- Home
- »
- Clinical Diagnostics
- »
-
U.S. Blood-based Biomarker For Parkinson’s Disease Market 2033GVR Report cover
![U.S. Blood-based Biomarker For Parkinson’s Disease Market Size, Share & Trends Report]()
U.S. Blood-based Biomarker For Parkinson’s Disease Market (2025 - 2033) Size, Share & Trends Analysis Report By Biomarker Type (Protein Biomarkers, Inflammatory Markers), By Technology (ELISA/Immunoassay, Multiplex Platforms), By End Use, And Segment Forecasts
- Report ID: GVR-4-68040-661-5
- Number of Report Pages: 150
- Format: PDF
- Historical Range: 2021 - 2024
- Forecast Period: 2025 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
Market Size & Trends
The U.S. blood-based biomarker for parkinson’s disease market size was estimated at USD 45.75 million in 2024, and is projected to grow at a CAGR of 18.33% from 2025 to 2033. The U.S. Parkinson’s disease (PD) blood-based biomarkers market is driven by a growing patient population, with 90,000 new diagnoses annually, and increasing demand for early, non-invasive diagnostic tools. Expanding research efforts, especially in academic and pharmaceutical settings, are accelerating biomarker discovery and validation. Technological advancements in multiplex assays and AI-based platforms are further supporting adoption. In addition, collaborations involving the NIH, MJFF, and Verily are helping standardize biomarker use. As assays such as α-synuclein and NfL progress toward regulatory approval, their integration into clinical practice is becoming increasingly viable.
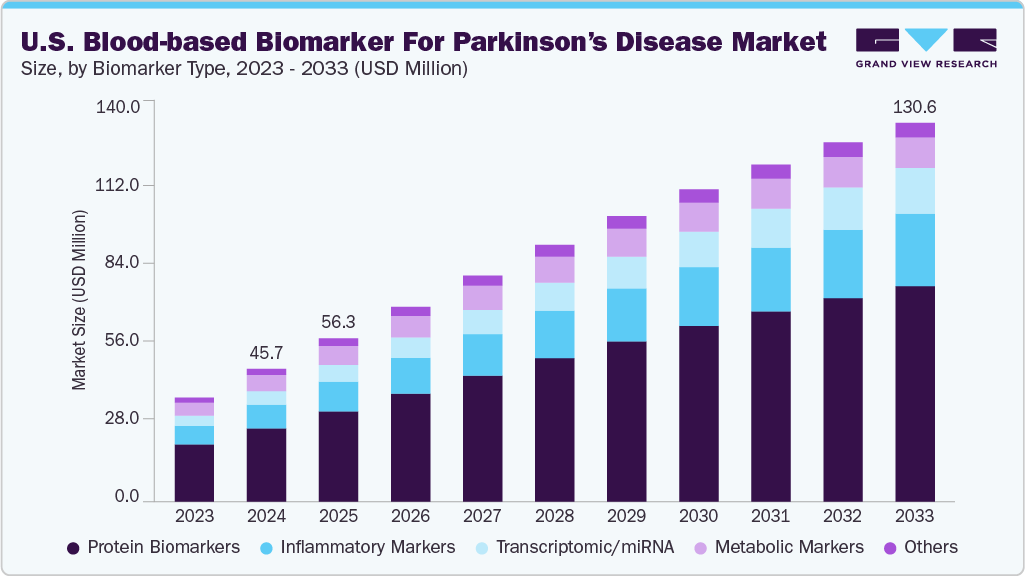
The U.S. blood-based biomarker for parkinson’s disease market is gaining significant momentum, fueled by the need for earlier diagnosis, improved disease monitoring, and more precise patient stratification in clinical trials. With over 1.1 million Americans currently living with Parkinson’s disease and an estimated 90,000 new diagnoses each year-a 50% increase over prior estimates-there is growing urgency for scalable, non-invasive diagnostic tools that go beyond traditional clinical evaluation and imaging.
Historically, PD diagnosis has relied heavily on the presence of motor symptoms, leading to frequent misdiagnoses and delayed treatment. Blood-based biomarkers offer a non-invasive, accessible alternative that can identify disease onset years before symptoms emerge. Key biomarkers under investigation include misfolded α-synuclein, neurofilament light chain (NfL), DJ-1, tau, inflammatory cytokines (e.g., IL-6, CRP), and oxidative stress markers. These are being developed across multiple technological platforms, including ELISA, multiplex proteomics, exosome-based assays, and AI-integrated multi-omics.
Amprion, a U.S.-based diagnostics company, is advancing a blood-based version of its SynRT™ test, which detects misfolded α-synuclein-a hallmark of PD pathology. Although currently approved only for cerebrospinal fluid (CSF), this test is undergoing clinical studies for blood validation. C2N Diagnostics, known for its Alzheimer’s PrecivityAD™ test, is extending its platform to PD with targeted assays for NfL and tau using mass spectrometry.
Pharmaceutical companies are also integrating biomarkers into therapeutic development. Denali Therapeutics and Biogen are utilizing phosphorylated Rab10 as a pharmacodynamic biomarker in trials for their LRRK2 inhibitors. Roche/Genentech is partnering with AC Immune to co-develop antibody-based therapies, while simultaneously validating associated α-synuclein biomarkers. These biomarkers are essential not only for determining therapeutic response but also for selecting appropriate patient subgroups-particularly those with LRRK2 or GBA mutations.
On the technology front, Olink Proteomics has deployed its Proximity Extension Assay (PEA) platform to identify PD-relevant protein panels, in partnership with the Michael J. Fox Foundation (MJFF). Meanwhile, Verily Life Sciences (an Alphabet subsidiary) is building AI-based multi-omic platforms to analyze blood, cerebrospinal fluid, and digital biomarkers as part of the MJFF-funded Parkinson’s Progression Markers Initiative (PPMI).
Academic institutions such as the University of Pennsylvania, Mayo Clinic, and Massachusetts General Hospital are also central to the biomarker ecosystem. They are running NIH- and MJFF-funded studies to validate exosomal α-synuclein, miRNA panels, and blood-based NfL as part of longitudinal cohorts. These efforts are critical for advancing biomarker candidates toward CLIA certification, FDA validation, and eventual CMS reimbursement. As the market moves toward clinical utility, the next 5–10 years will likely witness a transition from research to routine care, with hospitals and specialty neurology clinics beginning to incorporate biomarker-based blood tests for diagnosis, treatment selection, and disease monitoring.
Market Concentration & Characteristics
The U.S. blood-based biomarker for parkinson’s disease market demonstrates a high degree of innovation, driven by the shift from symptom-based diagnosis to molecular and precision diagnostics. Innovations include misfolded α-synuclein seed amplification assays (e.g., Amprion’s SynRT), multiplex proteomic panels (Olink), exosomal biomarkers, and AI-integrated multi-omic platforms (Verily). These advances aim to detect PD in preclinical stages, differentiate subtypes, and monitor progression non-invasively. Pharma companies are also pioneering the use of biomarkers as companion diagnostics in clinical trials, supporting targeted drug development and personalized treatment strategies for PD patients.
The level of M&A activity in this space is moderate but rising, reflecting growing strategic interest in blood-based biomarker technologies for neurodegenerative diseases. Recent acquisitions have focused on gaining access to advanced proteomics platforms, multiplex assay capabilities, and AI-integrated biomarker discovery tools. Consolidation is being driven by the need to integrate diagnostics with therapeutic development, especially as biomarkers become essential in clinical trial stratification and early disease detection. Strategic partnerships between diagnostics developers and large reference labs or hospital systems are also increasing, aimed at expanding access to emerging blood-based assays through CLIA-certified channels.
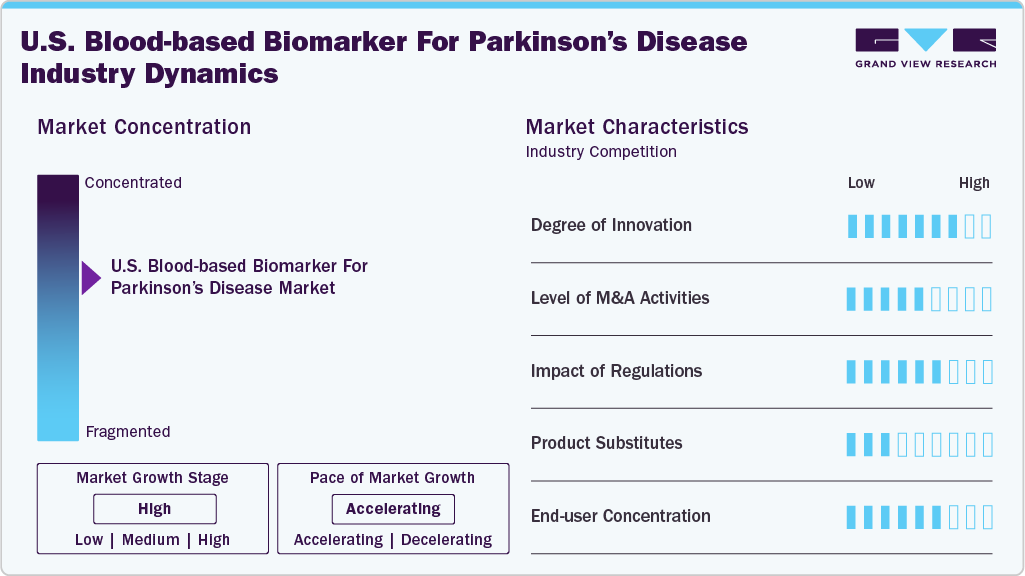
Regulation significantly influences the adoption of blood-based biomarkers in Parkinson’s disease. The FDA’s proposed oversight of lab-developed tests (LDTs) will require stricter validation and clinical evidence, potentially slowing approval but improving test reliability. Medicare coverage through CMS depends on analytical validity and demonstrated clinical utility, making reimbursement challenging without robust data. Recent FDA approval of Alzheimer’s blood tests offers a regulatory blueprint, suggesting a pathway for PD biomarkers. Additionally, CLIA certification ensures lab-based quality standards. Collectively, these frameworks are critical for clinical integration, market access, and payer acceptance.
Biomarker Type substitutes for blood-based biomarkers in Parkinson’s disease include several established and emerging diagnostic approaches. The most prominent alternatives are cerebrospinal fluid (CSF) biomarkers, which offer higher sensitivity but require invasive lumbar puncture, limiting routine use. Neuroimaging techniques, such as DaTscan (dopamine transporter imaging), are widely used to support diagnosis but are expensive and not disease-specific. Genetic testing is useful for identifying risk mutations (e.g., LRRK2, GBA) but lacks diagnostic value for sporadic cases. Additionally, clinical rating scales and symptom assessments remain the standard of care but are subjective and limited in early disease detection.
End User concentration in this market is currently moderate, with demand primarily concentrated among academic research centers, pharmaceutical companies, and select hospital-based neurology clinics. A small number of top-tier institutions-such as major universities, NIH-funded sites, and movement disorder centers-account for a significant share of test usage due to their involvement in clinical trials and biomarker validation studies. However, as blood-based biomarker tests move toward clinical adoption and reimbursement, the market is expected to diversify, with growing uptake by large health systems, clinical laboratories, and eventually community neurologists.
Biomarker Type Insights
Protein biomarkers segment dominates the market with a revenue share of 55.10% in 2024, driven by their strong biological relevance, diagnostic utility, and clinical validation. Proteins such as α-synuclein, neurofilament light chain (NfL), and DJ-1 are directly associated with Parkinson’s disease pathology, making them highly sought after for early detection and monitoring. Technological advancements in ELISA, mass spectrometry, and multiplex immunoassays have improved sensitivity and specificity for detecting these markers in blood. In addition, pharmaceutical companies increasingly use protein biomarkers in clinical trials for patient stratification and therapeutic monitoring, accelerating their integration into research and clinical workflows.
The transcriptomic/miRNA biomarkers segment is the fastest-growing in the Parkinson’s disease blood-based biomarker market, projected to expand at a CAGR of over 18% from 2025 to 2033. This rapid growth is driven by the ability of circulating miRNAs-such as miR-29c, miR-132, and miR-7-to reflect early neurodegenerative changes, even before clinical symptoms emerge. These small, stable RNA molecules can be easily detected in blood and offer high specificity for disease stage and subtype differentiation. Advancements in qPCR, NGS, and AI-powered bioinformatics are further accelerating their discovery, validation, and use in clinical research and diagnostic development.
Technology Insights
ELISA/Immunoassay platforms represent the largest technology segment in the Parkinson’s disease blood-based biomarker market, accounting for the highest revenue share of 44.87% in 2024. Their dominance is attributed to their widespread availability, cost-effectiveness, scalability, and proven performance in detecting key protein biomarkers such as α-synuclein, DJ-1, and NfL. These assays are well-established in both research and clinical laboratories, supported by a broad reagent ecosystem and regulatory familiarity. In addition, ELISA-based formats are often used as the gold standard in early-stage biomarker validation studies and clinical trials, making them a cornerstone technology across the biomarker development pipeline.
Multiplex platforms are the fastest-growing technology segment in the Parkinson’s disease (PD) blood-based biomarker market, with an estimated CAGR of over 8.89% from 2025 to 2033. These platforms enable simultaneous detection of multiple biomarkers-such as α-synuclein, NfL, and inflammatory proteins-in a single assay, improving diagnostic accuracy and reducing testing costs. Technologies such as Olink’s Proximity Extension Assay (PEA) and Luminex bead-based systems are gaining rapid adoption in PD research and clinical trials due to their high throughput, minimal sample requirements, and applicability in longitudinal monitoring. Their scalability and AI compatibility further accelerate their use in precision neurology.
End use Insights
Academic and research laboratories represent the largest End User segment in the U.S. blood-based biomarker for parkinson’s disease market as of 2024. These institutions account for a significant share due to their central role in biomarker discovery, validation, and translational research. Major universities and NIH-funded centers such as UPenn, UCSF, and Mayo Clinic are leading longitudinal studies investigating markers such as α-synuclein, NfL, and exosomal miRNAs. Supported by grants from the Michael J. Fox Foundation and NIH, these labs operate at the forefront of innovation, often generating the foundational data required for future clinical adoption and regulatory approvals.
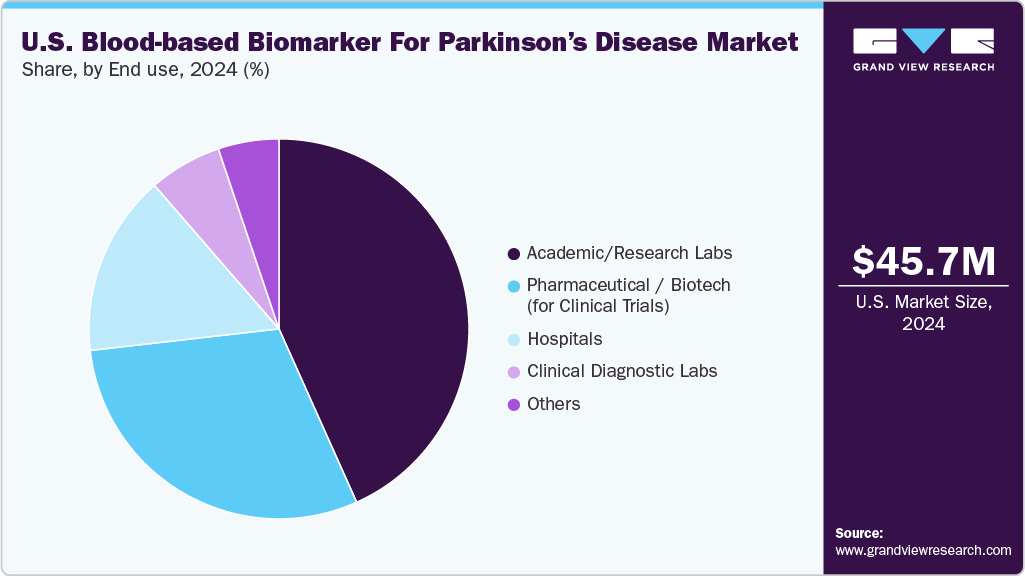
Pharmaceutical and biotech companies (for clinical trials) represent the fastest-growing End User segment in the U.S. Parkinson’s disease (PD) blood-based biomarker market. This growth is fueled by the increasing reliance on biomarkers for patient stratification, target engagement validation, and treatment response monitoring in neurodegenerative drug trials. Companies such as Denali Therapeutics, Biogen, and Roche are integrating markers such as phosphorylated Rab10, α-synuclein, and NfL into trials for LRRK2 and GBA-targeted therapies. Biomarkers are now essential tools in adaptive trial designs, accelerating drug development timelines while supporting companion diagnostic strategies, thereby driving rapid adoption in the pharmaceutical R&D space.
Country Insights
The U.S. market for blood-based biomarkers in Parkinson’s disease (PD) is evolving beyond traditional protein targets to encompass inflammatory and immune biomarkers, reflecting the growing recognition of PD as an inflammatory neurodegenerative disorder. Emerging evidence suggests that chronic neuroinflammation-mediated by activated microglia, cytokines, and CD4+ T cell subsets-plays a central role in PD pathogenesis even before clinical symptoms emerge.
Biomarkers such as IFN-γ, TNF-α (Th1-derived), IL-17 (Th17), and IL-10 (Th2) are being studied in the blood to assess their correlation with PD severity, progression, and non-motor symptoms such as cognitive decline or anxiety. However, inconsistencies in cytokine profiles across patient cohorts pose challenges in standardization. Companies such as Olink Proteomics and SomaLogic are actively developing multiplex inflammatory panels, using blood-based assays that include these cytokines to support both clinical research and trial enrollment.
Academic institutions such as Massachusetts General Hospital and Mayo Clinic are investigating inflammatory signatures in PD patients undergoing surgical procedures, noting that anesthesia and perioperative stress may temporarily amplify neuroinflammatory markers, potentially accelerating disease progression.
As inflammation-targeted therapies move into development, these blood-based inflammatory biomarkers are expected to become key tools in patient stratification, therapeutic monitoring, and possibly even drug response prediction, significantly expanding the market potential over the next decade.
Key U.S. Blood-based Biomarker for Parkinson’s Disease Company Insights
The U.S. Parkinson’s disease blood-based biomarker market is currently led by a mix of specialized diagnostics firms and large pharmaceutical companies, each contributing to distinct areas such as assay development, clinical validation, and trial integration. Amprion, C2N Diagnostics, and Olink Proteomics are front-runners in assay innovation, collectively accounting for a significant share of research-focused testing. Pharma players like Biogen, Denali Therapeutics, and Roche dominate biomarker use in clinical trials. While no single company holds overwhelming market control, early innovators in α-synuclein and NfL assays are capturing early adoption, giving them a strategic advantage as the market moves toward clinical use.
Key U.S. Blood-based Biomarker for Parkinson’s Disease Companies:
- Amprion
- C2N Diagnostics
- Olink Proteomics
- Denali Therapeutics
- Biogen
- Roche / Genentech
- Verily Life Sciences
- AC Immune
- SomaLogic
- QuanterixGE Healthcare
Recent Developments
-
In October 2024, Amprion secured $6 million in Series B funding to scale its SAAmplify seed amplification assay for detecting misfolded α-synuclein in blood-a hallmark of PD pathology. Previously validated in cerebrospinal fluid (CSF), this funding accelerates efforts to bring the first non-invasive α-synuclein blood test to the clinical market.
-
In April 2025, The Michael J. Fox Foundation (MJFF) expanded its landmark Parkinson’s Progression Markers Initiative (PPMI) to enroll individuals as young as age 40 and enable remote blood sample collection. This move supports broader biomarker discovery by increasing access to preclinical PD participants and integrating blood-based and digital biomarkers.
-
In March 2025, C2N Diagnostics, a leader in blood-based Alzheimer’s testing, was awarded a $1.5 million grant from MJFF to adapt its LC-MS/MS platform to Parkinson’s disease. The project aims to identify NfL, tau, and inflammatory proteins in archived PPMI biosamples to build multi-marker diagnostic and progression panels.
U.S. Blood-based Biomarker For Parkinson’s Disease Market Report Scope
Report Attribute
Details
Market size value in 2025
USD 56.30 million
Revenue forecast in 2033
USD 130.63 million
Growth rate
CAGR of 18.33% from 2025 to 2033
Actual data
2021 - 2024
Forecast period
2025 - 2033
Quantitative units
Revenue in USD billion/million and CAGR from 2025 to 2033
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Biomarker type, technology, and end use
Country scope
U.S.
Key companies profiled
Amprion; C2N Diagnostics; Olink Proteomics; Denali Therapeutics; Biogen; Roche / Genentech; Verily Life Sciences; AC Immune; SomaLogic; Quanterix
Customization scope
Free report customization (equivalent up to 8 analyst’s working days) with purchase. Addition or alteration to country, & segment scope
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
U.S. Blood-based Biomarker For Parkinson’s Disease Market Segmentation
This report forecasts revenue growth at country levels and provides an analysis of the latest industry trends in each of the sub-segments from 2021 to 2033. For this study, Grand View Research has segmented U.S. blood-based biomarker for Parkinson’s disease market report on the basis of biomarker type, technology, and end use:
-
Biomarker Type Outlook (Revenue, USD Million, 2021 - 2033)
-
Protein biomarkers
-
Inflammatory markers
-
Metabolic markers
-
Transcriptomic/miRNA
-
Others
-
-
Technology Outlook (Revenue, USD Million, 2021 - 2033)
-
ELISA/Immunoassay
-
Multiplex platforms
-
NGS/qPCR (genetic)
-
Mass spectrometry
-
Others
-
-
End Use Outlook (Revenue, USD Million, 2021 - 2033)
-
Academic/Research Labs
-
Pharmaceutical / Biotech (for Clinical Trials)
-
Hospitals
-
Clinical Diagnostic Labs
-
Others
-
Frequently Asked Questions About This Report
b. The U.S. blood-based biomarker for Parkinson’s disease market is expected to grow at a compound annual growth rate of 18.33% from 2025 to 2033 to reach USD 130.63 million by 2033.
b. Protein biomarkers segment dominates the market with a revenue share of 55.10% in 2024 by biomarker type, driven by their strong biological relevance, diagnostic utility, and clinical validation. Proteins like α-synuclein, neurofilament light chain (NfL), and DJ-1 are directly associated with Parkinson’s disease pathology, making them highly sought after for early detection and monitoring. Technological advancements in ELISA, mass spectrometry, and multiplex immunoassays have improved sensitivity and specificity for detecting these markers in blood. Additionally, pharmaceutical companies increasingly use protein biomarkers in clinical trials for patient stratification and therapeutic monitoring, accelerating their integration into research and clinical workflows.
b. Some key players operating in the U.S. blood-based biomarker for Parkinson’s disease market include Amprion, C2N Diagnostics, Olink Proteomics, Denali Therapeutics, Biogen, Roche / Genentech. Verily Life Sciences, AC Immune, SomaLogic, Quanterix
b. The U.S. Parkinson’s disease (PD) blood-based biomarkers market is driven by a growing patient population, with 90,000 new diagnoses annually, and increasing demand for early, non-invasive diagnostic tools. Expanding research efforts, especially in academic and pharmaceutical settings, are accelerating biomarker discovery and validation. Technological advancements in multiplex assays and AI-based platforms are further supporting adoption. Additionally, collaborations involving the NIH, MJFF, and Verily are helping standardize biomarker use. As assays like α-synuclein and NfL progress toward regulatory approval, their integration into clinical practice is becoming increasingly viable.
b. The U.S. blood-based biomarker for Parkinson’s disease market size was estimated at USD 45.75 million in 2024 and is expected to reach USD 56.30 million in 2025.
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










