- Home
- »
- Medical Devices
- »
-
North America 2-way Foley Catheter Market Report, 2035GVR Report cover
![North America 2-way Foley Catheter Market Size, Share & Trends Report]()
North America 2-way Foley Catheter Market (2026 - 2035) Size, Share & Trends Analysis Report By Material (Silicone 2-way Foley Catheters, Latex 2-way Foley Catheters), By Indication, By Usage Type (Short-Term Use, Long-Term Use), By End-use, And Segment Forecasts
- Report ID: GVR-4-68040-865-0
- Number of Report Pages: 150
- Format: PDF
- Historical Range: 2018 - 2025
- Forecast Period: 2026 - 2035
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
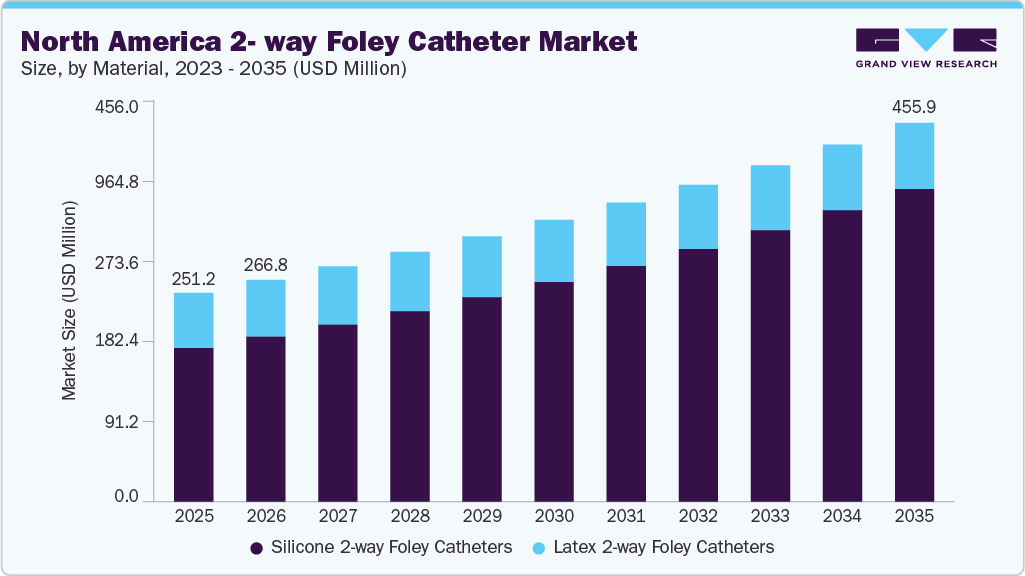
North America 2-way Foley Catheter Market Summary
The North America 2-way foley catheter market size was estimated at USD 251.20 million in 2025 and is projected to reach USD 455.92 million by 2035, growing at a CAGR of 6.14% from 2026 to 2035. The market growth is primarily driven by the rising prevalence of urological disorders, including benign prostatic hyperplasia, urinary incontinence, chronic kidney disease, and prostate cancer.
Key Market Trends & Insights
- The U.S. dominated the North America 2-way foley catheters market with the largest revenue share in 2025.
- By indication, urinary incontinence dominates the North America 2-way Foley catheters market in 2025.
- By material, silicone 2-way foley catheters segment led the market with the largest revenue share in 2025.
- By usage type, the long-term use segment led the market with the largest revenue share in 2025.
- By end use, the hospitals segment led the market with the largest revenue share in 2025.
Market Size & Forecast
- 2025 Market Size: USD 251.20 Million
- 2035 Projected Market Size: USD 455.92 Million
- CAGR (2026-2035): 6.14%
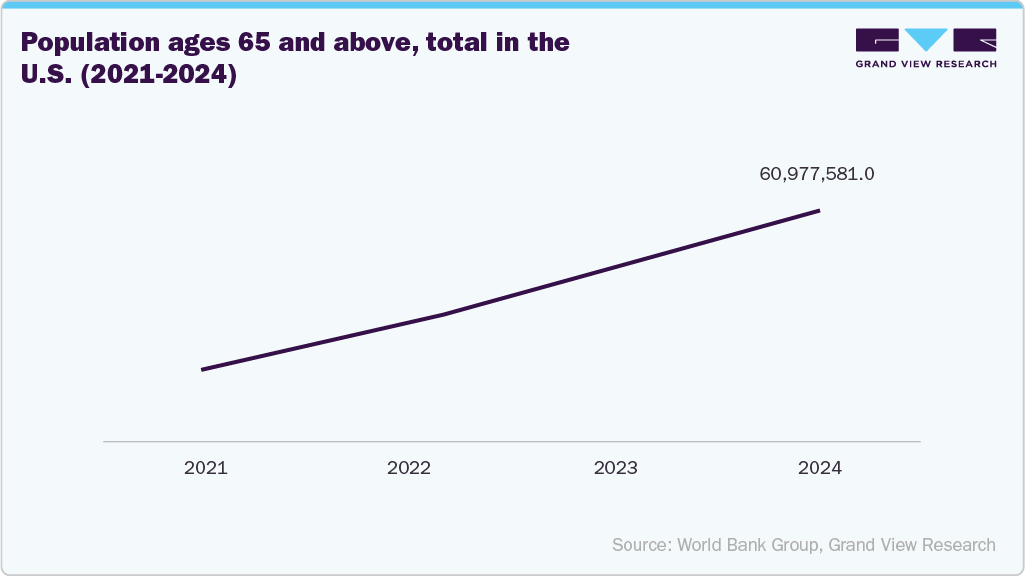
The expanding geriatric population in the U.S. and Canada further increases the need for short- and long-term catheterization. In addition, growing surgical procedures and hospital admissions support sustained demand for urinary drainage solutions. Technological advancements such as silicone-based and antimicrobial-coated catheters also contribute to market growth by enhancing infection prevention and patient safety.The increasing prevalence of urological diseases is a major driver for the North America 2-way foley catheters industry. The growing cases of urinary incontinence, enlarged prostate gland/benign prostatic hyperplasia, spinal cord injury, chronic kidney disease, and bladder dysfunction are significantly increasing the need for both short- and long-term catheterization. The expanding geriatric population in the U.S. and Canada, which is more prone to these conditions, further strengthens demand. Consequently, rising urological procedures and hospital admissions are supporting sustained regional market growth.
The rising prevalence of spinal cord injuries in North America contributes significantly to the demand for 2-way foley catheters, as many patients develop neurogenic bladders requiring long-term urinary drainage. This sustained need for chronic catheterization supports consistent product utilization and drives market growth, particularly in rehabilitation and long-term care settings. For instance, according to the United Spinal Association, in September 2025, an estimated 250,000 to 390,000 Americans were living with a spinal cord injury, with approximately 18,000 new cases reported annually, reflecting a significantly increasing patient population requiring ongoing medical management and supportive care.
Causes of Spinal Cord Injury in the U.S.
Cause
Percentage of Cases
Vehicle accidents
39.30%
Falls
31.80%
Violence
13.20%
Sports-related injuries
8.00%
Medical or surgical causes
4.30%
Source: United Spinal Association
Benign prostatic hyperplasia (BPH), a common age-related enlargement of the prostate in men, significantly contributes to urinary retention and other lower urinary tract symptoms that often require continuous bladder drainage. In North America, the high and increasing prevalence of BPH, especially among older males, leads to frequent clinical scenarios where catheterization is necessary to relieve obstruction, manage postoperative care, or support long-term urinary management. 2-way Foley catheters are the standard indwelling urinary drainage device for routine cases. The widespread prevalence of BPH directly increases demand for them across hospitals, long-term care facilities, and outpatient settings in the region. This trend, alongside a large aging population and well-established healthcare infrastructure, is helping to grow the 2-way Foley catheters market in North America.
Catheter-Associated Urinary Tract Infection (CAUTI) prevention initiatives across North America present a significant opportunity for the 2-way foley catheters market, as hospitals prioritize products that reduce infection risk and improve patient outcomes. Under value-based reimbursement models in the U.S., healthcare providers face financial penalties for hospital-acquired infections, driving demand for advanced silicone and antimicrobial-coated 2-way foley catheters. This shift encourages procurement of evidence-backed, low-complication devices, creating growth potential for manufacturers offering clinically differentiated solutions.
U.S. Hospital Infection Statistics - CAUTI by State and Location, 2024
2024
Catheter-associated urinary tract infections (CAUTI)
Inpatient Locations
State
Total
ICU
Wards
Alabama
531
146
385
Alaska
56
10
46
Arizona
468
100
368
Arkansas
325
65
260
California
2,667
566
2,101
Colorado
408
80
328
Connecticut
Upgrade the report license to gain access to the complete analysis.
D.C.
Delaware
Florida
Georgia
Guam
Hawaii
Idaho
Illinois
Indiana
Iowa
Kansas
Kentucky
Louisiana
Maine
Maryland
Massachusetts
Michigan
Minnesota
Mississippi
Missouri
Montana
Nebraska
Nevada
New Hampshire
New Jersey
New Mexico
New York
North Carolina
North Dakota
Ohio
Oklahoma
Oregon
Pennsylvania
Puerto Rico
Rhode Island
South Carolina
South Dakota
Tennessee
Texas
Utah
Vermont
Virgin Islands
Virginia
Washington
West Virginia
Wisconsin
Wyoming
All US
28,427
5,721
22,706
Source: CDC, 2026
Market Concentration & Characteristics
The North America 2-way Foley catheter market is growing, primarily driven by the high prevalence of urinary retention conditions such as benign prostatic hyperplasia, increasing surgical procedures, and sustained hospitalization volumes. The growing focus on infection prevention, particularly the reduction of catheter-associated urinary tract infections, has strengthened demand for high-quality indwelling catheter solutions. Hospitals currently account for the largest share of the market due to acute care catheter use, while long-term care facilities and home healthcare settings are emerging as essential growth segments, supported by an aging population and the chronic disease burden.
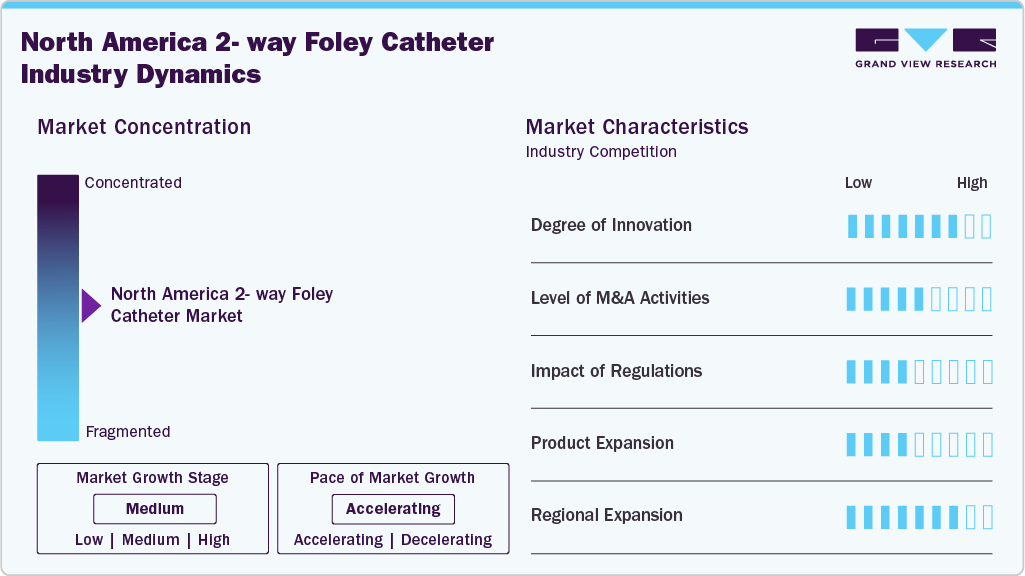
The degree of innovation in the North America 2-way Foley catheters market is moderate. Since the fundamental design of 2-way Foley catheters (drainage lumen + balloon inflation lumen) is well established, most advancements focus on material improvement, infection reduction, and patient comfort rather than structural redesign.
Key innovation areas include:
-
Material advancements: Shift from latex to 100% silicone to reduce allergic reactions, encrustation, and long-term complications.
-
Antimicrobial technologies: Silver coatings, antibiotic-impregnated surfaces, and antimicrobial hydrogels to reduce CAUTI risk.
-
Surface coatings: Hydrogel and silicone elastomer coatings to improve insertion comfort and reduce urethral trauma.
-
Closed drainage integration: Enhanced closed systems to minimize infection risk during handling.
- Improved balloon and tip designs: Better retention stability and reduced mucosal irritation.
Regulations have a significant impact on the North America market for 2-way Foley catheters, shaping product approval, manufacturing standards, infection-control practices, and competitive dynamics. In the U.S., 2-way Foley catheters are classified as Class II medical devices. They are regulated by the U.S. Food and Drug Administration under the 510(k) pathway, requiring manufacturers to demonstrate substantial equivalence, sterility validation, biocompatibility, and performance compliance before commercialization. Additionally, infection prevention guidelines issued by the Centers for Disease Control and Prevention, along with hospital reimbursement policies linked to CAUTI reduction, strongly influence procurement and usage patterns. In Canada, Health Canada's oversight of product approvals and licensing further supports safety and quality standards.
The level of mergers and acquisitions (M&A) activity in the North America 2-way Foley catheters industry has been moderate and strategically driven rather than highly consolidative in recent years. As established medical device companies dominate the market, acquisitions are typically aimed at portfolio expansion, strengthening distribution, or entering adjacent urology and infection-prevention segments rather than purely volume-driven consolidation.
-
In November 2025, Amsino International Inc. announced the successful completion of its acquisition of the assets of MedXL Inc. and Liebel-Flarsheim Canada Inc., both located in Montreal, Quebec, Canada. Through this transaction, Amsino significantly expands its North American manufacturing footprint and effectively doubles its production capacity.
Regional expansion in the North America 2-way Foley catheters market is primarily driven by strategic distribution strengthening, portfolio expansion, and deeper penetration into non-acute care settings, rather than by entering entirely new geographic territories. Regional expansion in this market reflects competitive positioning and channel optimization within a mature healthcare ecosystem rather than rapid geographic diversification.
-
In October 2024,HR HealthCare, a U.S. provider of healthcare consumables, announced the launch of a new division, HR HealthCare Patient Services, to enhance its urological care portfolio through expanded medical supply processing capabilities. This new division will serve as the company’s dedicated supply arm, offering patients and healthcare providers streamlined access to medical supplies through a fully integrated, single-team approach operating under a single unified brand.
Material Insights
The Silicone 2-way Foley catheters segment dominated the North America 2-way Foley catheters industry in 2025, due to its superior biocompatibility, lower risk of latex-related allergic reactions, and better suitability for long-term catheterization. Silicone catheters are associated with reduced encrustation, wider internal lumen for improved drainage, and lower incidence of urethral irritation compared to latex variants, making them clinically preferred in hospitals and long-term care settings. In addition, infection prevention initiatives and CAUTI-reduction protocols, encouraged by organizations such as the Centers for Disease Control and Prevention, have increased the adoption of higher-quality, non-latex materials. Regulatory oversight by the U.S. Food and Drug Administration and growing patient safety awareness have further accelerated the shift toward silicone-based products.
Latex 2-way Foley catheters are expected to capture a significant share of the North America 2-way Foley catheters industry, due to their cost-effectiveness and widespread use in short-term catheterization procedures. Despite the clinical shift toward silicone variants, latex catheters, particularly those coated with hydrogel or silicone elastomer, remain highly utilized in acute care settings where catheter dwell time is limited. Their lower unit cost makes them attractive for high-volume hospital procurement and budget-sensitive healthcare facilities.
Indication Insights
Urinary incontinence dominates the North America 2-way Foley catheters market in 2025 and is expected to grow at the fastest CAGR over the forecast period. The high prevalence of urinary incontinence among the elderly population, particularly in long-term care and home healthcare settings, significantly drives demand for indwelling catheterization. Increasing rates of age-related conditions, neurological disorders, post-surgical complications, and mobility limitations further contribute to sustained catheter utilization. In addition, the expanding geriatric population across the U.S. and Canada, along with improved diagnosis and management of bladder dysfunction, supports market growth. Clinical guidance and infection-control recommendations from organizations also support appropriate catheter use in medically necessary incontinence cases.
Spinal cord injury is expected to grow at a significant CAGR over the forecast period, due to the high incidence of neurogenic bladder dysfunction associated with traumatic and non-traumatic spinal injuries. Patients with spinal cord injuries require long-term or intermittent bladder management, leading to sustained demand for indwelling catheter solutions, including 2-way Foley catheters in acute and rehabilitation settings. The growing survival rate of spinal cord injury patients, improved access to long-term care, and expanding home healthcare services further support market expansion.
Usage Type Insights
The long-term use segment dominated the North America 2-way Foley catheters market in 2025 due to the high prevalence of chronic urinary retention, neurogenic bladder, urinary incontinence, and age-related urological conditions requiring extended catheterization. A growing geriatric population, particularly in long-term care facilities and home healthcare settings, has significantly increased demand for indwelling catheters designed for prolonged use. Patients with conditions such as benign prostatic hyperplasia, spinal cord injuries, and advanced neurological disorders often require continuous bladder management, supporting higher volume utilization in the long-term category. Furthermore, the preference for silicone-based catheters in extended-dwell applications has contributed to a greater revenue share in this segment.
The short-term use segment is expected to grow at the fastest CAGR over the forecast period, due to rising surgical procedures, increasing hospital admissions, and higher utilization in acute care settings. Short-term catheterization is commonly required for perioperative bladder management, critical care monitoring, emergency interventions, and temporary urinary retention management. The growing volume of minimally invasive and same-day surgical procedures has further expanded the need for temporary indwelling catheterization. In addition, heightened focus on early catheter removal protocols and reduced-dwell-time strategies has supported higher turnover and repeat use in short-term applications.
End Use Insights
Hospitals dominated the North America 2-way Foley catheters market in 2025 due to high inpatient admissions, surgical procedure volumes, and intensive care utilization requiring continuous urinary drainage. Indwelling 2-way Foley catheters are routinely used in perioperative care, critical care monitoring, emergency treatment, and the management of acute urinary retention, making hospitals the largest consumption setting. The presence of established procurement systems, higher patient turnover, and standardized catheterization protocols further supported volume demand within hospital environments.
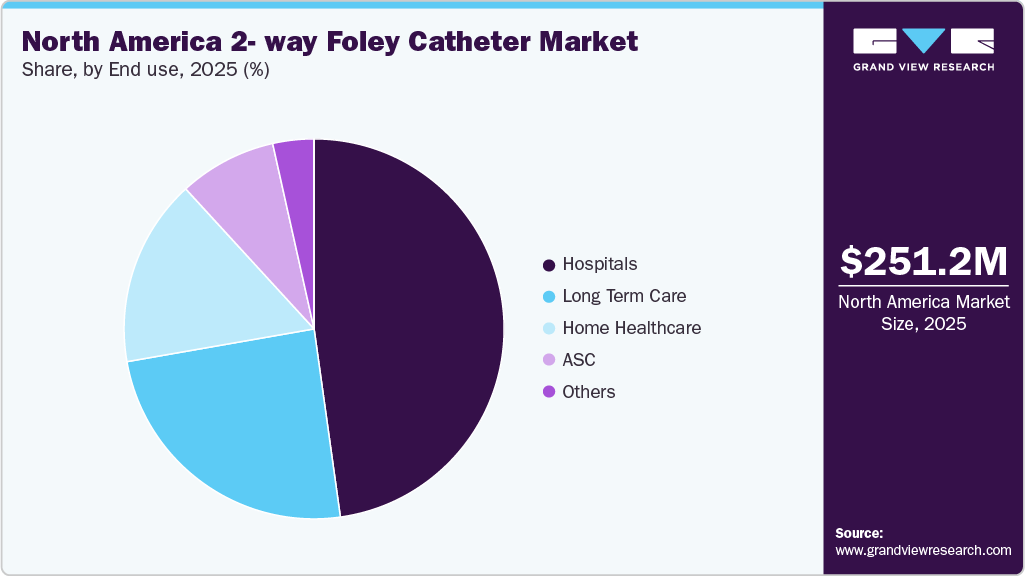
Home care is expected to grow at the fastest CAGR during the forecast period, due to rising preference for at-home chronic care management, an increasing geriatric population, and the growing prevalence of long-term urinary conditions. Patients with neurogenic bladder, mobility limitations, and chronic urinary retention are increasingly managed outside hospital settings to reduce healthcare costs and improve quality of life. Expansion of reimbursement support, availability of trained home care providers, and improved access to catheter supplies through distributors have further accelerated adoption in this segment. As a result, the shift toward decentralized care delivery is driving faster growth in home healthcare compared to traditional acute care settings.
Country Insights
U.S. 2-Way Foley Catheter Market Trends
The U.S. dominated the North America 2-way Foley catheters market due to strong hospital infrastructure, high surgical procedure volumes, and a large aging population requiring urinary catheterization for both acute and chronic conditions. The widespread prevalence of urinary retention disorders such as benign prostatic hyperplasia, urinary incontinence, and neurogenic bladder significantly supports sustained demand for 2-way Foley catheters. In addition, advanced healthcare spending, established reimbursement frameworks, and the presence of major medical device manufacturers contribute to market leadership.
In addition, the CDC framework highlights the need for continuous quality improvement programs, surveillance of catheter-associated infections, and the adoption of newer technologies to reduce infection burden across acute and non-acute care settings. This encourages hospitals and integrated delivery networks to standardize procurement toward infection-prevention-focused devices, indirectly supporting the adoption of catheter technologies across urology applications.

Canada 2-way Foley Catheters Market Trends
In Canada, 2-way Foley catheters are growing; access to them is supported by a combination of provincial disability programs, workplace insurance coverage, auto insurance benefits, and select municipal assistance initiatives. In Ontario, eligible individuals may receive support through the Ontario Disability Support Program (ODSP) under the Mandatory Special Necessities (MSN) benefit, which covers certain surgical and incontinence supplies, subject to medical and financial eligibility criteria. Families caring for children with severe disabilities may obtain financial assistance through the Assistance for Children with Severe Disabilities Program (ACSD). At the same time, additional support for pediatric incontinence supplies may be available through Easter Seals Ontario. Individuals who require catheterization due to workplace injuries may be reimbursed by the Workplace Safety and Insurance Board (WSIB), subject to a physician's prescription and approval.
Key North America 2-way Foley Catheter Company Insights
The North America 2-way Foley catheters market is competitive, with key players holding significant positions. The major companies are undertaking various organic as well as inorganic strategies, such as new product development, collaborations, acquisitions, mergers, and regional expansion, to serve the unmet needs of their customers.
Key North America 2-way Foley Catheter Companies:
- BD (Bard)
- Medline Industries
- Cardinal Health
- Teleflex Incorporated
- Medtronic
- Coloplast Corp
- Amsino International, Inc.
- HR HealthCare
- Dynarex Corporation
- Sterimed Group.
- Well Lead Medical Co., Ltd.
Recent Developments
-
In December 2025, Medline Industries announced a supply agreement with Soundview Medical Supply, a Washington state-based provider of medical supplies serving adult family homes, long-term care facilities, and home medical equipment (HME) providers nationwide. This agreement strengthens Medline’s distribution network and enhances product accessibility across post-acute and home care settings throughout the U.S.
-
In April 2025, Amsino advanced its global healthcare mission with the establishment of Amsino Scientific Inc., a new medical device manufacturing, research, and development facility located in Franklin, Massachusetts. This strategic expansion represents a significant milestone in the company’s ongoing growth trajectory and reinforces its commitment to delivering innovative, reliable, cost-effective, and user-friendly medical devices to healthcare providers worldwide.
North America 2-way Foley Catheter Market Report Scope
Report Attribute
Details
Market size value in 2026
USD 266.75 million
Revenue forecast in 2035
USD 455.92 million
Growth rate
CAGR of 6.14% from 2026 to 2035
Actual data
2018 - 2025
Forecast period
2026 - 2035
Quantitative units
Revenue in USD million/billion and CAGR from 2026 to 2035
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Material, indication, usage type, end use, country
Regional scope
North America
Country scope
U.S.; Canada; Mexico
Key companies profiled
BD (Becton, Dickinson and Company) (Bard); Medline Industries; Cardinal Health; Teleflex Incorporated; Medtronic; Coloplast Corp; Amsino International, Inc.; HR HealthCare; Dynarex Corporation; Sterimed Group; Well Lead Medical Co., Ltd.
Customization scope
Free report customization (equivalent up to 8 analysts working days) with purchase. Addition or alteration to country & segment scope.
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
North America 2-way Foley Catheters Market Report Segmentation
This report forecasts revenue growth at the regional and country levels and provides an analysis of the latest industry trends and opportunities in each of the sub-segments from 2018 to 2035. For this study, Grand View Research has segmented the North America 2-way Foley catheters market report based on material, indication, usage type, end use, and country:
-
Material Outlook (Revenue, USD Million, 2018 - 2035)
-
Silicone 2-way Foley Catheters
-
Hydrophilic Coated
-
Hydrogel Coated
-
Anti-Microbial Coated
-
Others
-
-
Latex 2-way Foley Catheters
-
Hydrophilic Coated
-
Hydrogel Coated
-
Silicone Elastomer Coated
-
Anti-Microbial Coated
-
Others
-
-
-
Indication Outlook (Revenue, USD Million, 2018 - 2035)
-
Urinary Incontinence
-
Enlarged Prostate Gland/BPH
-
Spinal Cord Injury
-
Others
-
-
Usage Type Outlook (Revenue, USD Million, 2018 - 2035)
-
Short-Term Use
-
Long-Term Use
-
-
End Use Outlook (Revenue, USD Million, 2018 - 2035)
-
Hospitals
-
Ambulatory Surgical Centers (ASCs)
-
Long-Term Care (LTCs)
-
Home Care
-
Others
-
-
Country Outlook (Revenue, USD Million, 2018 - 2035)
-
U.S.
-
Canada
-
Mexico
-
Frequently Asked Questions About This Report
b. The North America 2-way Foley catheters market size was estimated at USD 251.20 million in 2025.
b. The North America 2-way Foley catheters market is expected to grow at a compound annual growth rate of 6.14% from 2026 to 2035, reaching USD 455.92 million by 2035.
b. U.S. dominated the North America 2-way Foley catheters market in 2025.
b. Some key players operating in the North America 2-way Foley catheters market are BD (Bard), Medline, Cardinal Health, Teleflex Incorporated, Medtronic, Coloplast Corp, Amsino International, Inc., HR HealthCare, Dynarex Corporation, Sterimed Group, Well Lead Medical Co., Ltd.
b. The North America 2-way Foley catheters market is driven primarily by the rising prevalence of urinary retention, benign prostatic hyperplasia, post-surgical urinary management needs, and the growing geriatric population, which is more susceptible to urological disorders.
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










