regional commercialization programs to strengthen market access, particularly in high-growth regions such as the Asia-Pacific and emerging markets.
Product Insights
Based on product, the injectors segment dominated the market with a revenue share of 59.3% in 2025 and is projected to register the fastest CAGR of 10.1% during the forecast period. This growth is driven by increasing demand for easy-to-use self-injection devices that enable patients with chronic diseases such as rheumatoid arthritis, multiple sclerosis, and diabetes to manage treatments at home. Autoinjectors provide a simplified, safe, and reliable delivery system with features such as hidden needles, automated dosing, and ergonomic designs that reduce user error and needle anxiety. The growing preference for self-administration and advancements in biologics are driving the adoption of autoinjectors. In addition, the rising prevalence of diabetes in low- and middle-income countries is increasing demand for injection devices, supporting the injectors segment, which holds the largest share of the market. For instance, in February 2024, Novo Nordisk A/S partnered with HemoCue to enhance point-of-care diagnostic testing for children with type 1 diabetes in low- & middle-income nations.
The pen segment is projected to witness the second fastest CAGR over the forecast period, driven by the increasing demand for convenient and accurate drug delivery systems for chronic disease management. Injection pens are widely used for therapies such as diabetes and hormone treatments due to their ease of use, portability, and precise dosing capabilities. In addition, the growing shift toward home-based treatment and self-administration is further supporting the adoption of pen-based devices. Technological advancements, including reusable and smart connected pens that enable dose tracking and adherence monitoring, are also contributing to the segment’s growth.
Therapeutic Use Insights
The diabetes mellitus segment dominated the market in 2025, primarily due to the high global prevalence of diabetes and the frequent need for injectable therapies such as insulin and GLP-1 drugs. According to the International Diabetes Federation (IDF), approximately 11.1% of adults worldwide, about 589 million people, were living with diabetes in 2024, highlighting the significant and growing patient population requiring injectable drug delivery systems. The long-term nature of diabetes management, which often requires daily or frequent injections, drives substantial demand for devices such as insulin pens, autoinjectors, and other injectable delivery systems. In addition, the increasing prevalence of diabetes, particularly in low- and middle-income countries, continues to support the dominance of this segment in the market.
The obesity segment is projected to grow at the fastest CAGR during the forecast period, driven by the rapid rise in global obesity prevalence and the increasing adoption of injectable anti-obesity therapies such as GLP-1 receptor agonists. According to the World Health Organization, more than 1 billion people worldwide were living with obesity in 2022, and the prevalence has more than doubled since 1990. This growing patient population is significantly increasing the demand for injectable drug delivery devices, particularly pens and autoinjectors used for weight-management medications. In addition, the expanding pipeline of injectable obesity drugs and increasing awareness of obesity-related health risks are further supporting the rapid growth of the obesity segment in the market.
End Use Insights
The homecare settings segment dominated the global market in 2025 and is projected to grow at the fastest CAGR during the forecast period. Patients are increasingly using injectable drug delivery devices in home care settings to manage their chronic conditions, as these devices provide greater autonomy, safety, and flexibility in treatment. The shift toward home-based care has been driven by the growing preference for convenient, efficient, and less invasive treatment options. Injectable devices, such as autoinjectors and prefilled pens, enable patients to administer medications accurately and safely with minimal supervision.
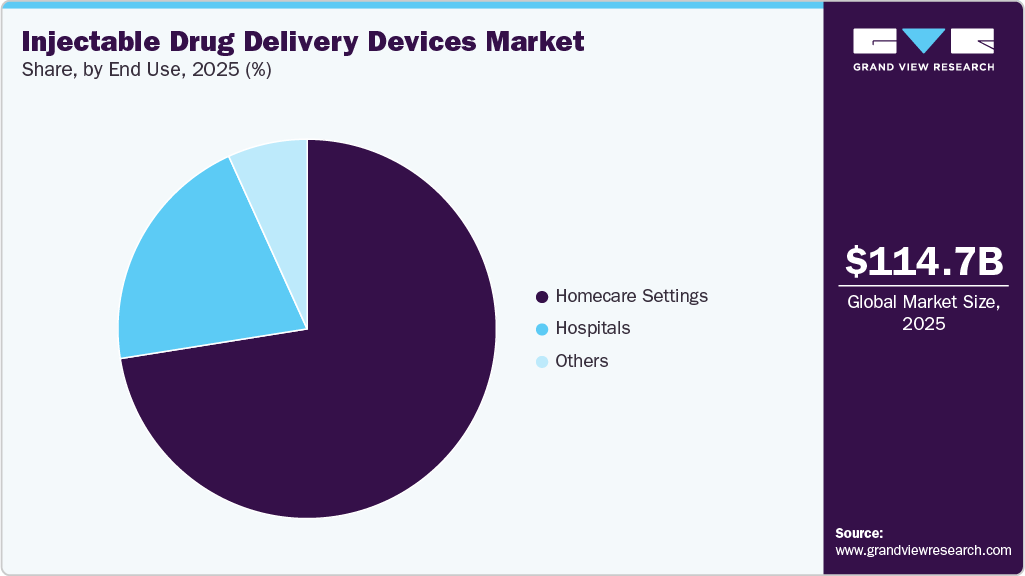
The hospitals segment is projected to grow at the second-fastest CAGR from 2026 to 2033. Injectable drug delivery devices are crucial in hospitals for precise, rapid, and effective medication administration across various therapeutic areas. Hospitals are a primary end-user for systems like autoinjectors and prefilled pens, driven by the high volume of acute care, emergency procedures, and chronic conditions treated within their facilities. The sterile and controlled hospital environment enables healthcare providers to administer injectable treatments that require precise dosing, sterility, and rapid action, such as biologics, vaccines, antibiotics, and pain relievers. Delivering these drugs directly into the bloodstream also ensures faster therapeutic effects, which are crucial in intensive care units, surgical recovery areas, and cancer treatment departments.
Regional Insights
The North America injectable drug delivery devices market held a 50.0% market share in 2025, driven by the presence of a well-established healthcare sector, commitment to innovation, advanced infrastructure, a strong emphasis on R&D, and a high prevalence of chronic conditions, such as diabetes and autoimmune disorders. According to the American Heart Association, as of 2024, 29.3 million adults in the U.S. have been diagnosed with diabetes, whereas 9.7 million cases remained undiagnosed. This data highlights the significant impact of diabetes on the American population, highlighting the critical need for accessible and effective management tools, including injectable drug delivery devices, to address both diagnosed & undiagnosed cases. Moreover, favorable government regulations and reimbursement policies contribute to market dominance. Regulatory bodies such as the FDA ensure rigorous quality standards, assuring healthcare professionals and patients of the safety & effectiveness of various healthcare products, including injectable drug delivery devices.
U.S. Injectable Drug Delivery Devices Market Trends
The injectable drug delivery devices market in the U.S. is driven by the need for better patient outcomes, easier treatment use, and the increasing number of people with chronic diseases like diabetes. U.S. companies are developing innovative injectable devices to meet the growing demand for treatments for chronic and rare conditions. This demographic presents a vast opportunity for self-injection devices. The CDC's report emphasizes the need for effective diabetes management tools, highlighting the market potential for growth. The competitive landscape is shaped by the ability of these devices to cater to the diverse needs of people suffering from diabetes, considering factors such as race, ethnicity, socioeconomic status, and education level.
Europe Injectable Drug Delivery Devices Market Trends
The injectable drug delivery devices market in Europe is experiencing strong growth, driven by the rising prevalence of chronic diseases, technological innovations, and an increasing patient preference for convenient, long-acting therapies. Chronic conditions such as diabetes and cancer are prevalent across Europe, necessitating advanced drug delivery solutions that improve treatment efficacy and reduce dosing frequency. The growing diabetes burden across Europe is significantly driving the demand for injectable drug delivery devices. With 66 million adults affected in 2024, and over one-third remaining undiagnosed, there is an urgent need for therapies that ensure sustained drug release, improve patient compliance, and reduce dosing frequency.
The UK injectable drug delivery devices market is evolving rapidly, propelled by technological innovations, advances in personalized medicine, and an increasing demand for treatment regimens that support better patient adherence and outcomes. With the growing burden of chronic diseases, the need for simplified and effective drug delivery systems is more urgent than ever. As a professor from the University of Nottingham’s School of Pharmacy highlighted,
“The future of prescribed medication lies in a personalized approach, and we know that up 50% of people in the UK alone don’t take their medicines correctly, and this has an impact on poorer health outcomes with conditions not being controlled or properly treated. A single pill approach would simplify taking multiple medications at different times, and this research is an exciting step towards that.”
This underscores the importance of self-administering injectable technologies in addressing non-adherence and improving therapeutic success.
The injectable drug delivery devices market in Germany is witnessing significant growth due to advancements in drug delivery technologies. This growth is largely fueled by the demand for better patient outcomes, simplified treatment methods, and an increasing prevalence of chronic illnesses like diabetes and cardiovascular diseases. German pharmaceutical and medical device firms are at the forefront of this innovation, developing cutting-edge injectable solutions such as prefilled syringes, autoinjectors, and wearable injectors to meet the rising need for treatments targeting both chronic and rare diseases. Furthermore, the rising incidence of cancer in Germany is intensifying the demand for sophisticated injectable drug delivery systems. This includes a focus on biologics and targeted oncology therapies that require careful and precise administration via pen injectors and infusion devices, contributing to the overall expansion of the market.
Asia Injectable Drug Delivery Devices Market Trends
The injectable drug delivery devices market in Asia is expected to grow at the fastest CAGR from 2025 to 2033, driven by a combination of rising chronic disease prevalence, healthcare modernization, and growing adoption of biologics and biosimilars. Across the region, conditions such as diabetes, cancer, and rheumatoid arthritis, among other autoimmune diseases and chronic conditions, are becoming increasingly common, creating sustained demand for advanced injectable therapies. Governments in both developed and emerging Asian economies are investing heavily in universal health coverage, digital health transformation, and patient-centered care models, which have accelerated the transition from conventional vial-and-syringe methods to prefilled pens and autoinjectors. The shift toward self-administration and home-based treatment, especially for diabetes and autoimmune diseases, is one of the strongest market drivers, supported by greater patient awareness and physician acceptance of self-injection techniques.
China injectable drug delivery devices market is growing rapidly, driven by the rising prevalence of chronic diseases such as diabetes, cancer, and autoimmune disorders, which continue to increase the need for advanced injectable treatments. Furthermore, strong government support and substantial R&D investments have played a pivotal role in advancing China’s self-injection device market. Various initiatives, such as Made in China 2025, which prioritize the advancement of high-tech sectors, have allocated significant resources to biotechnology & biomanufacturing, fostering a conducive environment for the development of medical technologies. In addition, the Chinese government has undertaken initiatives to enhance domestic medical device production. For instance, as reported in a 2022 article, the Volume-based Procurement (VBP) Act was introduced in 2020. This act focuses on the volume-based procurement of high-value medical supplies to address rising costs. This government initiative is expected to create opportunities for drug delivery devices, thereby driving the market.
Key Injectable Drug Delivery Devices Company Insights
Many brands in the global market are focusing on strong growth opportunities through innovation, strategic partnerships, and expanding product portfolios. Leading players are developing advanced delivery technologies such as smart connected injectors, wearable injectors, and reusable drug delivery systems to improve patient convenience and treatment adherence. Companies are also investing heavily in research and development to introduce digitally enabled devices with features such as Bluetooth connectivity and dose tracking, which help enhance dosing accuracy and patient monitoring. These innovations are supporting the broader adoption of injectable therapies across multiple therapeutic areas.
Key Injectable Drug Delivery Devices Companies:
The following key companies have been profiled for this study on the injectable drug delivery devices market.
- Novo Nordisk A/S
- Sanofi
- Eli Lilly and Company
- AstraZeneca plc
- Pfizer, Inc.
- Merck KGaA
- Teva Pharmaceuticals
- Amgen, Inc.
- Ferring Pharmaceuticals
- AbbVie Inc.
Recent Developments
-
In October 2025, Stevanato Group S.p.A. announced a major expansion of its drug delivery system manufacturing capacity in Germany. This move strengthens its presence in Europe. The company added over 2,500 square meters of new production space to its Bad Oeynhausen facility. This includes an ISO 8 cleanroom for injection molding and automated assembly. The multi-million-euro investment improves scalability, supply chain resilience, and support for both proprietary and contract manufacturing of drug delivery devices, including autoinjector and pen injector platforms.
-
In May 2025, IDE Group introduced OKO, a new auto-injector for eye care markets. Its goal is to improve drug delivery accuracy, make procedures more efficient, and enhance patient safety. The device overcomes the limitations of existing intravitreal injection methods used for treating retinal diseases. Developed with the help of ophthalmologists from Australia, the U.S., and Europe, OKO provides up to 59 times better dosing accuracy, reduces procedural steps by 45%, and increases patient throughput, cutting per-injection costs by up to 78%.
Injectable Drug Delivery Devices Market Report Scope
Report Attribute
Details
Market value size in 2026
USD 124.9 billion
Revenue forecast in 2033
USD 236.5 billion
Growth rate (revenue)
CAGR of 9.6% from 2026 to 2033
Actuals
2021 - 2025
Forecast period
2026 - 2033
Quantitative units
Revenue in USD million/billion, volume in units, and CAGR from 2026 to 2033
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Product, therapeutic use, end use, region
Regional scope
North America; Europe; Asia
Country scope
U.S.,; Canada; Mexico; Germany; UK; France; Italy; Spain; Norway; Sweden; Denmark; China; India; Japan; Thailand; South Korea; Saudi Arabia; UAE; Kuwait
Key companies profiled
Novo Nordisk A/S; Sanofi; Eli Lilly and Company; AstraZeneca plc; Pfizer, Inc.; Merck KGaA; Teva Pharmaceuticals; Amgen, Inc.; Ferring Pharmaceuticals; AbbVie Inc.
Customization scope
Free report customization (equivalent up to 8 analysts’ working days) with purchase. Addition or alteration to country, regional & segment scope.
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
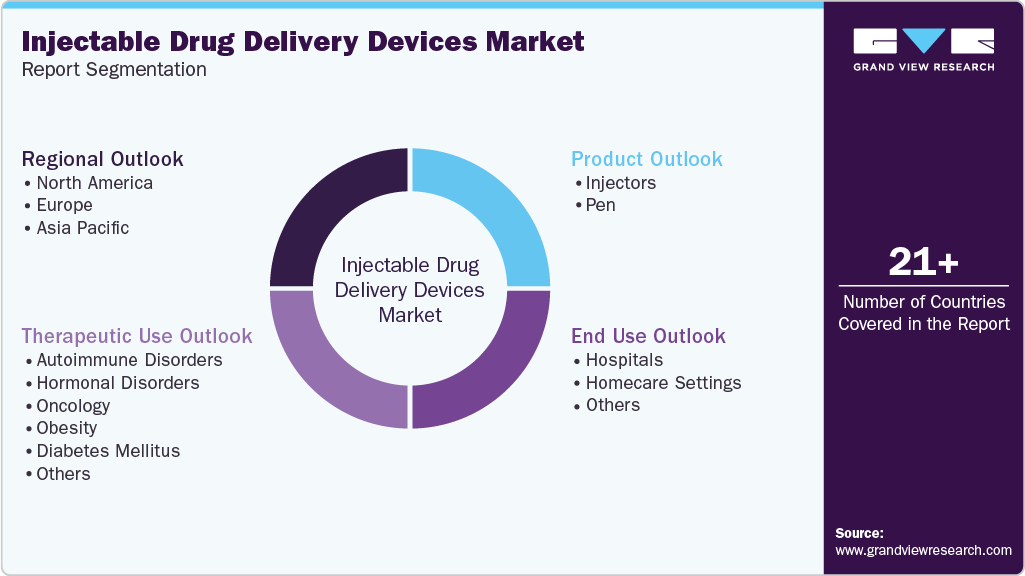
Global Injectable Drug Delivery Devices Market Report Segmentation
This report forecasts revenue growth at the global, regional, and country levels and provides an analysis of the latest industry trends and opportunities in each of the sub-segments from 2021 - 2033. For this study, Grand View Research has segmented the global injectable drug delivery devices market report based on product, therapeutic use, end use, and region:
-
Product Outlook (Volume, Units; Revenue, USD Billion, 2021 - 2033)
-
Injectors
-
GLP-1 Agonists
-
Hormone Therapies
-
Biologics
-
Others
-
-
Pen
-
GLP-1 Agonists
-
Insulin
-
Hormone Therapies
-
Others
-
-
-
Therapeutic Use Outlook (Volume, Units; Revenue, USD Billion, 2021 - 2033)
-
Autoimmune Disorders
-
Hormonal Disorders
-
Oncology
-
Obesity
-
Diabetes Mellitus
-
Others
-
- End Use Outlook (Volume, Units; Revenue, USD Billion, 2021 - 2033)
-
Hospitals
-
Homecare Settings
-
Others
-
-
Regional Outlook (Volume, Units; Revenue, USD Billion, 2021 - 2033)
-
North America
-
U.S.
-
Canada
-
Mexico
-
-
Europe
-
UK
-
Germany
-
France
-
Italy
-
Spain
-
Norway
-
Sweden
-
Denmark
-
-
Asia
-
China
-
Japan
-
India
-
Thailand
-
South Korea
-
Saudi Arabia
-
UAE
-
Kuwait
-
-
Frequently Asked Questions About This Report
Key factors that are driving the market growth include increasing application of injectables in the treatment of diseases, such as Wilson’s disease, autoimmune diseases, cystic fibrosis, hepatitis C, and hemophilia.
The global injectable drug delivery devices market size was estimated at USD 114.7 billion in 2025 and is expected to reach USD 124.9 billion in 2026.
The global injectable drug delivery devices market is expected to grow at a compound annual growth rate of 9.6% from 2026 to 2033 to reach USD 236.5 billion by 2033.
North America dominated the injectable drug delivery devices market with a share of 50.0% in 2025. This is attributable to the rising prevalence of chronic disorders and the growing demand for targeted drug delivery systems.
Some key players operating in the injectable drug delivery market include Novo Nordisk A/S, Sanofi, Eli Lilly and Company, AstraZeneca plc, Pfizer, Inc., Merck KGaA, Teva Pharmaceuticals, Amgen, Inc., Ferring Pharmaceuticals, and AbbVie, Inc.
About the authors:
Author: GVR Medical Devices Research Team | Last Updated:
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.









