- Home
- »
- Clinical Diagnostics
- »
-
Middle East Liquid Biopsy Market Size, Industry Report, 2033GVR Report cover
![Middle East Liquid Biopsy Market Size, Share & Trends Report]()
Middle East Liquid Biopsy Market (2025 - 2033) Size, Share & Trends Analysis Report By Biomarker, By Technology, By Sample Type, By Application, By Clinical Application, By End Use, By Product, By Region, And Segment Forecasts
- Report ID: GVR-4-68040-825-7
- Number of Report Pages: 150
- Format: PDF
- Historical Range: 2021 - 2023
- Forecast Period: 2025 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
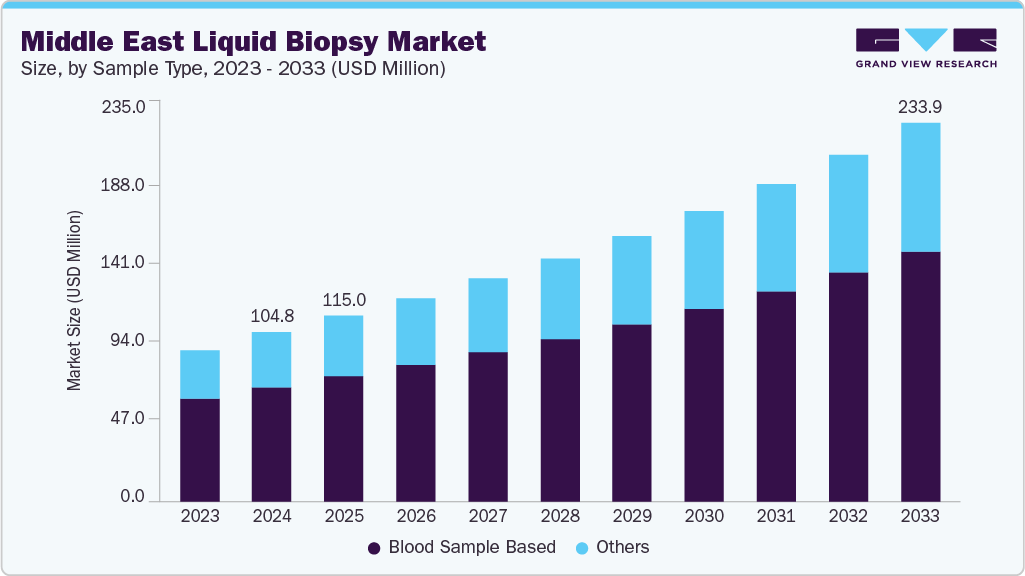
Middle East Liquid Biopsy Market Summary
The Middle East liquid biopsy market size was valued at USD 104.76 million in 2024 and is projected to reach USD 233.95 billion by 2033, growing at a CAGR of 9.28% from 2025 to 2033. The market is driven by increased emphasis on early detection and population-level screening initiatives across the Middle East, fueled by rising cancer prevalence.
Key Market Trends & Insights
- By product, the instruments segment led the liquid biopsy market in 2024 with a share of 46.74%
- By sample type, the blood sample segment led the market with the largest revenue share of 67.59% in 2024
- By technology, the multi-gene parallel analysis (NGS) segment represented the largest share of 79.02% in 2024
- By biomarker, the circulating nucleic acids segment led the liquid biopsy market in 2024 with a share of 35.99%
Market Size & Forecast
- 2024 Market Size: USD 104.76 million
- 2033 Projected Market Size: USD 233.95 million
- CAGR (2025-2033): 9.28%
For example, in December 2024, Abu Dhabi launched a new blood-based colon cancer test as part of a proactive strategy to improve early detection and survival rates. The program addresses a significant gap in early diagnosis.In addition, according to data from 2023, 67% of cases of colorectal cancer in Abu Dhabi were discovered at a late stage, which limited treatment options and decreased survival rates. The program, which offers a non-invasive alternative to traditional screening methods, is expected to enhance patient participation and support the UAE broader efforts to improve early detection and outcomes. The market for non-invasive early detection technologies is growing beyond advanced-stage cancer, and the Middle East liquid biopsy market is being boosted by new blood-based screening programs for colorectal and lung cancer.
Rising government and health-system adoption, supported by national pilot programs, is fueling market growth by encouraging wider testing and strengthening healthcare readiness. For instance, in June 2025, M42 entered into a strategic partnership with AstraZeneca and SOPHiA GENETICS to bring advanced liquid biopsy testing to the UAE, supporting cancer diagnosis, guiding treatment choices, and enabling quicker and more accurate biomarker analysis across various cancer types. Through this collaboration, M42 adopted SOPHiA GENETICS’ MSK-ACCESS, powered by the SOPHiA DDM platform, within its testing network, allowing for non-invasive genomic profiling using a simple blood sample.
Government-supported screening initiatives and hospital-led validation studies, such as evaluation by Cleveland Clinic Abu Dhabi and the Abu Dhabi colorectal cancer program, are accelerating early clinical adoption, increasing in test reliability, and moving reimbursement awareness forward throughout the region. The new testing initiative will initially focus on lung, ovarian, breast, colorectal, and pancreatic cancers, among others, addressing some of the region's most pressing cancer issues. Over the next year, the partnership will expand access to liquid biopsy testing to selected major hospitals in the UAE, providing patients with faster and less invasive biomarker insights than traditional tissue biopsies, while also increasing the country's testing capacity and cancer diagnostic capabilities.
Moreover, regional pharmaceutical and medical groups are entering exclusive distribution and partnership agreements that expand commercial availability and strengthen regional laboratory services. For instance, in June 2022, Imagia Canexia Health (Canada) signed a Memorandum of Understanding (MOU) with the Kingdom of Saudi Arabia to distribute its liquid biopsy testing products throughout Saudi Arabia and Middle East region. The company offers liquid biopsy solutions for therapy selection and patient monitoring that can be performed at or near the point of care, eliminating the need to send samples to international centralized laboratories. This approach allows cancer patients all across the region to access advanced precision oncology treatments at a lower cost and faster turnaround time. Such collaborations remove key access barriers, standardize test delivery across local markets, and accelerate the liquid biopsy market's growth.
Furthermore, growing strategic partnerships and commercial rollouts are fueling the Middle East liquid biopsy market, enabling global companies to work closely with regional clinical stakeholders. For instance, in January 2024, Hikma Pharmaceuticals PLC (UK) entered into an agreement with Guardant Health, Inc. (US) for commercialization and marketing of Guardant Health’s portfolio of liquid and tissue biopsy tests for cancer screening, recurrence monitoring and tumor mutation profiling across all solid cancers in the Middle East. In addition, the portfolio features tests such as Shield for early cancer screening, Guardant Reveal for detecting minimal residual disease and tracking recurrence, and Guardant360 along with Guardant360 TissueNext for comprehensive genomic profiling (CGP) across all solid tumors. These alliances help to launch and expand liquid biopsy products across the Middle East, improving accessibility, streamlining distribution, and significantly accelerating market entry and scale-up.
Market Concentration & Characteristics
The Middle East liquid biopsy market is undergoing a transformation with the integration of cutting-edge AI and cloud-based genomics. For instance, in June 2025, M42 collaborated with AstraZeneca and SOPHiA GENETICS to integrate SOPHiA’s MSK-ACCESS platform, powered by SOPHiA DDM (a cloud-based analytics platform), into the UAE clinical infrastructure. This enables scalable, non-invasive genomic profiling and real-time disease monitoring using blood samples. Meanwhile, in May 2024, ONCOHOST stated that AI-driven analytics will improve predictive biomarker interpretation and therapy response modeling, significantly burgeoning the sophistication and clinical utility of liquid biopsy assays across the Middle East region.
Strategic M&A is increasingly influencing the Middle East's liquid biopsy market. To increase innovation and scale, international precision oncology companies are collaborating with or purchasing local pharmaceutical and diagnostic shares. These transactions connect local networks to top-notch liquid biopsy capabilities, facilitate technology transfer, and improve lab infrastructure. By improving distribution, making advanced genomic testing more available, and helping to build lasting diagnostic systems in the region, this merging of companies promotes long-term growth through shared research and development.
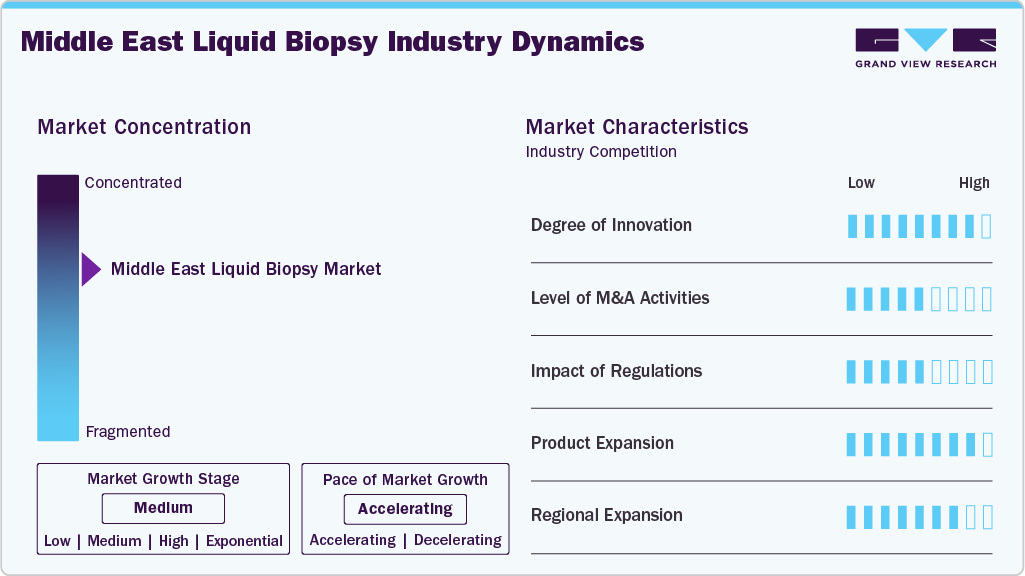
The Middle East liquid biopsy market is significantly shaped by regulatory dynamics. Health authorities are tightening regulations, quality standards, and approval processes for cutting-edge diagnostic technologies as countries in the region place a high priority on precision medicine. Enhanced regulatory clarity promotes the adoption of validated tests by healthcare providers, increases confidence in test accuracy and reliability, and facilitates the integration of liquid biopsy into standard clinical practice. By ensuring that new technologies meet established standards and can be implemented safely and effectively across hospitals and laboratories, this changing regulatory environment helps accelerate market growth.
The growing need for more comprehensive and clinically actionable tests is driving the rapid expansion of liquid biopsy product lines across the Middle East. Companies are introducing highly sensitive minimal residual disease (MRD) tests, multi-cancer early detection panels, and improved ctDNA assays. AI-powered interpretation and automated workflows are other innovations that improve turnaround, lower error rates, and enable broader use in hospitals. This broadening of the test menu from early detection to full genomic profiling enables oncologists to tailor diagnostics to a broader spectrum of cancers, thereby increasing the clinical reach and utility of liquid biopsy in the region.
Regional expansion is gaining strong momentum as major global players strengthen their presence in the Middle East to deliver advanced liquid biopsy services. Companies are increasingly collaborating with regional distributors, hospital networks, and diagnostic providers to accelerate market penetration. For instance, in January 2024, Guardant Health, Inc. and Hikma Pharmaceuticals, Inc. announced a strategic partnership to introduce Guardant’s portfolio of cancer screening and genomic profiling tests including Shield, Guardant Reveal, and Guardant360 across multiple Middle Eastern markets. This collaboration not only enhances access to cutting-edge liquid biopsy technologies but also supports the establishment of regional testing infrastructure, thereby advancing precision oncology adoption throughout the region.
Product Insights
The instruments segment led the liquid biopsy market in 2024 with a share of 46.74%, owing to the increasing installation of advanced NGS and PCR platforms, increased adoption of automated workflow systems, and higher expenditures by hospitals and laboratories in modern diagnostic infrastructure. Rising demand for high-throughput analysis, increased precision in cancer detection, and the need for dependable equipment to support large testing volumes have all contributed to this segment's dominance. Furthermore, expanding national screening programs and regional partnerships encouraged healthcare facilities to upgrade their equipment, which contributed to the instruments' strong market share.
In addition, the instruments segment is projected to show the fastest growth over the forecast period in the liquid biopsy market. The rapid development of high-sensitivity sequencing platforms, the increased adoption of automated sample-processing systems, and the growing demand for multi-cancer early detection technologies are all key drivers. Growth is also aided by ongoing improvements in ctDNA, exosome, and CTC isolation instruments, which enable higher throughput and greater analytical accuracy. Furthermore, diagnostic manufacturers' investments in next-generation microfluidics and integrated workflows are hastening instrument upgrades in research laboratories and clinical testing facilities.
Sample Type Insights
The blood sample segment led the market with the largest revenue share of 67.59% in 2024. This dominance is primarily fueled by the widespread clinical use of blood-based biopsy tests for detecting CTC, circulating nucleic acids, and other cancer biomarkers. Blood collection is minimally invasive, highly standardized, and compatible with all major liquid biopsy technologies, making it the preferred sample type across oncology centers in the region. For instance, in early 2025, Abu Dhabi Public Health Center launched a blood-based colorectal cancer screening program using Guardant Health’s Shield test, improving early detection via a non-invasive method. In addition, strong adoption of blood-based assays for therapy selection, treatment monitoring, and recurrence tracking continues to reinforce its leading position.
The others segment is expected to grow the fastest, due to increasing interest in non-blood biofluids such as urine and saliva, especially for urologic and head-& neck cancers, is accelerating adoption. These sample types offer improved patient comfort, lower collection complexity, and the potential for frequent, at-home or outpatient monitoring. Growing research collaborations in the Middle East focusing on exosomal biomarkers, cell-free RNA, and tumor-derived vesicles in alternative fluids is further encouraging development. As precision oncology expands beyond traditional blood-based assays, demand for diversified sample types is set to rise rapidly.
Technology Insights
The multi-gene parallel analysis (NGS) segment represented the largest share of 79.02% in 2024, driven by the region’s accelerated transition toward comprehensive genomic profiling to support precision oncology initiatives. NGS-based liquid biopsy platforms enable simultaneous detection of multiple mutations, gene fusions, copy-number variations, and therapy-resistance markers from a single blood sample, making them highly valuable for both treatment selection and disease monitoring. Major cancer centers across the UAE, Saudi Arabia, and Israel are increasingly integrating NGS into routine clinical workflows as national precision medicine programs scale. Its high analytical sensitivity, broad biomarker coverage, and proven ability to inform targeted therapies and immunotherapies have solidified NGS as the preferred modality for advanced molecular diagnostics in the region’s oncology ecosystem.
The multi-gene parallel analysis (NGS) segment is expected to show the fastest growth during the forecast period, owing to region's increasing adoption of comprehensive genomic profiling, rising demand for early cancer detection, and expanding precision oncology programs across major healthcare systems. Growth is also aided by the establishment of advanced sequencing facilities, government-led investment in cancer genomics, and a preference for NGS panels that detect multiple biomarkers in a single test, improving clinical decision-making and treatment personalization.
Biomarker Insights
The circulating nucleic acids segment led the liquid biopsy market in 2024 with a share of 35.99%, driven by the accelerated installation of advanced NGS and PCR platforms across major hospitals and diagnostic laboratories. The growing adoption of automated workflow systems ranging from high-throughput sequencers to integrated sample preparation units has significantly improved operational efficiency and reduced manual variability, prompting greater investment in state-of-the-art diagnostic infrastructure. Rising demand for accurate, high-volume cancer testing and the need for reliable equipment capable of supporting expanding clinical workloads further reinforced the segment’s dominance. Moreover, national screening initiatives, government-funded diagnostic modernization programs, and strategic regional partnerships encouraged healthcare facilities to upgrade and standardize their technology, contributing to the strong and sustained market share of the segment.
Exosomes/Microvesicles segment is expected to show the fastest CAGR during the forecast period, owing to their rich molecular cargo (RNAs, proteins, and lipids) and stability in body fluids. According to the PubMed Central studies, exosomes/microvesicles can carry tumor-specific lipids and miRNAs, which hold strong promises for early detection and prognosis. In addition, BioMed Central reported that biopsies based on this biomarker are gaining traction in immuno-oncology: they can reflect immune status and treatment response via their cargo. Furthermore, a Saudi Arabia study profiling circular RNAs within extracellular vesicles linked to triple-negative breast cancer further underscores the region’s growing potential for developing exosomes/microvesicles based diagnostic applications.
Application Insights
The cancer segment accounted for the largest share, 86.32%, of the Middle East liquid biopsy market in 2024. This dominance is driven by the high regional burden of cancers such as lung, breast, colorectal, and prostate, which continue to increase demand for noninvasive diagnostic and monitoring tools. Meanwhile, the PubMed Central article published a study in May 2025 that prostate cancer incidence is rising with nearly 51,000 new cases reported across the region in 2022 and projections indicating further increase by 2050. These epidemiological trends are fueling demand for noninvasive, blood-based biopsy assays for cancer diagnosis, treatment monitoring, and recurrence tracking.
The reproductive health segment is expected to show the fastest growth during the forecast period, driven by rising demand for non-invasive prenatal testing (NIPT), carrier screening, and early fetal genetic assessment across Middle East countries. Increasing maternal age, greater awareness of hereditary and chromosomal risks, and wider availability of advanced prenatal diagnostics are accelerating market growth. Private fertility centers, IVF clinics, and specialized women’s health facilities are also integrating liquid biopsy–based genetic screening to enable safer, earlier, and more accurate evaluation of fetal health. In parallel, government-led maternal health initiatives and expanding insurance coverage for prenatal genetic tests are improving accessibility. As reproductive genomics becomes a key focus area within regional healthcare strategies, this segment is poised for strong, sustained, and long-term growth.
Clinical Application Insights
The therapy selection segment led the market with the largest revenue share of 33.69% in 2024, owing to rising focus on precision oncology, where ctDNA-based profiling plays a critical role in identifying actionable mutations and guiding targeted treatment decisions. Hospitals and cancer centers across the UAE, Saudi Arabia, and Qatar are increasingly adopting NGS-enabled liquid biopsy assays to support real-time therapy planning, particularly for lung, breast, colorectal, and prostate cancers. The technology’s ability to capture tumor heterogeneity, monitor emerging resistance mutations, and reduce the need for invasive tissue biopsies has made it a preferred clinical tool. As oncologists prioritize personalized, molecularly informed treatment strategies, liquid biopsy continues to strengthen its dominant position in therapy selection across the region.
Furthermore, the early cancer screening segment is expected to grow the fastest, driven by rising government-led screening initiatives and increasing adoption of non-invasive tests for early detection of high-burden cancers in the region. Growing awareness of colorectal, breast, and lung cancer prevalence has prompted healthcare systems in the Middle East to explore blood-based screening tools that enable earlier diagnosis with minimal patient discomfort. Pilot programs and hospital-level evaluations across the UAE, Saudi Arabia, and other GCC countries are accelerating acceptance of liquid biopsy for routine screening. As the region prioritizes early detection to improve survival outcomes, demand for screening-focused liquid biopsy solutions is expanding rapidly.
End Use Insights
The hospitals and laboratories segment led the market with the largest revenue share of 42.44% in 2024, supported by their advanced diagnostic infrastructure, higher patient volume, and rapid adoption of precision oncology tools. Major regional institutions are integrating liquid biopsy into routine cancer care, strengthening this segment’s dominance. For instance, in November 2023, King Faisal Specialist Hospital & Research Centre (KFSHRC) in Saudi Arabia recently adopted cutting-edge liquid biopsy technology capable of analyzing around 50 cancer-related genes and more than 3,000 clinically relevant mutations from a single 10 mL blood sample. The hospital also runs large-scale research projects to validate these technologies for therapy selection, monitoring, and early detection. Similar initiatives at top-tier oncology centers in the UAE and Qatar reinforce hospital’s and laboratorie’s roles as primary users of liquid biopsy solutions throughout the region.
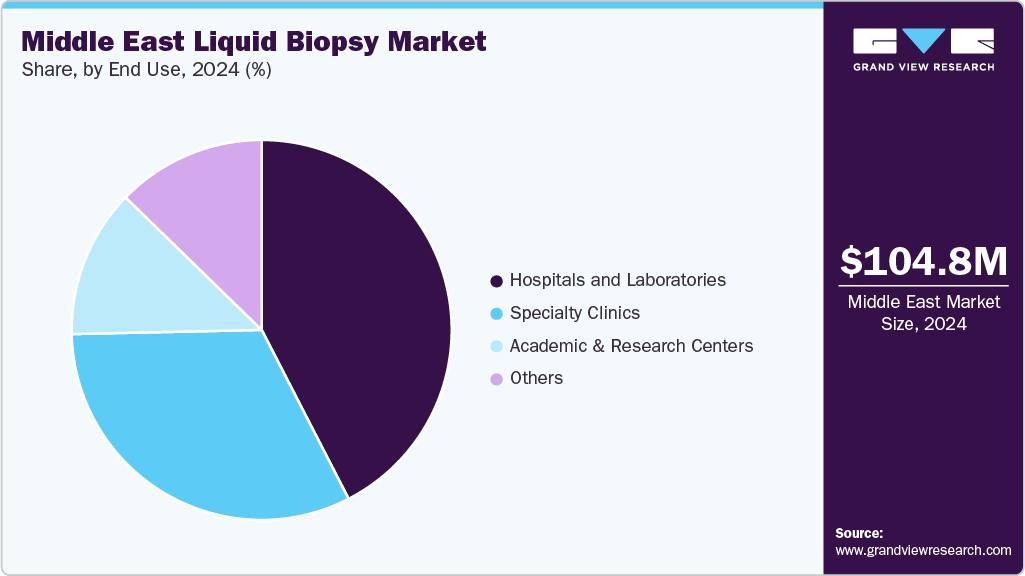
The specialty clinics segment is expected to expand at a significant CAGR of 9.89% owing to rising demand for personalized oncology care and a growing shift of cancer patients to specialized, high-quality diagnostic centers. Specialty clinics are increasingly using liquid biopsy for early detection, therapy selection, and real-time treatment monitoring, enhancing their role in precision oncology. The rapid expansion of private cancer clinics, especially in the UAE and Saudi Arabia, is increasing access to advanced genomic services. Many of these clinics are implementing next-generation sequencing-based liquid biopsy panels, which allow clinicians to detect multiple biomarkers with a single blood draw.
Country Insights
The liquid biopsy market in the Middle East is expanding rapidly, driven by advanced technology, growing clinical evidence, and strong global company investments. Cutting-edge platforms that combine AI, cloud-based bioinformatics, and highly sensitive ctDNA assays are improving detection accuracy and scalability. Academic and research institutions are also contributing, with more publications and real-world data validating liquid biopsy for cancer and prenatal diagnostics across the region. Moreover, global diagnostics firms are scaling up their Middle East presence, fostering the growth of the market. For instance, in May 2025, GC Genome (South Korea) entered into an agreement with UAE for supply agreement for its AI-enhanced non-invasive prenatal test G-NIPT. This regional entry not only raises awareness but also strengthens local access to advanced genomic testing, fueling market growth.
Saudi Arabia Liquid Biopsy Market Trends
The Saudi Arabia liquid biopsy market is experiencing strong growth, supported by increasing investment in precision oncology, rising cancer incidence, and rapid adoption of minimally invasive diagnostic technologies. Early detection and personalized treatment are being implemented as part of national health transformation programs, driving up demand for ctDNA, CTC, and multi-omics blood tests. Advancements in AI-powered analytics and integrated bioinformatics platforms improve test accuracy and clinical decision-making, encouraging widespread adoption across hospitals and specialty centers. International companies that enter Saudi Arabia through strategic collaborations helps to expand the market. For instance, in October 2024, Helio Genomics partnered with Halub Medical Co. to commercialize its AI-enabled liver cancer test, HelioLiver Dx, across Saudi Arabia.
UAE Liquid Biopsy Market Trends
The UAE liquid biopsy market is driven by the country’s rapid shift toward precision medicine, high cancer prevalence, and strong government support for advanced molecular diagnostics. The growing use of non-invasive testing for therapy selection, treatment monitoring, and early cancer detection is driving demand in major hospitals and cancer centers. The UAE's robust digital health infrastructure and investment in AI-powered genomics platforms are increasing the clinical adoption of liquid biopsy for real-time, personalized decision-making. Growing collaborations between global diagnostics companies and UAE healthcare providers, as well as increased availability of NGS and ctDNA-based assays, are driving market growth. Furthermore, the UAE's readiness to deploy next-generation liquid biopsy technologies is being enhanced by strategic initiatives such as the UAE National Biotechnology Strategy, which promote medical innovation.
Kuwait Liquid Biopsy Market Trends
The Kuwait liquid biopsy market is growing due to a rising cancer burden, increasing adoption of precision oncology, and the shift toward minimally invasive diagnostic solutions. The country continues to witness a notable increase in cancer incidence, creating strong demand for advanced early detection and monitoring tools. According to Global Cancer Observatory 2022, 4,347 new cancer cases in 2022 in Kuwait, with breast cancer leading at 892 cases (20.5%), followed by colorectal cancer with 436 cases (10%), prostate cancer with 274 cases (6.3%), non-Hodgkin lymphoma with 258 cases (5.9%), and thyroid cancer with 251 cases (5.8%). This rising prevalence is accelerating the adoption of liquid biopsy for genetic profiling, therapy selection, and recurrence monitoring. In addition, growing investments in molecular diagnostics and collaborations between global test developers and local healthcare institutions are strengthening market expansion across Kuwait.
Oman Liquid Biopsy Market Trends
The Oman liquid biopsy market is expanding as the country accelerates its shift toward advanced diagnostics, precision medicine, and technology-driven healthcare. Supported by Oman Vision 2040, which prioritizes digital transformation, innovation, and high-quality clinical services, healthcare facilities are increasingly incorporating molecular and genomic tools into routine practice. The government’s emphasis on strengthening research ecosystems, enhancing laboratory capabilities, and integrating AI-enabled platforms is creating a favorable environment for liquid biopsy adoption. Moreover, growing collaboration between Oman healthcare institutions and global diagnostics companies is improving access to ctDNA-based tests, minimally invasive monitoring solutions, and next-generation biomarker platforms. As hospitals modernize their diagnostic infrastructure and adopt cloud-based reporting systems, the adoption of liquid biopsy technologies across oncology and specialty care is expected to accelerate steadily.
Qatar Liquid Biopsy Market Trends
The Qatar liquid biopsy market is growing due to the country’s strong national commitment to becoming a regional leader in genomic medicine, supported by large-scale precision health programs such as the Qatar Genome Programme (QGP) and investments through the Qatar Foundation. These initiatives emphasize early detection, personalized therapy, and population-level genomic mapping, creating a favorable environment for liquid biopsy adoption. Major global players including F. Hoffmann-La Roche Ltd, Guardant Health, Inc., Thermo Fisher Scientific, Inc., and Illumina, Inc. are collaborating with Qatari research centers and hospital laboratories to provide advanced ctDNA assays, NGS platforms, and oncology-focused liquid biopsy tests. Their involvement supports clinical validation studies, strengthens local analytical capacity, and accelerates technology transfer. By offering training, cloud-based analytics, and high-throughput solutions, these companies play a critical role in enhancing Qatar’s diagnostic ecosystem and driving broader uptake of liquid biopsy technologies.
Key Middle East Liquid Biopsy Company Insights
The liquid biopsy market features several influential players that are shaping innovation, clinical adoption, and technological advancement across the region. Key companies include CelLBxHealth, Oncimmune Holdings PLC, Guardant Health, Myriad Genetics, F. Hoffmann-La Roche, QIAGEN, Illumina, Thermo Fisher Scientific, Epigenomics AG, and NeoGenomics Laboratories, all of which contribute significantly through advanced platforms, assay development, and oncology-focused solutions. These firms continue to expand their presence through product enhancements, AI-enabled analytics, multi-gene NGS panels, and improved biomarker detection technologies. In addition, they are strengthening their market position through strategic partnerships, regional collaborations, distributor agreements, and clinical validation programs with major hospitals and research institutions. Their ongoing investments in R&D, infrastructure, and precision oncology workflows collectively accelerate adoption and support the rapid evolution of the liquid biopsy landscape.
Key Middle East Liquid Biopsy Companies:
- CelLBxHealth
- Oncimmune Holdings PLC
- Guardant Health, Inc.
- Myriad Genetics, Inc.
- F. Hoffmann-La Roche Ltd
- QIAGEN
- Illumina, Inc.
- Thermo Fisher Scientific, Inc.
- Epigenomics AG
- NeoGenomics Laboratories.
Recent Developments
-
In June 2025, M42, AstraZeneca, and SOPHiA GENETICS launched a UAE initiative to deploy SOPHiA’s DDM-powered testing infrastructure. These entries promote regional competition, raise awareness of precision oncology, and increase infrastructure investment across the Middle East.
-
In January 2025, Senseera secured USD7.1 million in seed funding led by Lightspeed Venture Partners, with support from I-Next Capital and the Lowy Medical Research Institute. The investment will accelerate development of its GEM BIOMARKERS liquid biopsy platform, which uses cfChIP-seq technology to decode cell-specific gene expression and cell states from a simple blood sample. This approach delivers biopsy-level molecular insights through a non-invasive test, enhancing disease diagnosis and monitoring.
Middle East Liquid Biopsy Market Report Scope
Report Attribute
Details
Market size value in 2025
USD 115.01 million
Revenue forecast in 2033
USD 233.95 million
Growth rate
CAGR of 9.28% from 2025 to 2033
Base year for estimation
2024
Historical data
2021 - 2023
Forecast period
2025 - 2033
Quantitative units
Revenue in USD Billion/Million and CAGR from 2025 to 2033
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Product, sample type, technology, biomarker, application, clinical application, end use, country
Key companies profiled
CelLBxHealth; Oncimmune Holdings PLC; Guardant Health, Inc.; Myriad Genetics, Inc.; F. Hoffmann-La Roche Ltd; QIAGEN; Illumina, Inc.; Thermo Fisher Scientific, Inc.; Epigenomics AG; NeoGenomics Laboratories
Customization scope
Free report customization (equivalent up to 8 analysts’ working days) with purchase. Addition or alteration to country, country & segment scope
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
Middle East Liquid Biopsy Market Report Segmentation
This report forecasts revenue growth and provides an analysis of the latest trends in each of the sub-segments from 2021 to 2033. For the purpose of this report, Grand View Research has segmented the liquid biopsy market on the basis product, sample type, technology, biomarker, application, clinical application, end use, and country.
-
Product Outlook (Revenue, USD Million, 2021 - 2033)
-
Instruments
-
Consumables Kits and Reagents
-
Software and Services
-
-
Sample Type Outlook (Revenue, USD Million, 2021 - 2033)
-
Blood Sample
-
Others
-
-
Technology Outlook (Revenue, USD Million, 2021 - 2033)
-
Multi-gene parallel Analysis (NGS)
-
Single Gene Analysis (PCR Microarrays)
-
-
Biomarker Outlook (Revenue, USD Million, 2021 - 2033)
-
Circulating Nucleic Acids
-
CTC
-
Exosomes/Microvesicles
-
Circulating Proteins
-
-
Application Outlook (Revenue, USD Million, 2021 - 2033)
-
Cancer
-
Lung Cancer
-
Prostate Cancer
-
Breast Cancer
-
Colorectal Cancer
-
Leukemia
-
Gastrointestinal Cancer
-
Others
-
-
Reproductive Health
-
Others
-
-
Clinical Application Outlook (Revenue, USD Million, 2021 - 2033)
-
Therapy Selection
-
Treatment Monitoring
-
Early Cancer Screening
-
Recurrence Monitoring
-
Others
-
-
End Use Outlook (Revenue, USD Million, 2021 - 2033)
-
Hospitals and Laboratories
-
Specialty Clinics
-
Academic and Research Centers
-
Others
-
-
Country Outlook (Revenue, USD Million, 2021- 2033)
-
Saudi Arabia
-
UAE
-
Kuwait
-
Oman
-
Qatar
- Rest of Middle East
-
Frequently Asked Questions About This Report
b. The Middle East liquid biopsy market size was estimated at USD 104.6 million in 2024 and is expected to reach USD 115.01 million in 2025.
b. The Middle East liquid biopsy market is expected to grow at a compound annual growth rate of 9.28% from 2025 to 2033 to reach USD 233.95 million by 2033.
b. The instrument segment led the market in 2024 with a share of 46.74%, owing to the increasing installation of advanced NGS and PCR platforms, increased adoption of automated workflow systems, and higher expenditures by hospitals and laboratories in modern diagnostic infrastructure
b. Some key players operating in the point-of-care influenza diagnostics market include CelLBxHealth, Oncimmune Holdings PLC, Guardant Health, Inc., Myriad Genetics, Inc., F. Hoffmann-La Roche Ltd, QIAGEN, Illumina, Inc., Thermo Fisher Scientific, Inc., Epigenomics AG, and NeoGenomics Laboratories.
b. Key factors that are driving the market growth include factor such as the increased emphasis on early detection and population-level screening initiatives across the Middle East, fueled by rising cancer prevalence. For example, in December 2024, Abu Dhabi launched a new blood-based colon cancer test as part of a proactive strategy to improve early detection and survival rates
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










