- Home
- »
- Medical Devices
- »
-
Self-expanding Covered Stents Market Size Report, 2033GVR Report cover
![Self-expanding Covered Stents Market Size, Share & Trends Report]()
Self-expanding Covered Stents Market (2026 - 2033) Size, Share & Trends Analysis Report by Anatomy (External Iliac, SFA (Superficial Femoral Artery)), By Indication (Aneurysm Repair, Occlusive Disease (PAD)), By Material, By End Use, By Region, And Segment Forecasts
- Report ID: GVR-4-68040-908-1
- Number of Report Pages: 100
- Format: PDF
- Historical Range: 2021 - 2025
- Forecast Period: 2026 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
Self-expanding Covered Stents Market Summary
The global self-expanding covered stents market size was estimated at USD 743.8 million in 2025 and is projected to reach USD 1,091.7 million by 2033, growing at a CAGR of 5.0% from 2026 to 2033. The increasing prevalence of peripheral artery disease and aortoiliac occlusive disease, rising incidence of Aneurysm, are driving the demand for effective vascular interventions, thereby boosting the adoption of self-expanding covered stents.
Key Market Trends & Insights
- North America dominated the self-expanding covered stents market in 2025 with a revenue share of 42.8%.
- Theself-expanding covered stents industry in the U.S.dominated with a global revenue share of 80.5% in 2025.
- By anatomy, the superficial femoral artery (SFA) segment accounted for the largest revenue share of 44.8% in 2025.
- By indication, the aneurysm repair segment accounted for the largest revenue share of 38.2% in 2025.
- By end-use, the hospitals segment led the industry, accounting for the largest revenue share of 57.0% in 2025.
Market Size & Forecast
- 2025 Market Size: USD 743.8 Million
- 2033 Projected Market Size: USD 1,091.7 million
- CAGR (2026-2033): 5.0%
- North America: Largest market in 2025
The section below outlines the key factors driving the growth of the self-expanding covered stents (SX-CS) market, highlighting the rising prevalence of peripheral artery disease and aortoiliac occlusive disease, along with the increasing demand for minimally invasive vascular interventions. It also examines the role of technological advancements, such as Nitinol-based stent designs and improved Polytetrafluoroethylene coverings, as well as the expanding use of SX-CS in complex procedures, including Endovascular Aneurysm Repair, Fenestrated Endovascular Aneurysm Repair, and Branched Endovascular Aneurysm Repair, thereby strengthening their adoption across healthcare settings.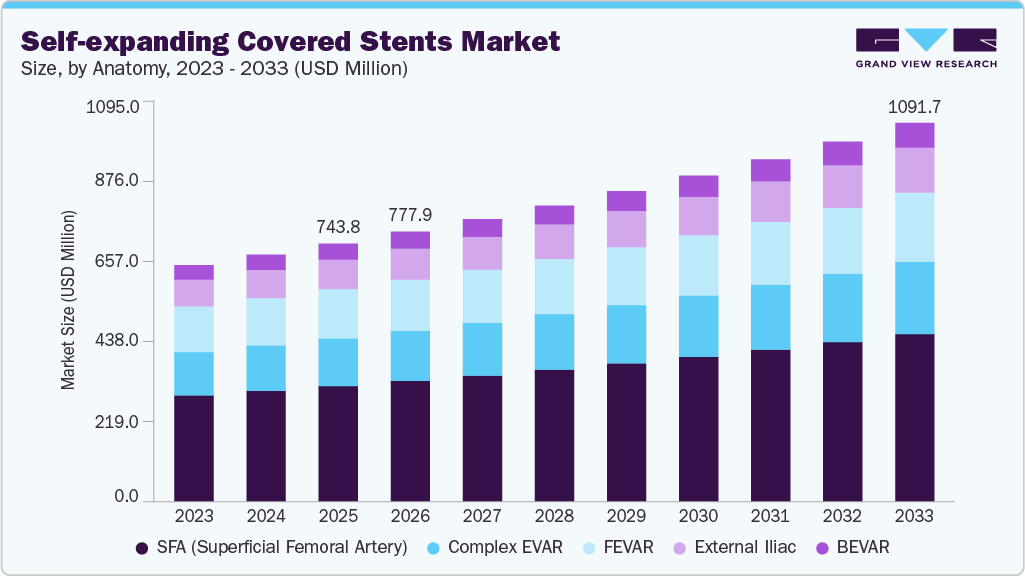
Rising prevalence of peripheral vascular diseases
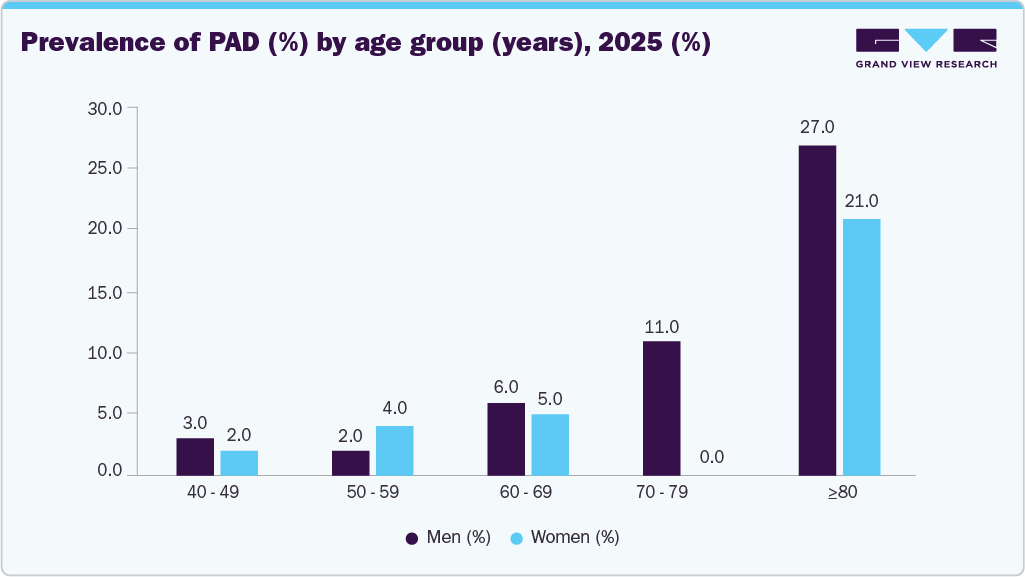
The increasing incidence of Peripheral Artery Disease and Aortoiliac Occlusive Disease is driving demand for durable and flexible stent solutions. SX-CS is widely preferred due to its ability to provide long-term patency and vessel support in complex lesions. In May 2024, the Centers for Disease Control and Prevention highlighted that PAD affects millions of adults in the U.S., significantly increasing the need for endovascular interventions.
Continuous innovation in stent design improves treatment outcomes
Ongoing advancements in stent technologies are enhancing flexibility, deliverability, and suitability for complex vascular anatomies. These innovations are improving procedural efficiency and expanding the use of advanced stents, thereby supporting overall market growth. BD Fluency Plus Endovascular Stent Graft is a self-expanding covered nitinol stent encapsulated with ePTFE, designed for iliac and femoral artery interventions, offering flexibility, accurate deployment, and durable patency in complex lesions. The system features a nitinol-based self-expanding design that provides sustained radial force, flexibility, and the ability to adapt to dynamic vascular anatomies, while the ePTFE covering acts as a barrier to prevent plaque prolapse and support effective sealing. Similarly, in March 2024, W. L. Gore & Associates continued advancements in its VIABAHN endoprosthesis platform, enhancing performance in complex peripheral artery interventions.
Post-Market Evidence Supporting SX-CS Safety, Patency, and Durability in Iliac Lesions
Post-market clinical results validating the safety and durability of SX-CS in challenging iliac anatomy boost market growth. According to an Endovascular Today article published in December 2025, a recent real-world clinical instance illustrating the performance of self-expanding covered stents is the Solaris SX iliac post-market clinical follow-up study, which reported excellent outcomes through 12 months. In this prospective multicenter study across nine European sites, the Solaris SX device achieved primary patency of over 95 % at 6 months and approximately 94 % at 12 months, along with a significant improvement in ankle brachial index from 0.547 at baseline to 0.927 at 12 months, and no device-related serious adverse events through 30 days, representing both safety and durability in treating challenging iliac artery lesions. These findings suggest strong potential for SX CS platforms in a range of peripheral applications, including external iliac and related segment interventions.
“The 12-month results from this multicenter study show consistent and durable performance of the Solaris SX covered stent across challenging iliac anatomy. We observed high patency, very few reinterventions, and strong safety outcomes across nine European sites. The performance of Solaris SX in this study reflects the integrity of the stent design and indicates potential utility in broader peripheral arterial applications.”
- Giacomo Isernia, MD, from Azienda Ospedaliera di Perugia in Perugia, Italy, is Principal Investigator of the study.
Technological advancements in stent design and materials
Innovations in Nitinol self-expanding frameworks and Polytetrafluoroethylene coverings are improving flexibility, durability, and clinical outcomes. These advancements support usage in complex anatomies and high-risk patients. In January 2026, W. L. Gore & Associates, Inc. received FDA approval for the VIABAHN FORTEGRA Venous Stent, a self-expanding device indicated for deep venous disease in the inferior vena cava and iliofemoral veins. The approval was supported by an 89-patient international study showing 83.4% 12-month primary patency with no major device-related safety events.
Increasing Clinical R&D Activities Driving Innovation and Product Development in SX-CS
An increased focus on clinical research and development is driving innovation and the adoption of advanced vascular devices, including self-expanding covered stents. Ongoing clinical trials are evaluating safety, effectiveness, and long-term outcomes, thereby accelerating product approvals and expanding treatment options for complex vascular diseases. In March 2024, Becton, Dickinson and Company initiated the AGILITY clinical study to evaluate its investigational self-expanding covered stent for the treatment of Peripheral Artery Disease, enrolling patients across multiple global sites to assess long-term safety and effectiveness.
Market Concentration & Characteristics
The chart below represents the relationship between industry concentration, industry characteristics, and industry participants. There is a high degree of innovation, a moderate level of merger & acquisition activity, a high impact of regulations, and low regional expansion.
The degree of innovation is high, supported by continuous advancements in stent materials and delivery systems, while M&A and partnership activities are moderate to high. The impact of regulations is moderate to high due to stringent approval requirements for vascular devices, and regional expansion remains steady, driven by the increasing prevalence of vascular diseases and improving healthcare infrastructure. For instance, in December 2025, Solaris Endovascular reported 12-month outcomes from its European Solaris SX iliac postmarket study at VEITHsymposium. The multicenter trial showed 94% primary patency at 12 months, improved ankle brachial index, and no device-related serious adverse events at 30 days, confirming sustained performance of the non-drug eluting self-expanding covered stent.
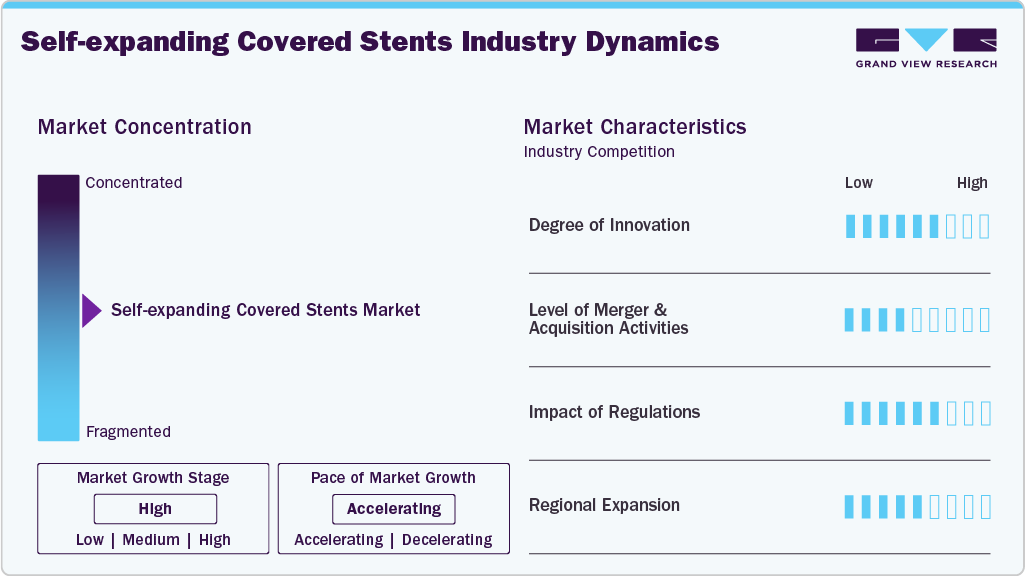
The level of mergers and acquisitions is medium in this market. To improve their market position, several companies participate in mergers and acquisitions. These activities boost their market expertise, diversify their product offerings, and enhance skills. For instance, in July 2025, Teleflex Incorporated announced that it had completed the acquisition of substantially all assets of the Vascular Intervention business from BIOTRONIK SE & Co. KG. This acquisition expands Teleflex’s portfolio with a broad range of therapeutic vascular products, strengthening its position in interventional access and enhancing its global presence in the cath lab.
Regulatory frameworks play a critical role in shaping the self-expanding covered stents market by influencing product approvals, clinical adoption, and commercialization timelines. Stringent approval pathways from agencies such as the U.S. Food and Drug Administration and evolving requirements under the European Union Medical Device Regulation (MDR) mandate extensive clinical evidence on safety, long-term patency, and durability, which can delay product launches but improve device quality and physician confidence. These regulations also increase development costs and compliance burden for manufacturers, particularly for smaller players, leading to market consolidation and favoring established companies with strong regulatory capabilities.
Regional expansion is a key growth driver in the self-expanding covered stents market, as manufacturers increasingly target high-growth geographies beyond traditional markets in North America and Europe. Expanding healthcare infrastructure, rising prevalence of vascular diseases, and improving access to minimally invasive procedures in emerging regions such as the Asia-Pacific and Latin America are creating significant opportunities. Countries such as India and China are witnessing increased investments in hospitals, growing physician expertise in endovascular techniques, and supportive government initiatives, all of which are accelerating adoption.
Anatomy Insights
The superficial femoral artery (SFA) segment accounted for the largest revenue share of 44.8% in 2025, driven by long-segment disease, restenosis, in-stent restenosis, and cases with significant plaque burden or calcification. In August 2023, an article published in the Journal of Invasive Cardiology highlighted a 57-year-old woman with critical limb-threatening ischemia who underwent endovascular therapy using a balloon-expandable covered stent in the superficial femoral artery, following a prior femoropopliteal bypass for chronic total occlusion performed four years earlier.
The BEVAR segment is anticipated to grow at the fastest CAGR over the forecast period. The growth in this market is driven by innovations by key players in the market and the rising number of regulatory approvals by regulatory authorities. For instance, in November 2025, Bentley InnoMed received CE Mark approval for its BeGraft Plus stent graft for use in branched endovascular aneurysm repair. The stent, available in cleared markets, offers a length of up to 78 mm to bridge complex anatomies, providing stable support and flexibility. Early clinical experience is positive, offering physicians a reliable on-label option for complex aortic repair.
Indication Insights
The aneurysm repair self-expanding covered stents segment accounted for the largest revenue share of 38.2% in 2025. The market is driven by the increasing adoption of minimally invasive endovascular aneurysm repair (EVAR) procedures, strong physician preference for self-expanding nitinol-based stents due to their flexibility and conformability in complex anatomies, and ongoing product innovation enhancing durability and procedural outcomes. For instance, in April 2026, Endospan received U.S. FDA approval for the NEXUS Aortic Arch Stent Graft System, the first shelf endovascular stent graft approved in the U.S. for aortic arch disease. It expands treatment options for high-risk aneurysm patients and further drives the adoption of advanced self-expanding covered stents.
The occlusive disease (PAD) segment is anticipated to grow at a notable CAGR over the forecast period, driven by the rising burden of peripheral artery diseases due to aging populations, diabetes, and cardiovascular risk, along with increasing adoption of minimally invasive endovascular interventions for below-the-knee and complex peripheral lesions. For instance, in March 2025, BD received FDA approval for the Revello vascular covered stent. This next-generation endovascular solution combines the flexibility of a nitinol self-expanding stent with the radial resistive force designed for the iliac arteries. Revello features an ultra-thin, expanded polytetrafluoroethylene covering.
Material Insights
The ePTFE-covered stents segment led the industry, accounting for the largest revenue share of 45.8% in 2025, largely attributed to clinical preference for ePTFE-covered stents due to their biocompatibility, thromboresistance, and long-term patency performance in complex vascular interventions. For instance, in April 2026, announced the application of the CE Mark for the Liverty TIPS Stent Graft. The Liverty TIPS Stent Graft is engineered to support individualized patient management, offering a 6-10 mm adjustable inner diameter and the broadest range of covered TIPS stent lengths currently available.
The polyurethane / hybrid coverssegment is anticipated to grow at the fastest CAGR over the forecast period. This growth is driven by rising demand for next-generation covered stents that offer enhanced flexibility, durability, and biocompatibility. For instance, in January 2026, Solaris Endovascular, Inc., announced the completion of clinical trial enrollment for Solaris DEScover. The company stated that the study includes both native arteriovenous fistula (AVF) and prosthetic arteriovenous fistula graft (AVG) patients. It is designed to evaluate safety and target-lesion primary patency (TLPP) outcomes compared with standard percutaneous transluminal angioplasty in AVF patients, and to assess a single-arm treatment approach for AVG patients.
End Use Insights
The hospitals segment led the industry, accounting for the largest revenue share of 57.0% in 2025. The market is growing as a significant number of complex endovascular procedures are being conducted in hospital environments. There is also increased demand for self-expanding covered stents in emergency vascular repair, aneurysm exclusion, and the management of complex occlusive lesions. For instance, in July 2025, W. L. Gore & Associates announced an expanded CE Mark indication for the GORE VIABAHN VBX Balloon Expandable Endoprosthesis, authorizing its use as a bridging stent in branched and fenestrated endovascular aneurysm repair (BEVAR/FEVAR). Hospitals have since initiated the implantation of this device in complex aneurysm procedures
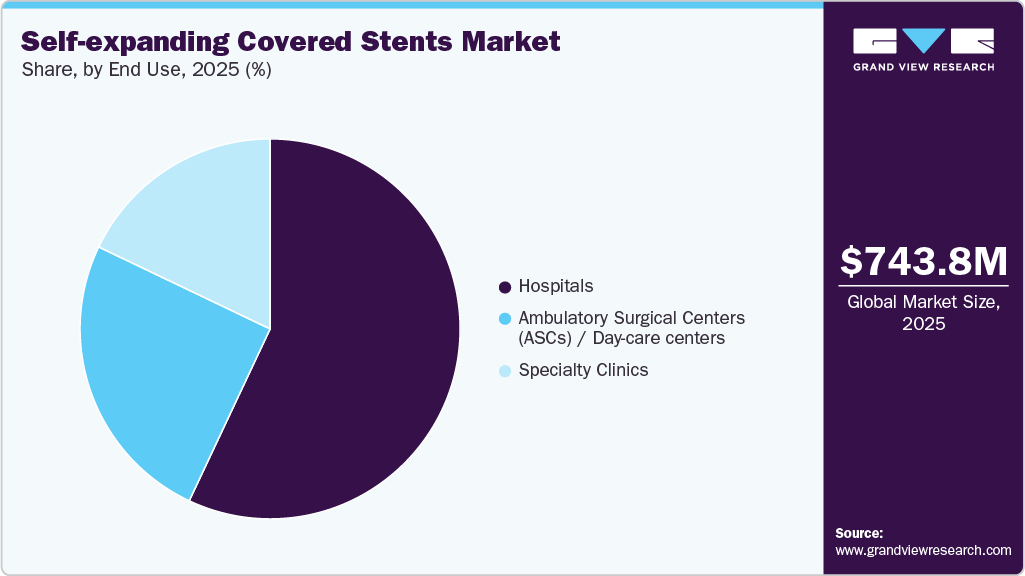
The ambulatory surgical centers (ASCs) / day-care centers segment is anticipated to grow at the fastest CAGR over the forecast period. The growth is driven by the shift of peripheral and vascular interventions from inpatient hospitals to lower-cost outpatient settings, rising demand for minimally invasive procedures with shorter recovery times, and the growing adoption of self-expanding covered stents for same-day endovascular treatments. Growth is further supported by expanding ASC infrastructure, favorable reimbursement trends for outpatient vascular procedures, technological advances enabling complex interventions in day-care settings, and increasing physician preference for ASCs.
Regional Insights
The North America self-expanding covered stents industry dominated with a global revenue share of 42.8% in 2025, driven by the high prevalence of Peripheral Artery Disease, advanced healthcare infrastructure, and strong adoption of minimally invasive vascular procedures. The presence of leading players and continuous clinical research further supports market growth. For instance, in January 2024, W. L. Gore & Associates, Inc. secured FDA clearance for a reduced-profile Gore Viabahn VBX balloon expandable endoprosthesis. The 1 Fr smaller delivery system increases 6 Fr sheath compatibility for complex iliac artery treatment, with over 375000 devices implanted globally.
U.S. Self-Expanding Covered Stents Market Trends
Theself-expanding covered stents industry in the U.S.dominated with a global revenue share of 80.5% in 2025. The U.S. dominates the regional market due to robust healthcare spending, early adoption of advanced endovascular technologies, and a large patient pool with chronic vascular conditions. In July 2025, The Sage Group reported that over 190 million people in the U.S. suffer from CVD, with more than 50 million affected by CVI, highlighting a substantial unmet need for vascular treatment solutions.
Europe Self-Expanding Covered Stents Market Trends
The self-expanding covered stents industry in Europe is mature, supported by strong regulatory frameworks, an aging population, and therising incidence of vascular diseases. Demand is further driven by advancements in endovascular therapies and the presence of key manufacturers.
The Germany self-expanding covered stents industry dominates the market in Europe, due to advanced medical device manufacturing and high adoption of minimally invasive vascular procedures. The country benefits from strong clinical expertise and healthcare infrastructure supporting complex interventions. According to the International Trade Administration data published in 2025, Germany’s healthcare expenditure reached approximately €492 billion ($577), accounting for ~11.7% of GDP, the highest among EU countries, highlighting strong financial capability for adopting advanced medical devices.
The self-expanding covered stents industry in Italy is witnessing steady growth due to the increasing adoption of endovascular treatments and the improving hospital infrastructure, which are supporting demand for advanced stent technologies. The country benefits from rising procedural volumes, particularly in peripheral and aortoiliac interventions, supported by growing awareness of vascular diseases and preference for minimally invasive therapies.
Asia Pacific Self-Expanding Covered Stents Market Trends
The self-expanding covered stents industry in the Asia Pacific is expected to grow at a considerable CAGR during the forecast period, driven by a large patient population, rising prevalence of Peripheral Artery Disease, and expanding healthcare infrastructure. Increasing awareness, improving affordability, and growing access to minimally invasive procedures are accelerating market expansion across emerging economies. Over the past 20 years, Wuhan Asia Heart Hospital has treated more than 3.6 million patients with heart disease and successfully completed over 307,000 cardiac surgeries, ranging from simple to complex.
The Japan self-expanding covered stents industry is a key market in the region due to its rapidly aging population and high prevalence of complex vascular conditions such as aortic aneurysms and dissections. The country’s advanced healthcare system, strong regulatory framework, and high adoption of innovative endovascular and hybrid procedures are driving demand for technologically advanced stent systems. In July 2023, Terumo Aortic received approval from the Pharmaceuticals and Medical Devices Agency for its Thoraflex Hybrid device in Japan. The system integrates a nitinol self-expanding stent graft with a surgical graft, enabling effective treatment of complex aortic arch diseases, including aneurysms and dissections.
Latin America Self-Expanding Covered Stents Market Trends
Theself-expanding covered stents industry in Latin Americais expected to witness considerable growth over the forecast period, driven by increasing investments in healthcare infrastructure, rising awareness of vascular diseases, and improving access to minimally invasive procedures. Countries such as Brazil and Mexico are experiencing enhancements in hospital capabilities and diagnostic services, which support the growing adoption of advanced endovascular treatments, including self-expanding covered stents.
The Brazil self-expanding covered stents industry holds a significant share in the regional market, supported by expanding healthcare investments and increasing procedural volumes for conditions such as Peripheral Artery Disease and aneurysms.
Middle East Self-Expanding Covered Stents Market Trends
Theself-expanding covered stents industry in the Middle East & Africais driven by increasing demand for advanced vascular treatment solutions and rising investments in healthcare modernization. Governments in countries such as Saudi Arabia and the United Arab Emirates are focusing on upgrading hospital infrastructure and expanding access to minimally invasive procedures as part of broader healthcare transformation initiatives.
The UAE self-expanding covered stents industry is witnessing growing adoption of advanced endovascular technologies due to improved availability of medical devices, an increasing number of specialized healthcare facilities, and a rising focus on high-quality patient care. In February 2025, UAE-based Crescent Enterprises led a USD 30 million funding round in XCath, a company developing robotic systems for minimally invasive vascular procedures. This investment highlights the UAE’s growing focus on advancing endovascular technologies, which is expected to support the adoption of devices such as self-expanding covered stents in complex vascular interventions.
Key Self-expanding Covered Stents Company Insights
Key players operating in the self-expanding covered stents market are undertaking various initiatives to strengthen their presence and increase the reach of their products and services. Strategies such as expansion activities and partnerships are key in propelling market growth.
Key Self-expanding Covered Stents Companies:
The following key companies have been profiled for this study on the self-expanding covered stents market.
- W. L. Gore & Associates, Inc.
- Solaris Endovascular
- BD (Angiomed GmbH & Co. Medizintechnik KG)
- LeMaitre Vascular, Inc.
- Endologix LLC
- Artivion, Inc
- Merit Medical Systems
Recent Developments
-
In April 2026, Getinge’s iCast Covered Stent System received FDA approval for new sizes. The approval includes a 10-mm diameter by 59-mm length, a 32-mm length, and a 12-mm diameter. Newly approved sizes enable the treatment of a broader range of vessel and lesion diameters and lengths.
-
In January 2026, W. L. Gore & Associates received FDA approval for the GORE VIABAHN FORTEGRA Venous Stent, a self-expanding nitinol and ePTFE-based device designed to treat deep venous disease in the inferior vena cava and iliofemoral veins, demonstrating strong clinical safety and efficacy outcomes.
-
In December 2025, Solaris Endovascular reported 12-month outcomes from its European Solaris SX iliac postmarket study at VEITHsymposium. The multicenter trial showed 94% primary patency at 12 months, improved ankle brachial index, and no device-related serious adverse events at 30 days, confirming sustained performance of the non-drug eluting self-expanding covered stent.
-
In December 2025, Olympus Corporation of the Americas, W. L. Gore & Associates, Inc., and ConMed Corporation announced that US distribution of the Gore Viabil biliary endoprosthesis will transfer to Olympus on January 1, 2026, following a global rights agreement. ConMed will support the product through December 31, 2025.
Self-expanding Covered Stents Market Report Scope
Report Attribute
Details
Market size in 2026
USD 777.9 million
Revenue forecast in 2033
USD 1,091.7 million
Growth rate
CAGR of 5.0% from 2026 to 2033
Actual data
2021 - 2025
Forecast period
2026 - 2033
Quantitative units
Revenue in USD million/billion, unit volume in thousand, procedure volume in thousand, and CAGR from 2026 to 2033
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Indication, material, anatomy, end use, region
Country scope
U.S.; Canada; Mexico; Germany; UK; France; Italy; Spain; Norway; Denmark; Sweden;Belgium; Netherlands; Luxembourg China; Japan; India; South Korea; Australia; Thailand; Brazil; Argentina; Saudi Arabia; South Africa; UAE; Kuwait
Key companies profiled
W. L. Gore & Associates, Inc.; Solaris Endovascular; BD (Angiomed GmbH & Co. Medizintechnik KG; LeMaitre Vascular, Inc.; Endologix LLC; Artivion, Inc; Merit Medical Systems
Customization scope
Free report customization (equivalent up to 8 analysts working days) with purchase. Addition or alteration to country, regional & segment scope.
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
Global Self-expanding Covered Stents Market Report Segmentation
This report forecasts revenue growth at the global level and provides an analysis of the latest industry trends in each of the sub-segments from 2021 to 2033. For this study, Grand View Research has segmented the global self-expanding covered stents market report based on indication, material, anatomy, end use, and region:
-
Anatomy Outlook (Unit Volume, Thousand, Procedure Volume, Thousand; Revenue, USD Million, 2021 - 2033)
-
External Iliac
-
SFA (Superficial Femoral Artery)
-
Complex EVAR
-
FEVAR
-
BEVAR
-
-
Indication Outlook (Unit Volume, Thousand, Procedure Volume, Thousand; Revenue, USD Million, 2021 - 2033)
-
Aneurysm Repair
-
Occlusive Disease (PAD)
-
Dissections
-
Trauma / Emergency Repair
-
-
Material Outlook (Unit Volume, Thousand, Procedure Volume, Thousand; Revenue, USD Million, 2021 - 2033)
-
ePTFE Covered Stents
-
Polyurethane / Hybrid Covers
-
Nitinol-based self-expanding stents
-
Others (Heparin-bonded / bioactive coated stents, Stainless steel-based systems)
-
-
End Use Outlook (Unit Volume, Thousand, Procedure Volume, Thousand; Revenue, USD Million, 2021 - 2033)
-
Hospitals
-
Ambulatory Surgical Centers (ASCs) / Day-care centers
-
Specialty Clinics
-
-
Regional Outlook (Unit Volume, Thousand, Procedure Volume, Thousand; Revenue, USD Million, 2021 - 2033)
-
North America
-
U.S.
-
Canada
-
Mexico
-
-
Europe
-
Germany
-
UK
-
France
-
Italy
-
Spain
-
Belgium
-
Netherlands
-
Luxembourg
-
Norway
-
Sweden
-
Denmark
-
-
Asia Pacific
-
China
-
Japan
-
India
-
South Korea
-
Australia
-
Thailand
-
-
Latin America
-
Brazil
-
Argentina
-
-
MEA
-
South Africa
-
Saudi Arabia
-
UAE
-
Kuwait
-
-
Frequently Asked Questions About This Report
b. The global self-expanding covered stents market size was estimated at USD 743.8 million in 2025 and is expected to reach USD 777.9 million in 2026.
b. The global self-expanding covered stents market is expected to reach USD 1,091.7 million by 2033, growing at a CAGR of 5.0% from 2026 to 2033.
b. Key drivers include increasing prevalence of peripheral artery disease and aortoiliac occlusive disease, rising incidence of Aneurysm.
b. The superficial femoral artery (SFA) segment accounted for the largest revenue share of 44.8% in 2025 and is anticipated to grow at the fastest CAGR over the forecast period, driven by long-segment disease, restenosis, in-stent restenosis, and cases with significant plaque burden or calcification.
b. Key players include W. L. Gore & Associates, Inc., Solaris Endovascular, BD (Angiomed GmbH & Co. Medizintechnik KG), LeMaitre Vascular, Inc., Endologix LLC, Artivion, Inc, and Merit Medical Systems.
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










