U.S. Artificial Organs And Bionics Market Summary
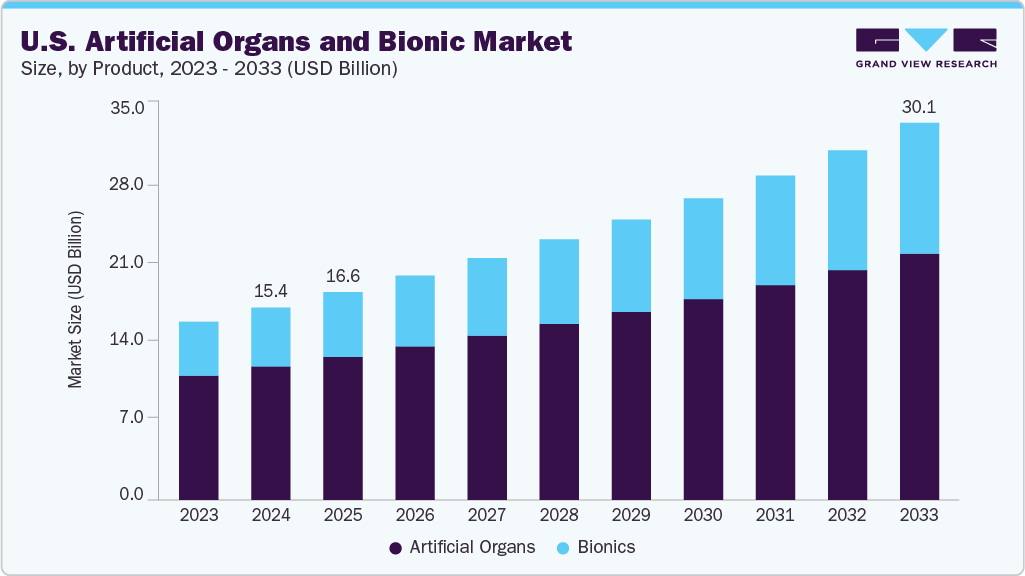
The U.S. artificial organs and bionics market size was estimated at USD 15.35 billion in 2024 and is projected to reach USD 30.11 billion by 2033, growing at a CAGR of 7.7% from 2025 to 2033. The market growth is attributed to the increasing number of transplants and the rising number of people waiting for donors.
Key Market Trends & Insights
- By product, the artificial organs segment accounted for a revenue share of 69.4% in 2024.
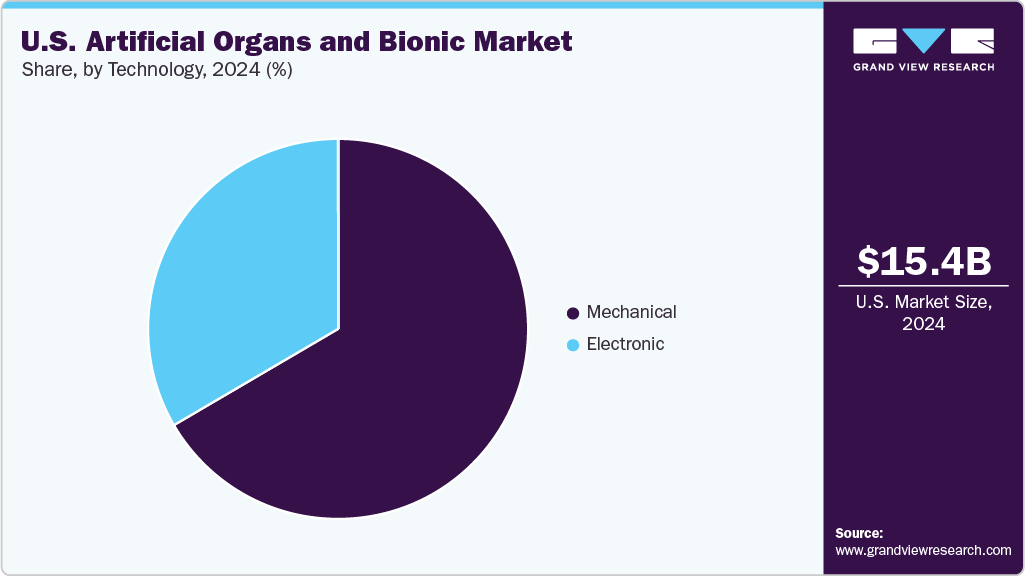
- By technology, the mechanical segment accounted for a revenue share of 66.5% in 2024.
Market Size & Forecast
- 2024 Market Size: USD 15.35 Billion
- 2033 Projected Market Size: USD 30.11 Billion
- CAGR (2025-2033): 7.7%
In 2023, as reported by the Human Resource and Services Administration, the national transplant waiting list in the U.S. exceeded 104,000 individuals. Currently, a new name is placed on the waiting list every ten minutes, underscoring the increasing need for artificial medical devices to address unmet demand. This includes various devices designed to restore function or fully replace failed organs. The National Institute of Biomedical Imaging and Bioengineering (NIBIB), part of the National Institutes of Health (NIH), actively supports the development of these technologies to help people live healthier, more independent lives.
The U.S. government plays a key role in fostering innovation in the U.S. artificial organs and bionic industry through significant funding for research. The NIH, through its various institutes such as the National Institute of Neurological Disorders and Stroke (NINDS), has funded research into next-generation neural prosthetics that can restore movement and sensation by using a computerized interface.
Regulatory Acceleration and Funding
The FDA expedited device approvals for artificial organs, including issuing breakthrough device designations to new-generation heart-assist and kidney devices, streamlining market entry and clinical availability for patients in need.
The National Institutes of Health (NIH) sharply increased funding for research in bioengineered organ tissues, bionic limbs, and implantable electronics. 2024 witnessed NIH support for projects using 3D bioprinting, gene editing, and smart prosthetics, especially for advancing lab-grown organ constructs and neural-controlled prosthetics.
Medicare and Insurance Support
Medicare reimbursement and private insurance program enhancements for approved artificial organ and bionic devices facilitated wider adoption of high-cost devices. These measures improved access for patients requiring life-saving implants such as mechanical hearts, cochlear or retinal implants, and exoskeletons.
Public-Private Research Alliances
Academic research centers joined forces with engineering and biomedical companies in federally supported collaborations to accelerate innovation in artificial organs and bionics. Flagship university hospitals and biotech firms received targeted grants for pivotal trials involving bioartificial kidneys, regenerative organs, and brain-computer interfaces.
Government-backed partnerships promoted the development and real-world adoption of personalized bionic solutions and AI-enabled prosthetics, enabling the full physiological integration of devices for mobility and patient independence.
National Prioritization
The shortage of donor organs, growing chronic disease burden, and aging population continued to make artificial organs and bionics a national health priority. Government policies encouraged scientific investment and streamlined regulatory hurdles to accelerate product introduction and meet transplant demand.
These initiatives collectively positioned the U.S. as a global leader in artificial organ and bionic innovation, market adoption, and advanced clinical treatments in 2024.
Product Insights
The artificial organ segment held the largest revenue share of 69.4% in 2024. The growth can be attributed to the critical shortage of human donor organs. This life-threatening gap between organ demand and supply has made artificial organs, such as hearts, kidneys, liver, and lungs, a vital solution for patients with end-stage organ failure. A rigorous federal approval process is required to bring these life-saving technologies to market. The NIH also funded ‘The Kidney Project’ at the University of California, San Francisco (UCSF), which is developing an implantable bio-artificial kidney designed to use the body's blood pressure to filter the blood, thereby eliminating the need for an external power source or continuous dialysis treatments.
The bionic segment is expected to grow at the fastest CAGR of 9.2% over the forecast period. Rapid advancements in robotics, neuroscience, and advanced materials drive the segment. These technologies have enabled the creation of sophisticated artificial bionic devices that restore function and improve the quality of life for a broad patient population. For instance, the U.S. FDA has approved various cochlear implants, such as MED-EL, that directly stimulate the auditory nerve, allowing individuals with severe hearing loss to regain a sense of sound. In addition, government-funded research has led to the development of powered exoskeletons that can assist people with mobility impairments, as supported by the NIH.
Technology Insights
The mechanical segment dominated the market with a revenue share of 66.5% in 2024. Advancements in materials, science and biomechanics drive growth. This technology is critical for creating durable physical structures, precise mechanisms, and reliable power systems that allow artificial organs and bionics to integrate seamlessly with the human body.
Federal agencies are heavily involved in R&D to ensure the safety and efficacy of these complex systems. For example, the U.S. FDA has sponsored initiatives to validate and standardize Computational Fluid Dynamics (CFD) to design and analyze blood-contacting devices. These FDA-led projects, which involve collaborations with various research groups, focus on evaluating how CFD can improve the mechanical design of devices such as artificial heart valves and blood pumps to minimize the risk of blood clots.
Furthermore, the NIH actively funds research into using advanced biomaterials, such as specialized polymers and metals, to prevent rejection and improve the long-term viability of implanted devices. These mechanical innovations are essential for ensuring the longevity and functional effectiveness of both artificial organs and bionic limbs.
The electronic segment is expected to grow at the fastest rate over the forecast period. Breakthroughs in microprocessors, biosensors, and power management drive growth. This technology is essential for the intelligence and functionality of artificial organs and bionic devices, enabling them to communicate with the body and make real-time adjustments. For example, the National Institute of Diabetes and Digestive and Kidney Diseases (NIDDK) has championed the development of a fully electronic artificial pancreas that uses a continuous glucose monitor and an insulin pump to manage blood sugar levels.
Key U.S. Artificial Organs And Bionic Company Insights
Some of the key companies in the U.S. artificial organs and bionic industry include ABIOMED (Johnson & Johnson), Berlin Heart, SynCardia Systems, LLC., and others.
-
ABIOMED is a Johnson & Johnson medical technology firm specializing in bionic circulatory support devices. It offers implantable cardiac pumps, such as the Impella system, for patients with heart failure.
Key U.S. Artificial Organs And Bionic Companies:
- ABIOMED
- Berlin Heart
- Zimmer Biomet
- Boston Scientific Corporation
- Cochlear Ltd.
- Edwards Lifesciences Corporation
- Ekso Bionics
- Medtronic
- Jarvik Heart, Inc
- SynCardia Systems, LLC
Recent Developments
-
In October 2024, Beta Bionics, Inc. announced the launch of a new integration: ILet Bionic Pancreas with Abbott's FreeStyle LIbre 3 Plus sensor. This integration allows users to manage their diabetes, reducing their burden.
-
In April 2024, United Therapeutics Corporation announced the first successful transplant of a UThymoKidney into a living person.
-
In February 2024, SynCardia Systems, LLC announced the publication of a patent application for its next-generation Total Artificial Heart, signaling innovation and intellectual property growth.
U.S. Artificial Organs And Bionic Market Report Scope
|
Report Attribute
|
Details
|
|
Market size value in 2025
|
USD 16.58 billion
|
|
Revenue forecast in 2033
|
USD 30.11 billion
|
|
Growth rate
|
CAGR of 7.7% from 2025 to 2033
|
|
Base year for estimation
|
2024
|
|
Historical data
|
2021 - 2023
|
|
Forecast period
|
2025 - 2033
|
|
Quantitative units
|
Revenue in USD million/billion and CAGR from 2024 to 2033
|
|
Report coverage
|
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
|
|
Segments covered
|
Product, technology
|
|
Key companies profiled
|
ABIOMED; Berlin Heart; Zimmer Biomet; Boston Scientific Corporation; Cochlear Ltd.; Edwards Lifesciences Corporation; Ekso Bionics; Medtronic; Jarvik Heart, Inc; SynCardia Systems, LLC
|
|
Customization scope
|
Free report customization (equivalent up to 8 analysts working days) with purchase. Addition or alteration to country & segment scope.
|
|
Pricing and purchase options
|
Avail customized purchase options to meet your exact research needs. Explore purchase options
|
U.S. Artificial Organs And Bionic Market Segmentation
This report forecasts revenue growth at country level and provides an analysis of the latest industry trends in each of the sub-segments from 2021 to 2033. For this study, Grand View Research has segmented the U.S. artificial organs and bionic market report based on product and technology:
-
Product Outlook (Revenue, USD Billion, 2021 - 2033)
-
Artificial Organs
-
Kidney
-
Heart
-
Liver
-
Lungs
-
Pancreas
-
Bionic
-
Cochlear Implant
-
Exoskeleton
-
Bionic Limbs
-
Brain Bionic
-
Vision Bionic
-
Technology Outlook (Revenue, USD Billion, 2021 - 2033)













