- Home
- »
- Medical Devices
- »
-
Neurovascular Devices Market Size, Industry Report, 2033GVR Report cover
![Neurovascular Devices Market Size, Share & Trends Report]()
Neurovascular Devices Market (2026 - 2033) Size, Share & Trends Analysis Report By Device (Neurothrombectomy Devices, Support Devices), By Therapeutic Applications (Stroke, Cerebral Artery), By End Use (Hospitals, Specialty Clinics), By Region, And Segment Forecasts
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
Market Size, 2025
$7.8BMarket Estimate, 2026
$8.2BMarket Forecast, 2033
$11.9BCAGR, 2026–2033
5.4%Neurovascular Devices Market Summary
The global neurovascular devices market size was estimated at USD 7.78 billion in 2025 and is projected to reach USD 11.91 billion by 2033, growing at a CAGR of 5.40% from 2026 to 2033. The market is driven by the rising prevalence of neurovascular disorders, advancements in medical technology, and an increasing focus on minimally invasive procedures.

Key Market Trends & Insights
- North America dominated the neurovascular devices market with the largest revenue share in 2025.
- By device, the cerebral embolization and aneurysm coiling devices segment led the market with the largest revenue share in 2025.
- By application, the stroke segment led the market with the largest revenue share in 2025.
- By end use, the hospitals segment led the market with the largest revenue share in 2025.
Market Size & Forecast
- 2025 Market Size: USD 7.78 Billion
- 2033 Projected Market Size: USD 11.91 Billion
- CAGR (2026-2033): 5.40%
- North America: Largest market in 2025
- Asia Pacific: Fastest-growing market
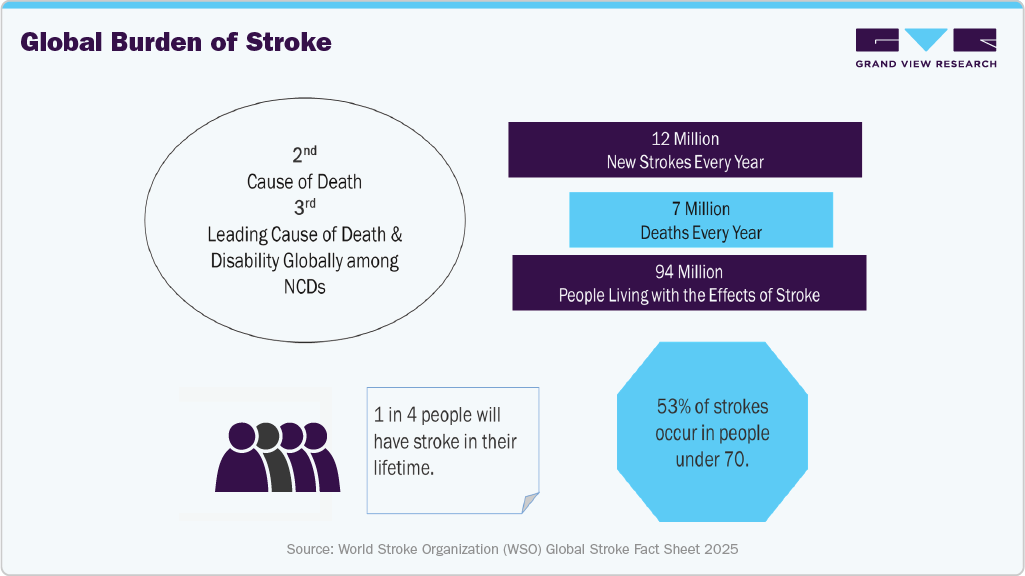
As conditions such as stroke, cerebral aneurysms, and Arteriovenous Malformations (AVM) become more common, the demand for effective treatment options also increases. The increasing prevalence of neurovascular diseases such as strokes and aneurysms is a major driving force behind the market. Various neurovascular devices, including clot retrieval devices, microcatheters, and flow diversion coils, are utilized to treat these conditions. According to the Global Stroke Fact Sheet 2024 published by the World Stroke Organization (WSO), more than 12 million new strokes occur annually, and about 25% of the global population will experience a stroke in their lifetime.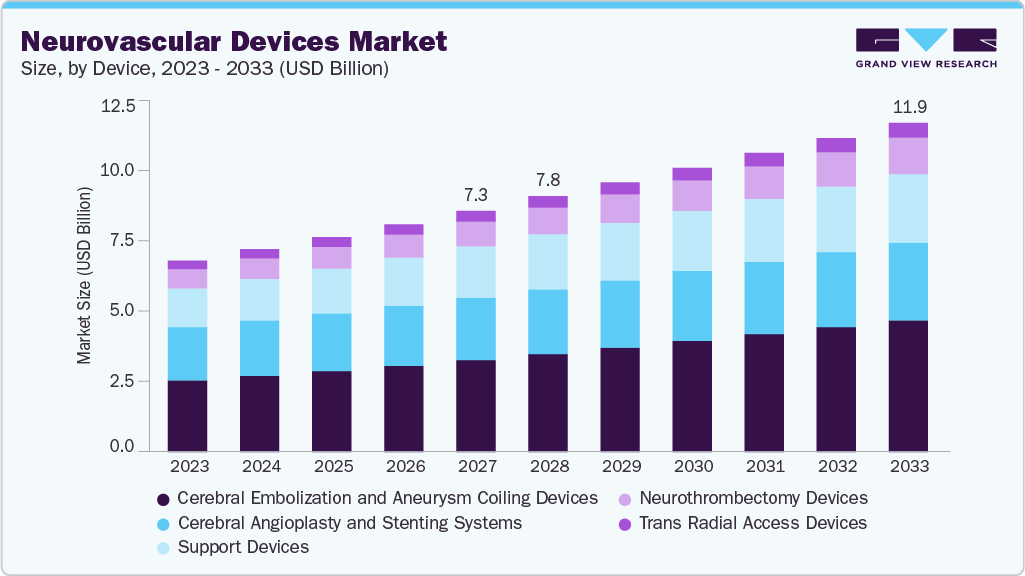
The burden of neurovascular disorders such as strokes is increasing significantly, which is anticipated to propel the demand for neurovascular devices such as neurothrombectomy devices like clot retrievers and suction devices, among others.
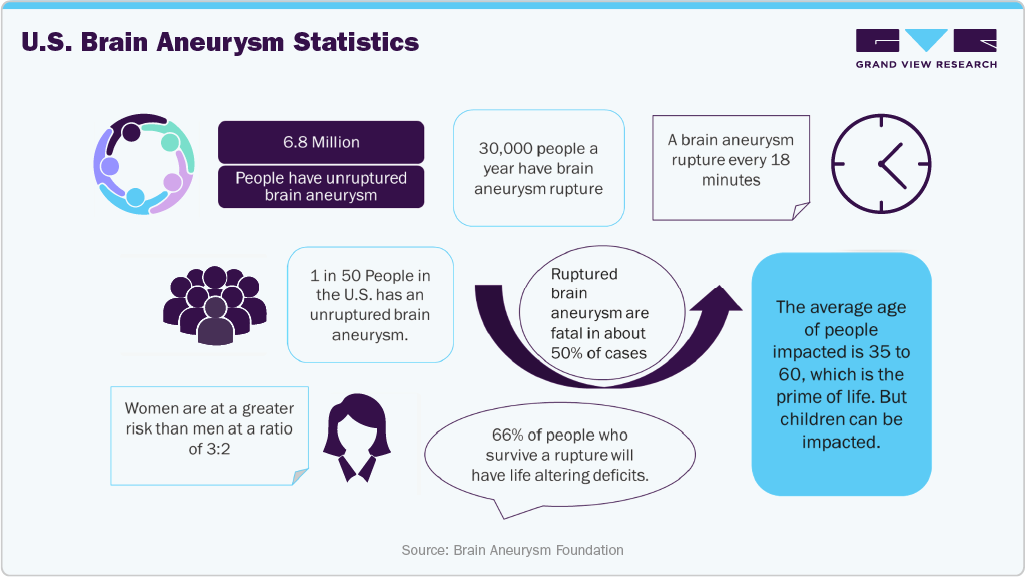
In addition, data released by the Brain Aneurysm Foundation in August 2024 indicates that an unruptured brain aneurysm affects an estimated 6.8 million people in the U.S., with around 30,000 individuals suffering a brain aneurysm rupture each year. As a result, the significant number of patients affected by neurovascular diseases and the increasing use of neurovascular devices in their treatment are expected to drive market growth in the coming years.
Moreover, companies are using innovative technologies to develop neurovascular devices. For instance, in June 2024, Penumbra launched BMX 81 and BMX 96 neuro access catheters in Europe. These catheters are manufactured utilizing laser-cut stainless steel hypo tube technology to provide trackability and stability. Such product developments incorporating advanced technology are expected to drive market growth over the forecast period.
The market continues to expand, driven by rising procedural volumes, increasing stroke prevalence, and continuous technological advancements across interventional neurology. Market growth is supported by the strong adoption of minimally invasive therapies, particularly within the neurovascular thrombectomy devices segment, where demand is accelerating due to the growing clinical emphasis on rapid ischemic stroke intervention. The broader neurovascular devices landscape is further shaped by ongoing innovations in embolization, aneurysm management, and procedural support technologies. Segments including neurovascular embolization devices, neurovascular coiling assist devices, and neurovascular accessory devices play a critical role in enabling complex neurointerventional procedures. In addition, the expanding clinical focus on acute stroke management continues to reinforce demand for neurovascular devices used in ischemic stroke treatment, supporting sustained long-term market growth.
Key Opinion Leaders
Company Name
KOLs
Growth Opportunities
Dr. Zaidat, Director of the Neuroscience and Stroke Center at Mercy Hospital in Toledo, Ohio.
“I’m privileged to be one of the first users of the new Cerepak detachable coils. The advancement of greater tools in neurology may improve patient outcomes, and my team looks forward to continuing the utilization of Cerepak coils in our treatment plans.”
-
- Early adoption of innovative tools
- Growing demand for advanced neurology solutions
Joan Kristensen, head of the Europe, Middle East, and Africa region for Penumbra, Inc.
“Physicians in Europe now have more options to serve their patients using our expanded portfolio. With five innovative devices introduced in Europe in a matter of months, our dedication to improving stroke care enables us to provide physicians with customised stroke solutions. In all modesty, Penumbra provides physicians with the widest aspiration thrombectomy stroke portfolio on the market.”
-
- Demand for customized devices
- Expansion in European markets
- Growth in thrombectomy adoption
Jeff Hopkins, CEO of FlowPhysix.
"Our game-changing technology has the potential to alter the current standard of care for thrombectomy. This partnership provides FlowPhysix with an expanded reach in delivering our unique solutions to patients worldwide. We strongly believe that 3comma's proven leadership and deep expertise will drive commercial success internationally and increase shareholder value for the company."
-
- Global expansion of thrombectomy solutions
- Increased access to innovative technologies worldwide
- Strengthened international market presence
Source: Grand View Research Analysis
Market Characteristics
The market growth stage is high, and the pace of growth is accelerating. The market is characterized by a high degree of growth due to the increasing prevalence of neurovascular diseases such as strokes and aneurysms.
The neurovascular devices industry demonstrates a high degree of innovation, driven by continuous advancements in device engineering, material science, and procedural precision. Industry participants increasingly prioritize technological differentiation through improved deliverability, enhanced safety profiles, and optimized clinical performance. Innovations in thrombectomy systems, embolization platforms, and coiling assist technologies highlight the focus on addressing complex neurovascular anatomies. In addition, research collaborations and clinical validation efforts support the introduction of next-generation solutions, reinforcing competitive dynamics and accelerating the adoption of advanced minimally invasive neurointerventional therapies.
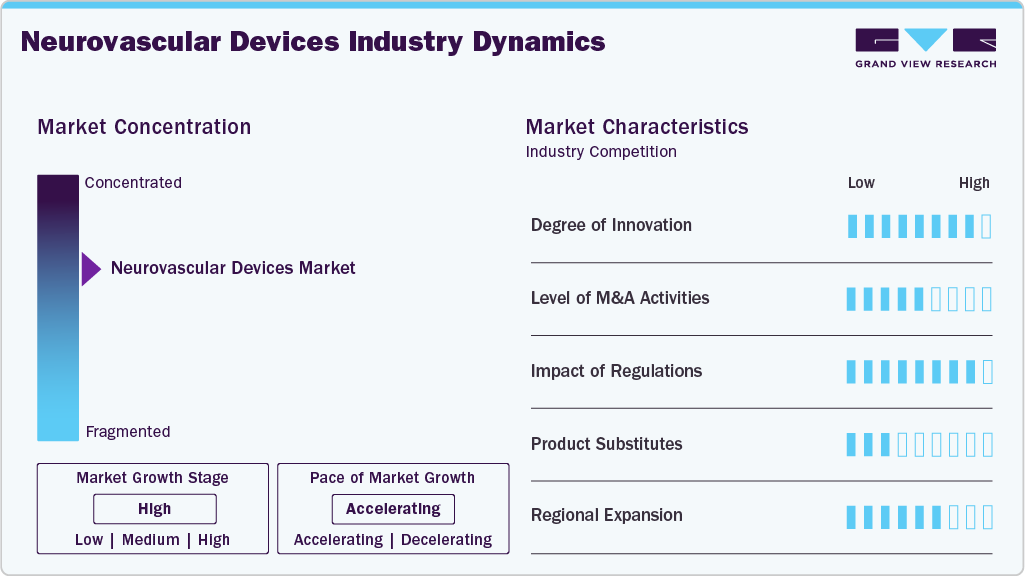
Regulatory bodies, such as the European Medicines Agency (EMA) and the U.S. Food and Drug Administration (FDA), play a crucial role in shaping the regulatory framework for the neurovascular devices industry. Stricter requirements for clinical trials and post-market surveillance ensure patient safety and product efficacy, but they can also pose challenges for companies seeking approvals.
U.S. FDA Classification For Neurovascular Devices:
Classification
Product Category
Class II
Embolization Device
Class III
Intracranial Aneurysm Flow Diverter
Class III
Stent, carotid
Class II
Neurothrombectomy Devices
Source: U.S. Food and Drug Administration
The market is poised to experience robust mergers and acquisitions (M&A) activity as companies seek to expand their product portfolios, leverage synergies, and enter new markets. Established players are acquiring smaller innovators to gain access to advanced technologies. For instance, in January 2025, Kaneka Corporation acquired 96.8% of shares in EndoStream Medical Ltd (ESM), a company developing the innovative Nautilus device for treating cerebral aneurysms.
Neurovascular device companies are increasingly focusing on regional expansion to meet growing demand. In September 2024, FlowPhysix Inc. collaborated with 3comma Medical GmbH to serve as the international commercial partner for the FLOWRUNNER Aspiration System. This collaboration will offer global access to advanced thrombectomy technology, allowing FlowPhysix to concentrate on U.S. commercialization while 3comma drives international market growth.
Device Insights
The cerebral embolization and aneurysm coiling devices segment held the largest share in 2025. These devices are further categorized into embolic coils, flow diversion coils, and liquid embolic agents. Ongoing research and development of advanced solutions, such as smart aneurysm coils, are expected to drive segment growth. For instance, in November 2024, a study published by the National Library of Medicine evaluated the safety and efficacy of the SMART coil system for treating brain aneurysms. This next-generation embolic coil includes a detachment pusher, bare platinum coil, and detachment handle. The study found the SMART coil to be a safe and effective option for managing intracranial aneurysms, offering advantages over existing treatment methods. These promising clinical results are anticipated to fuel embolic device growth further.
The neurothrombectomy devices segment is expected to grow fastest during the forecast. Industry players' increasing focus on expanding their product portfolios and the rising prevalence of acute ischemic stroke are expected to drive the segment's growth in the coming years.
Some of the recently launched neurothrombectomy devices are mentioned below:
Company Name
Product Name
Launch Date
Emboa Medical
Thrombus Retrieval Aspiration Platform (TRAP Catheter)
February 2025
CERENOVUS, Inc., part of Johnson & Johnson MedTech
CEREGLIDE 71 Intermediate Catheter
February 2024
MicroVention
Eric Retrieval Device
June 2023
Source: Grand View Research Analysis
Application Insights
The stroke segment held the largest share in 2025. This dominance can be attributed to the growing prevalence of stroke across the globe. Moreover, healthcare facilities and industry players are focusing on developing innovative neurovascular devices that can help treat strokes. For instance, in November 2024, sixteen Indian hospitals, led by AIIMS Delhi, began testing a new stent-retriever for acute ischemic stroke, developed by U.S.-based Gravity Medical Technology. The GRASSROOT trial aims to evaluate this device, designed specifically for stroke clot characteristics in Indian patients, addressing a significant health burden in India. These developments of products for stroke treatment are anticipated to drive the segment growth.
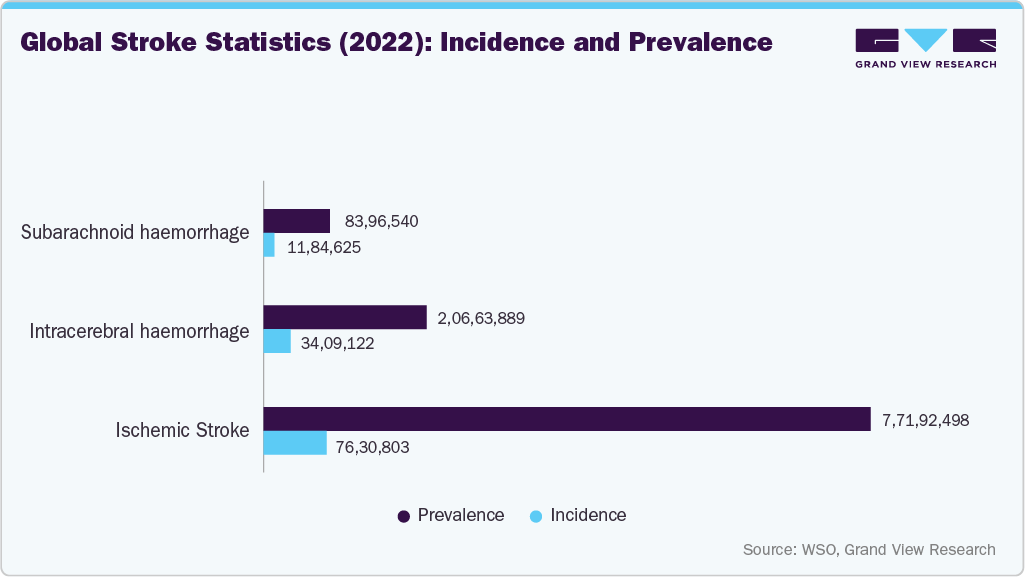
The cerebral aneurysm segment is projected to witness the fastest growth during the forecast period, driven by increasing incidental detection rates, rising adoption of minimally invasive endovascular therapies, and continuous advancements in embolization technologies. Growing clinical preference for coil embolization and flow diversion procedures, coupled with expanding neurointerventional capabilities, supports segment expansion. In addition, improving diagnostic imaging utilization and heightened awareness of aneurysm rupture risks continue to accelerate procedural volumes, reinforcing strong demand for advanced neurovascular treatment solutions.
End Use Insights
The hospital segment dominated the market in 2025, accounting for the largest share. This dominance is primarily driven by the high volume of neurovascular procedures performed in hospitals, the availability of advanced infrastructure, and the presence of skilled healthcare professionals. In addition, the growing number of hospital-based clinical trials and collaborations with medical device companies further strengthens the hospital segment's position in the market.
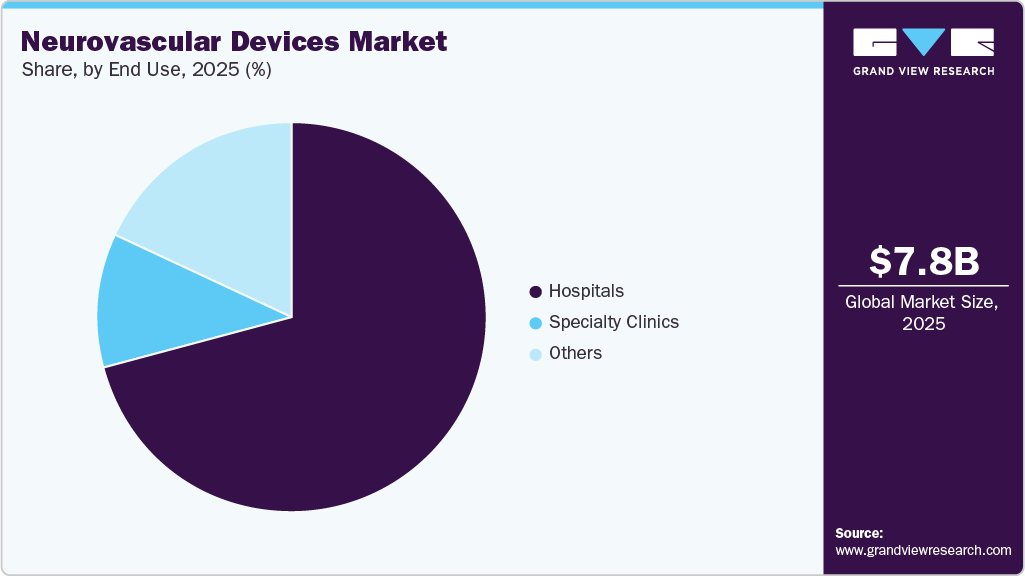
The other segment is projected to witness notable market growth from 2026 to 2033. This segment comprises ambulatory surgery centers (ASCs), emergency care centers, and long-term care facilities. Growth is primarily driven by the expanding number of ASCs and the increasing shift toward minimally invasive, outpatient neurointerventional procedures. In addition, rising investments in modern healthcare infrastructure and the growing emphasis on procedural efficiency, cost optimization, and patient-centric care models continue to support demand within these alternative treatment settings.
Regional Insights
North America accounted for the largest share of the neurovascular devices market in 2025, supported by advanced healthcare infrastructure, high procedural volumes, and strong adoption of minimally invasive neurointerventional therapies. The region benefits from widespread availability of specialized stroke centers, favorable reimbursement frameworks, and early integration of innovative technologies. Rising prevalence of ischemic stroke and cerebral aneurysms further sustains demand for thrombectomy, embolization, and coiling assist devices. Continuous product innovation and the strong presence of leading device manufacturers reinforce North America’s dominant market position.
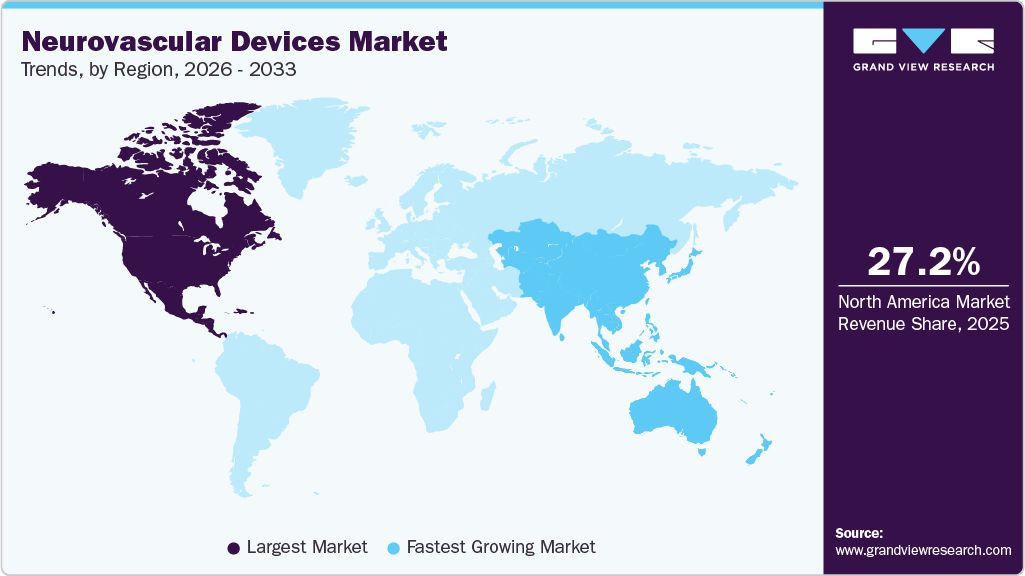
U.S. Neurovascular Devices Market Trends
The neurovascular devices market in the U.S. represents a leading segment within interventional medicine, driven by high stroke prevalence, advanced healthcare infrastructure, and strong adoption of minimally invasive neurointerventional procedures. Growth is supported by increasing utilization of mechanical thrombectomy, embolization, and aneurysm management technologies. Continuous innovation in device design, imaging integration, and procedural precision further accelerates market expansion. Favorable reimbursement frameworks, expanding neurointerventional specialist availability, and rising awareness of early stroke intervention contribute to sustained demand, positioning the U.S. as a key contributor to global neurovascular device revenues.
Canada neurovascular devices market is expected to grow significantly in the coming years. The country's healthcare coverage and compulsory insurance enable the adoption of advanced technologies. In addition, increasing awareness and the presence of organizations such as the Canadian Neurological Sciences Federation and the Canadian Neurovascular Health Society, which focus on neurovascular research, are expected to support market growth in the future.
Europe Neurovascular Devices Market Trends
The neurovascular devices market in Europe is witnessing substantial growth, fueled by a rising geriatric population and rapid advancements in neurovascular technology. The increasing adoption of innovative devices and a growing number of neurovascular surgeries across the region are further driving demand. In addition, supportive healthcare infrastructure and growing awareness of stroke management contribute to market expansion.
The UK neurovascular devices market is expected to grow significantly, driven by rising awareness about acute ischemic stroke treatment and the strong presence of key industry players. According to NHS England, stroke admissions in England increased to 111,137 in 2023/24, highlighting the growing demand for effective neurovascular interventions.
The neurovascular devices market in Germany represents a key segment within Europe, supported by advanced healthcare infrastructure, strong clinical expertise, and high adoption of minimally invasive neurointerventional procedures. Growth is driven by consistent demand for thrombectomy, embolization, and aneurysm management technologies. The country benefits from a well-established network of specialized stroke centers and interventional facilities. Continuous technological integration, favorable reimbursement mechanisms, and growing procedural volumes further reinforce Germany’s position as a major contributor to the European neurovascular devices landscape.
Asia Pacific Neurovascular Devices Market Trends
The neurovascular devices market in the Asia Pacific is expected to register the fastest growth, driven by the high prevalence of neurological disorders and rapid advancements in healthcare infrastructure. The availability of innovative products and a large patient population affected by brain aneurysms and strokes further contributes to this growth. In addition, the rising demand for advanced yet cost-effective healthcare solutions is creating substantial regional opportunities.
China neurovascular devices market is growing with a lucrative growth rate, owing to the presence of several local & key players in the market and an increase in the number of stroke cases. For instance, a study published in JAMA Network Open in March 2023 estimated the incidence and mortality of stroke in China. The study found that the overall stroke incidence was 500 per 100,000 person-years for individuals older than 40 years. Thus, the rising incidence of stroke in China is expected to boost the country's market in the coming years.
The neurovascular devices market in Indiais experiencing significant growth, driven by the increasing adoption of advanced technologies, rising clinical trials, and the development of innovative devices. In addition, an increase in domestic manufacturing is boosting the market. For instance, in March 2025, the Technology Development Board (TDB) authorized funding for S3V Vascular Technologies Limited, Mysuru, to establish India's first indigenous mechanical thrombectomy device manufacturing facility. This initiative marks a significant step toward strengthening the country's neurovascular capabilities.
Latin America Neurovascular Devices Market Trends
The neurovascular devices market in Latin America is driven by increasing government investments in healthcare infrastructure, rising awareness of neurological disorders, and growing demand for advanced medical technologies. Countries like Brazil are expanding healthcare budgets, improving access to high-cost neurovascular devices in public hospitals. In addition, the rising incidence of stroke and brain aneurysms, coupled with improvements in diagnostic capabilities and minimally invasive treatment options, is further accelerating market growth across the region.
Middle East and Africa Neurovascular Devices Market Trends
The neurovascular devices market in the MEA is experiencing steady growth, primarily driven by the rising incidence of neurovascular diseases such as strokes, aneurysms, and other cerebrovascular conditions. This surge is primarily attributed to the increasing prevalence of risk factors, including hypertension, diabetes, obesity, and sedentary lifestyles. As these health challenges become more widespread across the region, the demand for effective neurovascular interventions and advanced medical devices continues to grow.
UAE neurovascular devices market is growing rapidly, driven by rising stroke incidence and significant investments in advanced healthcare infrastructure. Annually, 8,000 to 10,000 people in the UAE suffer strokes, often resulting in long-term disability. Facilities like Sheikh Shakhbout Medical City (SSMC) are enhancing stroke care. In June 2022, SSMC launched a state-of-the-art Biplane Angio Suite for diagnosing and treating ischemic strokes. This service, part of its Interventional Neuroradiology division, is vital in improving stroke outcomes across Abu Dhabi.
Key Neurovascular Devices Company Insights
Medtronic, Stryker, Johnson & Johnson and its affiliates, Penumbra, Inc., MicroPort Scientific Corporation, Terumo Corporation, ZYLOX-TONBRIDGE MEDICAL TECHNOLOGY CO., LTD., NeuroSafe Medical Co., Ltd., Rapid Medical, phenox GmbH, Lepu Medical Technology(Beijing) Co., Ltd., Evasc Neurovascular Enterprises, and Acandis GmbH are some of the major market players. Companies are launching novel products and also focusing on gaining regulatory approvals. Moreover, industry players are also forming distribution partnerships to gain a competitive advantage.
Key Opinion Leader
-
“The TRAP design demonstrated a greater than 200% increase in blockage removal force compared to a traditional catheter. In addition, the TRAP catheter showed significant benefits in removing clots on the first attempt in a worst-case neurovascular model. It achieved a 40% success rate compared to 10% for conventional smooth inner diameter catheters.” Said CEO Ángel Enríquez of Emboa Medical
-
"I’ve really enjoyed getting to use and know EVO. With improved visibility and the ability to shelf the stent across the aneurysm neck, it’s become my tool-of-choice for stent-assisted coiling," said Dr. Charles Matouk, Vice Chair of the Department of Neurosurgery at Yale University.

Key Neurovascular Devices Companies:
The following key companies have been profiled for this study on the neurovascular devices market.
- Medtronic
- Stryker
- Johnson & Johnson and its affiliates
- Penumbra, Inc.
- MicroPort Scientific Corporation
- Terumo Corporation
- ZYLOX-TONBRIDGE MEDICAL TECHNOLOGY CO., LTD.
- NeuroSafe Medical Co., Ltd.
- Rapid Medical
- Evasc Neurovascular Enterprises
- Lepu Medical Technology(Beijing) Co., Ltd.
- Acandis GmbH
- phenox GmbH (Wallaby Medical)
Recent Developments
-
In January 2026, Boston Scientific Corporation announced an agreement to acquire Penumbra, Inc. The acquisition strengthens Boston Scientific’s cardiovascular portfolio and provides entry into fast-growing mechanical thrombectomy and neurovascular segments.
-
In January 2026, Zylox-Tonbridge Medical Technology Co., Ltd., a Hong Kong-listed company specializing in neurovascular and peripheral vascular interventional devices, agreed to acquire German medical technology firm Optimed Medizinische Instrumente GmbH.
-
In February 2025, Emboa Medical Inc., a medical device startup, introduced TRAP (Thrombus Retrieval Aspiration Platform), a biomimetic catheter inspired by a boa constrictor’s teeth. Designed to enhance clot retrieval without fragmentation, this innovative device has shown promising results in improving stroke treatment outcomes.
-
In January 2025, Lauxera Capital Partners invested in German neurovascular medical device company Acandis GmbH. The transaction represents the first investment from Lauxera Growth II, the firm’s second-generation healthcare growth buyout fund.
-
In June 2024, Terumo Corporation introduced the LVIS EVO Intraluminal Support Device for treating wide-neck intracranial aneurysms in the U.S. market.
Neurovascular Devices Market Report Scope
Report Attribute
Details
Market size value in 2026
USD 8.24 billion
Revenue forecast in 2033
USD 11.91 billion
Growth rate
CAGR of 5.40% from 2026 to 2033
Actual data
2021 - 2025
Forecast period
2026 - 2033
Quantitative units
Revenue in USD million/billion and CAGR from 2026 to 2033
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Device, therapeutic application, end use, region
Regional scope
North America; Europe; Asia Pacific; Latin America; MEA
Country scope
U.S.; Canada; Mexico; UK; Germany; France; Italy; Spain; Denmark; Sweden; Norway; Japan; China; India; South Korea; Australia; Thailand; Brazil; Argentina; South Africa; Saudi Arabia; UAE; Kuwait
Key companies profiled
Medtronic; Stryker; Johnson & Johnson and its affiliates; Penumbra, Inc.; MicroPort Scientific Corporation; Terumo Corporation; ZYLOX-TONBRIDGE MEDICAL TECHNOLOGY CO., LTD.; NeuroSafe Medical Co., Ltd.; Rapid Medical; phenox GmbH; Lepu Medical Technology (Beijing) Co., Ltd.; Evasc Neurovascular Enterprises; Acandis GmbH; phenox GmbH
Customization scope
Free report customization (equivalent up to 8 analysts working days) with purchase. Addition or alteration to country, regional & segment scope.
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
Global Neurovascular Devices Market Report Segmentation
This report forecasts revenue growth at global, regional, and country levels and provides an analysis of the industry trends in each of the sub-segments from 2021 to 2033. For this study, Grand View Research has segmented the global neurovascular devices market report based on device, application, end use, and region:

-
Device Outlook (Revenue USD Million, 2021 - 2033)
-
Cerebral Embolization and Aneurysm Coiling Devices
-
Embolic coils
-
Flow diversion devices
-
Liquid embolic agents
-
-
Cerebral Angioplasty and Stenting Systems
-
Carotid artery stents
-
Embolic protection systems
-
-
Neurothrombectomy Devices
-
Clot retrieval devices
-
Suction devices/aspiration catheters
-
Vascular snares
-
-
Support Devices
-
By Size
-
0.027"
-
0.021"
-
0.071"
-
0.017"
-
0.019"
-
0.013"
-
0.058"
-
0.068"
-
Others
-
-
By Product
-
Micro catheters
-
Micro guidewires
-
-
-
Trans Radial Access Devices
-
-
Therapeutic Application Outlook (Revenue USD Million, 2021 - 2033)
-
Stroke
-
Cerebral Artery
-
Cerebral Aneurysm
-
Aneurysmal Subarachnoid Hemorrhage
-
Others
-
-
Others
-
-
End Use Outlook (Revenue USD Million, 2021 - 2033)
-
Hospitals
-
Specialty Clinics
-
Others
-
-
Regional Outlook (Revenue USD Million, 2021 - 2033)
-
North America
-
U.S.
-
Canada
-
Mexico
-
-
Europe
-
Germany
-
UK
-
France
-
Italy
-
Spain
-
Norway
-
Denmark
-
Sweden
-
-
Asia Pacific
-
Japan
-
China
-
India
-
Australia
-
South Korea
-
Thailand
-
-
Latin America
-
Brazil
-
Argentina
-
-
Middle East and Africa (MEA)
-
South Africa
-
Saudi Arabia
-
UAE
-
Kuwait
-
-
Frequently Asked Questions About This Report
The neurovascular devices market is driven by the rising prevalence of neurovascular disorders, advancements in medical technology, and an increasing focus on minimally invasive procedures. As conditions such as stroke, cerebral aneurysms, and Arteriovenous Malformations (AVM) become more common, the demand for effective treatment options also increases.
Based on device, the cerebral embolization and aneurysm coiling devices segment held the largest share of the neurovascular devices market in 2025. These devices are further categorized into embolic coils, flow diversion coils, and liquid embolic agents.
The stroke segment held the largest share of the neurovascular devices market in 2025. This dominance can be attributed to the growing prevalence of stroke across the globe.
Key companies in the global neurovascular devices market include Medtronic, Stryker, Johnson & Johnson and its affiliates, Penumbra, Inc., MicroPort Scientific Corporation, Terumo Corporation, ZYLOX-TONBRIDGE MEDICAL TECHNOLOGY CO., LTD., NeuroSafe Medical Co., Ltd., Rapid Medical, phenox GmbH, Lepu Medical Technology(Beijing)Co.,Ltd., Evasc Neurovascular Enterprises , and Acandis GmbH are some of the major players in the neurovascular devices market.
The global neurovascular devices market size was estimated at USD 7.78 billion in 2025 and is expected to reach USD 8.24 billion in 2026.
The global neurovascular devices market is projected to grow at a CAGR of 5.40% from 2026 to 2033 to reach USD 11.91 billion by 2033.
About the Author(s)
Medical Devices Research Team
Healthcare · Medical DevicesThis report was authored by the medical devices Research Team at Grand View Research - comprising two research analysts, one senior research analyst, and one industry expert - with specialized expertise in the medical devices segment of the healthcare industry. All findings are based on proprietary healthcare databases, executive interviews, and regulatory analysis, subject to internal peer review prior to publication.
Last Updated:
Speak to Author
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.









