- Home
- »
- Pharmaceuticals
- »
-
UK Alzheimer’s Therapeutics Market, Industry Report 2030GVR Report cover
![UK Alzheimer’s Therapeutics Market Size, Share & Trends Report]()
UK Alzheimer’s Therapeutics Market (2025 - 2033) Size, Share & Trends Analysis Report By Product (Cholinesterase Inhibitors, NMDA Receptor Antagonist Combination Drug), By End Use (Hospital Pharmacy, Retail Pharmacy), And Segment Forecasts
- Report ID: GVR-4-68040-622-3
- Number of Report Pages: 110
- Format: PDF
- Historical Range: 2021 - 2023
- Forecast Period: 2025 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
UK Alzheimer’s Therapeutics Market Trends
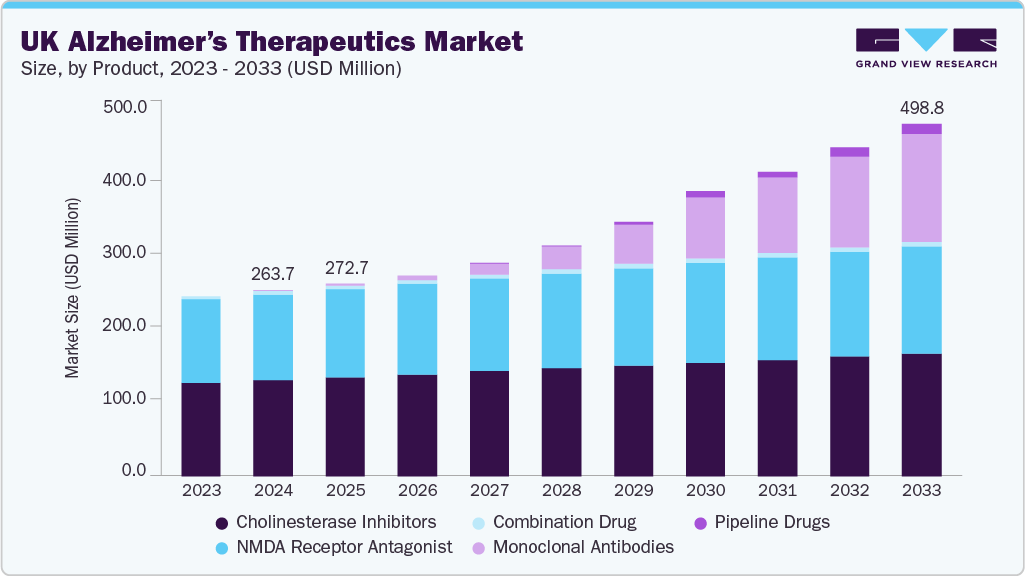
The UK Alzheimer’s therapeutics market size was estimated at USD 263.66 million in 2024 and is projected to reach USD 498.80 million by 2033, growing at a CAGR of 7.84% from 2025 to 2033. The increasing prevalence of Alzheimer’s disease in the UK is driving demand for effective and innovative treatment solutions. The growing proportion of elderly individuals is leading to a surge in cognitive health issues, resulting in more frequent diagnoses. The expanding over-65 population is directly linked to a rising number of Alzheimer’s cases, necessitating expanded access to advanced therapeutics. The trend toward early screening and better public awareness allows healthcare systems to identify the condition at earlier stages.
Drug development and clinical research innovations support market growth across the forecast period. Pharmaceutical companies are focusing on targeted therapies, including monoclonal antibodies and disease-modifying treatments, which are demonstrating potential in clinical trials. Strong engagement in research collaborations with academic institutions is accelerating the pace of therapeutic innovation. For instance, in August 2024, Eisai Co., Ltd. and Biogen Inc. announced that Leqembi received Marketing Authorization from the Medicines and Healthcare products Regulatory Agency (MHRA) for use in Great Britain. The UK’s established clinical trial environment attracts international drug developers to test novel Alzheimer’s treatments. Advanced imaging techniques and biomarker research aid in early detection and effective treatment monitoring. These advancements facilitate the development of precision therapies tailored to disease stages and individual responses. As a result, the treatment landscape is gradually shifting toward more personalized and outcome-driven approaches.
Private sector investments and strategic partnerships are further strengthening the market outlook. Key players are expanding their product pipelines and engaging in licensing deals to enhance their competitiveness. Pharmaceutical companies are launching new products and reformulating existing drugs to extend their clinical benefits. In May 2024, Takeda and AC Immune SA entered into an exclusive global option and license agreement focused on AC Immune’s active immunotherapy programs targeting toxic amyloid beta (Aβ) species. The deal includes ACI-24.060, a vaccine candidate designed for the treatment of Alzheimer’s disease.
Pipeline Analysis
Phase 3
The Alzheimer’s drug pipeline is steadily advancing, with several promising candidates currently in Phase 3 clinical trials. These experimental treatments address a range of disease stages, from early cognitive symptoms to more severe conditions such as agitation, psychosis, and tau-related pathologies. The table below highlights notable Phase 3 trials, detailing study identifiers, therapeutic agents, sponsoring organizations, and projected launch timelines. These timelines are subject to change based on trial results and regulatory decisions. The expanding pipeline demonstrates the sector’s commitment to disease-modifying therapies and symptomatic interventions. This momentum reflects a growing effort to address the significant unmet needs in Alzheimer’s treatment, particularly within global healthcare frameworks like the UK.
Table 1 Phase 3 Drugs
NCT Number
Conditions
Interventions
Sponsor
Estimated Launch
NCT04437511
Alzheimer Disease
DRUG: Donanemab|DRUG: Placebo
Eli Lilly and Company
2027
NCT05531526
Alzheimer Disease
DRUG: AR1001|DRUG: Placebo
AriBio Co., Ltd.
2029
NCT03116126
Alzheimer Disease
DRUG: Guanfacine|DRUG: Placebo
Imperial College London
2026
NCT06384573
Alzheimer's Disease|Dementia|Alzheimer's Disease, Familial
DRUG: lecanemab
Washington University School of Medicine
2032
NCT06937229
Alzheimer's Disease|Agitation
DRUG: KarXT|DRUG: KarX-EC
Bristol-Myers Squibb
2030
NCT06653153
Alzheimer's Disease
DRUG: Remternetug|DRUG: Placebo
Eli Lilly and Company
2032
NCT04468659
Preclinical Alzheimer's Disease|Early Preclinical Alzheimer's Disease
DRUG: Lecanemab|DRUG: Placebo
Eisai Inc.
2033
Source: Clinicaltrials.gov
Market Concentration & Characteristics
The UK Alzheimer's therapeutics industry is highly innovative, especially with the rise of monoclonal antibodies and disease-modifying therapies. Pipeline drugs from players such as Biogen, Eli Lilly, and Alzheon target beta-amyloid and tau protein pathways, shifting focus from symptom relief to disease progression. Traditional products such as cholinesterase inhibitors remain, but innovation has moved toward biologics and precision medicine. Companies are also exploring combination drugs and novel delivery mechanisms. Research partnerships and clinical trials are intensifying, particularly in academic hubs like Cambridge and Oxford.
The market has high entry barriers due to strict regulatory requirements, long clinical trial durations, and substantial capital investment. Approval for Alzheimer's drugs in the UK requires extensive efficacy data and long-term safety profiles, often taking over a decade to bring a product to market. Existing brand loyalty toward therapies from companies like Eisai and Roche further complicates entry for new players. Intellectual property protections on biologics and newer drugs add legal and financial hurdles. Moreover, market access is tightly controlled through NHS pricing and reimbursement evaluations.
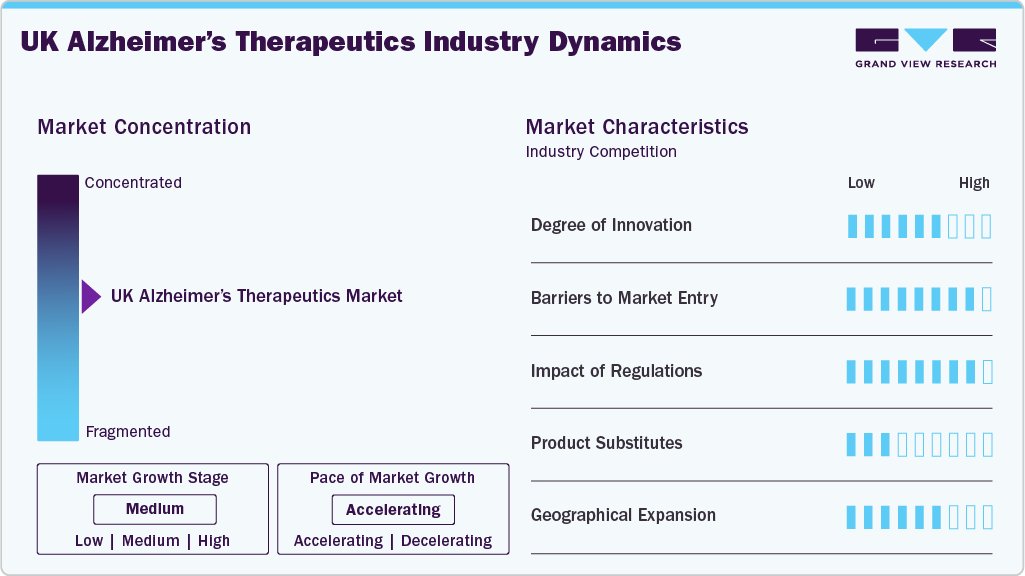
UK regulatory frameworks, driven by the MHRA and NICE, heavily influence drug approval, pricing, and market access for Alzheimer’s therapeutics. Regulatory scrutiny is particularly strong for high-cost biologics and monoclonal antibodies, which require proven cost-effectiveness. Post-Brexit adjustments have introduced some delays and uncertainties in approval pathways. However, the UK government supports accelerated reviews for breakthrough therapies, benefiting innovative pipeline drugs. Compliance with pharmacovigilance standards and clinical trial protocols remains essential for product availability.
There are limited direct substitutes for advanced Alzheimer’s therapeutics, but non-drug interventions such as cognitive therapy, caregiving technology, and nutritional supplements provide partial alternatives. Generic cholinesterase inhibitors and NMDA receptor antagonists offer cost-effective treatment but lack disease-modifying effects. Herbal remedies and off-label psychiatric drugs are sometimes used, though without robust clinical validation. Due to efficacy gaps, these substitutes pose minimal threat to monoclonal antibodies and newer drug classes. Nonetheless, they attract patients unwilling or unable to afford advanced therapies.
Product Insights
Cholinesterase inhibitors dominated the market, accounting for 52.0% of the share in 2024, driven by the continued use of Donepezil. Commonly prescribed for mild-to-moderate Alzheimer’s disease, Donepezil enhances acetylcholine levels in the brain to help ease symptoms such as memory loss and cognitive decline. Available in branded forms like Aricept and generics post-patent, it is typically started at 5 mg daily, increasing to 10 mg based on patient response. Prescribed under specialist supervision, the drug is regularly reviewed to assess effectiveness and tolerance, as side effects such as nausea and sleep disturbances are more frequent during dosage increases. Its cost-efficiency and broad availability under the NHS make it a first-line choice, with alternatives like rivastigmine and galantamine reserved for cases of intolerance. Endorsed in UK clinical guidelines, Donepezil remains a core component of the nation’s Alzheimer’s treatment protocols.
The pipeline drugs segment is projected to grow at the fastest CAGR over the forecast period, driven by a diverse array of late-stage candidates targeting various stages and symptoms of Alzheimer’s disease. Key drivers include Donanemab (Eli Lilly), expected between 2027 and 2029, and Lecanemab (Eisai), slated for 2029 to 2033, aimed at reducing amyloid plaques. Additional contributors include AR1001 (AriBio, 2029), Remternetug (Eli Lilly, 2032), and ALZ-801 (Alzheon, 2028), all focusing on early intervention. Symptom management therapies such as Guanfacine (Imperial College London, 2026) and KarXT (Bristol-Myers Squibb/Karuna Therapeutics, 2028-2030) address agitation and psychosis. Semaglutide (Novo Nordisk), being studied for early Alzheimer’s, may launch by 2028. The expansion of novel therapies across disease-modifying and symptomatic areas underpins the robust growth outlook for this segment.
End Use Insights
Hospital pharmacies dominate the end use segment and hold a revenue share of 56.00% in 2024 due to their critical role in managing moderate to severe cases and administering complex therapies. They ensure proper storage, dispensing, and initiation of treatment for patients diagnosed in NHS memory clinics. Key hospitals like UCLH and King’s College Hospital rely on their pharmacies for medication supply and adherence support. These pharmacies also guide prescribing, funding approvals, and formulary compliance under NICE and NHS frameworks. Their coordination with dementia units ensures ongoing treatment monitoring and care continuity. This makes them essential to effective Alzheimer’s management within the NHS.
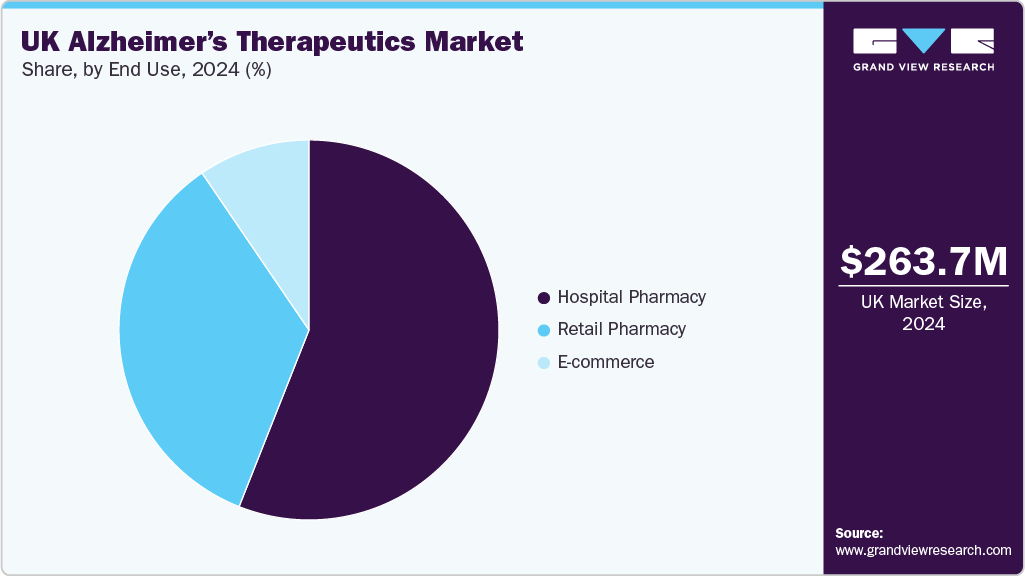
The retail pharmacy segment is projected to grow at the fastest CAGR over the forecast period, fueled by its expanding role in Alzheimer's care across the UK. Retail pharmacies serve as primary access points for outpatient and community-based patients, ensuring the timely distribution of symptomatic treatments and emerging therapies. Their widespread presence in urban and rural areas enhances medication availability, supporting adherence and improving patient outcomes. Pharmacists also provide crucial counseling on drug use, side effects, and the importance of consistent therapy, helping to manage expectations for treatments that primarily address symptoms. In addition, retail pharmacies act as intermediaries between patients and the healthcare system, assisting with prescription renewals and reimbursement navigation under the NHS. Leading chains like Boots, LloydsPharmacy, and Well Pharmacy are advancing their role in Alzheimer’s care, with initiatives such as Boots’ Advantage Card program aiding clinical trial recruitment and accelerating therapeutic development.
Key UK Alzheimer’s Therapeutics Company Insights
Major players such as Eisai Co., Ltd, Novartis AG, and AbbVie Inc. hold significant market share, capitalizing on strong brand reputation, successful clinical trial outcomes, and established regulatory approvals. Competitive dynamics in the UK market are shaped by pricing strategies, innovation in drug mechanisms, and efficient distribution channels. Emerging firms like AC IMMUNE SA and Alzheon are gaining traction by developing novel therapies and focusing on unmet patient needs. Furthermore, regional manufacturers and biotech startups are introducing cost-effective treatment options to broaden accessibility. As the UK healthcare system increasingly emphasizes effective and affordable dementia care, competition among manufacturers is expected to intensify. Overall, the UK Alzheimer’s therapeutics industry will experience robust growth during the forecast period.
Key UK Alzheimer’s Therapeutics Companies:
- Eisai Co., Ltd
- Novartis AG
- AbbVie Inc
- Supernus Pharmaceuticals, Inc.
- H. Lundbeck A/S
- Biogen
- AC IMMUNE SA
- F. Hoffmann-La Roche Ltd
- Daiichi Sankyo Company, Limited
- Johnson & Johnson Services, Inc.
- TauRx Pharmaceuticals Ltd.
- Eli Lilly and Company
- Alzheon
Recent Developments
-
In April 2025, Biogen Inc. and Eisai Co., Ltd. revealed that the European Commission (EC) granted Marketing Authorization (MA) for Leqembi (lecanemab), an amyloid-beta (Aβ) monoclonal antibody, within the European Union (EU). This approval represents the first instance of an Alzheimer’s disease (AD) treatment aimed at the root cause of the condition receiving MA in the EU.
-
In December 2024, AbbVie Inc. entered into a definitive agreement to acquire Aliada Therapeutics, a biotech firm specializing in treatments for complex central nervous system (CNS) disorders. Aliada’s primary investigational candidate, ALIA-1758, an antibody targeting pyroglutamate amyloid beta (3pE-Aβ), is being developed as a potential therapy for Alzheimer’s.
-
In July 2024, TauRx Pharmaceuticals Ltd announced the submission of a UK Marketing Authorisation Application (MAA) for hydromethylthionine mesylate (HMTM) to treat mild cognitive impairment due to Alzheimer’s disease (MCI-AD) and mild to moderate stages of Alzheimer’s dementia.
UK Alzheimer’s Therapeutics Market Report Scope
Report Attribute
Details
Market size value in 2025
USD 272.69 million
Revenue forecast in 2033
USD 498.80 million
Growth rate
CAGR of 7.84% from 2025 to 2033
Base year for estimation
2024
Historical data
2021 - 2023
Forecast period
2025 - 2033
Quantitative units
Revenue in USD million/billion and CAGR from 2025 to 2033
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, trends
Segments covered
Product and end use
Key companies profiled
Eisai Co., Ltd; Novartis AG; AbbVie Inc; Supernus Pharmaceuticals, Inc.; H. Lundbeck A/S; Biogen; AC IMMUNE SA; F. Hoffmann-La Roche Ltd; Daiichi Sankyo Company, Limited; Johnson & Johnson Services, Inc.; TauRx Pharmaceuticals Ltd.; Eli Lilly and Company; Alzheon.
Customization scope
Free report customization (equivalent up to 8 analysts working days) with purchase. Addition or alteration to country, regional & segment scope.
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
UK Alzheimer’s Therapeutics Market Report Segmentation
This report forecasts revenue growth at a country level and provides an analysis of the latest industry trends in each of the sub-segments from 2021 to 2033. For this study, Grand View Research has segmented the UK Alzheimer’s therapeutics market report based on product and end use:
-
Product Outlook (Revenue, USD Million, 2021 - 2033)
-
Cholinesterase Inhibitors
-
Donepezil
-
Galantamine
-
Rivastigmine
-
-
NMDA Receptor Antagonist
-
Combination Drug
-
Monoclonal Antibodies
-
Pipeline Drugs
-
-
End Use Outlook (Revenue, USD Million, 2021 - 2033)
-
Hospital Pharmacies
-
Retail Pharmacies
-
E-commerce
-
Frequently Asked Questions About This Report
b. The global UK Alzheimer’s therapeutics market size was estimated at USD 263.66 million in 2024 and is expected to reach USD 272.69 million in 2025.
b. The global UK Alzheimer’s therapeutics market is projected to grow at a CAGR of 7.84% from 2025 to 2033 to reach USD 498.80 billion by 2033.
b. Based on product, Cholinesterase inhibitors dominated the UK Alzheimer’s therapeutics market, accounting for 52.0% of the share in 2024, driven by the continued use of Donepezil. Commonly prescribed for mild to moderate Alzheimer’s disease, Donepezil enhances acetylcholine levels in the brain to help ease symptoms such as memory loss and cognitive decline.
b. Key players in the market include Eisai Co., Ltd, Novartis AG, AbbVie Inc., Supernus Pharmaceuticals, Inc., H. Lundbeck A/S, Biogen, AC IMMUNE SA. among others
b. The growth of the UK Alzheimer’s therapeutics market is primarily driven by the increasing prevalence of Alzheimer’s disease and the development of innovative treatment solutions. The rising proportion of elderly individuals is contributing to a surge in cognitive health issues, leading to more frequent diagnoses.
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










