- Home
- »
- Medical Devices
- »
-
Middle East Small Molecule Contract Manufacturing Market Report 2033GVR Report cover
![Middle East Small Molecule Contract Manufacturing Market Size, Share & Trends Report]()
Middle East Small Molecule Contract Manufacturing Market (2025 - 2033) Size, Share & Trends Analysis Report By Type (Clinical, Commercial), By Service, By Therapeutic Area (Oncology, Infectious Diseases), By End-use, By Country, And Segment Forecasts
- Report ID: GVR-4-68040-827-8
- Number of Report Pages: 180
- Format: PDF
- Historical Range: 2021 - 2024
- Forecast Period: 2025 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
Middle East Small Molecule Contract Manufacturing Market Summary
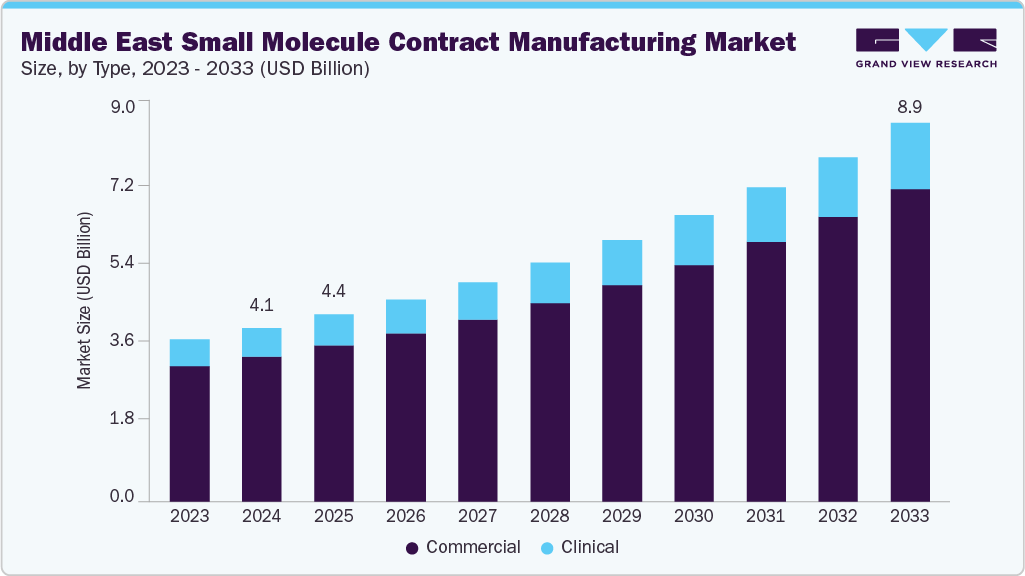
The Middle East small molecule contract manufacturing market size was estimated at USD 4.07 billion in 2024 and is projected to reach USD 8.90 billion by 2033, growing at a CAGR of 9.24% from 2025 to 2033. The market growth is driven by increasing prevalence of chronic diseases, growing emphasis on R&D in the Middle East’s pharmaceutical sector, expanding costs for establishing and maintaining in-house manufacturing facilities and stringent regulatory requirements.
Key Market Trends & Insights
- Saudi Arabia small molecule contract manufacturing industry held the largest share of 34.13% of the market in 2024.
- The small molecule contract manufacturing industry in the UAE is expected to grow lucratively over the forecast period.
- By type, the commercial segment held the largest market share of 83.29% in 2024.
- By service, the API manufacturing segment held the largest market share in 2024.
- By therapeutic area, the oncology segment held the largest revenue share in 2024.
Market Size & Forecast
- 2024 Market Size: USD 4.07 Billion
- 2033 Projected Market Size: USD 8.90 Billion
- CAGR (2025-2033): 9.24%
- Saudi Arabia: Largest market in 2024
- UAE: Fastest growing market
Some other factors contributing to segment growth are rising government initiatives to localize pharmaceutical production, growing generic drug development, and increasing partnerships among regional manufacturers and global CDMOs. In addition, the growing expansion of GMP-compliant facilities, improved logistics infrastructure, and strong investment from Gulf countries in building advanced pharmaceutical clusters are expected to enable faster, cost-effective, and high-quality production of small molecules. Such factors are expected to drive the market growth over the estimated time period.The increasing prevalence of chronic diseases such as diabetes, cardiovascular disorders, and cancer in the Middle East has caused a surge in demand for pharmaceutical products. A study published in August 2025 highlighted that diabetes mellitus is the most common chronic condition in Saudi Arabia, ranking first in three out of five studies reporting disease-specific patterns. Overall, multimorbidity prevalence ranged from 7.1% to 77.6%, with women, particularly older adults, experiencing notably higher rates, underscoring the growing burden of chronic diseases in the population. The rising disease conditions are putting pressure on domestic pharmaceutical companies to scale up production, which in turn is contributing to the outsourcing of manufacturing to contract manufacturing organizations. Thus, the small molecule contract manufacturing market is experiencing growth as companies utilize specialized expertise, advanced facilities, and cost efficiencies offered by contract manufacturing to meet the growing patient needs and ensure the timely availability of essential medications across the region.
In addition, the growing emphasis on R&D in the Middle East’s pharmaceutical sector has caused companies to seek partnerships with specialized contract manufacturers. Besides, outsourcing small molecule synthesis and formulation enables companies to accelerate drug development timelines, access advanced technologies, and utilize scientific expertise without significant capital investment. For instance, in July 2025, Saudi Arabia invited Indian pharmaceutical and medical device contract manufacturing companies to explore strategic partnerships and acquisitions, promoting the localization of APIs and small molecule manufacturing to enhance healthcare resilience, reduce import dependency, and support the objectives of Vision 2030. This increased collaboration with contract manufacturing drives the growth of the small molecule contract manufacturing industry, enabling companies to bring innovative therapies to market more quickly, reduce time-to-market, and maintain competitiveness in an increasingly dynamic and regulated pharmaceutical environment.
Furthermore, the increasing costs of establishing and maintaining in-house manufacturing facilities have led pharmaceutical companies to seek more efficient alternatives. By outsourcing small molecule production to contract manufacturers, companies reduce capital expenditures, operational risks, and compliance burdens. This shift allows companies to focus on R&D and commercialization while benefiting from the economies of scale and advanced technological capabilities of contract manufacturing units. As a result, the demand for small molecule contract manufacturing rises, enabling faster production cycles, improved product quality, and enhanced competitiveness in the regional pharmaceutical market.
Middle East region has increasingly stringent regulatory requirements for pharmaceutical manufacturing, including adherence to GMP, ISO, and regional health authority guidelines. This has caused domestic companies to face challenges in maintaining compliance in-house, prompting them to outsource production to specialized contract manufacturers. Moreover, the contract manufacturing units offer expertise in navigating complex regulatory frameworks, ensuring quality control, documentation, and timely approvals. Thus, most pharmaceutical companies are focusing on reducing compliance risks, avoiding production delays, and driving innovation, which further propels the small molecule contract manufacturing industry across the region.
Market Opportunities
The Middle East small molecule contract manufacturing industry presents significant growth opportunities driven by increasing pharmaceutical product requirements, the rising prevalence of chronic diseases, and growing regional efforts to enhance healthcare self-sufficiency. Furthermore, government bodies are promoting the local production of Active Pharmaceutical Ingredients (APIs), Key Starting Materials (KSMs), and finished small-molecule drugs to reduce import dependency and enhance healthcare resilience.
Furthermore, strategic partnerships with international pharmaceutical companies offer opportunities for technology transfer, joint ventures, and capacity expansion. In addition, growing investment in modern manufacturing facilities, regulatory support, and favorable free zone policies attract foreign players. The expanding demand for generic and specialty small molecule drugs, combined with initiatives under Vision 2030 and similar programs in neighboring countries, further reinforces opportunities for contract manufacturers to serve both regional and export markets.
Technology Landscape
The Middle East small molecule contract manufacturing industry is witnessing significant technological advancements. Continuous manufacturing is replacing traditional batch processes, enhancing efficiency, consistency, and scalability while reducing waste. Process Analytical Technology (PAT) tools are increasingly being integrated to monitor critical quality attributes in real-time, ensuring batch consistency and regulatory compliance. Besides, automation and smart manufacturing, including robotics, digital control systems, and predictive maintenance, improve precision, throughput, and cost-effectiveness.

In addition, the growing adoption of sustainability practices by contract manufacturers, such as solvent recycling, energy-efficient processes, and waste minimization, which align with environmental and regulatory standards, is further contributing to market growth. Moreover, advanced analytical and quality control technologies, such as HPLC, LC-MS/MS, and spectroscopy, enable real-time monitoring and predictive analytics, ensuring product quality and reliability. Thus, these technological innovations enable Middle East contract manufacturing to enhance productivity, maintain high-quality standards, meet regional and global regulatory requirements, and support the growing demand for small-molecule pharmaceuticals.
Pricing Analysis
The Middle East small molecule contract manufacturing industry employs a diverse range of pricing models to suit client needs and project complexities. Milestone-based pricing links payments to specific achievements, such as API synthesis, formulation, or regulatory approvals. This approach incentivizes contract manufacturers to meet deadlines and quality targets, while providing pharmaceutical clients with predictable, performance-driven costs aligned with project progress. Value-based pricing sets costs according to the perceived value of the product or service, considering therapeutic significance, market potential, and efficiency gains. It allows contract manufacturers to charge premiums for faster timelines, innovative processes, or superior quality, enhancing client competitiveness in regional markets.
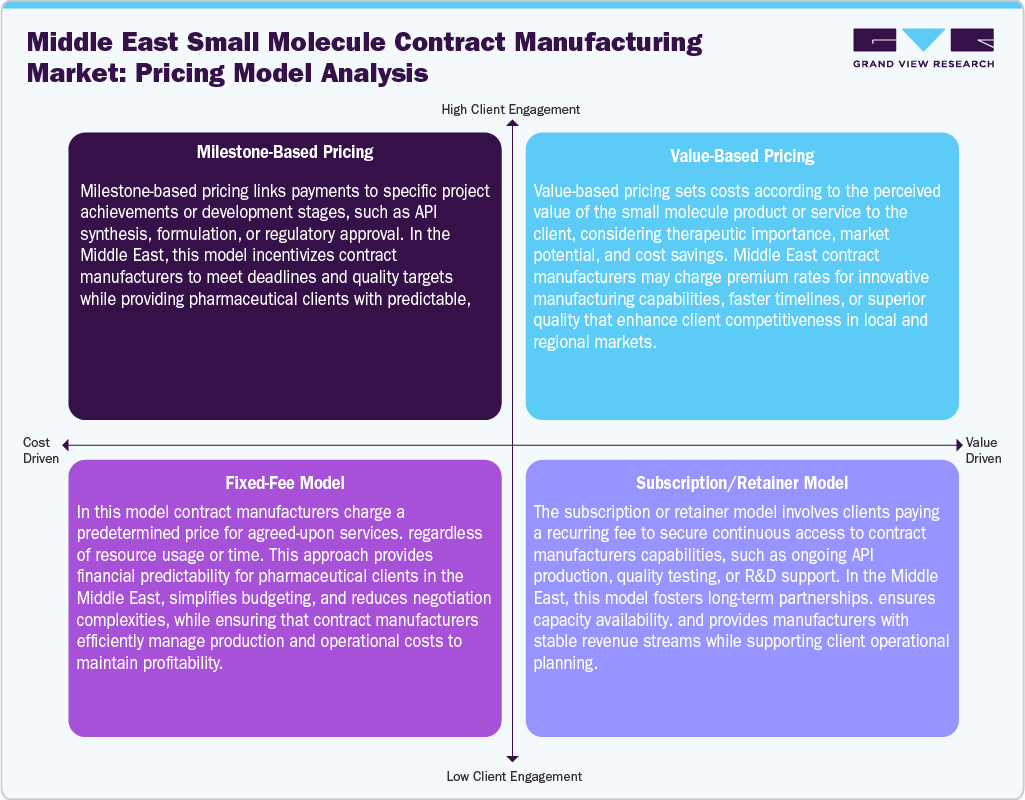
The fixed-fee models involve predetermined pricing for defined services, offering financial predictability, simplified budgeting, and reduced negotiation complexities. Contract manufacturers efficiently manage operational costs while ensuring profitability. Besides, subscription or retainer models offer recurring access to manufacturing capabilities, R&D support, or quality testing. These long-term partnerships secure capacity and ensure stable revenue streams for manufacturers while enabling clients to plan their operations reliably. Thus, these flexible pricing strategies support growth, attract global and regional clients, and enhance the competitiveness of Middle East contract manufacturing services.
Market Concentration & Characteristics
The market growth stage is medium and the pace is accelerating, characterized by a growing focus on local API and KSM production, increasing adoption of advanced manufacturing technologies, and strong government support. The market is moderately fragmented, with domestic CDMOs and international players competing on quality, regulatory compliance, capacity, and cost-efficiency.
The market is witnessing increasing innovation, with contract manufacturers adopting continuous manufacturing, advanced process analytical technologies, and automation to improve efficiency, quality, and scalability. Innovative capabilities such as green chemistry, eco-friendly processes, and advanced analytical techniques differentiate players, enabling faster time-to-market, regulatory compliance, and enhanced competitiveness in producing high-quality small molecule APIs. For instance, in August 2025, MS Pharma inaugurated a USD 50 million biologics facility in Saudi Arabia, offering formulation, fill & finish, lyophilization, and in-house bioanalytical testing, supporting monoclonal antibodies, peptides, and regional contract manufacturing.

Regulatory compliance plays a critical role with stringent GMP, ISO, and local health authority standards governing production. In addition, adherence to these frameworks ensures product quality, safety, and market approval. Companies failing to meet regulatory requirements face delays, penalties, or market exclusion.
Mergers and acquisitions are moderately active, as global pharmaceutical companies acquire or partner with contract manufacturers to expand capacity, enhance technological capabilities, and access regional markets. Strategic deals focus on capacity expansion, API diversification, and regulatory expertise.
Service expansion in the market includes enhanced R&D support, formulation development, analytical testing, and end-to-end manufacturing solutions. For instance, in January 2025, Gaelic Laboratories launched Gaelic Drug Store LLC in the UAE, providing local regulatory, distribution, and contract manufacturing support to meet Middle East pharmaceutical demand and assist international companies entering the region.
End use concentration is moderately high, with the majority of demand coming from pharmaceutical companies focused on generics, specialty drugs, and high-volume small molecule production. Leading clients often maintain long-term contracts with contract manufacturers, ensuring production stability.
Type Insights
By type, in 2024, the commercial segment held the largest market share, accounting for a revenue share of 83.29%. The segment's growth is driven by the increasing demand for the large-scale production of generic and specialty pharmaceuticals. Pharmaceutical companies outsource high-volume manufacturing to contract manufacturers to optimize costs, enhance efficiency, and comply with regulatory standards. This segment encompasses the full-scale synthesis, formulation, and production of finished dosage forms for regional and international markets. In addition, the segment benefits from long-term contracts, streamlined supply chains, and advanced manufacturing technologies.
The clinical segment is expected to grow at the fastest CAGR during the forecast period. The segment growth is driven by increasing demand for early-phase drug development and clinical trial support. Besides, the pharmaceutical companies outsource synthesis, formulation, and analytical services to contract manufacturers to accelerate time-to-market while ensuring compliance with GMP and regulatory standards. Thus, rising investment in R&D, expansion of clinical trial networks, and the region's strategic focus on establishing itself as a hub for pharmaceutical innovation are expected to drive the market growth over the estimated time period.
Service Insights
In terms of service, the API manufacturing segment dominated the market in 2024. The segment growth is driven by growing requirements for affordable generics, rising chronic diseases, and strong government efforts to localize the pharmaceutical production. Besides, the growing demand for high-quality active pharmaceutical ingredients for generics, specialty drugs, and treatments for chronic diseases is expected to drive market growth over the forecast period. In addition, the segment is growing, supported by local API production, rising healthcare infrastructure, and the region’s strategic goal of reducing import dependency, which are further contributing to segment growth. Moreover, enhanced R&D investments, the expansion of industrial clusters in the GCC, and improved regulatory frameworks further strengthen the region’s role as a manufacturing hub, supporting both domestic supply security and export-focused growth.
The finished dose formulation manufacturing segment is expected to witness a significant CAGR over the forecast period. The growth of this segment is driven by rising demand for generic drugs, expanding pharmaceutical exports, and pressure to shorten the time-to-market. The contract manufacturers offer formulation development, packaging, labeling, and quality testing services, enabling pharmaceutical companies to accelerate product commercialization without investing in in-house facilities. In addition, regulatory compliance, technology transfer capabilities, and high-quality manufacturing standards attract regional and international clients.
Therapeutic Area Insights
In terms of therapeutic area, the oncology segment dominated the market in 2024. The segment growth is driven by increasing prevalence of cancer, rising requirement for high-quality, affordable oncology therapies, expanding healthcare infrastructure, government initiatives promoting local production, and rising investment in oncology drug innovations. In addition, growing complexity of oncology drugs, and increasing regulatory sensitivity of oncology drug production, pharmaceutical companies are expected to drive the market growth. For instance, in September 2025, IAEA signed a USD 600,000 partnership with Qatar Fund for Development to enhance nuclear medicine services at Al-Biruni Hospital, Syria, benefiting over 3,000 cancer patients annually. Such factors are expected to drive the segment growth over the estimated time period.
The cardiovascular disease segment is expected to witness a significant CAGR over the forecast period. The segment growth is driven by the increasing prevalence of heart disease, hypertension, and related metabolic disorders. For instance, in Saudi Arabia, more than 30% of adults face a heightened risk of cardiovascular events due to smoking, unhealthy diets, and limited physical activity. By 2035, cases are projected to rise by about 480,000, significantly increasing healthcare costs. The Vision 2030, the Kingdom aimed to reduce the clinical and economic impact of heart disease, improve overall well-being, and raise life expectancy from 75 to 80 years. Moreover, pharmaceutical companies are outsourcing the production of CVD drugs including antihypertensives, anticoagulants, and lipid-lowering agents to CDMOs to ensure high-quality, scalable, and compliant manufacturing.
End-use Insights
On the basis of end use, the small and medium pharmaceutical companies segment accounted for the largest share in 2024. The segment is driven by their strong reliance on CDMOs for cost-effective production, regulatory compliance, and capacity expansion. These companies often lack extensive in-house manufacturing infrastructure, prompting them to outsource API synthesis, formulation, and finished-dose production to accelerate market entry. The growing need for flexibility, reduced operational risk, and access to specialized technologies further strengthens the segment growth.
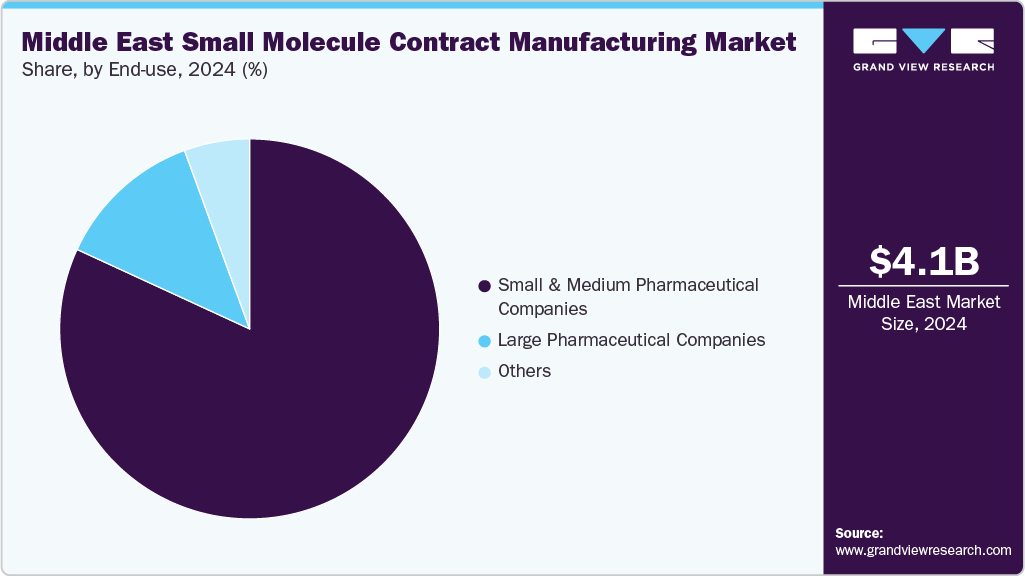
The large pharmaceutical companies segment represented the second-fastest-growing segment in the market, driven by their increasing use of contract manufacturers to enhance efficiency, expand their regional presence, and optimize production costs. As these companies focus on complex molecules, high-volume manufacturing, and rapid commercialization, they rely on contract manufacturers with advanced capabilities, regulatory expertise, and robust quality systems. Furthermore, expansion into the Middle East, supported by Vision 2030 initiatives, localization incentives, and free-zone manufacturing hubs, is expected to accelerate outsourcing. Such factors are expected to drive the segment growth over the estimated time period.
Country Insights
The Middle East small molecule contract manufacturing industry is driven by expanding healthcare investments, growing burdens of chronic diseases, and rising demand for cost-efficient pharmaceutical production. Government bodies are encouraging local manufacturing through incentives, free-zone benefits, and technology-transfer programs. The competitive landscape comprises regional contract manufacturers and global contract manufacturers entering the market, as well as partnerships between global pharmaceutical companies and local players aimed at enhancing capabilities. Competition centers on regulatory compliance, capacity expansion, and the adoption of advanced manufacturing technologies. The regulatory environment is strengthening, with agencies such as the SFDA, DHA, and GCC authorities enforcing higher quality, GMP standards, and mandatory local representation, transforming market entry and operational models.
Saudi Arabia Small Molecule Contract Manufacturing Market Trends
The small molecule contract manufacturing industry in Saudi Arabia held the largest revenue share of 34.13% in 2024, driven by rapid healthcare expansion, and strong government incentives supporting pharmaceutical localization. The competitive scenario features emerging regional manufacturers, multinational CDMOs entering through joint ventures, and free-zone facilities offering advanced capabilities. For instance, in October 2025, WuXi AppTec signed MoUs with NEOM and Saudi Arabia’s Ministry of Health to explore localizing pharmaceutical R&D and manufacturing, aiming to establish excellent facilities and strengthen the Kingdom’s biotechnology ecosystem.
UAE Small Molecule Contract Manufacturing Market Trends
The UAE small molecule contract manufacturing industry is the fastest-growing country over the forecast period, driven by increasing partnerships with global drug developers and investment in advanced manufacturing technologies. For instance, in May 2025, PharmaPrimes Laboratories partnered with Globalpharma to develop advanced UAE capabilities for biosimilar batch release and analysis, supporting the delivery of high-quality, cost-efficient therapies across the GCC and strengthening the region’s pharmaceutical and healthcare landscape.
Key Middle East Small Molecule Contract Manufacturing Company Insights
Middle East small-molecule contract manufacturing industry is dominated by regional CDMOs such as Julphar, Tabuk Pharmaceuticals, SPIMACO, and Globalpharma, alongside global players partnering locally. Market share is fragmented, with companies expanding capabilities in formulation, API production, and regulatory-compliant manufacturing to serve growing GCC and MENA demand. For instance, in April 2025, Hikma Pharmaceuticals signed an exclusive licensing deal with Vienna-based pharmaand GmbH to commercialize rucaparib, an innovative oral oncology therapy, across the MENA region, supporting Hikma’s strategy to expand its regional oncology leadership.
Key Middle East Small Molecule Contract Manufacturing Companies:
- Neopharma
- Tabuk Pharmaceuticals
- SPIMACO
- Sudair Pharma
- MISA
- AHT Health SARL
- Abdi Ibrahim Remede Pharma
- Saidal
- El Kendi
- Julphar
Recent Developments
-
In August 2025, MS Pharma opened a USD 50 million biologics plant in Saudi Arabia with mAb, peptide, and bioanalytical capabilities, enabling advanced regional manufacturing. The facility enhances MENA biopharma self-sufficiency and global partnership potential.
-
In July 2025, Saudi Arabia is engaging Indian pharma and medtech companies through an August 18 webinar to promote investment, localization, and partnerships under Vision 2030. The initiative strengthens bilateral cooperation and regional healthcare manufacturing goals.
-
In April 2025, Gaelic Laboratories established Gaelic Drug Store LLC in Dubai to meet regulatory requirements, expand Middle East market access, and support registration, import, and distribution of medicines. The move strengthens its regional growth strategy.
Middle East Small Molecule Contract Manufacturing Market Report Scope
Report Attribute
Details
Market size value in 2025
USD 4.39 billion
Revenue forecast in 2033
USD 8.90 billion
Growth rate
CAGR of 9.24% from 2025 to 2033
Actual data
2021 - 2024
Forecast period
2025 - 2033
Quantitative units
Revenue in USD million/billion, and CAGR from 2025 to 2033
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends, Middle East small molecule contract manufacturing outlook, volume analysis
Segments covered
Type, service, therapeutic area, end use, country
Regional scope
Middle East
Country Scope
Saudi Arabia; UAE; Kuwait; Oman; Qatar
Key companies profiled
Neopharma; Tabuk Pharmaceuticals; SPIMACO; Sudair Pharma; MISA; AHT Health SARL; Abdi Ibrahim Remede Pharma; Saidal; El Kendi; Julphar
Customization scope
Free report customization (equivalent up to 8 analysts working days) with purchase. Addition or alteration to country, & segment scope.
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
Middle East Small Molecule Contract Manufacturing Market Report Segmentation
This report forecasts revenue growth at country levels and provides an analysis of the latest industry trends in each of the sub-segments from 2021 to 2033. For this study, Grand View Research has segmented the Middle East small molecule contract manufacturing market report based on type, service, therapeutic area, end-use, and country.
-
Type Outlook (Revenue, USD Million, 2021 - 2033)
-
Clinical
-
Commercial
-
-
Service Outlook (Revenue, USD Million, 2021 - 2033)
-
API Manufacturing
-
Traditional API
-
HP-API
-
Controlled Substances/Specialty API
-
-
Finished Dose Formulations Manufacturing
-
Oral Solid
-
Injectables
-
Topical
-
Others
-
-
Packaging & Labelling
-
Others
-
-
Therapeutic Area Outlook (Revenue, USD Million, 2021- 2033)
-
Oncology
-
Cardiovascular Disease
-
Neurological Disorders
-
Infectious Diseases
-
Metabolic Disorders
-
Hormonal Disorders
-
Autoimmune Diseases
-
Gastrointestinal Disorders
-
Respiratory Diseases
-
Ophthalmology
-
Hematological Disorders
-
Others
-
Autoimmune/Inflammation
-
-
End-use Outlook (Revenue, USD Million, 2021 - 2033)
-
Large Pharmaceutical Companies
-
Small & Medium Pharmaceutical Companies
-
Others
-
-
Country Outlook (Revenue, USD Million, 2021 - 2033)
-
Middle East Small Molecule Contract Manufacturing
-
Saudi Arabia
-
UAE
-
Kuwait
-
Oman
-
Qatar
-
Rest of Middle East
-
-
Frequently Asked Questions About This Report
b. The Middle East small molecule contract manufacturing market size was estimated at USD 4.07 billion in 2024 and is expected to reach USD 4.39 billion in 2025.
b. The Middle East small molecule contract manufacturing market is expected to grow at a compound annual growth rate of 9.24% from 2025 to 2033 to reach USD 8.90 billion by 2033.
b. Saudi Arabia dominated the Middle East small molecule contract manufacturing market with a share of 34.13% in 2024. The market growth is driven by rapid healthcare expansion and strong government incentives supporting pharmaceutical localization. The competitive scenario features emerging regional manufacturers, multinational CDMOs entering through joint ventures, and free-zone facilities offering advanced capabilities.
b. Some key players operating in the Middle East small molecule contract manufacturing market include Neopharma, Tabuk Pharmaceuticals, SPIMACO, Sudair Pharma, MISA, AHT Health SARL, Abdi Ibrahim Remede Pharma, Saidal, El Kendi, Julphar
b. Key factors that are driving the market growth include increasing prevalence of chronic diseases, growing emphasis on R&D in the Middle East’s pharmaceutical sector, expanding costs for establishing and maintaining in-house manufacturing facilities and stringent regulatory requirements.
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










