- Home
- »
- Medical Devices
- »
-
Neurointerventional Devices Market, Industry Report, 2033GVR Report cover
![Neurointerventional Devices Market Size, Share & Trends Report]()
Neurointerventional Devices Market (2025 - 2033) Size, Share & Trends Analysis Report By Product (Diagnostic Devices, Therapeutic Devices), By Indication (Acute Ischemic Stroke (AIS), Intracranial Aneurysm), By End Use, By Region, And Segment Forecasts
- Report ID: GVR-4-68040-776-9
- Number of Report Pages: 300
- Format: PDF
- Historical Range: 2021 - 2023
- Forecast Period: 2025 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
Neurointerventional Devices Market Summary
The global neurointerventional devices market size was estimated at USD 2.95 billion in 2024 and is projected to reach USD 4.96 billion by 2033, growing at a CAGR of 5.9% from 2025 to 2033. The market is primarily driven by the rising prevalence of stroke, brain aneurysms, vascular malformations and other neurovascular disorders, coupled with the rapidly growing aging population prone to such conditions.
Key Market Trends & Insights
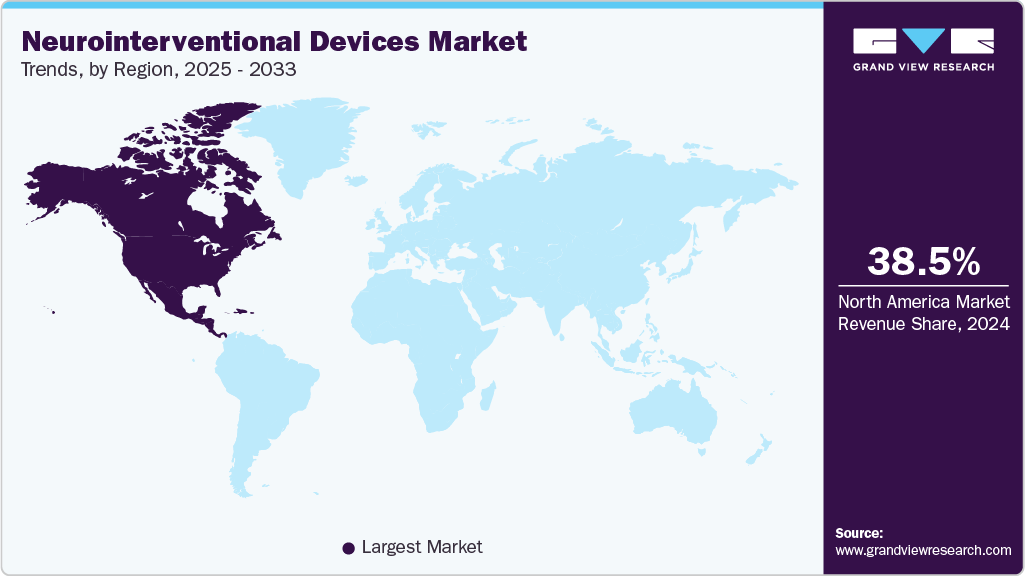
- North America dominated the neurointerventional devices market with the largest revenue share of 38.48% in 2024.
- The neurointerventional devices market in the U.S. accounted for the largest market revenue share of 78.09% in North America in 2024.
- By product, the therapeutic devices segment led the market with the largest revenue share of 65.84% in 2024.
- By indication, acute ischemic stroke segment led the market with the largest revenue share of 44.94% in 2024.
- By end use, the hospitals segment led the market with the largest revenue share of 54.28% in 2024.
Market Size & Forecast
- 2024 Market Size: USD 2.95 Billion
- 2033 Projected Market Size: USD 4.96 Billion
- CAGR (2025-2033): 5.9%
- North America: Largest market in 2024
Increasing demand for minimally invasive procedures, which offer faster recovery and fewer complications than open surgery, further accelerates market growth. The rising incidence of neurological disorders such as stroke, aneurysms, and other neurovascular conditions is a key driver of the neurointerventional devices market, as it increases the demand for effective, minimally invasive treatments that can reduce disability, improve survival rates, and enhance patient outcomes. According to the World Federation of Neurology (WFN), neurological disease prevalence varies across age groups, including preterm, neonatal, childhood, adolescent, adult, and older adults who often experience age-related multimorbidity and frailty. The 2024 Global Burden of Disease study estimated that 43% of the world’s population is affected by a neurological disorder, underscoring the significant global demand for neurointerventional devices and treatments.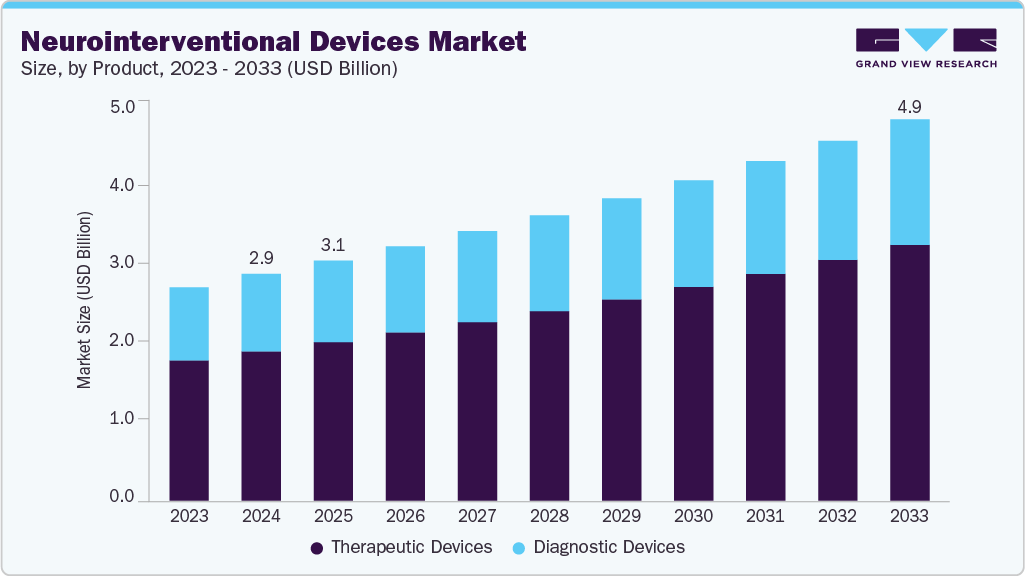
Technological advancements, including next-generation stent retrievers, flow diverters, and advanced imaging solutions, enhance treatment outcomes. Favorable regulatory approvals, improved reimbursement policies, and expanding healthcare infrastructure in emerging markets are supporting wider adoption of neurointerventional devices. For instance, in April 2024, Neurology journals have highlighted projections estimating that the number of brain disorder cases could reach 4.9 billion globally by 2050, marking a 22% increase compared to 2021. The all-age Disability-Adjusted Life Years (DALYs) rate of brain disorders is also expected to rise significantly by 10% globally, from 5,139 DALYs per 100,000 in 2021 to 5,666 DALYs per 100,000 in 2050. Headaches, Alzheimer’s disease, depressive and anxiety disorders, and strokes are identified as the primary contributors to this anticipated surge in prevalence and disease burden.
Distribution of Neurological Diseases By Age Groups
Age Group
Key Neurological Disorders
Prenatal
- Developmental malformations
Neonatal & Early Childhood (0-5 years)
- Hypoxic-ischemic encephalopathy
- Neuroinfections (meningitis, encephalitis, cerebral malaria)
- Neurodevelopmental disorders (autism, microcephaly, spina bifida)
Childhood & Adolescence (5-18 years)
- Epilepsy
- Neurodevelopmental disorders (attention deficit hyperactivity disorder, autism, dyslexia)
- Trauma-related neurological sequelae
- Increasing prevalence of mental health disorders (depression, anxiety, post-traumatic stress disorder)
Adulthood (18-65 years)
- Migraine
- Stroke
- Epilepsy
- Traumatic brain injury
- Neuroinfections (HIV-associated neurocognitive disorder, tuberculosis meningitis)
Older Adults (65+ years)
- Dementia (Alzheimer's, vascular dementia)
- Parkinson's disease
- Stroke
- Other neurodegenerative diseases
Source: World Federation of Neurology
The rising incidence of stroke, particularly ischemic stroke is propelling the neurointerventional devices market by increasing the demand for efficient and minimally invasive clot removal procedures. For instance, in 2024, CDC data highlighted, as presented in the table below, that over 795,000 people in the U.S. suffer a stroke each year, emphasizing the growing need for advanced neurointerventional treatment options.[ According to the WHO, the global incidence of stroke is estimated to be approximately 15 million people annually, with ischemic stroke representing about 87% of all stroke cases.
Statistic
Value
Stroke-related deaths as % of cardiovascular deaths (2022)
17.5% (1 in 6)
Stroke death rate
39.5 per 100,000 (2022); 39.0 per 100,000 (2023)
Frequency of stroke occurrence
Every 40 seconds
Frequency of stroke-related death
Every 3 minutes 14 seconds
Annual number of strokes
>795,000
First or new strokes
~610,000
Recurrent strokes
~185,000 (nearly 1 in 4)
Ischemic strokes
~87% of all strokes
Source: Centers for Disease Control and Prevention (CDC)
According to the World Stroke Organization 2022, the table below shows the incidence and prevalence of ischemic stroke.
Measure
Category
Number
Crude rate per 100,000 per year (95% UI)
Age-adjusted rate per 100,000 per year (95% UI)
Key Statement
Incidence
Ages (all), Sexes (both)
7,630,803
94.51 (81.91-110.76)
98.62 (84.90-115.80)
Over 7.6 million new ischemic strokes each year globally; >62% of all incident strokes are ischemic.
15-49 years
865,723
22.00 (16.37-29.50)
-
Over 11% of ischemic strokes occur in people 15-49 years old.
<70 years
4,427,351
60.87 (49.72-72.93)
-
Over 58% of ischemic strokes occur in people under 70 years.
Men (all ages)
3,445,762
88.79 (76.15-103.81)
90.91 (78.52-106.55)
45% of ischemic strokes occur in men.
Women (all ages)
4,185,041
108.52 (93.57-127.20)
97.22 (84.12-113.94)
55% of ischemic strokes occur in women.
Prevalence
Ages (all), Sexes (both)
77,192,498
997.65 (889.92-1,117.39)
950.97 (849.82-1,064.06)
Over 77 million people are currently living who have experienced ischemic stroke.
15-49 years
14,480,207
367.97 (306.12-440.40)
-
19% of people who have experienced an ischemic stroke are aged 15-49.
<70 years
47,161,262
648.37 (569.07-730.52)
-
61% of people who have experienced an ischemic stroke are under 70 years old.
Men (all ages)
33,216,442
855.88 (759.25-967.92)
863.51 (767.96-974.62)
43% of people who have experienced an ischemic stroke are men.
Women (all ages)
43,976,056
1,140.31 (1,020.45-1,273.16)
1,025.52 (918.48-1,144.69)
57% of people who have experienced an ischemic stroke are women; globally, women account for just over half (56%).
Source: World Stroke Organization
According to the Journal of Experimental Stroke & Translational Medicine, Hemorrhagic stroke accounts for approximately 10-15% of cases and results from the rupture of a blood vessel, causing bleeding within the brain tissue (intracerebral hemorrhage) or into the space surrounding the brain (subarachnoid hemorrhage).
Arteriovenous malformations (AVMs) are a significant factor driving the growth of the neurointerventional devices market, as these complex vascular abnormalities require precise and minimally invasive treatment approaches. The increasing detection of AVMs through advanced imaging techniques and the growing preference for endovascular procedures over open surgery fuel the demand for specialized neurointerventional devices such as embolization systems, catheters, and coils. Rising awareness among healthcare professionals about early intervention to prevent hemorrhagic complications further accelerates the adoption of advanced neurointerventional solutions for AVM management. According to a research article published by the National Center for Biotechnology Information, arteriovenous malformations (AVMs) are often asymptomatic in about 15% of cases until a clinical event occurs. Between 41% and 79% of AVMs are present with intracranial hemorrhage. They are the second most common cause of intracranial bleeding after cerebral aneurysms, accounting for approximately 10% of all subarachnoid hemorrhages.
The growing aging population drives the neurointerventional devices market, as older adults are more prone to neurovascular conditions such as stroke, aneurysms, and age-related cerebrovascular diseases. This demographic trend increases the demand for minimally invasive interventions and advanced neurovascular devices. For instance, in June 2024, the Brain Aneurysm Foundation reported that an estimated 6.8 million people in the United States, about 1 in 50, have an unruptured brain aneurysm, with an annual rupture rate of 8-10 per 100,000 people. Each year, roughly 30,000 Americans experience a brain aneurysm rupture, which occurs on average every 18 minutes. Globally, brain aneurysms cause nearly 500,000 deaths annually, with half of the victims under 50. While most aneurysms develop after age 40 and are most common between ages 35 to 60, they can also occur in children. Women are more likely than men to have a brain aneurysm (3:2 ratio), and women over 55 faces approximately 1.5 times higher risk of rupture than men.
Key Opinion Leaders Responses
Company
KoL Response
About KoL
Terumo Neuro
“Our hemorrhagic portfolio leads the way in delivering innovative solutions, grounded in strong clinical outcomes, that empower physicians to confidently treat a wide range of aneurysms. The Aneurysm Treatment Alliance brings this commitment to life by providing physicians across EMEA with practical tools and collaborative opportunities to elevate aneurysm treatment. This launch also reflects our laser-focused dedication to physician partnerships, innovation, and improving patient outcomes worldwide.”
Sebastien Chabeauti, Vice President and General Manager, EMEA, Terumo Neuro.
Terumo Neuro
“As we introduce the SOFIA 88 Neurovascular Support Catheter, we are building on a decade of performance that physicians around the world have come to trust. This launch reflects our continued investment in neurovascular innovation, made possible through direct collaboration with physicians and our strong R&D and manufacturing capabilities. Together, we are delivering meaningful advancements in stroke care designed to improve outcomes for patients worldwide.”
Carsten Schroeder, President and CEO of Terumo Neuro
Boston Scientific Corporation
"Boston Scientific is pleased to offer this new technology, which is designed to assist physicians simplify these complex neurovascular procedures. The Neuroform EZ Stent System reinforces our commitment to providing advanced treatment options focused on improving patient care."
Mark Paul, President of Boston Scientific's Neurovascular Division
Philips
“In stroke care, minutes matter - and confidence is everything. With this latest generation of SmartCT for intelligent 3D imaging applications, we’re giving neuro interventionalists the imaging tools they need at the exact moment they need them - improving workflow, reducing delays, and ultimately helping to save lives.”
Dr. Atul Gupta, Chief Medical Officer Diagnosis & Treatment at Philips
Boston Scientific Corporation
"The TCAR platform developed by Silk Road Medical is a notable advancement in the field of vascular medicine, which has revolutionized stroke prevention and the treatment of carotid artery disease. We believe the addition of this clinically differentiated technology to our vascular portfolio demonstrates our continued commitment to provide meaningful innovation for physicians who care for patients with peripheral vascular disease."
Cat Jennings, president, Vascular, Peripheral Interventions, Boston Scientific
Source: Company Websites
Market Concentration & Characteristics
The degree of innovation in the neurointerventional devices market is high, driven by advancements in AI-assisted imaging, neuro-robotics, and minimally invasive technologies. These innovations enhance procedural precision, safety, and patient outcomes across neurovascular conditions. For instance, in June 2025, Philips launched the latest generation of SmartCT intelligent 3D imaging in Europe, enabling neurointerventionists to view brain tissue, blood vessels, and interventional devices in real time directly in the Angio suite. By eliminating the need for separate CT scans, SmartCT allows care teams to treat patients faster with greater clarity and confidence, supporting more efficient and effective stroke care. In addition, for instance, in May 2025, Terumo Neuro announced the U.S. commercial availability of the SOFIA 88 Neurovascular Support Catheter, expanding its stroke treatment portfolio with a large-bore catheter designed for enhanced reliability, procedural flexibility, and physician control in neurovascular interventions.
Regulations significantly impact the neurotechnology market by ensuring device safety and efficacy while guiding innovation. For instance, in February 2025, the International Journal of Biology, Pharmacy and Allied Sciences highlighted that the FDA’s regulatory framework for neurological devices covers premarket pathways such as Investigational Device Exemptions (IDEs), Premarket Approval (PMA), 510(k) clearance, and post market surveillance. It highlights the balance between fostering innovation and protecting public health, providing a valuable resource for industry professionals and researchers navigating device development and market approval.
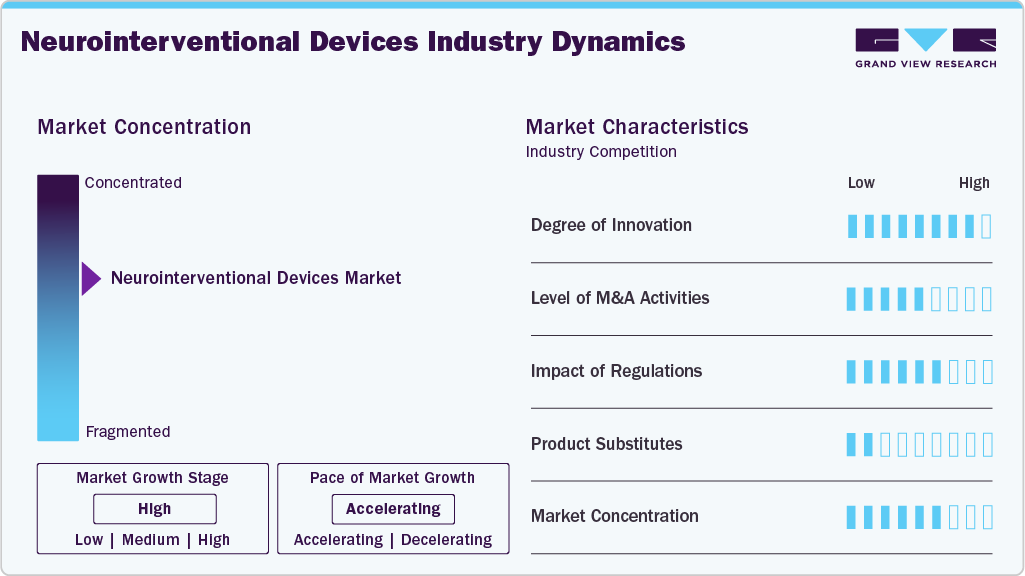
The neurointerventional devices market has witnessed high mergers and acquisitions, driven by companies seeking to expand product portfolios and geographic reach. Strategic acquisitions focus on integrating innovative technologies and enhancing competitive positioning in the growing neurovascular sector. For instance, in June 2024, Boston Scientific announced an agreement to acquire Silk Road Medical for 1.16 billion, strengthening its vascular portfolio with Silk Road’s transcarotid artery revascularization (TCAR) technology for minimally invasive stroke prevention.
The neurointerventional devices market faces moderate risk from product substitutes, including pharmaceutical therapies, conventional surgery, and non-invasive treatment options. These alternatives can limit device adoption, especially when they offer lower cost or reduced procedural complexity.
The neurointerventional devices market is moderately concentrated, with a few leading players such as Terumo Neuro, NeuroSafe Medical Co., Ltd, Medtronic, among others holding significant market share. These key companies drive innovation, influence pricing, and shape industry standards through advanced technology and strategic collaborations. Emerging players continue to enter the market, fostering competition and accelerating technological advancements.
Product Insights
The therapeutic devices segment held the largest revenue share in 2024 and is also expected to register the fastest growth over the forecast period. This dominance is driven by the rising prevalence of neurovascular disorders such as ischemic stroke, aneurysms, and arteriovenous malformations, coupled with increasing adoption of minimally invasive treatment procedures. Continuous advancements in thrombectomy devices, stents, and embolization systems have enhanced procedural efficacy and patient outcomes, further propelling segment growth. Moreover, growing clinical evidence supporting the effectiveness of mechanical thrombectomy and embolic devices encourages wider use across healthcare facilities globally. For instance, in July 2025, Boston Scientific Corporation launched the Neuroform EZ Stent System in the U.S. and Europe, its fourth-generation intracranial aneurysm stent designed for use with endovascular coiling to treat wide-necked aneurysms. The system features enhanced flexibility and conformability for tortuous brain anatomy, employs Segmental Expansion technology for improved anchoring and stability, and integrates a simplified single-operator delivery mechanism, building on the proven hybrid cell design of earlier Neuroform stents.
The diagnostic devices segment is anticipated to experience a significant growth within the neurointerventional devices market. This growth is fueled by the increasing demand for accurate and minimally invasive diagnosis of cerebrovascular disorders, such as stroke, aneurysms, and vascular malformations. Innovations in high-precision guidewires and advanced imaging technologies, including 3D and real-time imaging systems, improve procedural accuracy and patient safety. The rising number of diagnostic procedures and early detection initiatives globally is expected to further drive the adoption of these devices in hospitals and specialized neurointerventional settings. For instance, in February 2025, Johnson & Johnson MedTech announced the launch of the CEREGLIDE 92 Catheter System, a next-generation .092” catheter featuring the INNERGLIDE 9 delivery aid. Designed for use in the neurovascular system, the CEREGLIDE 92 enables large distal access and reduces flow in the M1 during device insertion for acute ischemic stroke treatment. The catheter’s larger lumen also supports multiple neurovascular tools for treating aneurysms and other hemorrhagic complications, and it integrates seamlessly with Johnson & Johnson MedTech’s Stroke Solutions portfolio, including CEREGLIDE 71, PROWLER EX, and EMBOTRAP III.
Indication Insights
The acute ischemic stroke (AIS) segment dominated the neurointerventional devices market in 2024 and is expected to be the fastest-growing segment during the forecast period. This growth is driven by the increasing global prevalence of ischemic strokes, rising awareness of early intervention benefits, and the growing adoption of minimally invasive mechanical thrombectomy procedures. Advanced neurointerventional devices, such as aspiration catheters and stent retrievers, have significantly improved patient outcomes and procedural success rates, further boosting market demand. Expanding healthcare infrastructure and favorable reimbursement policies in key regions support the widespread use of AIS-focused interventions, making this segment a key driver of the overall neurointerventional market. For instance, according to the WHO, the global incidence of stroke is estimated to be approximately 15 million people annually, with ischemic stroke representing about 87% of all stroke cases.
The arteriovenous malformation (AVM) segment is experiencing significant growth in the neurointerventional devices market. This expansion is driven by increasing diagnoses of AVMs due to advancements in imaging technologies and heightened awareness of potential complications such as hemorrhagic stroke. Minimally invasive endovascular procedures, including embolization using specialized catheters and coils, are becoming the preferred treatment approach, offering improved safety and efficacy compared to traditional surgical methods. Ongoing R&D and the introduction of innovative AVM-specific devices are further accelerating adoption, contributing to the segment’s notable growth in the market. In addition, according to an article published by Radiopaedia.org in November 2024, the incidence of solitary AVM is 98% and multiple AVM is 2%. Thus, growing AVM cases are a primary contributor to market growth.
End Use Insights
The hospitals segment held the largest revenue share in 2024, driven by the availability of advanced infrastructure, skilled specialists, and high patient volumes for complex neurovascular procedures. Hospitals serve as primary centers for acute care, including stroke interventions, aneurysm treatments, and arteriovenous malformation management, making them the largest end user of neurointerventional devices. The segment’s dominance is further supported by the increasing adoption of minimally invasive procedures, investment in state-of-the-art imaging and surgical equipment, and growing awareness among patients and clinicians of early intervention benefits. This leadership underscores hospitals as key drivers of market growth, facilitating wider access to advanced neurointerventional therapies.
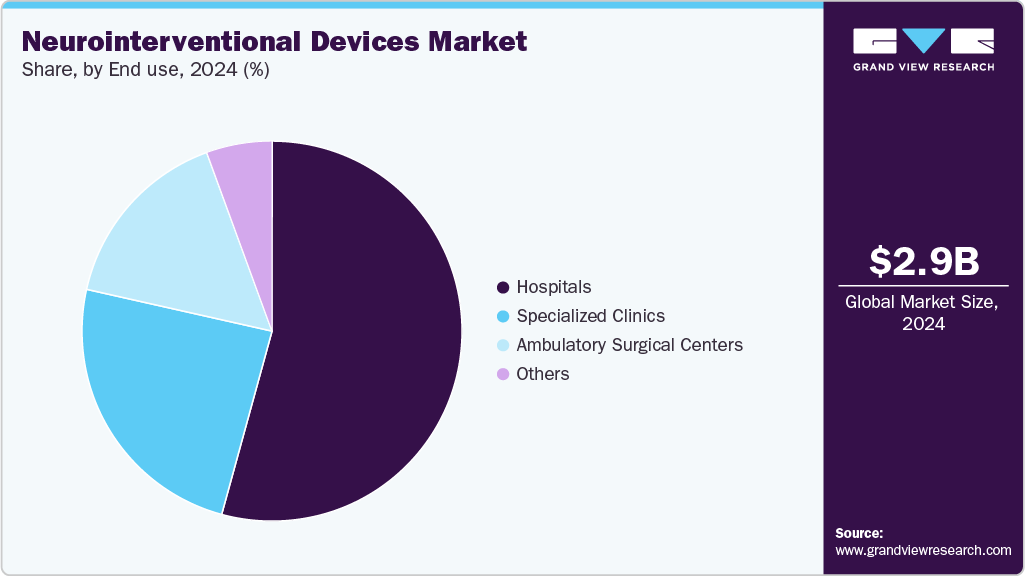
The ambulatory surgery centers segment is anticipated to experience fastest growth over the forecast period. This growth is driven by the increasing preference for minimally invasive procedures that can be performed on an outpatient basis, offering reduced hospital stays and lower treatment costs. Advancements in portable imaging systems, compact neurointerventional devices, and streamlined procedural techniques enable ASCs to perform complex interventions safely and efficiently. In addition, rising patient awareness, favorable reimbursement policies, and the expansion of specialized outpatient facilities support the accelerated adoption of neurointerventional procedures in ASCs, positioning the segment as a key growth area in the market.
Regional Insights
North America neurointerventional devices market accounted for the largest revenue share of 38.48% in 2024, driven by the rising prevalence of cerebrovascular diseases and increased adoption of minimally invasive procedures. Technological advancements in devices, along with growing awareness among healthcare professionals and patients about early diagnosis and treatment, are further fueling market expansion in the region.
The region is witnessing a strong shift toward minimally invasive procedures, as these approaches offer reduced recovery times, lower complication rates, and improved patient outcomes compared to traditional surgical indications. Continuous technological advancements in neurointerventional devices, including innovative catheters, stents, and embolization systems, are enabling more precise and effective treatments. In addition, growing awareness among healthcare professionals and patients about early diagnosis, preventive care, and advanced treatment options is further driving the adoption of these devices. Favorable reimbursement policies, coupled with ongoing investments in healthcare infrastructure and research, are also contributing to the robust expansion of the neurointerventional devices market in North America.
U.S. Neurointerventional Devices Market Trends
The U.S. held the largest share of 78.09% in 2024 in North America neurointerventional devices market. The gowth is driven by the rising incidence of cerebrovascular diseases such as stroke, aneurysms, and arteriovenous malformations. Increasing adoption of minimally invasive procedures, technological advancements in devices, and a growing focus on early diagnosis and timely intervention fuel market expansion. The market is further supported by favorable healthcare infrastructure, high awareness among physicians and patients, and continuous innovation in neurovascular treatment solutions, making the U.S. a leading region in neurointerventional care. For instance, in July 2025, Terumo Neuro expanded its global stroke portfolio with the commercial launch of the SOFIA 88 Neurovascular Support Catheter in the U.S. Engineered for reliable trackability, proximal stability, and vessel-friendly flexibility, the SOFIA 88 catheter is designed to enhance physician control and seamlessly integrate into a variety of aspiration and combination treatment strategies.
Europe Neurointerventional Devices Market Trends
The European neurointerventional devices market is witnessing significant growth, driven by the increasing prevalence of cerebrovascular disorders and the rising demand for minimally invasive treatments. Neurointerventional technologies, including innovative stents, catheters, and embolization systems, enable more effective and precise procedures. For instance, in June 2025, Philips launched its intelligent 3D imaging system in Europe to empower faster stroke care, assisting physicians save time, brain, and lives. Growing awareness among healthcare professionals, improved healthcare infrastructure, and an emphasis on early diagnosis and intervention contribute to the expanding adoption of neurointerventional devices across the region.
The UK neurointerventional devices market is showing significant growth, driven by the rising prevalence of cerebrovascular diseases and the increasing adoption of minimally invasive procedures. Technological advancements in neurointerventional devices, such as advanced catheters, stents, and embolization systems, are enhancing treatment precision and patient outcomes. In addition, growing awareness among healthcare professionals, improved hospital infrastructure, and government initiatives to support early diagnosis and stroke care are contributing to the expanding use of neurointerventional devices across the UK.
Asia Pacific Neurointerventional Devices Market Trends
The Asia Pacific neurointerventional devices market is witnessing strong growth, driven by the increasing prevalence of stroke and neurovascular disorders and rising healthcare infrastructure and awareness. Technological adoption, expanding minimally invasive procedures, and government initiatives to improve neurological care further boost regional market demand. For instance, MicroPort NeuroScientific Corporation and its subsidiaries are among China's pioneering medical device companies that have entered the neuro-interventional therapeutic field. Dedicated to researching and developing advanced medical devices within this specialty. Their comprehensive product portfolio for stroke intervention encompasses all three major categories of neurovascular diseases: hemorrhagic stroke, cerebral atherosclerotic stenosis, and acute ischemic stroke. Their products have been implemented in over 3,400 hospitals nationwide, supporting approximately 210,000 neurointerventional procedures.
The neurointerventional devices market in China is rapidly expanding due to the growing incidence of stroke and cerebrovascular diseases and increasing investments in advanced healthcare infrastructure. Rising adoption of minimally invasive neurovascular procedures and supportive government policies drive market growth. For instance, in the Chinese market, the MicroPort NeuroScientific Corporation products have been adopted by approximately 3,400 hospitals nationwide, supporting around 210,000 neuro-interventional surgeries. Benefiting from the VBP policy, the NUMEN Coil has accelerated market development, recently entered 520 new hospitals and now serving nearly 1,450 hospitals. The Tubridge Flow-diverting Stent has been introduced to 170 new hospitals, bringing its coverage to over 1,190. Launching the new generation Tubridge Plus Flow-diverting Stent further enhances their product offerings. The clinical application of the Bridge Vertebral Artery DES has increased significantly, providing new growth momentum. The APOLLO Intracranial Stent System has continued to strengthen its market position and has expanded to nearly 190 hospitals, with an overall reach of approximately 2,340 hospitals. In 2024, the Group ranked first among all domestic brands in China’s neuro-interventional medical device market based on sales, maintaining the leading market share.
Latin America Neurointerventional Devices Market Trends
The Latin America neurointerventional devices market is experiencing gradual growth, driven by increasing awareness of cerebrovascular diseases and the rising demand for minimally invasive treatment options. Key countries contributing to market expansion include Brazil, Mexico, Argentina, Colombia, and Chile, where improving healthcare infrastructure and growing adoption of advanced neurointerventional technologies, such as stents, catheters, and embolization systems, are supporting market growth. Initiatives to enhance early diagnosis, along with rising investments in healthcare facilities and specialist training, are further driving the adoption of neurointerventional devices across the region.
Middle East and Africa Neurointerventional Devices Market Trends
The Middle East neurointerventional devices market is witnessing significant growth, driven by the rising prevalence of cerebrovascular diseases and increasing demand for minimally invasive treatments. Countries such as Saudi Arabia, UAE, Qatar, and Israel are investing in advanced healthcare infrastructure and adopting innovative neurointerventional technologies, including stents, catheters, and embolization systems. Growing awareness among healthcare professionals and patients, coupled with initiatives to promote early diagnosis and timely intervention, is further fueling the adoption of neurointerventional devices across the region.
Key Neurointerventional Devices Company Insights
The neurointerventional devices market is highly competitive, with several key players driving innovation and capturing significant market share. Leading companies, including Medtronic, Stryker, Terumo Neuro, Philips, and Boston Scientific, focus on developing advanced catheters, stents, and embolization systems to improve procedural outcomes and patient safety. These companies leverage continuous product innovation, strategic partnerships, and regional expansions to strengthen their presence in major markets. Their strong R&D capabilities and extensive distribution networks enable them to maintain a competitive edge, collectively holding a substantial share of the global neurointerventional devices market.
Key Neurointerventional Devices Companies:
The following are the leading companies in the neurointerventional devices market. These companies collectively hold the largest market share and dictate industry trends.
- Terumo Neuro
- NeuroSafe Medical Co., Ltd
- ZYLOX-TONBRIDGE MEDICAL TECHNOLOGY CO., LTD.
- Brain Navi Biotechnology Co., Ltd.
- Medtronic
- Stryker
- CERENOVUS (Johnson & Johnson)
- Integra LifeSciences Corporation
- Penumbra Inc
- Balt SAS
- B. Braun SE
- Phenox GmbH
- Shanghai MicroPort Medical (Group) Co. Ltd.
- Boston Scientific Corporation
- Meril
- Gem srl
- Sirtex (BlackSwan Vascular, Inc.)
- INVAMED
- Merit Medical Systems
- Abbott
- Acandis GmbH
- Cook Medical
- Imperative Care, Inc.
- Rapid Medical
- Koninklijke Philips N.V.
- KANEKA CORPORATION
Recent Developments
-
In September 2025, Terumo Neuro announced the launch of the Aneurysm Treatment Alliance at the 2025 European Society of Minimally Invasive Neurological Therapy (ESMINT) 17th Annual Congress in EMEA to advance physician collaboration and improve aneurysm care through standardized tools and a comprehensive device portfolio. The Aneurysm Treatment Alliance is a new EMEA initiative designed to strengthen physician engagement, foster collaboration, and showcase Terumo Neuro’s differentiated hemorrhagic portfolio for aneurysm treatment. It provides standardized tools and a collaborative platform to support physicians in advancing patient care across the region. The Alliance highlights comprehensive portfolio access, cross-regional learning, and collaboration opportunities to share expertise, best practices, and education on integrated aneurysm therapy solutions.
-
In September 2025, SENTANTE Stroke System received FDA Breakthrough Device Designation, enabling trained neurointerventional physicians to remotely perform mechanical neurothrombectomy for acute ischemic stroke patients at hospitals without local thrombectomy access or timely transfer options.
-
In September 2025, Penumbra received CE Mark approval for its SwiftPAC neuroembolisation coils in Europe, offering a soft, space-seeking design for precise vessel occlusion, compatibility with microcatheters, and enhanced repositioning capabilities for improved neurovascular treatment.
-
In September 2025, Rapid Medical announced that its DRIVEWIRE 24 steerable guidewire surpassed 1,000 neurovascular procedures in North America and received CE Mark approval under the EU MDR, enabling its use across Europe. The 0.024-inch guidewire offers real-time tip control for navigating complex anatomy in stroke, aneurysm, and other neurovascular interventions, with a multicenter analysis showing a 94% success rate in reaching target sites without device-related complications.
-
In July 2025, Imperative Care announced that its Zoom 7X Aspiration Catheter received FDA 510(k) clearance for use in aspiration thrombectomy procedures for acute ischemic stroke. This device is the first in the Zoom Stroke Solution system to incorporate CenTRX technology, featuring a 96-degree pre-shaped tip designed to self-orient within the vessel, enhancing navigation and maximizing clot ingestion. The Zoom 7X catheter aims to streamline procedures, reduce the need for additional devices, and improve overall efficiency in treating large vessel occlusions.
-
In June 2025, Philips launched its SmartCT intelligent 3D imaging solution in Europe for the Azurion neuro biplane system, enabling real-time, high-resolution imaging in the Angio suite to accelerate stroke care and support timely neurovascular interventions.
-
In May 2025, Terumo Neuro announced the U.S. commercial availability of the SOFIA 88 Neurovascular Support Catheter, expanding its stroke treatment portfolio. This new large-bore catheter, with a 0.088-inch inner diameter, is engineered for reliability, procedural flexibility, and physician control. It builds upon the proven clinical performance and global legacy of the SOFIA Catheter line, which has supported more than 500,000 procedures across 170 countries over the past decade.
-
In May 2025, Zylox-Tonbridge Medical Technology received regulatory approval in Indonesia for its ZYLOX Unicorn Vascular Closure Device, China’s first domestically developed solution capable of handling sheath sizes from 5F to 22F, offering a two-unit pre-close technique for large-bore access sites, and marking a key step in expanding its presence in Southeast Asia.
-
In February 2025, Johnson & Johnson MedTech announced the launch of the CEREGLIDE 92 Catheter System, a next-generation .092” catheter featuring the INNERGLIDE 9 delivery aid. Designed for use in the neurovascular system, the CEREGLIDE 92 enables large distal access and reduces flow in the M1 during device insertion for acute ischemic stroke treatment. The catheter’s larger lumen also supports multiple neurovascular tools for treating aneurysms and other hemorrhagic complications, and it integrates seamlessly with Johnson & Johnson MedTech’s Stroke Solutions portfolio, including CEREGLIDE 71, PROWLER EX, and EMBOTRAP III.
-
In January 2025, Kaneka Corporation acquired 96.8% of the shares of EndoStream Medical Ltd., an Israeli medical equipment company. Combining Kaneka's manufacturing with ESM's technology, likely to develop new medical devices, mainly for cerebrovascular treatment, including the Nautilus device for aneurysm treatment, which is currently under development. We aim to achieve sales of over 20 billion yen by 2030.
-
In June 2024, Boston Scientific announced an agreement to acquire Silk Road Medical for USD 1.16 billion, strengthening its vascular portfolio with Silk Road’s transcarotid artery revascularization (TCAR) technology for minimally invasive stroke prevention.
-
In September 2022, Penumbra, Inc. announced that its RED Reperfusion Catheters received CE Mark approval and became commercially available in Europe. These catheters are part of the Penumbra System, a fully integrated mechanical aspiration thrombectomy system designed to restore blood flow in patients experiencing acute ischemic stroke (AIS). The approval enables broader access to Penumbra’s advanced stroke treatment solutions across European markets.
Neurointerventional Devices Market Report Scope
Report Attribute
Details
Market size value in 2025
USD 3.12 billion
Revenue forecast in 2033
USD 4.96 billion
Growth rate
CAGR of 5.9% from 2025 to 2033
Base year for estimation
2024
Historical data
2021 - 2023
Forecast period
2025 - 2033
Quantitative units
Revenue in USD million/billion and CAGR from 2025 to 2033
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Product, indication, end use, region
Regional scope
North America; Europe; Asia Pacific; Latin America; Middle East & Africa (MEA)
Country scope
U.S.; Canada; Mexico; UK; Germany; France; Italy; Spain; Denmark; Sweden; Norway; Japan; China; India; Australia; Thailand; South Korea; Brazil; Argentina; South Africa; Saudi Arabia; Kuwait; UAE.
Key companies profiled
Terumo Neuro; NeuroSafe Medical Co., Ltd; ZYLOX-TONBRIDGE MEDICAL TECHNOLOGY CO., LTD.; Brain Navi Biotechnology Co., Ltd.; Medtronic; Stryker; CERENOVUS (Johnson & Johnson); Integra LifeSciences Corporation; Penumbra Inc; Balt SAS; B. Braun SE; Phenox GmbH; Shanghai MicroPort Medical (Group) Co. Ltd.; Boston Scientific Corporation; Meril; Gem srl; Sirtex (BlackSwan Vascular, Inc.); INVAMED; Merit Medical Systems; Abbott; Acandis GmbH; Cook Medical; Imperative Care, Inc.; Rapid Medical; Koninklijke Philips N.V.; KANEKA CORPORATION
Customization scope
Free report customization (equivalent up to 8 analysts working days) with purchase. Addition or alteration to country, regional & segment scope.
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
Global Neurointerventional Devices Market Report Segmentation
This report forecasts revenue growth at global, regional, and country levels and provides an analysis of the latest industry trends in each of the sub-segments from 2021 to 2033. For this study, Grand View Research has segmented the global neurointerventional devices market report based on product, indication, end use, and region:
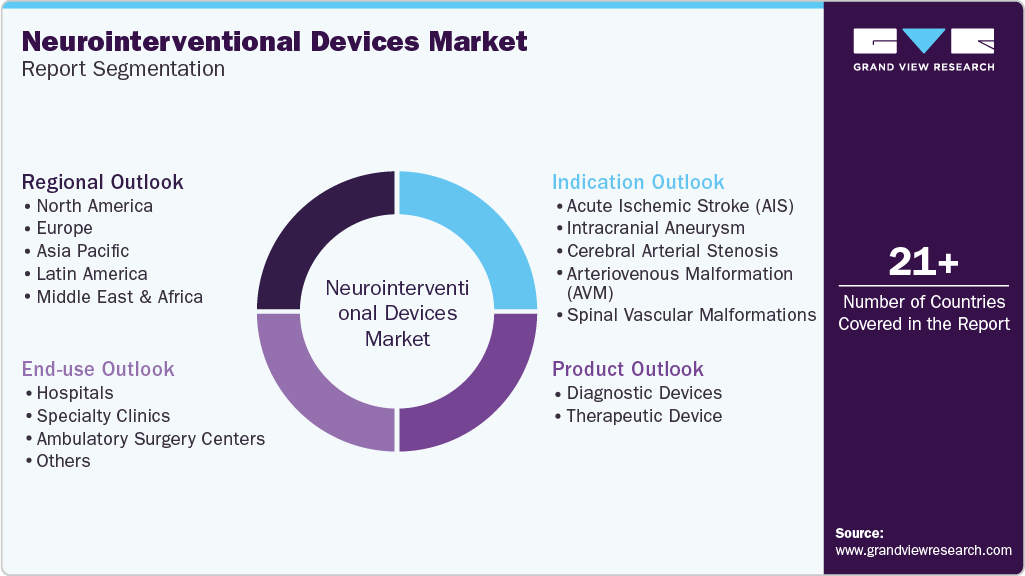
-
Product Outlook (Revenue, USD Million, 2021 - 2033)
-
Diagnostic Devices
-
Catheters
-
Distal Access Catheters
-
Microcatheters
-
Others
-
-
Guidewires
-
Imaging Systems
-
-
Therapeutic Devices
-
Embolic Materials
-
Embolic Coils
-
Embolic Agents
-
-
Neurovascular Stents
-
Self-Expanding Stents
-
Balloon-Expandable Stents
-
Flow Diverter Stents
-
Coil-Assist Stents
-
-
Neurothrombectomy Devices
-
Intrasaccular Devices
-
Embolic Protection Devices
-
Others
-
-
-
Indication Outlook (Revenue, USD Million, 2021 - 2033)
-
Acute Ischemic Stroke (AIS)
-
Intracranial Aneurysm
-
Cerebral Arterial Stenosis
-
Arteriovenous Malformation (AVM)
-
Spinal Vascular Malformations
-
-
End Use Outlook (Revenue, USD Million, 2021 - 2033)
-
Hospitals
-
Specialty Clinics
-
Ambulatory Surgery Centers
-
Others
-
-
Regional Outlook (Revenue, USD Million, 2021 - 2033)
-
North America
-
U.S.
-
Canada
-
Mexico
-
-
Europe
-
UK
-
Germany
-
France
-
Italy
-
Spain
-
Denmark
-
Sweden
-
Norway
-
-
Asia Pacific
-
Japan
-
China
-
India
-
Australia
-
Thailand
-
South Korea
-
-
Latin America
-
Brazil
-
Argentina
-
-
Middle East & Africa
-
South Africa
-
Saudi Arabia
-
Kuwait
-
UAE
-
-
Frequently Asked Questions About This Report
b. The global neurointerventional devices market size was estimated at USD 2.95 billion in 2024 and is projected to reach USD 3.12 billion by 2025.
b. The global neurointerventional devices market is projected to grow at a CAGR of 5.94% from 2025 to 2033 to reach around USD 4.96 billion by 2033.
b. On the basis of product, therapeutic devices held the largest revenue share in 2024 and is also expected to register the fastest growth over the forecast period. This dominance is driven by the rising prevalence of neurovascular disorders such as ischemic stroke, aneurysms, and arteriovenous malformations, coupled with increasing adoption of minimally invasive treatment procedures.
b. The key players includeTerumo Neuro, NeuroSafe Medical Co., Ltd, ZYLOX-TONBRIDGE MEDICAL TECHNOLOGY CO., LTD., Brain Navi Biotechnology Co., Ltd., Medtronic, Stryker, CERENOVUS (Johnson & Johnson), Integra LifeSciences Corporation, Penumbra, Inc, Balt SAS, B. Braun SE, Phenox GmbH, Shanghai MicroPort Medical (Group) Co. Ltd., Boston Scientific Corporation, Meril, Gem srl, Sirtex (BlackSwan Vascular, Inc.), INVAMED, Merit Medical Systems, Abbott, Acandis GmbH, Cook Medical, Imperative Care, Inc., Rapid Medical, and Koninklijke Philips N.V., among others
b. The neurointerventional devices market is primarily driven by the rising prevalence of stroke, brain aneurysms, vascular malformations and other neurovascular disorders, coupled with the rapidly growing aging population prone to such conditions. Increasing demand for minimally invasive procedures, which offer faster recovery and fewer complications than open surgery, further accelerates market growth.
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










