- Home
- »
- Pharmaceuticals
- »
-
Meningococcal Vaccines Market Size, Industry Report, 2033GVR Report cover
![Meningococcal Vaccines Market Size, Share & Trends Report]()
Meningococcal Vaccines Market (2026 - 2033) Size, Share & Trends Analysis Report By Type (Bivalent, Quadrivalent), By Brand (Menveo, Menactra, Nimenrix, Trumenba), By Serotype (W-135), By Age Group (Infants (0 to 2 years), By Sales Channel, By Region, And Segment Forecasts
- Report ID: GVR-4-68039-045-9
- Number of Report Pages: 150
- Format: PDF
- Historical Range: 2021 - 2024
- Forecast Period: 2026 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
Meningococcal Vaccines Market Summary
The global meningococcal vaccines market size was estimated at USD 4.05 billion in 2025 and is projected to reach USD 5.96 billion by 2033, growing at a CAGR of 4.5% from 2026 to 2033. The increasing prevalence of meningitis is one of the major factors expected to drive the market.
Key Market Trends & Insights
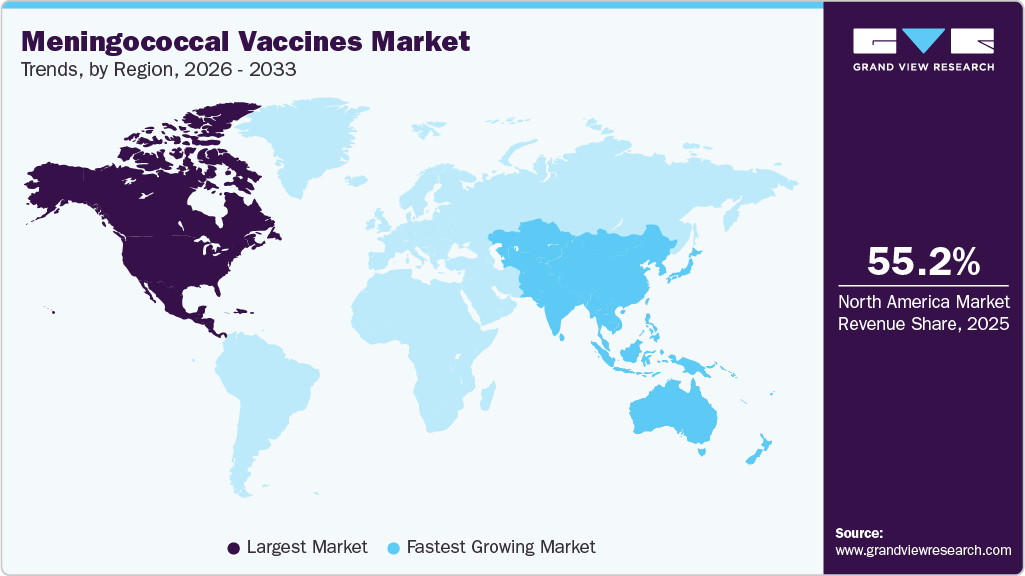
- North America dominated the global meningococcal vaccines market with the largest revenue share of 55.16% in 2025.
- The meningococcal vaccines industry in the U.S. is expected to grow at the significant CAGR over the forecast period.
- By type, the bivalent segment led the market with the largest revenue share of 80.30% in 2025.
- By brand, the menveo segment led the market with the largest revenue share of 29.56% in 2025.
- By serotype, the Y segment led the market with the largest revenue share of 32.98% in 2025.
Market Size & Forecast
- 2025 Market Size: USD 4.05 Billion
- 2033 Projected Market Size: USD 5.96 Billion
- CAGR (2026-2033): 4.5%
- North America: Largest market share in 2025
- Asia Pacific: Fastest growing market
According to the Centers for Disease Control and Prevention (CDC), approximately 1.2 million cases of bacterial meningitis are projected worldwide each year. Moreover, expanding immunization programs, greater awareness of the disease, and increased R&D activities for meningococcal vaccines are expected to fuel market growth during the forecast period.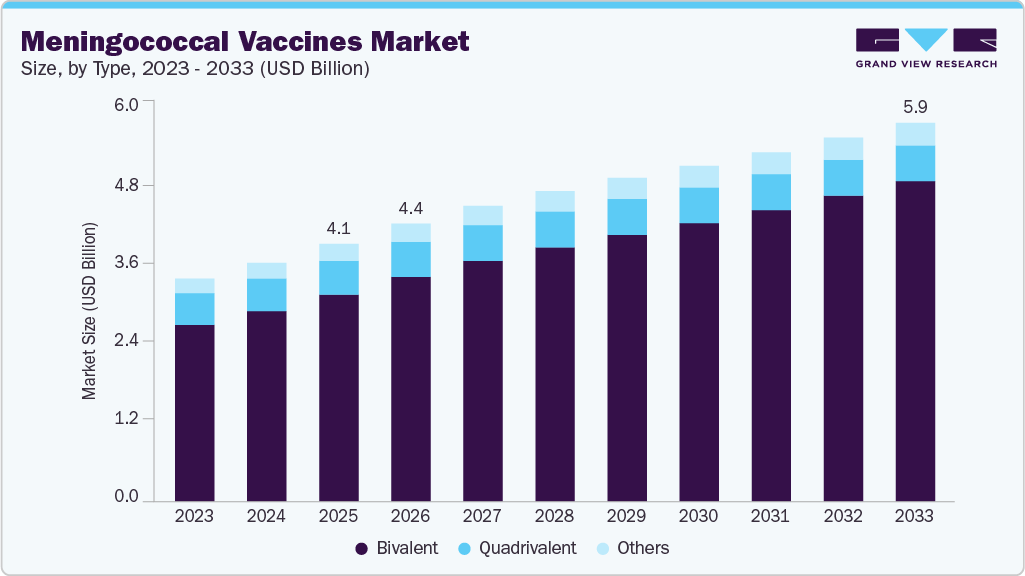
Meningococcal vaccines are critical tools in preventing invasive Neisseria meningitidis infections, which can cause life-threatening meningitis and sepsis. These vaccines target the serogroups most responsible for disease-A, B, C, W, and Y-through distinct formulations, including quadrivalent (MenACWY), serogroup B (MenB), and pentavalent (MenABCWY). As of 2025, regulatory agencies have expanded approvals to improve coverage across age groups, with MenQuadfi licensed for individuals from six weeks of age to provide protection against serogroups A, C, W, and Y. Routine recommendations by public health authorities continue to emphasize adolescent immunization as foundational to disease control. For instance, in July 2025, CDC guidance reinforced the importance of routine vaccination for adolescents and at-risk individuals.
Market and regulatory developments in 2024-2025 reflect a strategic shift toward broader protective vaccines that simplify immunization schedules and enhance uptake. Pentavalent vaccines such as Pfizer’s Penbraya and GSK’s Penmenvy, each covering serogroups A, B, C, W, and Y, have received approval for use in individuals aged 10-25 years, consolidating protection previously provided by multiple vaccines. In April 2025, ACIP recommended using pentavalent vaccines when both MenACWY and MenB immunizations are indicated during the same clinical visit, streamlining delivery protocols. For instance, in April 2025, a federal advisory committee recommended pentavalent meningococcal vaccines to support broader, integrated vaccination approaches.
Public health guidance and vaccine uptake efforts remain focused on balancing broad protection with targeted risk-based strategies. In 2024, the FDA updated dosing schedules for MenB vaccines, such as Bexsero, to optimize immunogenicity in recommended cohorts, underscoring evolving evidence and refinements to dosing schedules. Concurrently, efforts in lower-income settings, such as the 2024 rollout of WHO-recommended five-strain vaccines in parts of Africa, illustrate the global imperative to extend meningococcal prevention beyond high-income markets. Continuing enhancements in vaccine access and policy aim to reduce the burden of meningococcal disease globally through age-appropriate immunization.
Market Concentration & Characteristics
The meningococcal vaccine industry demonstrates moderate to high innovation driven by scientific advances in antigen design and delivery. Recent work focuses on combining serogroup coverage into single formulations and improving immunogenicity in younger age groups and immunocompromised patients. Manufacturers invest in conjugate technology and recombinant protein approaches to improve the breadth and durability of protection. Innovation also targets thermostability and simplified dosing to support immunization programs in resource-constrained settings. Incremental improvements such as extended age approvals and combination vaccines are commercially valuable because they reduce clinic visits and increase uptake. Competitive differentiation increasingly depends on real-world effectiveness data and cold chain logistics.
Entering the meningococcal vaccine industry requires substantial capital for research, development, clinical trials, and scale-up of manufacturing. Regulatory pathways demand extensive safety and efficacy evidence, which lengthens time to market and raises costs. Intellectual property protections for antigen constructs and manufacturing processes can limit new entrants. Economies of scale matter because production requires specialized facilities and rigorous quality control. Market access is influenced by procurement dynamics with large buyers, such as national immunization programs and global agencies, which negotiate prices and volumes. New entrants must also build distribution networks and trust with public health authorities to secure inclusion in immunization schedules.
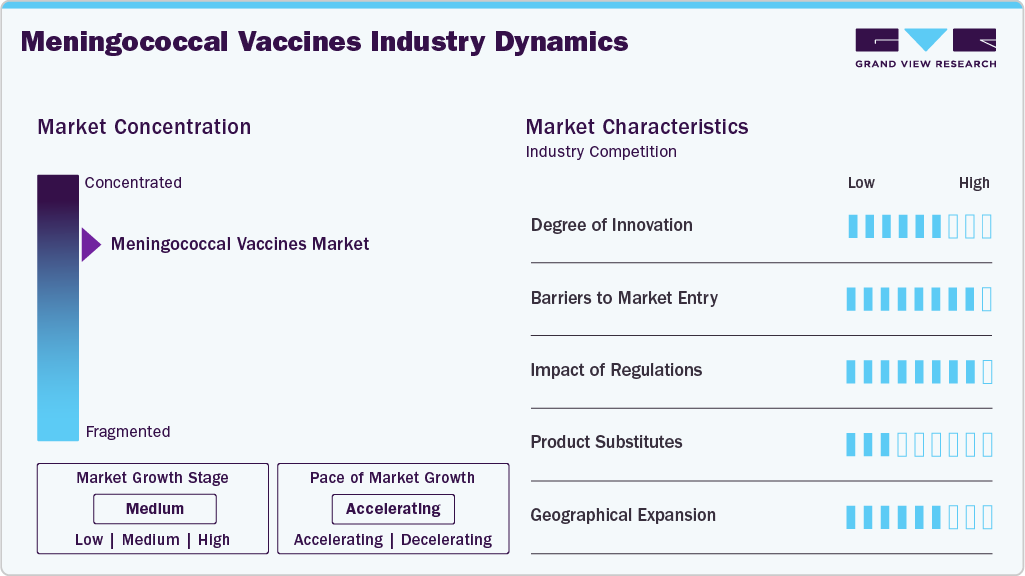
Regulation shapes product development, market access, and commercial strategy across the industry. Stringent licensure requirements ensure safety and efficacy but increase development timelines and costs. Regulatory decisions on age indications dosing schedules and interchangeability influence demand dynamics. Public health agency recommendations, including national advisory committee endorsements, determine reimbursement and procurement priorities. Compliance with lot-release pharmacovigilance and cold-chain standards adds operational complexity. Harmonization across jurisdictions can accelerate rollout, yet divergence forces tailored regulatory and clinical packages. In many markets, conditional approvals tied to post-market commitments are used to expedite access while ensuring continued evidence generation.
Substitutes for licensed meningococcal vaccines are limited, given the specific protection vaccines provide. Short-term substitutes can include prophylactic antibiotics to control outbreaks and clinical surveillance to detect cases early. For serogroup-specific gaps, some public health programs may rely on targeted immunization campaigns using alternative vaccine brands with similar serogroup coverage. Longer-term scientific substitutes could emerge from novel platforms, such as universal bacterial vaccines targeting conserved proteins across strains, though these remain investigational. Ultimately, vaccines remain the most effective preventive option, and substitutes typically play an adjunct role during supply shortfalls or urgent outbreak responses.
Geographic expansion is driven by disease epidemiology, procurement pathways, and local regulatory approval. High-income markets adopt new formulations faster due to payer capacity and stronger pharmacovigilance systems. Expanding into low- and middle-income regions requires demonstrating cost effectiveness and supporting technology transfer or tiered pricing to enable sustainable supply. Partnerships with global procurement agencies and participation in pooled procurement mechanisms facilitate access. Local regulatory approvals and cold chain readiness are prerequisites. Commercial strategy often sequences approvals by priority markets to leverage scale, then negotiates inclusion in national immunization programs based on evidence of disease burden and programmatic feasibility.
Type Insights
The bivalent segment led the market with the largest revenue share of 80.30% in 2025 and is expected to grow at the fastest CAGR over the forecast period, due to its long-standing clinical acceptance, streamlined dosing, and continued use in routine immunization and outbreak-response settings, particularly in cost-sensitive and public procurement-driven markets. Healthcare systems value bivalent vaccines for their established safety profiles, predictable supply chains, and operational simplicity, which support sustained utilization even as higher-valency options gain visibility. However, the segment operates within a dynamic competitive environment where manufacturers are progressively expanding portfolios toward broader serogroup coverage, signaling a gradual shift in innovation focus while maintaining bivalent products as foundational offerings in immunization programs.
For instance, in February 2025, GSK received approval from the U.S. Food and Drug Administration for its pentavalent meningococcal vaccine Penmenvy, covering serogroups A, B, C, W, and Y, for individuals aged 10-25 years. This development underscores how manufacturers are complementing existing bivalent and lower-valency vaccines with broader formulations to address evolving public health needs, while bivalent vaccines continue to play a role in established immunization strategies.
Brand Insights
The menveo segment led the market with the largest revenue share of 29.56% in 2025, due to sustained regulatory support and product enhancements that improve usability and administration. Its long-standing approval for active immunization against Neisseria meningitidis serogroups A, C, W, and Y across broad age groups (from infants through adults) reinforces its use in both routine immunization and catch-up campaigns. Menveo’s role as a foundational quadrivalent vaccine continues to anchor provider preference and procurement frameworks, even as higher-valency options emerge. Ongoing investments in simplifying presentation and delivery further support market adoption.
For instance, in November 2024, the European Commission approved a fully liquid, single-vial presentation of Menveo, eliminating the need for reconstitution before use and enhancing operational ease for healthcare providers. This regulatory milestone reflects industry focus on improving vaccine delivery and accessibility, which supports continued uptake in Europe and adjacent markets.
The nimenrix segment is projected to grow at the fastest CAGR of 11.3% over the forecast period, as public health bodies broaden recommendations for quadrivalent meningococcal vaccination across age groups and immunization settings. Its established profile protecting against serogroups A, C, W, and Y supports integration into routine schedules and catch-up campaigns in regions where meningococcal disease burden or policy emphasis on broad coverage is rising. Uptake is further reinforced by evidence on co-administration with other pediatric vaccines and supportive guidance from regional advisory committees, enhancing convenience and compliance for caregivers and providers.
For instance, in July 2024, the French National Authority for Health (HAS) issued a favourable opinion supporting continued inclusion of Nimenrix in its immunization recommendations, reaffirming its role within national vaccine strategies and influencing market uptake dynamics.
Serotype Insights
The Y segment led the market with the largest revenue share of 32.98% in 2025, driven by its growing association with invasive meningococcal disease among adolescents, older adults, and high-risk populations, particularly in North America and parts of Europe. Public health agencies continue to prioritize vaccines targeting serogroup Y as part of quadrivalent immunization strategies, given its role in sporadic cases and outbreaks. Demand is supported by routine adolescent vaccination programs, booster policies, and heightened awareness in institutional settings such as colleges, military facilities, and long-term care environments. The segment benefits from consistent inclusion within ACWY formulations, reinforcing its relevance across prevention frameworks.
For instance, in August 2024, the Centers for Disease Control and Prevention reported sustained circulation of serogroup Y-associated invasive meningococcal disease in the United States, reaffirming the importance of ACWY vaccination strategies in routine and risk-based immunization recommendations. This epidemiological update reinforced the continued emphasis on serogroup Y coverage in national vaccination policies and underscored its role in shaping vaccine demand across key markets.
The B segment is projected to grow at the fastest CAGR of 5.8% over the forecast period, as health authorities and clinicians intensify focus on addressing disease burden associated with Neisseria meningitidis serogroup B, particularly among adolescents and young adults, where incidence rates remain significant. This segment’s growth is supported by expanding recommendations in routine immunization frameworks and rising public awareness of serogroup B’s impact relative to other serotypes. Increased adoption is further driven by continued investment in B vaccine formulations that demonstrate broader immunogenicity and compatibility with co-administration schedules, enhancing programmatic feasibility and uptake in diverse immunization settings.
For instance, in February 2025, the U.S. Food and Drug Administration approved GSK’s pentavalent meningococcal vaccine Penmenvy. This integrated formulation provides robust serogroup B protection alongside A, C, W, and Y, underscoring advancing regulatory support for vaccines that include robust serogroup B protection. This approval highlights the strategic importance of serogroup B coverage within broader market dynamics and sets a precedent for expanded indication profiles that may accelerate segment growth.
Age Group Insights
The children and adults (2 years and above) segment led the market with the largest revenue share of 63.19% in 2025, due to its alignment with routine immunization schedules, booster policies, and risk-based vaccination strategies. This age group encompasses school-aged children, adolescents, and adults in institutional or high-exposure settings, including colleges, military facilities, and travelers. Demand is reinforced by established clinical evidence, broad regulatory approvals, and strong integration of quadrivalent and serogroup-specific vaccines into national programs. Compared with infant vaccination, uptake in this segment benefits from clearer policy mandates, higher awareness, and structured delivery channels through schools and primary care networks.
For instance, in July 2024, the Centers for Disease Control and Prevention reaffirmed meningococcal vaccination recommendations for children, adolescents, and adults aged 2 years and above who are at increased risk, including those with underlying medical conditions and individuals in outbreak-prone settings. This guidance reinforced the continued use of licensed meningococcal vaccines in this age group and supported sustained demand through routine and catch-up immunization pathways.
The infants (0 to 2 years) segment is projected to grow at the fastest CAGR of 5.7% over the forecast period, driven by expanding regulatory approvals, rising inclusion of meningococcal vaccines in early childhood immunization schedules, and increasing emphasis on preventing invasive meningococcal disease in high-risk infants. Public health authorities are prioritizing early protection due to the higher disease severity and mortality observed in this age group. Growth is further supported by improvements in vaccine formulations suitable for infant immune systems, expanded age indications, and integration with routine pediatric visits, which improve compliance and programmatic feasibility across developed and select emerging markets.
For instance, in May 2025, the U.S. Food and Drug Administration approved expanded use of Sanofi’s MenQuadfi vaccine for infants as young as 6 weeks, marking a significant regulatory milestone for meningococcal prevention in early childhood. This development reflects a broader industry and policy shift toward earlier immunization, directly supporting growth momentum in the infant segment.
Sales Channel Insights
The private segment led the market with the largest revenue share of 64.90% in 2025 and is anticipated to grow at the fastest CAGR over the forecast period, due to higher vaccine uptake through private hospitals, pediatric clinics, travel medicine centers, and corporate healthcare providers. This channel benefits from faster adoption of newly approved vaccines, broader brand availability, and greater pricing and procurement flexibility compared with public programs. Demand is further supported by rising health awareness, out-of-pocket spending capacity, and physician-driven recommendations, particularly for adolescents, travelers, and high-risk adults.
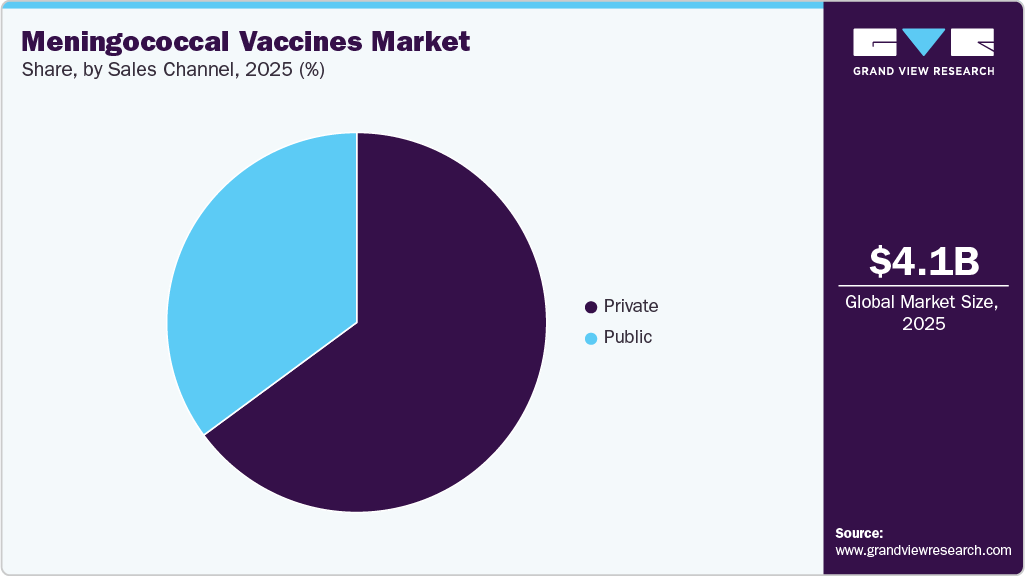
The private segment is also a key launchpad for premium and combination vaccines, enabling manufacturers to accelerate early adoption before wider public tender inclusion. For instance, in February 2025, GSK announced an expanded commercial rollout of its pentavalent meningococcal vaccine, Penmenvy, across private healthcare networks in the United States following regulatory approval. The launch strategy prioritized private clinics and hospital systems, reflecting the segment’s importance in driving early uptake of next-generation meningococcal vaccines and reinforcing its growth momentum over the forecast period.
Regional Insights
North America dominated the global meningococcal vaccines market with the largest revenue share of 55.16% in 2025, supported by strong regulatory oversight, high awareness, and structured immunization schedules. Demand is anchored by routine adolescent vaccination, booster compliance, and risk-based immunization among travelers and immunocompromised populations. The region benefits from rapid adoption of newly approved vaccines through private and public channels, supported by favorable reimbursement and strong provider engagement. Ongoing surveillance of serogroup trends and outbreak preparedness continues to shape procurement decisions. Manufacturers prioritize North America for early launches, lifecycle management, and portfolio expansion due to predictable demand and pricing stability.
U.S. Meningococcal Vaccines Market Trends
The meningococcal vaccines market in the U.S. is a key revenue and innovation hub within the North America, driven by CDC recommendations and ACIP guidance that strongly influence uptake. Routine vaccination in adolescents, expanded risk-based use, and increasing adoption of combination vaccines support sustained demand. The private healthcare channel plays a critical role in early uptake of newly approved products. Strong pharmacovigilance systems and real-world evidence generation further reinforce provider confidence. Market dynamics are shaped by regulatory approvals, payer coverage decisions, and school or college vaccination requirements, making the U.S. a priority market for strategic launches.
Europe Meningococcal Vaccines Market Trends
The meningococcal vaccines market in Europe is experiencing steady growth, supported by national immunization programs and centralized procurement mechanisms. Demand varies by country based on epidemiology, policy priorities, and budget allocation. The region has shown openness to adopting improved formulations such as liquid presentations and broader age indications. Public tenders dominate volume while private clinics contribute to incremental growth, particularly for travel-related vaccination. Regulatory harmonization through the EMA supports multi-country launches, though pricing pressure remains a key challenge. Europe remains strategically important for manufacturers due to stable volumes and long-term programmatic commitments.
The UK meningococcal vaccines market is anticipated to grow at a significant CAGR during the forecast period. The UK maintains a well-structured meningococcal vaccination landscape driven by NHS-led immunization policies. Strong public funding supports routine pediatric and adolescent vaccination, while targeted campaigns address outbreak risks. The UK has historically been an early adopter of meningococcal vaccines, particularly in response to epidemiological shifts. Centralized procurement ensures volume stability but limits pricing flexibility. Manufacturers focus on demonstrating cost-effectiveness and public health impact to secure inclusion. Continued surveillance and policy responsiveness support sustained demand across age groups within a predominantly public market framework.
The meningococcal vaccines market in Germany is characterized by a mixed public-private healthcare structure that supports both routine and elective vaccination. Recommendations from national advisory bodies influence eligibility for reimbursement, while private insurance enables broader access. Demand is supported by travel vaccination, occupational risk coverage, and increasing awareness among adults. The market values high-quality clinical evidence and safety data, influencing procurement decisions. Manufacturers benefit from Germany’s strong distribution infrastructure and physician-driven prescribing behavior, making it an important European market despite moderate pricing pressure.
The France meningococcal vaccine market is characterized by a policy in which national health authority recommendations strongly shape uptake. Inclusion in the national immunization schedule ensures high coverage in targeted age groups, particularly children and adolescents. Public reimbursement dominates demand, though private uptake exists for catch-up and travel indications. Regulatory evaluations emphasize public health benefits and population impact. Recent policy reaffirmations have supported the continued use of established vaccines, providing stability in demand. France remains attractive for manufacturers due to predictable volumes and long-term immunization planning.
Asia Pacific Meningococcal Vaccines Market Trends
The meningococcal vaccines market in the Asia Pacific is expected to register at the fastest CAGR of 7.4% over the forecast period, driven by expanding immunization coverage, rising healthcare expenditure, and increasing awareness of meningococcal disease. Market maturity varies significantly across countries, with developed markets focusing on routine schedules while emerging economies emphasize outbreak control and catch-up vaccination. Private healthcare channels are playing an increasingly important role, particularly in urban centers. Governments are progressively strengthening national immunization programs, creating long-term opportunities. Manufacturers pursue partnerships, tiered pricing, and phased launches to address diverse regulatory and economic environments across the region.
The Japan meningococcal vaccines market is relatively niche but strategically important due to stringent regulatory standards and high clinical expectations. Demand is driven primarily by travel, occupational risk, and specific medical indications rather than routine population-wide programs. Physician awareness and patient willingness to pay influence uptake, particularly through private channels. Market access requires strong safety and quality credentials. While overall volumes are limited, Japan offers value for premium products and serves as a benchmark market for regulatory credibility in the Asia Pacific region.
The meningococcal vaccines market in China is large, volume-driven, and supported by government-led immunization programs and domestic manufacturing capacity. Meningococcal vaccination is well established within public health initiatives, particularly for pediatric populations. Growth is influenced by policy updates, urbanization, and improved access to healthcare. The private segment is gradually expanding in major cities, supporting the uptake of differentiated products. Regulatory reforms and emphasis on vaccine quality are reshaping competitive dynamics. China remains strategically significant due to scale, though pricing and tender-based procurement define commercial outcomes.
Latin America Meningococcal Vaccines Market Trends
The meningococcal vaccines market in Latin America is experiencing moderate to strong growth, driven by public immunization programs and regional disease surveillance initiatives. Governments prioritize meningococcal vaccination in response to outbreak risks, particularly among children and adolescents. Budget constraints influence product selection and the timing of adoption. Private healthcare contributes to incremental demand in urban areas. Market access depends heavily on public tenders and international funding mechanisms. Manufacturers focus on demonstrating epidemiological relevance and affordability to expand their footprint across the region.
The Brazil meningococcal vaccines market held a significant share in Latin America in 2025, supported by a robust national immunization program. Public procurement dominates demand, ensuring high coverage in priority populations. The country has experience with large-scale vaccination campaigns, supporting volume stability. The private sector complements demand through travel and catch-up vaccination. Regulatory oversight and local production capabilities influence competitive positioning. Brazil remains a strategic market due to scale, program continuity, and government commitment to infectious disease prevention.
Middle East & Africa Meningococcal Vaccines Market Trends
The meningococcal vaccines market in the Middle East and Africa exhibits heterogeneous dynamics, shaped by disease burden, healthcare infrastructure, and funding availability. Demand is driven by outbreak prevention, religious travel requirements, and donor-supported immunization programs. Gulf countries show higher adoption through private and public channels, while parts of Africa rely on international partnerships. Market growth is supported by increasing awareness and regional vaccination initiatives. Manufacturers emphasize supply reliability and programmatic suitability to address diverse regional needs.
The Saudi Arabia meningococcal vaccines market is strategically important due to mandatory vaccination requirements for Hajj and Umrah pilgrims. Strong government enforcement ensures consistent demand across residents and international travelers. The healthcare system supports both public and private vaccination delivery. Seasonal vaccination cycles drive predictable procurement patterns. Regulatory standards and centralized purchasing shape market access. Saudi Arabia remains a key Middle Eastern market where public health policy directly translates into sustained vaccine utilization.
Key Meningococcal Vaccines Company Insights
The meningococcal vaccines industry is highly consolidated, with a small group of multinational companies driving innovation, supply, and global access. Leading players compete on portfolio breadth, regulatory approvals across age groups, and reliability in large-scale public procurement. Strategic emphasis is placed on lifecycle management, including expanded indications, improved formulations, and combination vaccines to align with evolving immunization policies.
Strong manufacturing capabilities and long-standing relationships with public health authorities support sustained market leadership. Competitive differentiation is increasingly shaped by the ability to meet diverse programmatic needs across public and private channels while maintaining consistent quality, safety, and supply continuity.
Key Meningococcal Vaccines Companies:
The following key companies have been profiled for this study on the meningococcal vaccines market.
- GlaxoSmithKline
- Pfizer
- Sanofi
- Merck & Co.
- Novartis
- Serum Institute of India
- Walvax Biotechnology
- Baxter International
- Bio-Med Pvt. Ltd.
- Incepta Vaccine Ltd.
Recent Developments
-
In May 2025, the FDA expanded Sanofi’s MenQuadfi approval to include infants as young as six weeks, strengthening early-age meningococcal prevention and supporting growth in the pediatric segment.
-
In April 2025, Gavi supported the rapid deployment of meningococcal vaccines in Nigeria to address outbreak risk, highlighting continued demand for large-scale public health vaccination in emerging markets.
-
In February 2025, the U.S. FDA approved GSK’s pentavalent meningococcal vaccine Penmenvy, enabling single-shot coverage against five major serogroups and supporting simplified immunization strategies for adolescents and young adults.
Meningococcal Vaccines Market Report Scope
Report Attribute
Details
Market size value in 2026
USD 4.37 billion
Revenue forecast in 2033
USD 5.96 billion
Growth rate
CAGR 4.5% from 2026 to 2033
Base year for estimation
2025
Historical data
2021 - 2024
Forecast period
2026 - 2033
Quantitative units
Revenue in USD million/billion, and CAGR from 2026 to 2033
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, trends
Segments covered
Type, brand, serotype, age group, sales channel, region
Regional scope
North America; Europe; Asia Pacific; Latin America; Middle East & Africa
Country scope
U.S.; Canada; Mexico; UK; Germany; France; Italy; Spain; Denmark; Sweden; Norway; China; Japan; India; Australia; South Korea; Thailand; Brazil; Argentina; Saudi Arabia; South Africa; UAE; Kuwait
Key company profiled
GlaxoSmithKline; Pfizer; Sanofi; Merck & Co.; Novartis; Serum Institute of India; Walvax Biotechnology; Baxter International; Bio-Med Pvt. Ltd.; Incepta Vaccine Ltd.
Customization scope
Free report customization (equivalent up to 8 analysts working days) with purchase. Addition or alteration to country, regional & segment scope.
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
Global Meningococcal Vaccines Market Report Segmentation
This report forecasts revenue growth at the global, regional, and country levels and provides an analysis of the latest industry trends across sub-segments from 2021 to 2033. For this study, Grand View Research has segmented the global meningococcal vaccines market report based on type, brand, serotype, age group, sales channel, and region:
-
Type Outlook (Revenue, USD Million, 2021 - 2033)
-
Bivalent
-
Quadrivalent
-
Others
-
-
Brand Outlook (Revenue, USD Million, 2021 - 2033)
-
Menactra
-
Menveo
-
Nimenrix
-
Trumenba
-
Bexsero
-
Others
-
-
Serotype Outlook (Revenue, USD Million, 2021 - 2033)
-
A
-
B
-
C
-
W-135
-
Y
-
-
Age Group Outlook (Revenue, USD Million, 2021 - 2033)
-
Infants (0 to 2 years)
-
Children and Adults (2 years and above)
-
-
Sales Channel Outlook (Revenue, USD Million, 2021 - 2033)
-
Private
-
Public
-
-
Regional Outlook (Revenue, USD Million, 2021 - 2033)
-
North America
-
U.S.
-
Canada
-
Mexico
-
-
Europe
-
UK
-
Germany
-
France
-
Italy
-
Spain
-
Denmark
-
Sweden
-
Norway
-
-
Asia Pacific
-
Japan
-
China
-
India
-
Australia
-
South Korea
-
Thailand
-
-
Latin America
-
Brazil
-
Argentina
-
-
Middle East & Africa
-
South Africa
-
Saudi Arabia
-
UAE
-
Kuwait
-
-
Frequently Asked Questions About This Report
The global meningococcal vaccine market size was estimated at USD 4.05 billion in 2025 and is expected to reach USD 4.37 billion in 2026.
The global meningococcal vaccine market is expected to grow at a compound annual growth rate of 4.5% from 2025 to 2033 and is expected to reach USD 5.96 billion by 2033.
Some key players operating in the meningococcal vaccine market include GlaxoSmithKline plc, Pfizer Inc, Merck & co., Inc., and Serum Institute of India Ltd. among others.
Increasing prevalence of meningitis, increasing R&D activities to develop novel vaccines, and increasing government initiatives to reduce disease burden are the major factors driving the meningococcal vaccine market growth over the forecast period.
The bivalent segment dominated the market with the largest revenue share of 80.30% in 2025 and is expected to be the fastest growth over the forecast period. due to its long-standing clinical acceptance, streamlined dosing, and continued use in routine immunization and outbreak-response settings, particularly in cost-sensitive and public procurement–driven markets
North America held the largest share of 55.16% in 2025 and is expected to register a lucrative growth rate over the forecast period. It is attributable to the favorable recommendations & support from governments and well-established healthcare infrastructure in the region.
About the authors:
Author: GVR Pharmaceuticals Research Team | Last Updated:
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.









