- Home
- »
- Biotechnology
- »
-
Large & Small-scale Bioprocessing Market Size Report, 2033GVR Report cover
![Large And Small-scale Bioprocessing Market Size, Share & Trends Report]()
Large And Small-scale Bioprocessing Market (2026 - 2033) Size, Share & Trends Analysis Report By Scale, By Workflow (Downstream, Upstream), By Product, By Application, By Use Type, By Mode (In-house, Outsourced), By Region, And Segment Forecasts
- Report ID: GVR-2-68038-277-8
- Number of Report Pages: 120
- Format: PDF
- Historical Range: 2021 - 2023
- Forecast Period: 2026 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Interactive Charts
- Methodology
- Download FREE Sample
-
Download Sample Report
Large And Small-scale Bioprocessing Market Summary
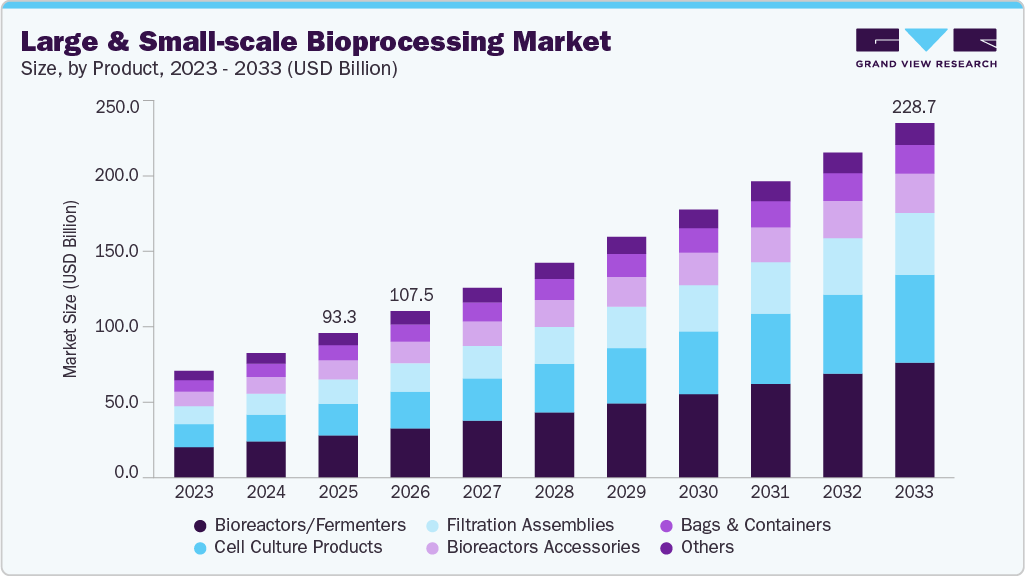
The global large and small-scale bioprocessing market size was estimated at USD 93.32 billion in 2025 and is expected to reach USD 228.73 billion by 2033, growing at a CAGR of 11.39% from 2026 to 2033. This substantial growth is primarily driven by the increasing global demand for biologics, including monoclonal antibodies, vaccines, and cell and gene therapies.
Key Market Trends & Insights
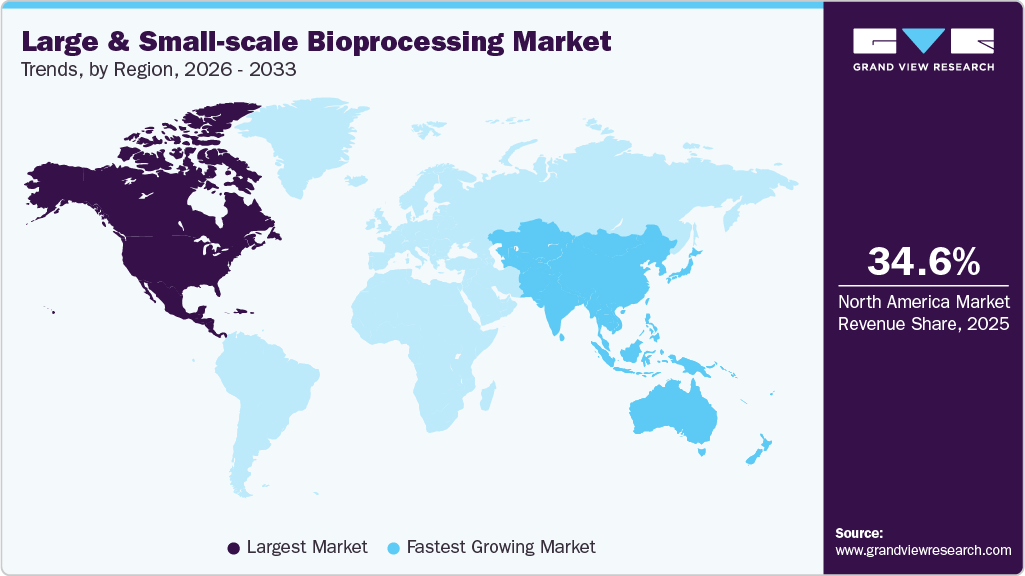
- The North America large and small-scale bioprocessing market held the largest share of 34.64% of the global market in 2025.
- The large- and small-scale bioprocessing industry in the U.S. is expected to grow significantly over the forecast period.
- Based on workflow, the downstream processing segment held the largest market share of 47.15% in 2025.
- By product, the bioreactors/fermenters segment held the largest market share of 29.4% in 2025.
- By scale, the industrial scale (over 50,000 liter) segment held the largest market share in 2025.
Market Size & Forecast
- 2025 Market Size: USD 93.32 Billion
- 2033 Projected Market Size: USD 228.73 Billion
- CAGR (2026-2033): 11.39%
- North America: Largest market in 2025
- Asia Pacific: Fastest growing market
Biopharmaceutical companies invest significantly in flexible manufacturing solutions, such as single-use technologies and modular bioprocessing systems, which help accelerate development timelines and reduce production costs.Increasing Demand for Biologics
One of the primary factors driving the global bioprocessing market is the rapid growth of biologics, which encompass cell and gene therapies, vaccines, recombinant proteins, and monoclonal antibodies. The need for both large-scale systems (for commercial production) and small-scale systems (used in R&D and clinical trials) is fueled by the complex therapies' need for accurate and scalable manufacturing processes. While large-scale bioprocessing guarantees consistent quality and compliance for the mass distribution of approved biologics, small-scale bioprocessing facilitates early-stage development through quick prototyping and process optimization.
Gene therapy pipeline: quarterly comparison
Global Status
Q1 2023
Q2 2023
Q3 2023
Q4 2023
Q1 2024
Preclinical
1,493
1,539
1,522
1,528
1,471
Phase I
245
240
256
270
301
Phase II
247
260
267
274
282
Phase III
30
30
30
33
35
Preregistration
7
6
7
6
4
Total
2,022
2,075
2,082
2,111
2,093
Source: American Society of Gene & Cell Therapy, Secondary Research, Grand View Research
The global increase in chronic illnesses, an aging population, and the drive for personalized medicine are all closely related to this biologics boom. Advanced therapies are becoming more and more necessary as conditions such as cancer, autoimmune disorders, and neurological diseases like Parkinson's and Alzheimer's become more common. CDMOs, biotech startups, and pharmaceutical companies are expanding their biomanufacturing capacity across all scales. The demand for effective, affordable bioprocessing technologies, such as microbial-based systems and single-use solutions, is further increased by the increasing use of biosimilars, which is fueled by the expiration of patents for branded biologics.
Rising investments in biotechnology research
Rising investments in biotechnology research also significantly boost the market. Governments and the private sector are directing substantial funding towards biotechnological innovations, promoting collaborations between academic institutions, research organizations, and industry players. For instance, according to the UK Bio Industry Association (BIA), UK biotechnology grant funding is expected to grow in 2025/2026. The Biotech and Medical sectors received financing for R&D and capital investments in equipment. Innovation funding experienced a five-fold increase, focusing on underfunded regions and sectors undergoing digitalization, such as healthcare.
The development and commercialization of cutting-edge bioprocessing technologies, such as single-use systems, continuous processing, and modular manufacturing, are being accelerated by increased funding. Besides, it is making it possible to establish new research facilities and pilot-scale units in previously underserved areas, which is assisting both startups and well-established biopharma companies in more effectively scaling novel therapies. Overall, these investments improve production flexibility, strengthen regulatory readiness, and enhance global competitiveness in a rapidly evolving biotech landscape.
Market Concentration & Characteristics
The high degree of innovation in the large- and small-scale bioprocessing industry is a key driver fueling the market growth. Continuous advancements such as automated bioreactor systems, real-time analytics, process intensification, and AI-driven process optimization are transforming biologics' development and manufacture. Innovations like continuous bioprocessing, disposable technologies, and modular facilities enable faster, more flexible, and cost-effective production. For instance, in April 2025, Thermo Fisher Scientific unveiled enhanced integrated platform solutions, including its Accelerator platform and a high-yield CHO K‑1 cell line, reducing investigational new drug (IND) filing timelines from 13 to 9 months. These offerings deliver up to 8 g/L protein expression and stable production, streamlining biologics development from gene to patient. These breakthroughs reduce development timelines, improve scalability, and ensure higher product consistency, which is especially critical for complex therapies such as monoclonal antibodies and cell and gene treatments.
Theindustry dynamics are greatly impacted by the growing number of mergers and acquisitions (M&A) in the bioprocessing and biopharmaceutical industries. To enhance their technology portfolios, expand manufacturing capacity, and gain access to cutting-edge bioprocessing platforms, larger pharmaceutical and biotech companies are actively acquiring smaller, innovative businesses. For instance, in October 2024, Lonza (Switzerland) finalized the acquisition of Genentech’s large-scale biomanufacturing site in Vacaville, California, for USD 1.2 billion, expanding its global mammalian production network. These strategic consolidations accelerate product development pipelines, reduce time-to-market, and foster integrated end-to-end biomanufacturing solutions, contributing to the overall growth and maturity of the global bioprocessing market.

Regulatory frameworks strongly shape the bioprocessing industry by setting strict requirements for quality, safety, and compliance. Guidelines from agencies such as the U.S. FDA and EMA ensure that bioprocessing systems meet high GMP, validation, and data-integrity standards. Although these regulations can be challenging for smaller firms, they ultimately drive demand for advanced, compliant technologies and greater process standardization.
As businesses continuously create and introduce cutting-edge machinery, consumables, and integrated solutions to meet the evolving demands of biologics manufacturing, product expansion in the large- and small-scale bioprocessing industry is a major factor driving market growth. To increase scalability, efficiency, and process control, manufacturers are adding next-generation bioreactors, filtration systems, sensors, and single-use technologies to their portfolios. For instance, in April 2025, Thermo Fisher Scientific launched the 5L DynaDrive Single-Use Bioreactor in Singapore, enhancing bioprocess development with scalable, high-performance, and sustainable solutions for biopharma and CDMO sectors. This expansion caters to traditional biologics, such as monoclonal antibodies, and emerging modalities, including cell and gene therapies, RNA-based therapeutics, and biosimilars. The continual introduction of high-performance, customizable, and automation-ready products further fuels demand across both small- and large-scale bioprocessing operations.
The market is largely driven by regional expansion, with equipment suppliers, CDMOs, and biopharmaceutical companies making significant investments in developing regions like the Middle East, Asia Pacific, and Latin America. These markets provide cost advantages, expanding patient populations, and favorable regulations that bolster regional biotech capacities. While North America and Europe continue to lead in cutting-edge technologies and robust regulations, nations such as China, India, and South Korea are establishing biotech hubs and attracting foreign investment. This geographic diversification increases global biologics production capacity, lowers supply chain risk, and enhances regional market access.
Scale Insights
Industrial scale dominated the global large- and small-scale bioprocessing industry and accounted for the largest revenue share in 2025. This growth can be attributed to the increasing demand for biopharmaceuticals and advancements in large-scale bioreactors. For instance, in June 2025, Univercells Technologies (Belgium) introduced its scale‑X nitro controller to enhance large-scale viral and advanced therapy manufacturing, offering compact, cost-effective, and scalable R&D-to-commercial production capabilities. Technological innovations such as automation enhance efficiency and reduce costs, making large-scale production more viable.
The small-scale segment is expected to grow at the fastest CAGR over the forecast period, owing to the need for flexible systems in personalized medicine. Moreover, single-use technologies enhance process control and productivity without compromising quality. For instance, in October 2024, Univercells Technologies launched its scale‑X nexo fixed‑bed bioreactor in Nivelles, Belgium. It optimized small‑scale cell culture workflows, reducing development timelines and costs while enabling seamless scalability for biopharma applications.
Workflow Insights
Downstream processing dominated the market, with the largest revenue share of 47.15% in 2025. Product purification, the key step in downstream processing, is considered the most challenging & crucial step and thus demands more attention. Moreover, any advancement in bioprocessing engineering directly affects the production yield, which positions the downstream processing segment as the center of significant attraction for several major pharma stakeholders.
The fermentation segment of the large- and small-scale bioprocessing industry is experiencing strong growth, driven by increasing adoption of microbial and cell-based expression systems across clinical and commercial manufacturing. Advancements in bioreactors, feed optimization, and process automation are enhancing yields, consistency, and real-time control, enabling manufacturers to meet stringent regulatory standards. As biopharma moves toward more efficient and sustainable production models, the fermentation segment is becoming increasingly essential across all scales of operation.
Product Insights
Bioreactors/Fermenters led the market, accounting for the largest revenue share of 29.41% in 2025, due to the extensive use of the product in the pharmaceutical sector and the portfolio expansion of major players. For instance, in September 2024, Chennai-based VFL Sciences (India) launched its GreatFlo benchtop fermentors and bioreactors, offering versatile, up to 10 L autoclavable and in‑situ sterilizable systems, enhancing process flexibility and affordability. Moreover, the availability of a wide range of bioreactors based on the operation mode, phase type, capacity, fluid flow type, use type, and applications is expected to boost the segment demand in the market.
The cell culture product segment is expected to be the fastest-growing segment over the forecast period. This is mainly attributed to the ongoing advancements in cell line development, including the development of the recombinant cell line. One of the most significant applications of cell culture in cell and molecular biology is to provide a platform for studying the biological, biochemical, physiological, and metabolic systems of wild-type and sick cells. The production of biopharmaceutical-derived cell lines from mammalian cells supports the projected growth of this segment.
Application Insights
Biopharmaceuticals held the dominant position in the market with the largest revenue share of 60.79% in 2025, primarily driven by the commercial success of biologics and biosimilars for clinical use in the current scenario. Moreover, companies are involved in enhancing their manufacturing capacity for biopharmaceutical development, which is expected to boost significant growth in the market.
Specialty industrial chemicals are projected to grow steadily due to rising demand for sustainable, bio-based alternatives to petrochemicals. These products have a smaller environmental footprint and are increasingly used in coatings, detergents, and personal care products. Ongoing technological advances and shifting consumer preferences toward high-performance, eco-friendly solutions further support this growth.
Use Type Insights
Multiple-use products for bioprocessing dominated the market, holding the largest share in 2025. The major factor attributed to its large share in the current market is the low environmental impact associated with implementing multiple-use instruments and a one-time investment. These systems have been highly stable in their operations for a long time and are the primary choice for large commercial bioprocesses due to the low risk of leakage or bag bursting. Their competitive lifecycle costs, along with the strong matching with large-scale productions, are the other factors that also support the usage of these systems.
Single-use systems are expected to grow at a notable CAGR over the forecast period due to their flexibility, reduced setup times, and lower contamination risks. They offer advantages in smaller-scale, personalized biomanufacturing, allowing quick product switches and minimizing cleaning needs. Moreover, innovations in materials and design make them increasingly attractive for biopharmaceutical production, particularly for complex molecules such as biologics. For instance, in October 2024, Belgium-based TekniPlex Healthcare showcased new single-use bioprocessing solutions, including custom barrier films, TPE/TPV tubing, silicone, and PVC components at CPHI Milan to support cell and gene therapy applications.
Mode Insights
The in-house segment led the market with the largest revenue share in 2025, owing to well-established international players capable of performing in-house biopharmaceutical production. Companies use their in-house capabilities to maintain the secrecy of their product development processes. Moreover, in-house facilities allow customized solutions, improved efficiency, and better integration with existing systems, supporting both large-scale and small-scale bioprocessing operations.

The outsourced segment is expected to grow at a considerable CAGR over the forecast period. This is due to the substantial growth in the budgets for outsourcing bio-manufacturing. Biopharma companies have started using contract manufacturers due to insufficient expertise, limited capacity to perform industrial-scale bioprocess applications, and the cost-saving advantages associated with contract services, which are expected to provide potential opportunities in the outsourcing sector of large- and small-scale bioprocessing.
Regional Insights
The North America large and small-scale bioprocessing market dominated the global market with the largest revenue share of 34.64% in 2025. The region's dominance in revenue generation is attributed to its focused, cutting-edge research and development in biomanufacturing. For instance, in June 2024, Ecolab Life Sciences and Repligen Corporation launched the DurA Cycle protein A chromatography resin in the U.S., enabling large‑scale monoclonal antibody manufacturing with reduced costs and enhanced purification efficiency. Increasing demand for biologics and cell therapies, and the presence of a substantial number of bio-manufacturing facilities in this region, have also contributed to the large share of this segment.
U.S. Large And Small-scale Bioprocessing Market Trends
The large- and small-scale bioprocessing industry in the U.S. led the North American market, driven by high investments in biotechnology research and a developed healthcare infrastructure. The prevalence of chronic diseases treated with biopharmaceuticals, and favorable government initiatives contribute to growth. For instance, in November 2024, Lonza completed its first GMP batch at its next-generation mammalian manufacturing facility in Portsmouth, USA, highlighting its commitment to advancing cell and gene therapy production, further driving market demand.
Europe Large And Small-scale Bioprocessing Market Trends
The large & small-scale bioprocessing industry in Europe is expected to grow significantly from 2026 to 2033, driven by a strong regulatory framework and significant R&D investments. For instance, in June 2025, Cytiva, a Danaher company, allocated USD 1.6 billion through 2028 to expand bioprocessing manufacturing capacity across North America, Europe, and Asia, reinforcing its regional strategy.
The UK large & small-scale bioprocessing industry held a significant share in 2025, primarily driven by the government funding for biotechnology research and a favorable regulatory environment. In addition, collaborations between academia and industry drive innovation in bioprocessing technologies.
The large & small-scale bioprocessing industry in Germany is growing rapidly in the Europe region driven by the deep expertise in process engineering and molecular biology. The country’s bioprocessing capabilities span from academic R&D labs to large-scale commercial manufacturing plants. German companies have been pioneers in developing upstream and downstream bioprocessing technologies, and their facilities are often outfitted with high degrees of automation and digital control. For instance, in December 2024, Eppendorf SE (Germany) and DataHow AG launched a strategic collaboration to integrate DataHowLab’s AI analytics into Eppendorf’s BioNsight cloud, enhancing global bioprocess data management efficiency. As biologics take a larger share of the global pharmaceutical market, Germany’s ability to integrate precision manufacturing with regulatory rigor places it in a strong competitive position.
Asia Pacific Large And Small-scale Bioprocessing Market Trends
The Asia Pacific large- and small-scale bioprocessing industry is anticipated to grow at the fastest CAGR of 12.80% over the forecast period. The region's economic development has led to increased investment in the healthcare sector. For instance, in April 2024, MilliporeSigma invested in launching a new bioprocessing production center in Daejeon, South Korea, to support biologics development, clinical research, and commercial manufacturing.
The large & small-scale bioprocessing industry in China led the Asia Pacific market in 2025, driven by a rapidly advancing biopharmaceutical sector and strong government support. Their competitive lifecycle costs, along with the strong matching with large-scale productions, are the other factors that also support the usage of these systems.
The Japan large & small-scale bioprocessing industry is growing steadily, supported by its precision-focused healthcare technologies and strong academia-industry collaboration in regenerative medicine and biopharmaceutical development.
Middle East And Africa Large and Small-scale Bioprocessing Market Trends
The large- and small-scale bioprocessing industry in the Middle East and Africa region is in the early but determined phase of building its bioprocessing ecosystem. Most activity focuses on foundational research, pilot-scale production, and public health-oriented biologics, such as vaccines and insulin. With support from international organizations, Africa is gradually moving toward self-sufficiency in bioproduction. Due to infrastructure constraints, the region focuses on small-scale bioprocessing; however, there is a clear ambition to expand into commercial-scale capabilities within the coming decade.
The Kuwait large- and small-scale bioprocessing industry is expected to grow steadily as the country pursues economic diversification and healthcare modernization. Although the current infrastructure is limited, interest in building capabilities for diagnostics, vaccines, and therapeutic biologics is rising.
Key Large And Small-scale Bioprocessing Company Insights
Companies such as Merck KGaA, Thermo Fisher Scientific, GE Healthcare, Sartorius AG, and Lonza have established substantial market presence through comprehensive product portfolios, large-scale manufacturing, and sustained investment in bioprocessing technology and automation.
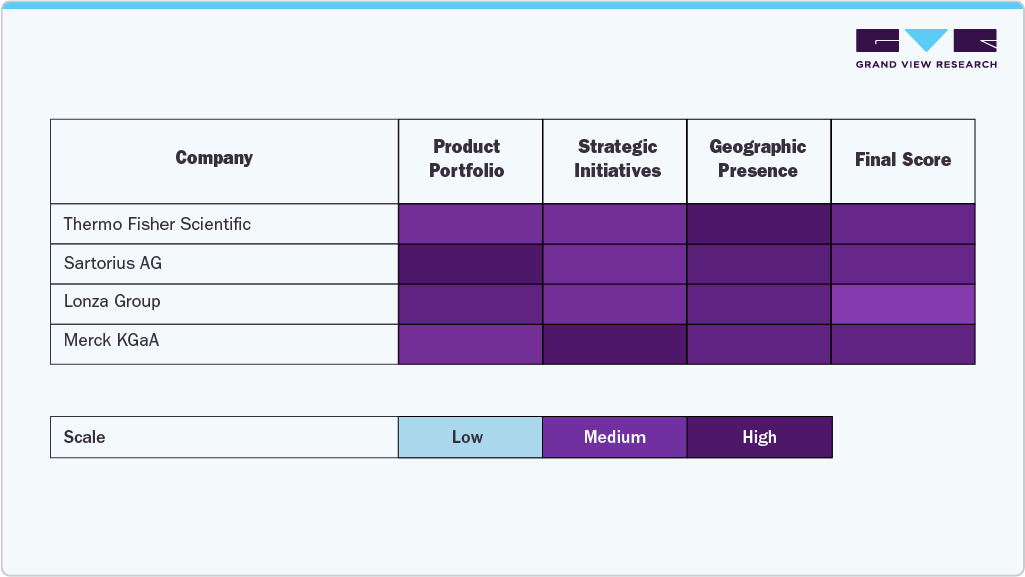
Asian and European firms, such as Samsung BioLogics, Binex Co., Ltd., Rentschler Biopharma, and Inno Biologics, are expanding globally by leveraging regional manufacturing strengths and forming strategic partnerships with multinational pharmaceutical companies. Meanwhile, niche players such as Bioprocess Control AB, Bio-Process Group, and TOYOBO Co., Ltd. support the market with specialized technologies in process analytics and upstream optimization, enabling efficient scale-up for smaller biotech and research organizations.
Key Large And Small-scale Bioprocessing Companies:
The following are the leading companies in the large and small-scale bioprocessing market. These companies collectively hold the largest market share and dictate industry trends.
- Merck KGaA
- Thermo Fisher Scientific, Inc.
- Danaher (Cytiva)
- Corning Inc.
- Sartorius AG
- Lonza
- CESCO BIOENGINEERING CO., LTD
- Bio-Process Group
- BPC Instruments AB
- Eppendorf AG
- Getinge AB
- PBS Biotech, Inc.
- Bio-Synthesis, Inc.
- Meissner Filtration Products, Inc.
- Entegris
- KUHNER AG
- Saint-Gobain
- ExcellGene SA
- Repligen Corporation
- Avantor, Inc
- CerCell A/S
- Univercells Technologies
- Distek, Inc.
- F. Hoffmann-La Roche Ltd
Recent Developments
-
In February 2025, Thermo Fisher Scientific announced plans to acquire Solventum’s Purification & Filtration business for USD 4.1 billion, enhancing its global bioproduction capabilities.
-
In March 2025, Cytiva expanded its Xcellerex X-platform bioreactor portfolio by introducing 500 L and 2,000 L single-use systems. These additions enable seamless scale-up from 50 L to 2,000 L, supporting the development of advanced therapeutics across the early clinical to commercial stages.
-
In July 2024, Thermo Fisher Scientific launched biobased films for single use bioprocessing containers, reducing greenhouse gas emissions and supporting recycling efforts. These films received ISCC PLUS certification.
Large And Small-scale Bioprocessing Market Report Scope
Report Attribute
Details
Market size value in 2026
USD 107.49 billion
Revenue forecast in 2033
USD 228.73 billion
Growth rate
CAGR of 11.39% from 2026 to 2033
Base year for estimation
2025
Historical data
2021 - 2023
Forecast period
2026 - 2033
Quantitative units
Revenue in USD million/billion, and CAGR from 2026 to 2033
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Scale, workflow, product, application, use type, mode, region
Regional scope
North America; Europe; Asia Pacific; Latin America; Middle East and Africa
Country scope
U.S.; Canada; Mexico; UK; Germany; France; Italy; Spain; Denmark; Sweden; Norway; India; China; Japan; Australia; South Korea; Thailand; Brazil; Argentina; Saudi Arabia; UAE; South Africa; Kuwait
Key companies profiled
Merck KGaA; Thermo Fisher Scientific, Inc.; Danaher (Cytiva); Corning Inc.; Sartorius AG; Lonza; CESCO BIOENGINEERING CO., LTD; Bio-Process Group; BPC Instruments AB; Eppendorf AG; Getinge AB; PBS Biotech, Inc.; Bio-Synthesis, Inc. ; Meissner Filtration; Products, Inc.; Entegris; KUHNER AG; Saint-Gobain; ExcellGene SA; Repligen Corporation; Avantor, Inc; CerCell A/S; Univercells Technologies; Distek, Inc.; F. Hoffmann-La Roche Ltd
Customization scope
Free report customization (equivalent up to 8 analyst’s working days) with purchase. Addition or alteration to country, regional & segment scope.
Pricing and purchase options
Avail customized purchase options to meet you exact research needs. Explore purchase options
Global Large And Small-scale Bioprocessing Market Report Segmentation
This report forecasts revenue growth and provides an analysis on the latest trends in each of the sub-segments from 2021 to 2033. For this report, Grand View Research has segmented the global large- and small-scale bioprocessing market report on the basis of scale, workflow, product, application, use type, mode, and region.
-
Scale Outlook (Revenue, USD Million, 2021 - 2033)
-
Industrial Scale (Over 50,000 Liter)
-
Small Scale (Less Than 50,000 Liter)
-
-
Workflow Outlook (Revenue, USD Million, 2021 - 2033)
-
Downstream Processing
-
Fermentation
-
Upstream Processing
-
-
Product Outlook (Revenue, USD Million, 2021 - 2033)
-
Bioreactors/Fermenters
-
Cell Culture Products
-
Filtration Assemblies
-
Bioreactors Accessories
-
Bags & Containers
-
Others
-
-
Application Outlook (Revenue, USD Million, 2021 - 2033)
-
Biopharmaceuticals
-
Specialty Industrial Chemicals
-
Environmental Aids
-
-
Use Type Outlook (Revenue, USD Million, 2021 - 2033)
-
Multi-Use
-
Single-Use
-
-
Mode Outlook (Revenue, USD Million, 2021 - 2033)
-
In-house
-
Outsourced
-
-
Regional Outlook (Revenue, USD Million, 2021 - 2033)
-
North America
-
U.S.
-
Canada
-
Mexico
-
-
Europe
-
UK
-
Germany
-
France
-
Italy
-
Spain
-
Denmark
-
Sweden
-
Norway
-
-
Asia Pacific
-
Japan
-
China
-
India
-
Australia
-
Thailand
-
South Korea
-
-
Latin America
-
Brazil
-
Argentina
-
-
Middle East & Africa
-
South Africa
-
Saudi Arabia
-
UAE
-
Kuwait
-
-
Frequently Asked Questions About This Report
b. The global large and small scale bioprocessing market size was estimated at USD 93.32 billion in 2025 and is expected to reach USD 107.49 billion in 2026
b. The global large and small scale bioprocessing market is expected to grow at a compound annual growth rate of 11.39% from 2026 to 2033 to reach USD 228.7 billion by 2033
b. By workflow, downstream processing segment dominated the large and small scale bioprocessing market with a share of 47.15% in 2024. This is driven by the growing need for high-purity biologics and efficient product recovery solutions.
b. Some prominent players operating in the market include Merck KGaA (Sigma-Aldrich Co. LLC), Thermo Fisher Scientific, Inc.; Corning, Inc.; Sartorius AG; CESCO BIOENGINEERING Co., Ltd.; Bioprocess Control AB; Eppendorf AG; Applikon Biotechnology; Lonza; and Meissner Filtration Products, Inc.
b. Key driving factors of the market include huge success of biologics, technological advancements in single-use bioprocessing, advent of single-use technology, and facility expansion by contract manufacturing organizations
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










