- Home
- »
- Pharmaceuticals
- »
-
Lysosomal Storage Disease Treatment Market Report, 2033GVR Report cover
![Lysosomal Storage Disease Treatment Market Size, Share & Trends Report]()
Lysosomal Storage Disease Treatment Market (2025 - 2033) Size, Share & Trends Analysis Report By Treatment Type (Enzyme Replacement Therapy, Substrate Reduction Therapy), By Disease Type, By Region, And Segment Forecasts
- Report ID: GVR-4-68040-649-3
- Number of Report Pages: 150
- Format: PDF
- Historical Range: 2021 - 2023
- Forecast Period: 2025 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
Lysosomal Storage Disease Treatment Market Summary
The global lysosomal storage disease treatment market size was estimated at USD 4.54 billion in 2024 and is projected to reach USD 6.68 billion by 2033, growing at a CAGR of 4.54% from 2025 to 2033, driven by rising diagnostic capabilities, advancements in enzyme replacement and gene therapies, and increased awareness of rare genetic disorders. The lysosomal storage disease (LSD) treatment market is experiencing steady growth due to expanding research and development initiatives and rising awareness about these rare genetic conditions.
Key Market Trends & Insights
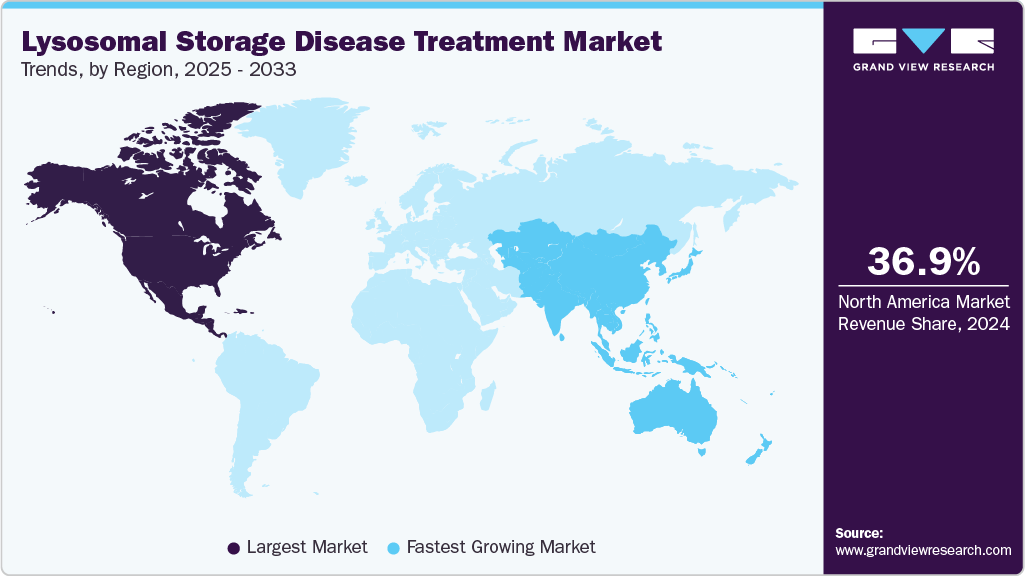
- North America lysosomal storage disease treatment market held the largest share of 36.98% of the global market in 2024.
- The lysosomal storage disease treatment industry in the U.S. is expected to grow significantly over the forecast period.
- By treatment type, the enzyme replacement therapy segment held the highest market share of 83.99% in 2024.
- By disease type, the gaucher disease segment held the highest market share in 2024.
Market Size & Forecast
- 2024 Market Size: USD 4.54 Billion
- 2033 Projected Market Size: USD 6.68 Billion
- CAGR (2025-2033): 4.54%
- North America: Largest market in 2024
- Asia Pacific: Fastest growing market
Significant R&D investments have enhanced therapeutic advancements, helping address unmet medical needs. A study published by the National Library of Medicine in July 2022 noted that LSDs, as a group, are more prevalent than previously assumed, with a collective incidence ranging from 1 in 5,000 to 1 in 8,000 individuals. It also highlighted the influence of ethnicity and geography on disease prevalence. For instance, Gaucher disease appears in 1 in 40,000 to 60,000 people in the general population, while the rate increases to 1 in 800 among Ashkenazi Jews. Similarly, Tay-Sachs disease and Niemann-Pick A are more common in this demographic, while the Finnish population has a higher frequency of aspartylglucosaminuria at 1 in 18,500. These rising prevalence rates underscore the growing need for effective treatments, fueling market expansion.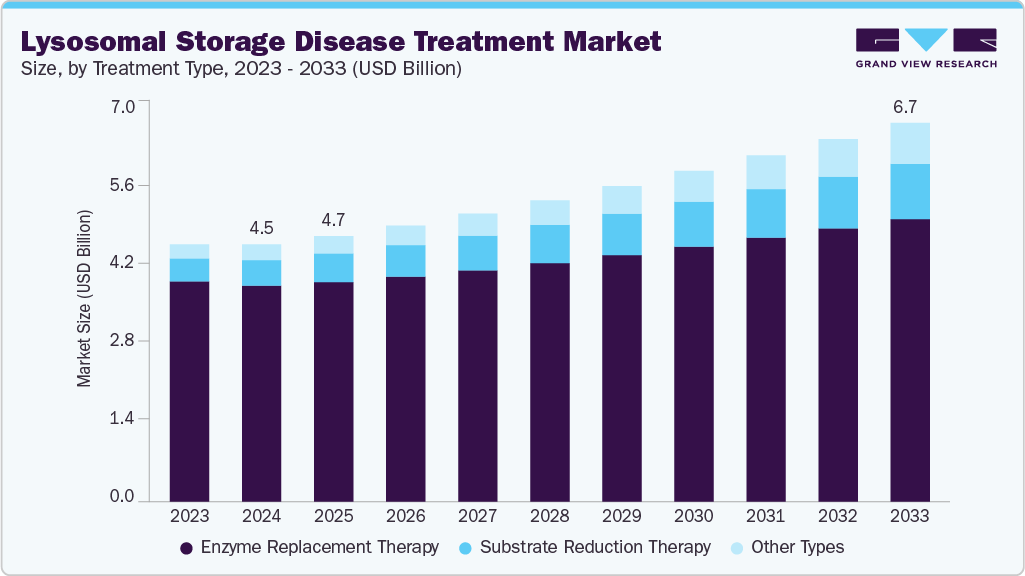
The introduction of innovative therapies is expected to strengthen the LSD treatment market further. Companies actively pursue novel drug development, supported by increasing global R&D spending. For example, in February 2021, Genethon initiated a gene therapy clinical trial targeting late-onset Pompe disease in collaboration with Spark Therapeutics, utilizing proprietary technologies. In parallel, awareness campaigns have gained momentum, playing a vital role in improving patient outcomes. In countries like the U.S. and the UK, awareness of Fabry disease has significantly increased through initiatives like Fabry Disease Awareness Month, observed every April, as promoted by the National Fabry Disease Foundation (NFDF), Fabry International Network, Fabry Australia, and the UK’s MPS Society.
Market Concentration & Characteristics
The lysosomal storage diseases (LSDs) treatment market is highly innovation-driven, with ongoing advancements in gene therapy, enzyme engineering, and precision medicine. Key players such as Sanofi, Takeda, and Amicus Therapeutics invest significantly in R&D to enhance therapeutic efficacy and patient outcomes. Enzyme replacement and substrate reduction therapies remain foundational, but pipeline innovations in gene editing and mRNA therapeutics are gaining momentum. Novel delivery mechanisms and personalized treatment approaches are being developed to overcome the limitations of existing therapies. Innovation remains essential due to the rare nature and complexity of LSDs, sustaining competitive differentiation and long-term growth.
Barriers to entry in the LSD treatment market are high due to extensive clinical validation, stringent regulatory requirements, and substantial capital investment. Developing and gaining approval for orphan drugs requires expertise in rare disease biology, long development cycles, and robust safety profiles. Existing players such as BioMarin and Johnson & Johnson maintain strong footholds through patented technologies, specialized manufacturing infrastructure, and established distribution channels. Additionally, the rarity of LSDs demands niche marketing strategies and disease-specific patient engagement programs. New entrants face significant challenges in competing with the clinical experience and reputational trust that incumbent firms have built.
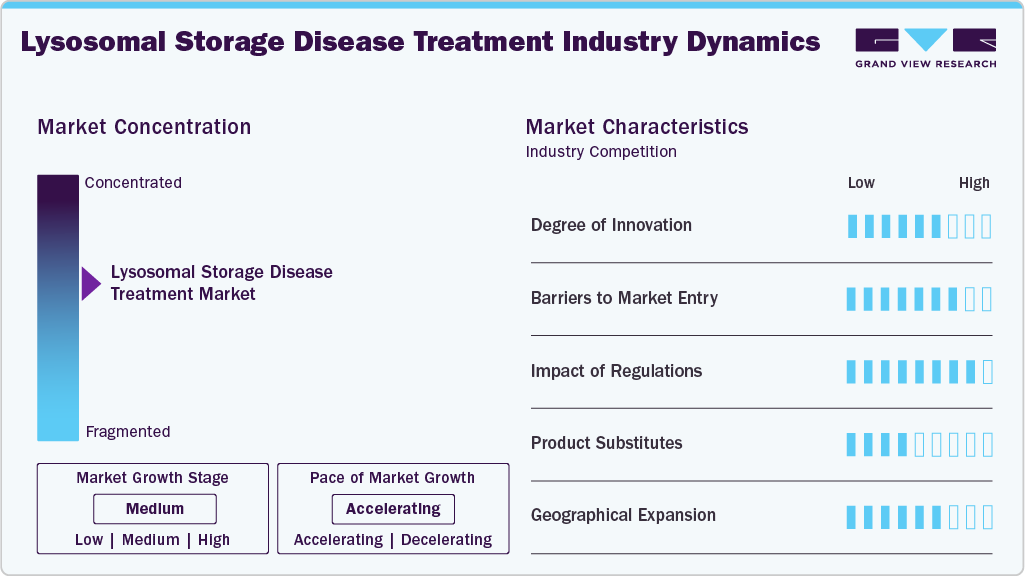
The regulatory landscape plays a crucial role in shaping the LSD treatment market, especially through orphan drug designations and accelerated approval pathways. Agencies like the FDA and EMA offer incentives to stimulate innovation in rare diseases, including tax credits, market exclusivity, and fee waivers. While these incentives support product development, compliance with safety, efficacy, and post-marketing surveillance requirements remains rigorous. Regulatory frameworks also necessitate long-term patient monitoring and real-world evidence to maintain product authorization. Companies like Alexion and Sanofi navigate complex international regulatory environments to achieve broad market access.
Product substitution in the LSD treatment market is limited due to the rarity and specificity of each disorder. Enzyme replacement therapies often have no direct generic alternatives due to their biological complexity and manufacturing sensitivity. Substrate reduction therapies provide some therapeutic flexibility, yet patient response variability limits interchangeability. Emerging modalities such as gene therapy may eventually serve as long-term substitutes for recurring treatments, potentially shifting treatment paradigms. However, clinical uncertainty, cost, and delivery challenges restrict immediate substitution, reinforcing reliance on existing therapies in the short term.
Geographical expansion in the LSDs market is shaped by unequal diagnostic capabilities, healthcare infrastructure, and reimbursement systems across regions. North America and Europe dominate access, with key players prioritizing regulatory approvals and partnerships in these markets. Expansion into Asia Pacific and Latin America is growing, driven by increased disease awareness, improved diagnostic tools, and supportive rare disease policies. However, high treatment costs and limited insurance coverage pose challenges in low- and middle-income countries. Companies like Takeda and Pfizer are gradually extending their global reach through localized operations and collaborative healthcare models.
Treatment Type Insights
The enzyme replacement therapy segment dominated the market with the largest revenue share of 83.99% in 2024 due to the high adoption of approved therapies and their proven clinical effectiveness. This approach offers a targeted solution by directly supplementing the deficient or missing enzymes in patients with lysosomal storage diseases (LSDs), resulting in symptom relief and disease management. For instance, in March 2021, Business Wire reported that JCR Pharmaceuticals received approval in Japan for IZCARGO (pabinafusp alfa) to treat MPS II (Hunter syndrome). It became the first approved enzyme replacement therapy for MPS II to penetrate the blood-brain barrier via intravenous administration, validating JCR’s J-Brain Cargo technology. The approval was based on clinical trials demonstrating improvements in both somatic and central nervous system symptoms.
The substrate reduction therapy segment is projected to grow at a significant CAGR over the forecast period, fueled by its rising use alongside or as an alternative to enzyme replacement therapy. These therapies help lower the accumulation of toxic substrates by reducing their production. Oral administration options like miglustat and eliglustat enhance convenience and patient adherence. They also benefit patients who are intolerant or unresponsive to enzyme therapies. Advancements in developing CNS-targeted substrate reducers are broadening treatment potential. These factors collectively support the growing relevance of this segment in LSD management.
Disease Type Insights
Fabry disease accounted for the largest revenue share of 29.31% in 2024, driven by increased diagnosis rates and the presence of multiple therapeutic options, including enzyme replacement and oral chaperone therapies. The availability of well-established biologics such as agalsidase beta (Fabrazyme) and the market adoption of oral migalastat (Galafold) have significantly contributed to commercial growth, especially in developed markets.
Moreover, heightened clinical attention toward heterozygous females and late-onset variants has expanded the diagnosed population base. According to a 2022 study published in Molecular Genetics and Metabolism, Fabry disease is one of the most commonly identified X-linked lysosomal disorders in newborn screening programs, with estimated birth prevalence rates ranging from 1 in 3,100 to 1 in 8,800 live births. This has translated into an expanding treated population and sustained therapy uptake, positioning Fabry disease as a key revenue-generating indication within the LSD market.
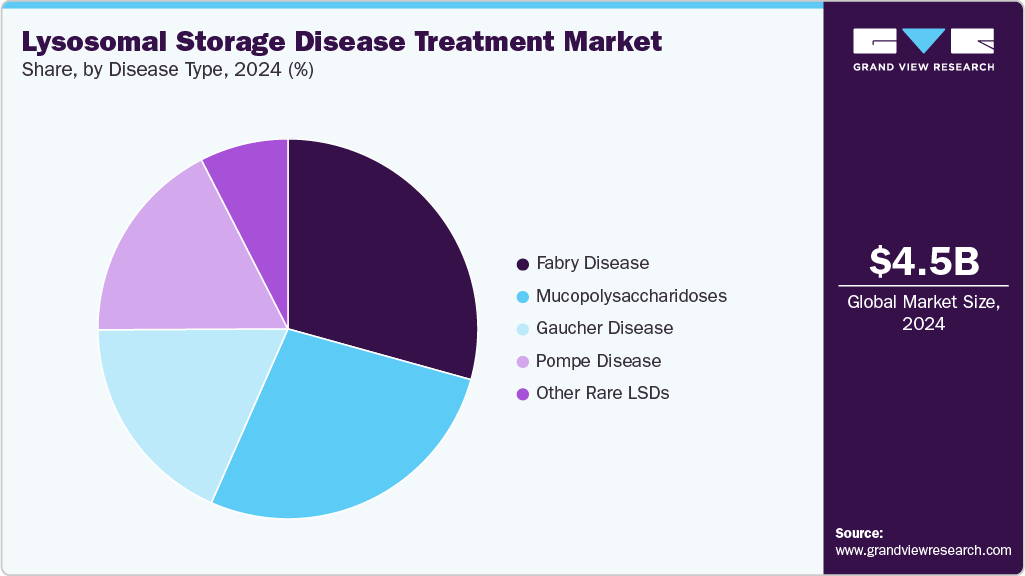
The Pompe disease segment is projected to grow at a rapid pace CAGR over the forecast period, due to rising diagnosis rates and expanding therapeutic options. Increasing awareness among clinicians and the availability of newborn screening in several regions have led to earlier detection and intervention. Approving next-generation therapies, such as cipaglucosidase alfa combined with miglustat, has improved clinical outcomes and broadened treatment access. For instance, in September 2023, Amicus Therapeutics announced FDA approval and the U.S. launch of Pombiliti (cipaglucosidase alfa-atga) + Opfolda (miglustat) for adults with late-onset Pompe disease. The therapy was approved under the accelerated approval pathway based on improved 6-minute walk distance. It is the first approved two-component treatment specifically for this condition in the U.S.
Regional Insights
North America lysosomal storage disease (LSDs) treatment market held the largest share of 36.98% of global revenue in 2024, due to the high prevalence of genetic disorders and strong diagnostic infrastructure. The region benefits from early disease detection and access to advanced therapeutic options. Major pharmaceutical companies based in North America continue to invest in clinical research and novel enzyme therapies. High awareness among physicians and patients supports the rapid adoption of approved treatments. Strategic collaborations between biotech firms and academic institutions are fueling innovation. Reimbursement policies for rare disease treatments further accelerate market penetration.
U.S. Lysosomal Storage Disease Treatment Market Trends
The U.S. lysosomal storage disease (LSDs) treatment market led North America market in 2024, holding a significant revenue share due to its robust biotechnology ecosystem and established regulatory framework. Increasing diagnosis rates of Gaucher, Fabry, and Pompe diseases contribute to market expansion. The presence of top-tier hospitals and research institutions drives clinical trials and drug development. High healthcare spending enables early access to new therapies for lysosomal storage disorders. Key players actively launch new enzyme replacement therapies and gene-based treatments targeting unmet needs. Digital health tools and patient registries enhance long-term disease monitoring and therapy optimization.
Europe Lysosomal Storage Disease Treatment Market Trends
Europe lysosomal storage disease (LSDs) treatment market held a substantial share in 2024 due to the presence of centralized rare disease networks and specialized healthcare facilities. Multinational pharmaceutical companies with strong R&D capabilities continue introducing innovative therapies globally. Cross-border patient registries and early screening programs contribute to improved disease identification. The growing number of clinical trials targeting LSDs is strengthening treatment pipelines. Increasing collaboration between the public and private sectors has accelerated access to cutting-edge therapies. Higher public awareness and early genetic testing are key enablers of market growth.
The UK lysosomal storage disease (LSDs) treatment market contributed significantly to the European market 2024, driven by enhanced genetic screening programs and rising patient advocacy. National healthcare systems are integrating specialized care pathways for rare diseases, including lysosomal disorders. Hospitals with metabolic and genetic expertise support timely diagnosis and initiation of treatment. Partnerships between biotech firms and academic centers are fostering clinical research in enzyme replacement and substrate reduction therapies. The availability of approved drugs and patient support initiatives further drives the market. The growing focus on precision medicine also aids treatment customization for better outcomes.
Germany lysosomal storage disease (LSDs) treatment market was strongly positioned in the lysosomal storage disease (LSDs) treatment market in 2024, supported by its advanced diagnostic infrastructure and pharmaceutical innovation. A high number of metabolic disorder centers across the country enhances early intervention. Domestic biotech firms are increasingly developing targeted therapies for Niemann-Pick, Fabry, and Gaucher diseases. Strong healthcare reimbursement systems allow access to expensive enzyme replacement therapies. Clinical research in gene therapy and pharmacological chaperones is expanding rapidly. Public-private collaborations are promoting sustainable long-term management of rare lysosomal disorders.
France lysosomal storage disease (LSDs) treatment market maintained a notable share in 2024 due to increasing research in rare metabolic diseases. National rare disease plans and medical centers of excellence provide a structured framework for managing LSDs. Pharmaceutical firms are leveraging partnerships with local hospitals to expand clinical trial activity. The widespread availability of diagnostic tools enables the timely identification and classification of various lysosomal conditions. Enzyme therapies for Pompe and Fabry diseases are gaining traction among specialists. Market expansion is supported by rising awareness through patient advocacy groups and education programs.
Asia-Pacific Lysosomal Storage Disease Treatment Market Trends
The Asia Pacific lysosomal storage disease (LSDs) treatment market is projected to grow at the fastest CAGR of 6.13% over the forecast period, fueled by increasing disease awareness and expanding diagnostic capabilities. Countries across the region are improving rare disease detection by adopting genetic testing technologies. Pharmaceutical firms are entering strategic alliances to boost therapy availability and scale distribution. Rising healthcare expenditure and urbanization are contributing to better access to specialist care. Growing participation in multinational clinical trials strengthens regional expertise in managing lysosomal disorders. Improved data collection and patient tracking systems are accelerating treatment development and uptake.
Japan lysosomal storage disease (LSDs) treatment market demonstrates steady growth in the market, focusing on precision medicine and rare disease care. The country’s advanced healthcare infrastructure supports early identification and treatment of lysosomal storage disorders. Local pharmaceutical firms are actively producing enzyme replacement therapies tailored to domestic needs. Regulatory pathways for orphan drugs are streamlined, facilitating quicker product approvals. Collaboration between academia and industry drives continuous innovation in therapy design. Patient registries and nationwide screening enhance treatment monitoring and improve outcomes.
China lysosomal storage disease (LSDs) treatment market is rapidly expanding its footprint due to increased healthcare investment and research focus on rare genetic diseases. Introducing newborn screening and diagnostic centers enables earlier identification of affected individuals. Biotech companies are investing in local production of enzyme therapies to improve affordability and access. Academic institutions are partnering with international firms to develop advanced treatments. Awareness campaigns and education programs are increasing knowledge among healthcare providers. Policy reforms and market liberalization are enhancing opportunities for international drug approvals.
Latin America Lysosomal Storage Disease Treatment Market Trends
Latin America lysosomal storage disease (LSDs) treatment market shows emerging potential due to rising diagnostic efforts and expanding specialist healthcare access. Countries are investing in metabolic disease infrastructure and clinical capacity building. Multinational companies are initiating collaborations with local partners to distribute enzyme therapies. Patient advocacy groups promote early detection and treatment access for rare lysosomal conditions. Educational outreach is improving physician familiarity with LSDs and available therapies. Growth in the market is supported by increased awareness and expansion of clinical programs.
Brazil lysosomal storage disease (LSDs) treatment market is a key market within Latin America, driven by a growing population base and improved healthcare reach. Hospitals are establishing genetic counseling and rare disease centers to improve diagnostic timelines. Domestic research in Fabry and Gaucher diseases is gaining visibility and encouraging local biotech innovation. International companies are launching enzyme therapies through public-private partnerships. Advances in clinical trial participation are providing new therapeutic options to patients. The country's focus on specialized care programs is creating long-term growth potential for the LSD treatment market.
Middle East & Africa Lysosomal Storage Disease Treatment Market Trends
The MEA lysosomal storage disease (LSDs) treatment market is gradually adopting lysosomal storage disease treatments due to improved healthcare infrastructure and diagnostic initiatives. Urban centers in key countries now host metabolic and genetic disorder clinics. International pharmaceutical firms are expanding operations through local collaborations and education campaigns. Access to enzyme therapies is growing with the establishment of rare disease treatment centers. Professional training programs equip physicians with the skills to manage complex lysosomal conditions. The region is expected to grow moderately as awareness and capacity improve.
Saudi Arabia lysosomal storage disease (LSDs) treatment market is strengthening its position with an enhanced focus on rare disease management and clinical genetics. Investment in healthcare modernization is leading to the development of advanced diagnostic labs and treatment centers. Hospitals are incorporating genetic screening for early detection of lysosomal disorders in pediatric populations. The market benefits from international collaborations focused on training and therapy access. Leading biopharma companies are expanding distribution networks across the country. Ongoing initiatives in data integration and patient monitoring are improving therapy outcomes and adherence.
Key Lysosomal Storage Disease Treatment Company Insights
The lysosomal storage diseases (LSDs) treatment market is led by Pfizer Inc., Takeda Pharmaceutical Company Limited, and Sanofi (Genzyme), with strong portfolios in enzyme replacement therapies. BioMarin and Johnson & Johnson (Actelion) enhance competitiveness through targeted treatments and rare disease research. Amicus Therapeutics and Alexion Pharmaceuticals focus on next-gen therapies, including chaperone and immune-based approaches. Emerging players like Sigilon Therapeutics and Orphazyme A/S are advancing innovative platforms for improved outcomes. Strategic partnerships, robust pipelines, and global reach drive market leadership and expansion.
Key Lysosomal Storage Disease Treatment Companies:
The following are the leading companies in the lysosomal storage disease treatment market. These companies collectively hold the largest market share and dictate industry trends.
- Pfizer Inc
- Takeda Pharmaceutical Company Limited (Shire Plc)
- Sanofi (Genzyme Corporation)
- BioMarin
- Johnson & Johnson (Actelion Pharmaceuticals Ltd)
- Amicus Therapeutics, Inc
- Alexion Pharmaceuticals, Inc
- Sigilon Therapeutics, Inc
- Orphazyme A/S
Recent Developments
-
In June 2025, ISU Abxis (a subsidiary of ISU Group) launched its Gaucher disease treatment Abcertin (a form of imiglucerase) across Middle Eastern and North African (MENA) markets, marking a significant expansion beyond its prior availability in South Korea. This strategic rollout underscores Abcertin's broader regional penetration, even though it is not authorized under the WHO/EMA biosimilar frameworks.
-
In February 2025, Denali Therapeutics announced that its investigational enzyme replacement therapy for Hunter syndrome (MPS II), tividenofusp alfa (DNL310), successfully met safety endpoints and achieved key biomarker reductions, including a ~90% decrease in cerebrospinal fluid heparan sulfate after 24 weeks. Treated patients also demonstrated long-term improvements in cognition and hearing, with sustained benefits observed out to more than 4 years. Building on this, Denali is preparing to submit a rolling accelerated BLA with the FDA in early 2025, aiming for a late 2025 to early 2026 launch.
-
In January 2022, Maze Therapeutics presented preclinical data at the ESPKU Congress highlighting MZE001, an oral GYS1 inhibitor for Pompe disease. The data showed reduced glycogen buildup, improved biomarkers, strong target engagement, and favorable pharmacokinetics.
-
In August 2021, Amicus Therapeutics announced European Commission approval of Pombiliti + Opfolda for AAV1 antibody-negative adults with late-onset Pompe disease. The decision followed April’s CHMP opinion and was based on positive Phase 3 PROPEL study results.
Lysosomal Storage Disease Treatment Market Report Scope
Report Attribute
Details
Market size value in 2025
USD 4.68 billion
Revenue forecast in 2033
USD 6.68 billion
Growth rate
CAGR of 4.54% from 2025 to 2033
Base year for estimation
2024
Historical data
2021 - 2023
Forecast period
2025 - 2033
Quantitative units
Revenue in USD million/billion and CAGR from 2025 to 2033
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Treatment type, disease type, region
Regional scope
North America; Europe; Asia Pacific; Latin America; Middle East & Africa
Country scope
U.S.; Canada; Mexico; UK; Germany; France; Italy; Spain; Denmark; Sweden; Norway; China; Japan; India; Australia; South Korea; Thailand; Brazil; Argentina; South Africa; Saudi Arabia; UAE; Kuwait
Key companies profiled
Pfizer Inc; Takeda Pharmaceutical Company Limited (Shire Plc); Sanofi (Genzyme Corporation); BioMarin; Johnson & Johnson (Actelion Pharmaceuticals Ltd); Amicus Therapeutics, Inc; Alexion Pharmaceuticals, Inc; Sigilon Therapeutics, Inc; Orphazyme A/S.
Customization scope
Free report customization (equivalent up to 8 analysts working days) with purchase. Addition or alteration to country, regional & segment scope.
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
Global Lysosomal Storage Disease Treatment Market Report Segmentation
This report forecasts revenue growth at global, regional, and country levels and provides an analysis of the latest industry trends in each of the sub-segments from 2021 to 2033. For this study, Grand View Research has segmented the global lysosomal storage disease treatment market report based on treatment type, disease type, and region:
-
Treatment Type Outlook (Revenue, USD Million, 2021 - 2033)
-
Enzyme Replacement Therapy
-
Velaglucerase Alfa
-
Alglucosidase Alfa (Myozyme/Lumizyme)
-
Idursulfase (Elaprase)
-
Imiglucerase (Cerezyme)
-
Others
-
-
Substrate Reduction Therapy
-
Others
-
-
Disease Type Outlook (Revenue, USD Million, 2021 - 2033)
-
Gaucher Disease
-
Mucopolysaccharidoses
-
Pompe Disease
-
Fabry Disease
-
Others
-
-
Regional Outlook (Revenue, USD Million, 2021 - 2033)
-
North America
-
U.S.
-
Canada
-
Mexico
-
-
Europe
-
UK
-
Germany
-
France
-
Italy
-
Spain
-
Denmark
-
Sweden
-
Norway
-
-
Asia Pacific
-
Japan
-
China
-
India
-
Australia
-
South Korea
-
Thailand
-
-
Latin America
-
Brazil
-
Argentina
-
-
Middle East & Africa
-
South Africa
-
Saudi Arabia
-
UAE
-
Kuwait
-
-
Frequently Asked Questions About This Report
b. The global lysosomal storage disease treatment market size was estimated at USD 4.54 billion in 2024 and is expected to reach USD 4.68 billion in 2025.
b. The global lysosomal storage disease treatment market is projected to grow at a CAGR of 4.54% from 2025 to 2033 to reach USD 6.68 billion by 2033.
b. Based on treatment type, enzyme replacement therapy segment dominated the market with the largest revenue share of 83.99% in 2024 due to the high adoption of approved therapies and their proven clinical effectiveness.
b. Key players in the Lysosomal Storage Disease Treatment Market include Sanofi Genzyme, Takeda Pharmaceutical, Pfizer Inc., Johnson & Johnson (Actelion), and BioMarin Pharmaceutical, which offer established enzyme replacement and substrate reduction therapies.
b. The lysosomal storage disease treatment market is driven by increasing awareness of rare genetic disorders, advancements in diagnostic technologies, and growing investments in orphan drug development.
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










