- Home
- »
- Medical Devices
- »
-
Middle East Terminal Sterilization Services Market Report, 2033GVR Report cover
![Middle East Terminal Sterilization Services Market Size, Share & Trends Report]()
Middle East Terminal Sterilization Services Market (2025 - 2033) Size, Share & Trends Analysis Report By Product (Equipment, Consumables, Service & Maintenance), By Method (Low Temperature And High Temperature), By End Use, And Segment Forecasts
- Report ID: GVR-4-68040-741-0
- Number of Report Pages: 80
- Format: PDF
- Historical Range: 2021 - 2023
- Forecast Period: 2025 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
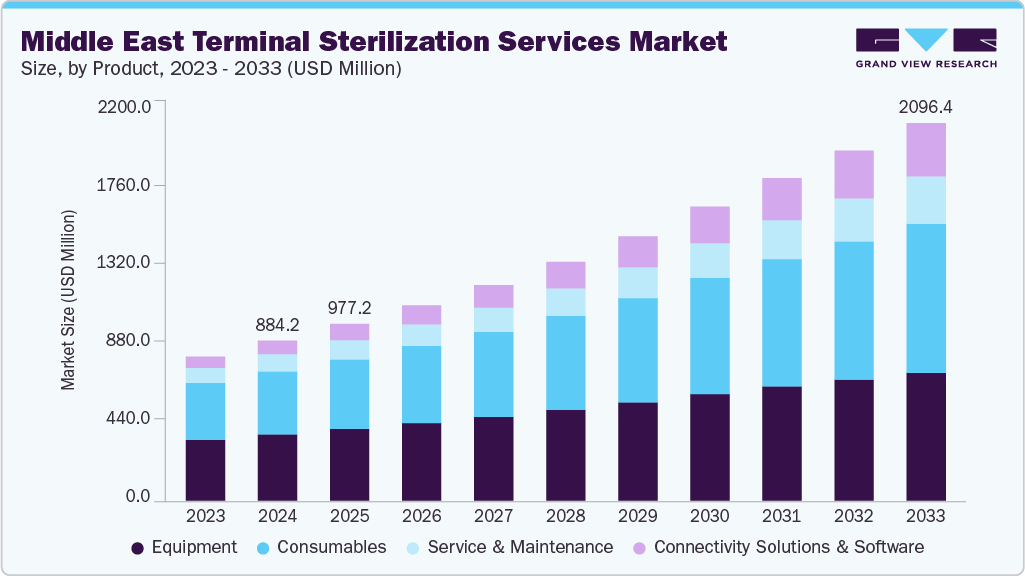
Middle East Terminal Sterilization Services Market Summary
The Middle East terminal sterilization services market size was estimated at USD 884.2 million in 2024 and is projected to reach USD 2,096.4 million by 2033, growing at a CAGR of 10.0% from 2025 to 2033. The growing incidence of healthcare-associated infections and the rising volume of surgical procedures across the Middle East are key drivers fueling the demand for terminal sterilization services in the region.
Key Market Trends & Insights
- Saudi Arabia dominated the Middle East terminal sterilization services market with the largest revenue share of 19.2% in 2024.
- Based on product, the equipment segment led the market with the largest revenue share of 41.3% in 2024.
- By method, the low temperature segment led the market with the largest revenue share of 40.6% in 2024.
- By end use, the hospitals segment led the market with the largest revenue share of 33.6% in 2024.
Market Size & Forecast
- 2024 Market Size: USD 884.2 Million
- 2033 Projected Market Size: USD 2,096.4 Million
- CAGR (2025-2033): 10.0%
The increasing prevalence of healthcare-associated infections (HAIs) is expected to significantly drive market growth in the Middle East. Hospitals and clinics across the region are under growing pressure to enhance sterilization protocols to prevent infections linked to inadequately sterilized medical equipment. Similar to global trends, HAIs in the Middle East represent a major public health concern, contributing to prolonged hospital stays, increased treatment costs, and higher morbidity and mortality rates.The booming pharmaceutical manufacturing, including sterile drugs, vaccines, and injectables, requires robust sterilization capabilities. This demand significantly uplifts the need for terminal sterilization services in the region. Adopting modern sterilization technologies such as low‑temperature methods (e.g., hydrogen peroxide, peracetic acid) and advanced irradiation enhances efficiency and expands applicability to heat-sensitive products. Eco-conscious approaches like non-toxic, sustainable sterilization techniques are gaining traction across the region.
Terminal sterilization, performed after final packaging, is critical for ensuring the complete inactivation of microbial contaminants. In the Middle East, where healthcare systems are undergoing rapid modernization and expansion, terminal sterilization is becoming an integral component of infection prevention protocols. The rising incidence of healthcare-associated infections (HAIs) and increased surgical volumes significantly boost the regional demand for terminal sterilization services.
Healthcare facilities in countries such as the United Arab Emirates, Saudi Arabia, and Qatar increasingly prioritize advanced sterilization methods to meet local and international infection control standards. As surgical procedures rise, particularly high-volume operations like cataract surgeries and cesarean sections, which mirror trends seen in EU hospitals, ensuring sterility of surgical instruments and medical devices has become paramount.
Regulatory alignment with global health authorities is also a major growth driver for terminal sterilization services in the Middle East. Regulatory bodies and Ministries of Health in the region are increasingly adopting guidelines and frameworks modeled after agencies such as the U.S. Food and Drug Administration (FDA), European Medicines Agency (EMA), and the World Health Organization (WHO). These authorities strongly advocate using terminal sterilization wherever feasible, as it offers the highest sterility assurance.
Major Brands Operating in the Market
Companies
Brand Name
Sterilization Method
ASP (Fortive)
STERRAD NX
Hydrogen Peroxide
STERRAD 100S
BIOTRACE
Steam Sterilization
VERISURE
SEALSURE
STERIS
V-PRO
Low Temperature Sterilization
Celerity HP
VERIFY V24
ConnectAssure
Getinge
GEE
Ethylene Oxide
Getinge Assured MI
Getinge Poladus 150
Hydrogen Peroxide
Steelco S.p.A.
PL 40
Hydrogen Peroxide
PL 70
AMS Series
Steam Sterilization
ASW Series
Sterigenics U.S., LLC - A Sotera Health company (Services)
Low Temperature, Service
Ethylene Oxide, Nitrogen Dioxide, and Gamma Sterilization
Source: Company Websites
Key Opinion Leaders Responses
KoL Response
About KoL
“Vaporized hydrogen peroxide’s addition as an established sterilization method helps us build a more resilient supply chain for sterilized devices that can help prevent medical device shortages. As innovations in sterilization advance, the FDA will continue to seek additional modalities that deliver safe and effective sterilization methods that best protect public health.”
Suzanne Schwartz, M.D., M.B.A., director of the Office of Strategic Partnerships and Technology Innovation in the FDA’s Center for Devices and Radiological Health.
"To deliver on our big bets on next-generation modalities like small molecules, biologics, and nucleic acid therapies, Lilly is investing in the state-of-the-art manufacturing infrastructure needed to deliver tomorrow's safe and reliable medicines,”
Edgardo Hernandez, executive vice president and president of Lilly Manufacturing Operations.
“The use of hydrogen peroxide gas plasma represents a significant shift in making it the gold standard of sterilization for duodenoscopes and thereby improving safety for patients, technicians, and the environment, The ULTRA GI Cycle sets a new benchmark for the entire industry on sterilization and environmental safety and will give healthcare professionals and their patients a heightened level of assurance that the duodenoscopes used are now being sterilized in a safe and effective way.”
Ivan Salgo, MD, Chief Medical & Scientific Officer ats ASP
“Our state-of-the-art X-Ray facility features innovative technology designed to maximize product safety and provide high-throughput processing while accommodating a wide range of product configurations-giving customers the flexibility and the geographic capacity they are looking for in a sterilization provider”
Mike Rutz, President, Sterigenics
Source: Company Websites
Market Concentration & Characteristics
The Middle East terminal sterilization services industry demonstrates a high degree of innovation, driven by growing regulatory requirements, the demand for safer and more eco-friendly methods, and advances in healthcare technology. For instance, in May 2025, MATACHANA announced the launch of its NEW S1000 Steam Sterilizer, featuring improved energy efficiency, faster cycle times, and advanced automation. This exemplifies the industry’s move toward smarter and more sustainable sterilization solutions.
Regulations play a critical role in shaping the Middle East terminal sterilization services industry by setting safety, efficacy, and environmental standards that manufacturers must meet. Stringent regulatory requirements drive innovation and the adoption of advanced sterilization technologies, ensuring patient safety while encouraging the development of more efficient and sustainable sterilization methods.

The terminal sterilization services industry across the Middle East and Africa has also seen strategic M&A moves, as companies aim to bolster their service capabilities, regional presence, and technology portfolios-particularly in advanced methods like electron beam (E‑beam) and vaporized hydrogen peroxide sterilization. In late 2023, a leading MEA-based sterilization service provider, Steris Middle East, completed the acquisition of a gamma irradiation sterilization facility in the United Arab Emirates from a European firm seeking to exit the regional market. This strategic move enabled Steris to quickly integrate high-capacity gamma sterilization technology, expand its geographic footprint in the Gulf region, and diversify its service portfolio to better serve pharmaceutical, medical device, and hospital clients across MEA.
Product substitutes for the terminal sterilization services industry include alternative methods such as aseptic processing, which is commonly used in pharmaceutical manufacturing to maintain sterility without applying terminal processes. High-level disinfection (HLD) is a substitute for semi-critical medical devices where complete sterility is not required. Sterile filtration (membrane filtration) is also another product substitute that helps remove microorganisms from liquids or gases before packaging.
The terminal sterilization services industry is moderately highly concentrated and dominated by key players like ASP (Fortive), STERIS, Getinge, and Sterigenics U.S., LLC. This Sotera Health company leverages extensive service networks and regulatory expertise. High entry barriers like strict regulations and costly infrastructure limit new competition, though regional and specialized providers add diversity, especially in emerging markets. For instance, in June 2025, Solventum launched the Attest Super Rapid Vaporized Hydrogen Peroxide (VH₂O₂) Clear Challenge Pack, a single-use test combining FDA-cleared biological and chemical indicators in a transparent pack, reflecting continued innovation in the sterilization space.
Product Insights
Based on product, the equipment segment held the largest revenue share of 41.3% in 2024. This segment includes steam autoclaves, ethylene oxide sterilizers, and hydrogen peroxide (H₂O₂) sterilizers, including plasma-based systems, the technological foundation of terminal sterilization services in healthcare, pharmaceutical, and medical device industries. These sterilization systems are essential for ensuring the sterility of medical instruments, drugs, and packaged devices, with each type designed to accommodate specific material sensitivities, sterility assurance requirements, and operational demands.
Modern terminal sterilization service equipment is evolving rapidly to meet the demands of efficiency, safety, and regulatory compliance. Key advancements include digital monitoring, shorter sterilization cycles, and automated validation systems, significantly improving workflow, reducing human error, and ensuring consistent sterilization performance. For instance, in June 2024, Getinge introduced the Poladus 150, its latest low-temperature sterilizer, to address the increasing need for safe and effective reprocessing of heat-sensitive surgical instruments. Utilizing vaporized hydrogen peroxide (VH₂O₂) and operating at a controlled temperature of up to 55°C, the Poladus 150 is engineered to sterilize non-lumen, flexible lumen, and rigid lumen instruments, making it highly suitable for the complex requirements of endoscopic and robotic surgeries.
Consumables is expected to witness a significant growth of 9.79% during the forecast period, driven by the rising demand for rapid sterilization verification, increased surgical procedures, and stricter infection control regulations. For instance, in August 2025, Advanced Sterilization Products (ASP) introduced the BIOTRACE Instant Read Steam Biological Indicator System, which delivers results in just 7 seconds. This innovation significantly enhances workflow efficiency by eliminating delays, supporting continuous operation, and ensuring speed, accuracy, and confidence in every sterilization cycle. Growing investments in terminal sterilization services and the shift toward automation in central sterile services departments (CSSDs) further support the strong demand for high-performance consumables.
Method Insights
Based on method, low temperature sterilization segment held the largest share in 2024. The low temperature sterilization method has a significant market share due to its effectiveness in sterilizing heat- and moisture-sensitive medical devices. Using chemical agents like ethylene oxide (EtO), hydrogen peroxide gas plasma, ozone, and vaporized peracetic acid ensures sterility without damaging delicate instruments. It is increasingly preferred for sterilizing minimally invasive surgical tools, flexible endoscopes, and implantable devices. Market growth is driven by the rise in reusable medical equipment, the expansion of outpatient surgical centers, and advancements that reduce cycle times and enhance sterilization efficiency.
The high temperature (steam sterilization) segment is expected to grow significantly over the forecast period. High temperature sterilization, primarily through steam sterilization (autoclaving), remains among the market's most established and widely used methods. Operating at 121°C to 134°C eliminates a broad range of microorganisms, including spores, making it ideal for heat- and moisture-resistant devices like surgical tools, stainless steel instruments, and reusable glassware. Known for its cost-effectiveness, reliability, and environmental safety, it is standard in hospitals, surgical centers, and labs. Despite the rise of low-temperature methods for sensitive devices, high-temperature sterilization continues to dominate due to its simplicity, short cycle times, and proven efficacy.
One of the major drivers of this segment is the increasing number of surgical procedures. As the volume of surgical procedures rises, there is a heightened need for effective sterilization methods to ensure patient safety and prevent healthcare-associated infections. According to a report by the NIH in March 2025, approximately 266 million surgical procedures are performed worldwide each year. In addition, the rising focus on infection control, particularly in the wake of hospital-acquired infections, has increased the demand for effective and validated sterilization systems.
End Use Insights
On the basis of end use, hospital & clinic held the largest revenue share of 33.6% in 2024. Hospitals & clinics drive demand for terminal sterilization services because it ensures the highest level of device sterilization, which is critical for patient safety by eliminating all microorganisms and reducing healthcare-associated infections. With rising surgeries, stricter regulations, and increased infection awareness, hospitals prioritize terminal sterilization services to meet standards, improve outcomes, and comply with laws, avoiding costly recalls and reputational damage.
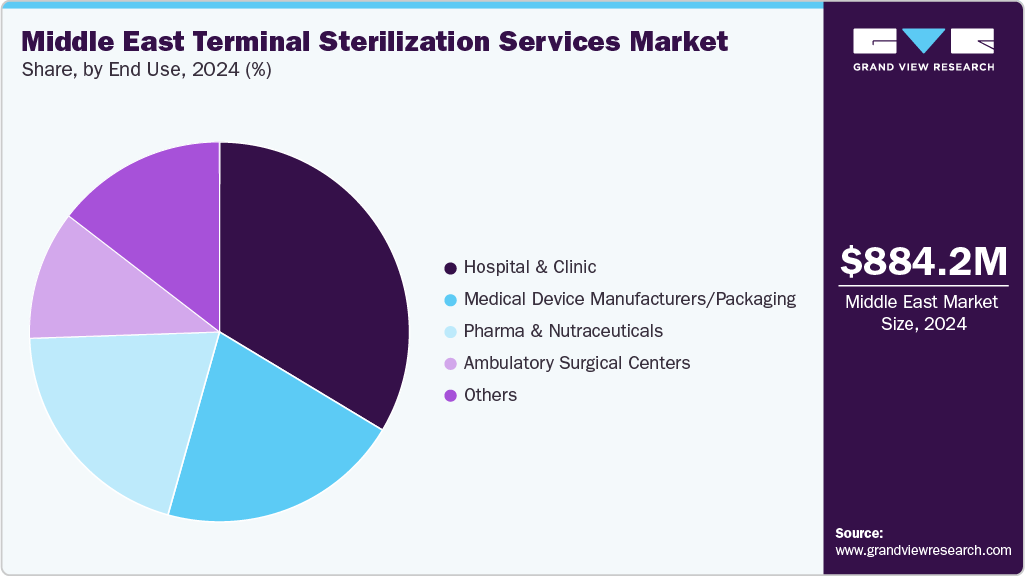
The ambulatory surgery centers segment is anticipated to experience the fastest growth over the forecast period, driven by increasing preference for outpatient procedures and expansion of ambulatory surgery center facilities, among others. For instance, according to UPMC HealthBeat, a publishing website for UPMC, an integrated health care system based in Pittsburgh, reports that many procedures and surgeries are now routinely performed on an outpatient basis.
Country Insights
Saudi Arabia Terminal Sterilization Services Market Trends
The region’s strong focus on improving healthcare quality, supported by national transformation programs such as Saudi Vision 2030, is pushing hospitals and manufacturers to shift toward outsourced, high-standard sterilization services. Regulatory pressure to meet stringent hygiene and sterility benchmarks is accelerating the adoption of terminal sterilization methods like ethylene oxide (EtO), gamma radiation, and steam sterilization.
Furthermore, the expansion of medical device manufacturing in the region, including disposables and implants, is creating additional demand for validated terminal sterilization processes. Investment in healthcare infrastructure, the rising elderly population, and the growth of ambulatory surgical centers are increasing procedural volumes, thereby driving the need for reliable and scalable sterilization services. As a result, terminal sterilization is emerging as a critical component in the region’s healthcare supply chain, with growing opportunities for service providers and technology innovators alike.
Kuwait Terminal Sterilization Services Market Trends
The terminal sterilization services market in Kuwait is witnessing notable growth, driven by the country’s commitment to enhancing healthcare quality and aligning with international best practices. Government-led healthcare reforms and strategic investments-particularly under Kuwait Vision 2035-are encouraging hospitals and medical device manufacturers to adopt outsourced, high-quality sterilization services.
The ongoing development of Kuwait’s medical device sector, especially in consumables and implantable devices, is further increasing demand for validated sterilization solutions. Modernization of healthcare infrastructure, coupled with an aging population and the expansion of outpatient and day-surgery centers, is contributing to higher surgical volumes and a greater need for efficient, scalable sterilization services.
UAE Terminal Sterilization Services Market Trends
The terminal sterilization services market in the UAE is experiencing robust growth, driven by the country's rapid expansion of its healthcare sector and its positioning as a leading medical tourism hub in the Middle East. Ongoing investments in state-of-the-art hospitals, specialty clinics, and pharmaceutical manufacturing facilities fuel demand for reliable and compliant sterilization solutions. Additionally, regulatory oversight by authorities such as the Ministry of Health and Prevention (MOHAP) and the Dubai Health Authority (DHA) is tightening infection control and product sterility standards, accelerating the shift toward professional terminal sterilization services.
Key Middle East Terminal Sterilization Services Company Insights
Leading players are actively pursuing strategic collaborations, new service offerings, mergers & acquisitions, and regional expansions to strengthen their market presence and capture a greater share of the growing demand for outsourced sterilization services. These strategies not only allow companies to scale operations efficiently but also enable faster access to regulatory-compliant sterilization infrastructure in key healthcare hubs. For instance, in 2023, Sterigenics (a Sotera Health company) expanded its EtO sterilization capacity in the UAE to meet the increasing regional demand for medical device sterilization, especially from Gulf Cooperation Council (GCC) countries. This move enhanced its logistical reach and improved turnaround times for clients in the Middle East region. The following are the leading companies in the Middle East terminal sterilization services market. These companies collectively hold the largest market share and dictate industry trends.
Key Middle East Terminal Sterilization Services Companies:
- ASP (Fortive)
- DE LAMA S.P.A.
- Getinge
- Andersen Sterilizers
- Labotronics Scientific
- Labtron Equipment Ltd
- STERIS
- Stryker
- Qingdao Antech Scientific Co., Ltd.
- MATACHANA
- Solventum
- Steelco S.p.A.
- RENOSEM CO., LTD.
- Tuttnauer
- MMM Group
- SOLSTEO
- Canon Singapore Pte. Ltd.
Recent Developments
-
In June 2025, Solventum announced the launch of its Attest Super Rapid Vaporized Hydrogen Peroxide (VH2O2) Clear Challenge Pack. This ready-to-use test combines two FDA-cleared indicators- a biological indicator (BI) that confirms microbial neutralization and a chemical indicator (CI) that verifies proper sterilizer performance- into a single-use pack featuring a transparent container.
-
In May 2025, MATACHANA launched its NEW S1000 Steam Sterilizer, featuring improved energy efficiency, faster cycle times, and advanced automation. This exemplifies the industry’s move toward smarter and more sustainable sterilization solutions.
-
In November 2024, ASP was recognized with four esteemed 2024 Medical Device Network Excellence Awards. These awards, in Innovation, Product Launch, Safety, and Environmental, reflect the company's dedication to advancing sterilization technology, enhancing healthcare safety, and promoting sustainability.
Middle East Terminal Sterilization Services Market Report Scope
Report Attribute
Details
Market size value in 2025
USD 977.24 million
Revenue forecast in 2033
USD 2,096.4 million
Growth rate
CAGR of 10.0% from 2025 to 2033
Base year for estimation
2024
Historical data
2021 - 2023
Forecast period
2025 - 2033
Quantitative units
Revenue in USD million/billion and CAGR from 2025 to 2033
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Product, method, end use
Regional scope
Middle East
Country scope
Saudi Arabia; Kuwait; UAE; Oman; Qatar
Key companies profiled
ASP (Fortive); DE LAMA S.P.A.; Getinge; Andersen Sterilizers; Labotronics Scientific; Labtron Equipment Ltd.; STERIS; Stryker; Qingdao Antech Scientific Co., Ltd.; MATACHANA; Solventum; Steelco S.p.A.; RENOSEM CO., LTD.; Tuttnauer; MMM Group; SOLSTEO; Canon Singapore Pte. Ltd.
Customization scope
Free report customization (equivalent up to 8 analysts working days) with purchase. Addition or alteration to country, regional & segment scope.
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
Middle East Terminal Sterilization Services Market Report Segmentation
This report forecasts revenue growth at country levels and provides an analysis of the latest industry trends in each of the sub-segments from 2021 to 2033. For this study, Grand View Research has segmented the Middle East terminal sterilization services market report based on product, method, end use, and country:
-
Product Outlook (Revenue, USD Million, 2021 - 2033)
-
Equipment
-
Consumables
-
Service & Maintenance
-
Connectivity Solutions and Software
-
-
Method Outlook (Revenue, USD Million, 2021 - 2033)
-
Low Temperature
-
Hydrogen Peroxide (H2O2)
-
Ethylene Oxide (EtO)
-
Others
-
-
High Temperature (Steam Sterilization)
-
Systems
-
Consumables
-
-
-
End Use Outlook (Revenue, USD Million, 2021 - 2033)
-
Hospitals & Clinic
-
Pharma and Nutraceuticals
-
Ambulatory Surgical Centres (ASCs)
-
Medical Device Manufacturers/Packaging
-
Others
-
-
Country Outlook (Revenue, USD Million, 2021 - 2033)
-
Middle East
-
Saudi Arabia
-
Kuwait
-
UAE
-
Oman
-
Qatar
-
-
Frequently Asked Questions About This Report
b. The Middle East terminal sterilization services market size was estimated at USD 884.2 million in 2024 and is expected to reach USD 977.24 million in 2025.
b. The Middle East terminal sterilization services market is expected to grow at a compound annual growth rate of 10.0% from 2025 to 2033 to reach USD 2,096.4 million by 2033.
b. The equipment segment held the largest revenue share of 41.3% in 2024. This segment includes steam autoclaves, ethylene oxide sterilizers, and hydrogen peroxide (H₂O₂) sterilizers, including plasma-based systems, which serve as the technological foundation of terminal sterilization services in healthcare, pharmaceutical, and medical device industries.
b. ASP (Fortive), DE LAMA S.P.A., Genist Technocracy Pvt. Ltd., Getinge, Andersen Sterilizers, Labotronics Scientific, Labtron Equipment Ltd, STERIS, Stryker , Qingdao Antech Scientific Co., Ltd., MATACHANA, Solventum, Steelco S.p.A., RENOSEM CO.,LTD., Tuttnauer, MMM Group, SOLSTEO, Sterigenics U.S., LLC – A Sotera Health company, Canon Singapore Pte. Ltd.
b. The growing incidence of healthcare-associated infections and the rising volume of surgical procedures across the Middle East are key drivers fueling the demand for terminal sterilization services in the region.
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










