- Home
- »
- Medical Devices
- »
-
UK Cardiovascular Clinical Trials Market Size Report, 2033GVR Report cover
![UK Cardiovascular Clinical Trials Market Size, Share & Trends Report]()
UK Cardiovascular Clinical Trials Market (2026 - 2033) Size, Share & Trends Analysis Report By Phase (Phase I, Phase II, Phase III, Phase IV), By Study Design Interventional, Observational), By Indication (Stroke, Cardiac Arrhythmias, Heart Failure), And Segment Forecasts
- Report ID: GVR-4-68040-904-7
- Number of Report Pages: 140
- Format: PDF
- Historical Range: 2021 - 2024
- Forecast Period: 2026 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
UK Cardiovascular Clinical Trials Market Summary
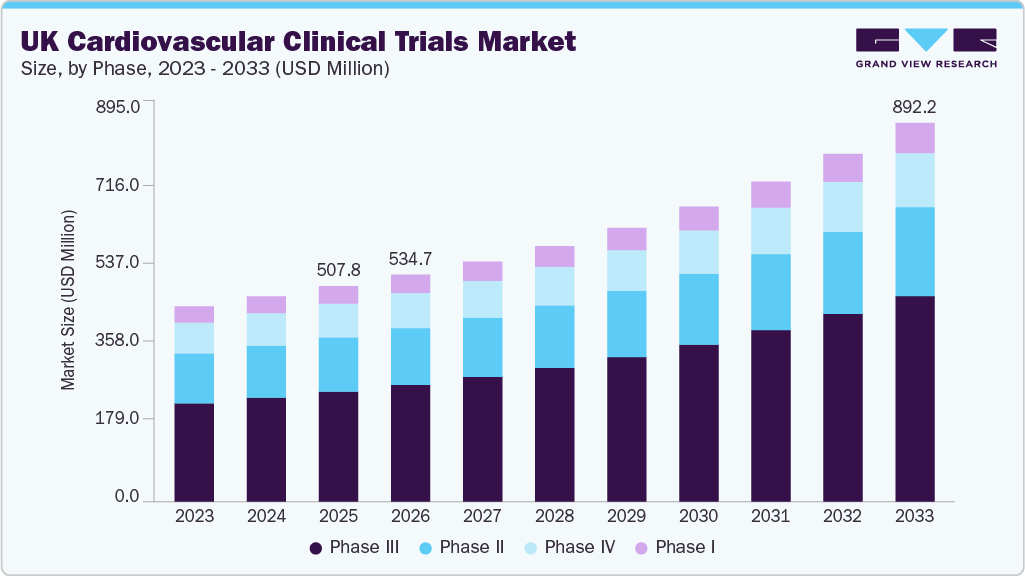
The UK cardiovascular clinical trials market size was estimated at USD 507.8 million in 2025 and is projected to reach USD 892.2 million by 2033, growing at a CAGR of 7.6% from 2026 to 2033. he market growth is driven by integrated healthcare data access, strong research funding, and regulatory acceleration.
Key Market Trends & Insights
- By phase, the phase III segment led the market with the largest revenue share of 51.03% in 2025.
- By study design, the interventional segment led the market with the largest revenue share in 2025.
- By indication, the heart failure segment dominated the market with the largest revenue share in 2025.
Market Size & Forecast
- 2025 Market Size: USD 507.8 Million
- 2033 Projected Market Size: USD 892.2 Million
- CAGR (2026-2033): 7.6%
In the country, the integrated infrastructure of the National Health Service (NHS) enables access to ~67 million patient records, which is expected to drive the recruitment and real-world evidence generation for cardiovascular clinical trials. In addition, the growing prevalence of cardiovascular disease (~7.6 million patients) has driven an increase in clinical trials, further contributing to market growth.The UK cardiovascular clinical trials industry is growing due to rising cardiovascular disease burden in the country, regulatory acceleration and adaptive trial frameworks in the UK, and increasing investment in cardiovascular drugs innovation and late-stage pipelines. Due to the rising incidence of cardiovascular disease across the UK, there is continued demand for long-term, large-scale clinical trials. Cardiovascular conditions like heart failure, coronary heart disease (CHD), atrial fibrillation, and ischemic heart disease require ongoing innovations in medications and treatment. This is mainly responsible for the growing number of clinical trials in the country, as these trials are usually conducted using evidence from real-world experiences from NHS databases.
The data published by the British Heart Foundation (BHF) in January 2026 stated that nearly 7 million people currently live with cardiovascular disease in England. The growing and aging population is further expected to boost the rate of CVDs, owing to improved survival rates from cardiovascular events. As the risk of CVD increases with age, by 2030, the population in England aged 65-84 is anticipated to increase by 29% and those aged over 85 by 61%. Furthermore, CVDs are also responsible for ~26% (140,000) of the total deaths in the country per year, which equates to 380 people per day. Amongst which, 38,000 people die under the age of 75 in England each year. Such a growing burden of CVDs in the country has increased the demand for new interventions and treatments, as the government and the public health authorities-led efforts aimed at reducing deaths from cardiovascular disease are providing a steady stream of financial support to clinical studies in the country. In addition, the demand for preventive cardiology therapies is increasing the clinical trial pipeline for cholesterol-lowering drugs and hypertension medications. All these factors contribute to market growth.
Furthermore, the growing investment in drugs for cardiometabolic diseases is boosting the pipeline of cardiovascular clinical trials in the UK, as pharmaceutical companies develop treatments for lipid disorders, heart failure, and inflammation. Besides, large-scale late-phase clinical trials are underway in various parts of the world, involving more than 10,000 participants, which is driving greater demand for UK-based clinical trial sites due to their specialized facilities. Moreover, research into new modes of action, such as inhibiting PCSK9 with RNA-based drugs and anti-inflammatories, is generating interest in early- and mid-phase clinical trials. Also, cooperation between international sponsors and domestic research networks is speeding up site activation and the patient enrollment process. Besides, there is intense competition among companies to demonstrate better cardiovascular outcomes, leading to a higher number of trials involving comparative and combination studies. Furthermore, regulatory pressure for the new drug discovery process is driving a rapid transition from Phase II to Phase III clinical trials.
In addition, government support, both public and private, is helping translational research progress to clinical phase trials. For example, in January 2026, Novo Nordisk announced the extension of its cardiovascular outcome studies in the UK, following increased clinical focus on reducing obesity-related cardiovascular risks. Thus, such an increase in the growing number of investments in the field of cardiology and late-stage pipelines is anticipated to propel the market growth.
Opportunity Analysis
There are substantial growth opportunities in the UK's cardiovascular clinical trials sector, driven by an increase in the use of data-enabled research methods and an emphasis on pre-emptive cardiology. Moreover, the growing access to patient data for high-risk patients, decentralization of clinical trials, early-stage interventions, and advancements in genomics and biomarkers are anticipated to provide numerous opportunities for market growth in the near future.
The access to patient data through the NHS allows for easy identification of high-risk patients and follow-up in the long run. The use of a decentralized trial design further accommodates increased participation by the rural and elderly populations that suffer from cardiovascular disease. There is also an increasing need to prevent cardiovascular disease, which in turn leads to more early-stage interventions targeting lipid regulation and metabolic risk factors. Also, advances in genomics and biomarker identification have enabled stratified trial designs, paving the way for the development of targeted drugs for conditions such as familial hypercholesterolemia and heart failure. Also, RNA and gene-editing therapy trials are becoming common at specialized UK centers, ultimately resulting in the need for niche clinical expertise.
Furthermore, closer cooperation between academia and industry is facilitating translational research, thereby reinforcing the flow from discovery to late-stage clinical development. Moreover, the centralization of the United Kingdom's healthcare system is enabling the conduct of larger outcome trials with more than 10,000 participants, thereby attracting international sponsors seeking strong clinical data. Also, the use of wearable and remote monitoring technology enables continuous cardiovascular data collection, thereby improving endpoint validity and patient adherence. Additionally, real-world evidence-based research offers opportunities to use hybrid models combining randomized and observational data. Moreover, contract research organizations are enhancing their technological capabilities and expanding their site networks, thereby fostering efficient, scalable cardiovascular research.
Therefore, these emerging trends have created a favorable environment for innovation and growth in the UK cardiovascular clinical trials market.
Technological Advancements
There have been tremendous developments in the UK cardiovascular clinical trial technology space, driven by advancements in digital health platforms and data-driven approaches, with connected platforms providing access to large-scale longitudinal patient data for trial design and patient recruitment. The adoption of electronic health record systems and real-world data has enhanced the feasibility analysis and patient stratification in complex cardiovascular trials. Similarly, wearable devices and remote monitoring technologies have enabled continuous monitoring of cardiovascular markers, such as heart rate, blood pressure, and activity.

Moreover, there has been increased uptake of hybrid and decentralized trial models, with the use of telemedicine and digital consent forms, which are improving patient participation from dispersed geographic locations. Also, there has been a rise in the use of artificial intelligence (AI) and machine learning (ML) to predict patient outcomes and optimize patient enrollment in trials. In addition, the use of genomics and biomarkers in cardiovascular trials is enabling the design of trials targeting specific subpopulations of cardiovascular conditions. The cloud-based data management systems and platforms that are interoperable have enabled smooth data sharing among sponsors, contract research organizations, and clinical sites, thereby improving collaboration and data integrity. Such advancements and the adoption of modern technologies in the clinical trials setting have positively impacted the market growth.
Pricing Model Analysis
The pricing model landscape for the UK cardiovascular clinical trials industry is a mix of performance and traditional models. The fee-for-service and milestone-based pricing approaches remain predominant in large-scale outcome-oriented trials conducted in NHS settings due to their compatibility with predetermined research designs and long-term follow-up. Additionally, such models provide sponsors with a clear budget perspective for trials with over 5,000 participants, though growing design complexities and adaptive endpoints are slowly favoring more flexible pricing models.
On the other hand, full-time equivalent pricing models have become increasingly popular in early-stage cardiovascular clinical trials, which require continuous scientific input and modifications throughout the research. The following figure depicts the key pricing models adopted by the companies in the UK cardiovascular clinical trials market.
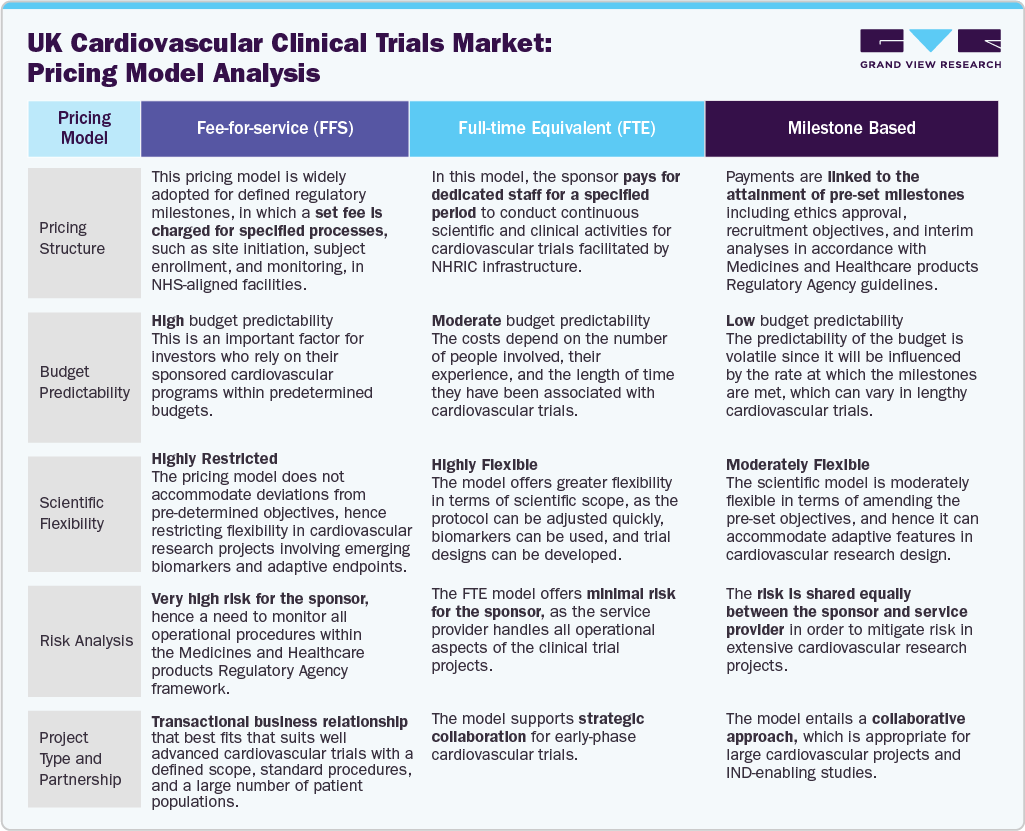
Besides, the growing use of decentralized, wearable trial components is driving new cost considerations for digital infrastructure and patient monitoring, leading to a shift towards more flexible pricing terms between sponsors and CROs. In addition, the range of services offered by CROs is expanding, including data management, decentralized trial monitoring, and patient recruitment, among others, resulting in the development of integrated pricing terms. Moreover, there is a growing demand for cost efficiency in long-term cardiovascular trials, which has led to the development of risk-sharing and performance-based contracts.
Market Concentration & Characteristics
The market growth stage is medium, and the pace of the market growth is accelerating. The UK cardiovascular clinical trials market is characterized by a high degree of innovation. Level of M&A activities, impact of regulations, service expansions, and regional expansions.
Innovation in the UK cardiovascular clinical trials industry is characterized by the implementation of decentralized and hybrid trials, as well as the use of analytics and AI-driven patient stratification, through the inclusion of wearable devices and real-world data to improve efficiency and accuracy of results. Additionally, trials incorporating biomarkers and genomics are facilitating the development of precision cardiology for subgroups such as heart failure phenotypes and lipid subgroups.
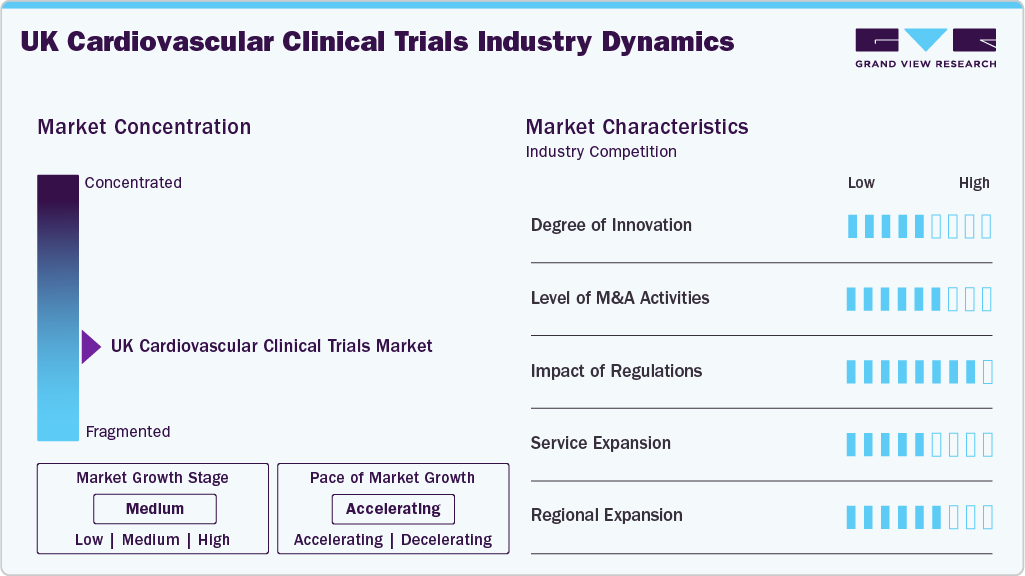
There is an intermediate level of merger and acquisition activity. This is due to global sponsors and contract research organizations increasingly focusing on enhancing their therapeutic capabilities and gaining access to NHS-linked trial facilities. Furthermore, mergers and acquisitions involving CROs, biotech firms, and academic institutions are more frequent compared to those involving large-scale acquisitions.
Regulatory changes initiated by the MHRA and the HRA have affected the market through joint reviews and shortened approval timelines. In addition, regulatory guidance related to transparency, data management, and patient safety is making compliance increasingly difficult, especially for lengthy cardiovascular outcomes trials. The regulatory changes have made the UK an attractive site for conducting complex cardiovascular research trials.
CROs are moving toward providing end-to-end clinical trial services, such as decentralized trials, digital data collection, and real-world evidence inclusion for cardiovascular treatment. This development enhances their scalability. In addition, increased investment in technologies, such as remote monitoring systems and innovative patient recruitment programs, has enabled CROs to handle complex cardiovascular trials that require the participation of large numbers of patients.
The clinical trials market for CVD in the UK possesses a relatively fragmented organizational framework, with international CROs, regional service providers, academic organizations, and specialized vendors participating across various phases of trials. Furthermore, large CROs have a commanding presence in later-stage outcome trials, while smaller firms and research facilities specialize in early-stage trials.
Phase Insights
The phase III segment dominated the UK cardiovascular clinical trials market, accounting for a revenue share of 51.03% in 2025. The segment’s growth is driven by the rising need to confirm efficacy and safety in large patient populations, the growing demand for late-stage validation of innovative therapies, and regulatory requirements for market approval. Besides, the increasing burden of cardiovascular disease and the focus on comparative effectiveness and outcomes-based evidence further accelerate Phase III clinical trial activity.According to ABPI, phase III accounted for the largest single category of MHRA submissions, 223 of 615 initial applications from September 2023 to August 2024, and grew 23.6% in initiation volumes in 2024 compared to 2023. Besides, the UK government has set an ambitious target to reduce the time from application to first participant from 250 days to 150 days by March 2026. Thus, this has led to low-risk phase III trials by the MHRA in 14 days instead of the statutory 30 days, PCI, a reform that directly compresses time-to-initiation for sponsors.
The phase I segment is expected to grow significantly during the forecast period. Phase I trials are driven by early-stage evaluation of safety, tolerability, and pharmacokinetics of novel therapies. In addition, the segment's growth is supported by increased innovation in cardiovascular drug development, the availability of specialized research units, rising investment in first-in-human studies, particularly for advanced cardiovascular and biologic therapies, and supportive regulatory data.
For instance, a strong regulatory framework under the Medicines and Healthcare Products Regulatory Agency (MHRA) and an integrated research infrastructure supported by the National Institute for Health and Care Research drive the country as a leading destination for early-phase trials. The agency has introduced a streamlined 14-day assessment pathway for Phase I trials, reinforcing the country’s commitment to accelerating first-in-human studies. Supporting this trend, data from the GOV.UK indicated that trials involving healthy volunteers were increased by 16% between January and November 2025 compared to the same period in 2024, highlighting growing early-stage research activity. Such factors are expected to drive the market growth.
Study Design Insights
The interventional segment dominated the UK cardiovascular clinical trials industry in 2025. The segment growth is driven by expanding therapeutic innovations across drugs, devices, and procedures. The adoption of adaptive trial designs, increased regulatory support, and demand for high-quality efficacy data are further driving market growth.In addition, the segment is expected to see new growth opportunities, with established market players such as IQVIA, ICON plc, Medpace, and MAC Clinical Research. These companies are well-positioned to offer interventional study mandates as sponsors shift toward adaptive, decentralized, and master-protocol designs. Such factors are expected to drive the market growth.
The observational segment is expected to grow significantly during the forecast period. The segment growth is driven by growing demand for real-world evidence to assess long-term safety and effectiveness. The increasing use of electronic health records, patient registries, and digital health tools supports large-scale data generation. Regulatory emphasis on post-marketing surveillance and value-based healthcare further contributes to their growth. In addition, the expansion of digital health technologies, including electronic health records, wearable devices, and AI-driven analytics, enables large-scale, longitudinal data collection at lower cost and is expected to drive market growth.
For instance, in September 2025, an international study reported a study underway to enhance early detection and management of patients at risk of pre-heart failure and cardiometabolic-renal multimorbidity. The research uses routinely collected healthcare data from the UK, Denmark, and the U.S., ensuring a broad, real-world representation of patients. This large, decentralized observational study is designed to mirror everyday clinical practice, helping uncover treatment gaps and unmet medical needs. Such strategic initiatives are expected to drive the market.
Indication Insights
The heart failure segment dominated the UK cardiovascular clinical trials market in 2025. The segment growth is driven by rising prevalence, aging population, and high hospitalization rates. The need for improved therapies, increasing focus on personalized medicine, and advancements in biomarkers and digital monitoring technologies are accelerating clinical trials in the market. It affects around 900,000 people in the UK and poses a significant burden on the National Health Service (NHS), accounting for one million bed days per year. The disease reduces life expectancy with a one-year survival rate of 75.9% post-diagnosis and a 10-year survival of 24.5%. Patients living with HF suffer from disability and reduced quality of life. With an aging population, increased survival from myocardial infarctions, and rising prevalence of risk factors, are expected to drive the need for cardiovascular drugs, further contributing to market growth.
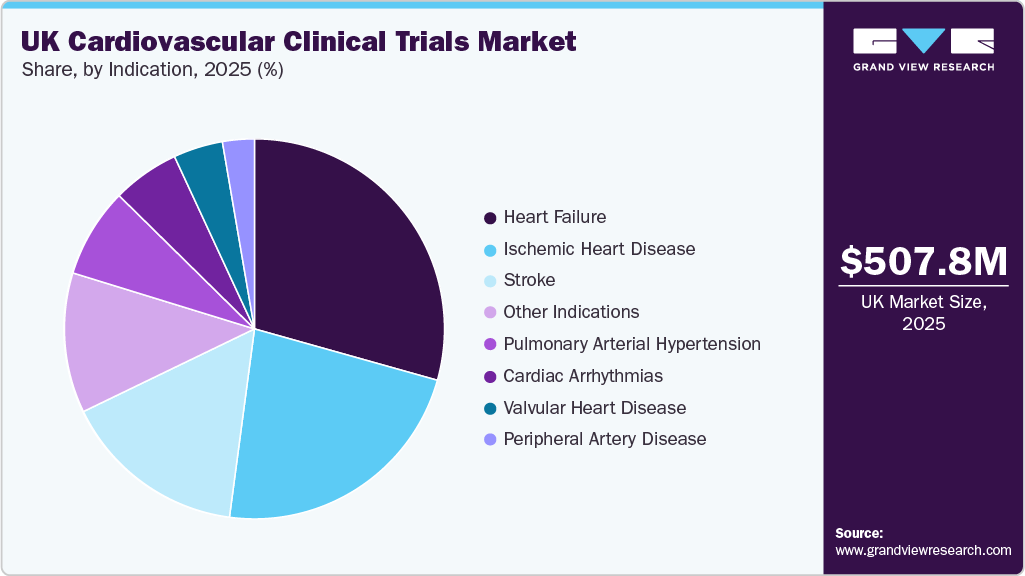
The ischemic heart disease segment is expected to grow significantly during the forecast period. The ischemic heart disease segment growth is driven by its high prevalence and mortality burden. Additionally, the growing demand for cardiovascular drugs, the increasing focus on prevention and early diagnosis, and the growing use of real-world data and large-scale outcome studies are key factors supporting research and market expansion. For instance, real-world data sources such as the Clinical Practice Research Datalink and NHS primary care databases are enabling advanced trial emulation studies. Thus, these factors are expected to drive the country as one of the major hubs for ischemic heart disease research and are expected to drive market growth over the forecast period.
Key UK Cardiovascular Clinical Trials Company Insights
Key players in the UK cardiovascular clinical trials market include IQVIA Inc.; Parexel International (MA) Corporation; Medpace Holdings, Inc.; Charles River Laboratories; AstraZeneca; Novartis; Pfizer; and Johnson & Johnson. Market share is driven by technological capabilities, regulatory compliance, and integrated services, with top companies commanding a significant portion of high-value contracts. For instance, in September 2025, IQVIA launched its AI-enabled Clinical Trial Financial Suite to streamline budgeting, contracting, and payments. Such an initiative enhances efficiency and transparency in clinical trials by accelerating financial workflows, reducing delays, and improving site payment management.
Key UK Cardiovascular Clinical Trials Companies:
- IQVIA Inc.
- ICON plc
- Parexel International (MA) Corporation
- Medpace Holdings, Inc.
- Charles River Laboratories
- MAC Research
- Sofpromed
- Syneos Health
- Worldwide Clinical Trials
- Laboratory Drug Development
- AstraZeneca
- Novartis
- Pfizer
- Bristol Myers Squibb
- Johnson & Johnson
Recent Developments:
-
In March 2026, AstraZeneca completed a wearable ECG-based observational heart failure study evaluating remote monitoring endpoints. Such advancements of decentralized clinical trials in the UK enable real-world data collection, improved patient monitoring, and faster cardiovascular drug development timelines.
-
In February 2026, Bristol Myers Squibb partnered with Evinova to deploy AI-enabled study design tools. Such a collaboration supports clinical trials by optimizing protocols, reducing timelines, and improving the efficiency of patient-centric trial execution in the UK.
UK Cardiovascular Clinical Trials Market Report Scope
Report Attribute
Details
Market size value in 2026
USD 534.7 million
Revenue forecast in 2033
USD 892.2 million
Growth rate
CAGR of 7.6% from 2026 to 2033
Actual data
2021 - 2024
Forecast period
2026 - 2033
Quantitative units
Revenue in USD million and CAGR from 2026 to 2033
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Phase, study design, indication
Country scope
UK
Key companies profiled
IQVIA Inc.; ICON plc; Parexel International (MA) Corporation; Medpace Holdings, Inc.; Charles River Laboratories; MAC Research; Sofpromed; Syneos Health; Worldwide Clinical Trials; Laboratory Drug Development; AstraZeneca; Novartis; Pfizer; Bristol Myers Squibb; Johnson & Johnson
Customization scope
Free report customization (equivalent up to 8 analyst’s working days) with purchase. Addition or alteration to country, regional & segment scope.
UK Cardiovascular Clinical Trials Market Report Segmentation
This report forecasts revenue growth at the country level and provides an analysis of the latest industry trends in each of the sub-segments from 2021 to 2033. For this study, Grand View Research has segmented the UK cardiovascular clinical trials market report based on phase, study design, and indication:
-
Phase Outlook (Revenue, USD Million, 2021 - 2033)
-
Phase I
-
Phase II
-
Phase III
-
Phase IV
-
-
Study Design Outlook (Revenue, USD Million, 2021 - 2033)
-
Interventional
-
Observational
-
Expanded Access
-
-
Indication Outlook (Revenue, USD Million, 2021 - 2033)
-
Valvular Heart Disease
-
Peripheral Artery Disease
-
Ischemic Heart Disease
-
Pulmonary Arterial Hypertension
-
Stroke
-
Cardiac Arrhythmias
-
Heart Failure
-
Other Indications
-
Frequently Asked Questions About This Report
b. The UK cardiovascular clinical trials market size was estimated at USD 507.8 million in 2025 and is expected to reach USD 534.7 million in 2026.
b. The UK cardiovascular clinical trials market is expected to grow at a compound annual growth rate of 7.6% from 2026 to 2033 to reach USD 892.2 million by 2033.
b. Phase III segment dominated the market for UK cardiovascular clinical trials and accounted for the largest revenue share of 51.03% in 2025. The dominance is driven by the rising need to confirm efficacy and safety in large patient populations, the growing demand for late-stage validation of innovative therapies, and regulatory requirements for market approval. Besides, the increasing burden of cardiovascular disease and the focus on comparative effectiveness and outcomes-based evidence further accelerate Phase III clinical trial activity.
b. Some key players operating in the UK cardiovascular clinical trials market include IQVIA Inc., ICON plc, Parexel International (MA) Corporation, Medpace Holdings, Inc., Charles River Laboratories, MAC Research, Sofpromed, Syneos Health, Worldwide Clinical Trials, Laboratory Drug Development, AstraZeneca, Novartis, Pfizer, Bristol Myers Squibb, and Johnson & Johnson among others.
b. Key factors driving the UK cardiovascular clinical trials market growth include integrated healthcare data access, robust research funding, and regulatory acceleration. In the country, the integrated infrastructure of the National Health Service (NHS) enables access to ~67 million patient records, which is expected to drive the recruitment and real-world evidence generation for cardiovascular clinical trials.
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










