- Home
- »
- Medical Devices
- »
-
U.S. Exoskeleton Market Size & Share, Industry Report 2033GVR Report cover
![U.S. Exoskeleton Market Size, Share & Trends Report]()
U.S. Exoskeleton Market (2026 - 2033) Size, Share & Trends Analysis Report By Mobility (Mobile, Fixed/Stationary), By Technology (Powered, Non-powered), By Extremity (Upper Body, Lower Body), By End-use (Healthcare, Military, Industry), By Region, And Segment Forecasts
- Report ID: GVR-4-68040-858-0
- Number of Report Pages: 150
- Format: PDF
- Historical Range: 2021 - 2025
- Forecast Period: 2026 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
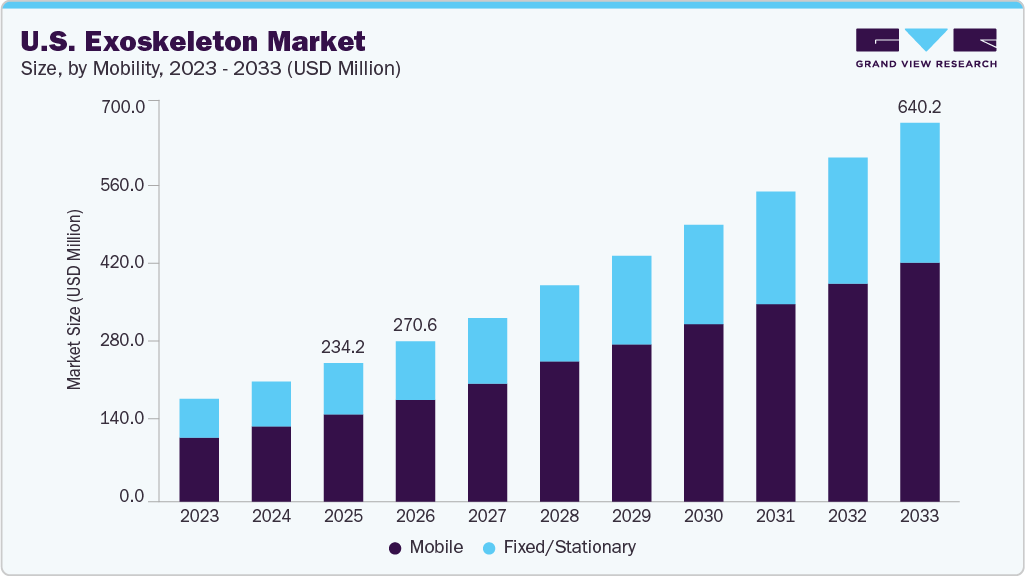
U.S. Exoskeleton Market Summary
The U.S. exoskeleton market size was estimated at USD 234.18 million in 2025 and is projected to reach USD 640.25 million by 2033, growing at a CAGR of 13.09% from 2026 to 2033. The rapidly growing geriatric population, rising adoption of medical devices across industries such as automotive, military, defense, and construction, and the rising incidence of stroke are key drivers of growth in the U.S. exoskeleton industry.
Key Market Trends & Insights
- Based on mobility, the mobile segment held the largest revenue share of 62.89% in 2025.
- Based on technology, the powered segment held the largest revenue share of 72.26% in 2025.
- Based on extremity, the lower body segment held the largest share of 41.11% in 2025.
Market Size & Forecast
- 2025 Market Size: USD 234.18 Million
- 2033 Projected Market Size: USD 640.25 Million
- CAGR (2026-2033): 13.09%
The growing incidence rate of traumatic spinal cord injuries (SCI) is expected to drive the demand across U.S. markets. For instance, in September 2025, the United Spinal Association reported that 250,000 to 390,000 people in the U.S. are living with spinal cord injury, with about 18,000 new cases each year, most often due to vehicle accidents and falls. It highlighted common secondary complications, including depression, chronic pain, spasticity, pressure injuries, and bowel, bladder, and respiratory issues.Exoskeleton solutions are increasingly being adopted to support mobility restoration and rehabilitation, particularly for individuals with spinal cord injuries. In the U.S., ongoing innovation in wearable robotics is encouraging manufacturers to introduce next-generation systems with improved personalization and digital integration. For instance, in 2025, Lifeward Ltd. announced the nationwide U.S. commercialization of its latest personal exoskeleton, ReWalk 7. The new system incorporates advanced features such as cloud-based connectivity, adaptive software, and adjustable walking speeds, enabling a more tailored gait experience for users with spinal cord injuries. This launch reflects the company’s strategic focus on enhancing user autonomy, clinical outcomes, and real-world usability of exoskeleton-assisted walking solutions in the U.S. market.
“The ReWalk 7 was developed over several years, integrating advanced technological innovations with feedback from clinicians and patients to build upon the ReWalk’s world-class reputation for industry leadership, The result is a device that is optimized for real-world use, with an unmatched user experience and freedom of movement. We are thrilled to be able to provide paralyzed individuals across the country with a new option for integrating walking to everyday life.” -Larry Jasinski, CEO of Lifeward
The increasing prevalence of traumatic spinal cord injury is boosting the growth of the U.S. exoskeleton market, as patients suffering from spinal cord injury are recommended to use an exoskeleton to improve their condition. This has become a key driver for the adoption of exoskeletons by patients and healthcare providers. For instance, as per the National Spinal Cord Injury Statistical Center (NSCISC) in 2024, in the U.S., traumatic spinal cord injury remains a significant public health concern, with recent estimates indicating that approximately 18,400 new cases are reported each year. This incident corresponds to around 54 newly diagnosed individuals per one million people annually, underscoring the persistent risk of severe spinal trauma across the population. The steady occurrence of these injuries reflects ongoing exposure to factors such as road traffic accidents, falls, sports-related injuries, and workplace incidents, contributing to a sustained patient pool requiring long-term rehabilitation, mobility assistance, and advanced supportive technologies.
Some of the most common musculoskeletal injuries and disorders affecting workers in physically demanding sectors such as construction include occupational overuse syndrome (OOS), cumulative trauma disorders (CTD), and repetitive strain injury (RSI). These conditions arise from repetitive motions, heavy lifting, awkward postures, and prolonged exertion on the job. According to the most recent data from the U.S. Bureau of Labor Statistics in August 2023, an estimated 502,380 workplace musculoskeletal disorder cases were reported over the 2021-2022 period, with an incidence rate of about 25.3 cases per 10,000 full-time equivalent workers, illustrating the widespread nature of these injuries across U.S. industries.
In addition, as per the WorkCare, Inc. article published in March 2025, broader estimates suggest that musculoskeletal disorders account for over 1 million workplace injuries in the U.S. annually, placing a substantial burden on employers and workers alike. These high rates of work-related musculoskeletal issues are key drivers for the growing adoption of exoskeleton solutions, as such technology can help augment or support physical tasks, improve worker health and productivity, and reduce fatigue among industrial laborers.
Rapid technological advancements in the U.S. exoskeleton market are driving innovation, accelerating adoption across health care, industrial, and consumer segments. For instance, in January 2026, a key development in the U.S. is the recent unveiling of new AI-enabled wearable robotic systems at major trade events such as CES 2026, where several companies showcased next-generation portable exoskeletons that incorporate real-time AI-driven assistance, including terrain adaptive controls and compact, user-centric designs that support mobility and reduce physical exertion for diverse users. These products signal a shift toward more intelligent, adaptive exoskeleton solutions designed to assist both daily mobility and demanding tasks, reflecting growing interest and commercial momentum in the U.S. wearable robotics space.
Case Studies & Insights
Case Study Title: “The Walking ICU: Wandercraft and Brigham and Women’s Test Robotics on Critical Patients”
Overview: According to the HIT Consultant Media article published in January 2026, this case study examines recent advances in U.S.-based medical exoskeleton development, focusing on mobility restoration for patients with spinal cord injuries, stroke, and neuromuscular disorders. The study highlights how innovations in self-balancing systems, artificial intelligence, and real-time motion control are expanding exoskeleton use beyond rehabilitation clinics into hospital and critical-care environments.
Key Findings:
-
Technological Integration: AI-driven motion control and sensor feedback enable exoskeletons to adapt dynamically to user posture and movement, improving balance and reducing reliance on external support.
-
Early Mobilization Benefits: U.S. clinical applications show that next-generation exoskeletons can support early standing and assisted walking in hospital settings, helping reduce complications associated with prolonged immobility and improving rehabilitation engagement.
A key instance is the personal exoskeleton developed by Wandercraft, which is undergoing clinical evaluation at leading U.S. hospitals and rehabilitation centers, including Brigham and Women’s Hospital. The self-balancing system is designed to enable hands-free standing and walking, reflecting a shift toward more autonomous and real-world mobility support.
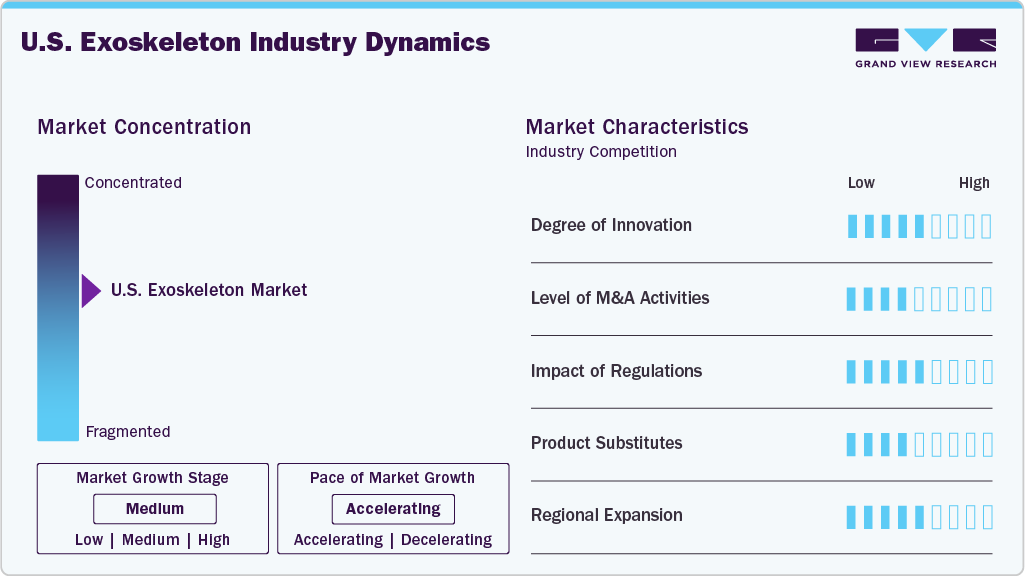
Market Concentration & Characteristics
The U.S. exoskeleton market is characterized by a high degree of innovation at the research and platform level, aimed at accelerating development and reducing system complexity. For instance, in June 2025, researchers at Northern Arizona University introduced OpenExo, an open-source robotic exoskeleton framework designed to support walking assistance research. The modular platform enables multiple exoskeleton configurations using a shared software and hardware architecture, significantly lowering development time and cost for researchers and developers. Such initiatives are expected to stimulate innovation, collaboration, and long-term commercialization opportunities within the U.S. exoskeleton ecosystem.
The U.S. exoskeleton industry has seen significant M&A activity as companies look to strengthen their technology and adapt to changing market conditions. In December 2025, Ekso Bionics, a leading developer of wearable exoskeletons including the Indego Personal system, announced a proposed merger with Applied Digital Corporation, a cloud and AI infrastructure firm. The deal could result in the separation or sale of Ekso’s exoskeleton business, highlighting how financial pressures and strategic restructuring are driving consolidation in the market, even for products with ongoing sales and Medicare coverage.
A well-developed regulatory framework and rising approvals for exoskeleton products from these regulatory bodies positively impact market growth. The certifications and standards issued by government authorities for exoskeletons in their manufacture, deployment, and use would enhance their adoption in the workplace. For instance, in January 2026, Baltimore‑based NextStep Robotics announced it is pursuing additional FDA authorization through a De Novo submission to expand the approved use of its AMBLE lower‑body exoskeleton beyond its current role as an exercise device for gait therapy; this bid follows NIH‑funded clinical trials that showed patients continued to make walking improvements months after completing treatment, and the company is also advancing a subscription‑based model to make the technology affordable for smaller community clinics.
Traditional rehabilitation methods remain prevalent, and exoskeletons are emerging as valuable alternatives. They offer advantages such as enhanced mobility and independence for patients with spinal cord injuries or neurological disorders. The high cost and specialized training requirements may limit their widespread adoption, positioning them as complementary rather than direct substitutes for conventional therapies.
Several market players are expanding their businesses by entering new geographic regions to strengthen their market positions and expand their product portfolios. Rising research and development activities create more opportunities for market players to enter new regions. In January 202 , NAVEE announced its geographical expansion from Europe to the U.S., unveiling its global intelligent mobility strategy at a Silicon Valley event. The company introduced five AI-enabled products, including Exo-Fit, a full-terrain wearable exoskeleton for human mobility, as well as solutions for land, air, and water travel.
Mobility Insights
The mobile segment led the U.S. exoskeleton industry in 2025, accounting for the largest revenue share of 62.89%. Factors such as rising innovation and increasing consumer interest in wearable mobility aids. Technologies that improve comfort, reduce exertion, and enhance everyday movement are driving broader adoption in both personal and recreational contexts. For instance, in Januar 2026, at CES 2026 in Las Vegas, personal exoskeletons such as the lightweight Wirobotics Wim S, a compact, belt-mounted robotic assist device designed to reduce leg fatigue and support longer walking endurance, were showcased with plans to expand into the U.S. market. The Wim S highlights growing consumer demand for mobile exoskeleton wearables that go beyond industrial and clinical use, fuels market growth.
The fixed/stationary segment is anticipated to register the fastest CAGR over the forecast period, driven by rising demand for structured rehabilitation and assisted mobility solutions. These systems are primarily used in controlled clinical environments to support standing balance, posture alignment, and repetitive gait training for patients with severe mobility limitations. Hospitals and rehabilitation centers increasingly prefer fixed-frame or tethered exoskeletons due to their suitability for supervised therapy, precise clinical monitoring, and extended rehabilitation sessions. For instance, in the U.S., Ekso Bionics continues to expand the clinical adoption of its EksoNR rehabilitation exoskeleton across U.S. hospitals and neurorehabilitation centers.
Technology Insights
The powered segment accounted for the largest revenue share of 72.26% in 2025, driven by the growing adoption of powered exoskeleton solutions across industries to enhance personal safety and boost productivity. In addition, the rise in innovative product launches and the benefits associated with powered technology products include smoother lifting motion, reduced strain on the worker’s body, increased strength, force multiplication, and enhanced productivity. For instance, in January 2026, in Las Vegas, where ULS Robotics showcased its VIATRIX powered exoskeleton, a range-extended wearable device designed to provide intelligent power assistance for outdoor mobility and general use, it offers advanced joint mechanisms, an AI gait learning system, and adaptable power support. This highlights how powered exoskeleton technology is transitioning to broader applications and gaining traction in U.S. markets beyond traditional industrial sectors.
Non-powered technology segment is anticipated to register the fastest CAGR over the forecast period, driven by increasing demand for cost-effective, lightweight wearable support solutions that assist with lifting and movement without relying on batteries or motors. These systems use mechanical elements such as springs, elastics, and structural linkages to store and return energy, helping reduce fatigue and musculoskeletal strain during repetitive tasks. Advantages that make them appealing in logistics, construction, healthcare, and other labor-intensive sectors. For instance, in January 2026, in the HeroWear Apex 2 passive back-assist exosuit, which has been deployed across multiple U.S. distribution centers and, in long-term real-world use, was associated with a significant reduction in strain and sprain injury rates among warehouse workers, indicating how non powered or soft passive devices are being adopted to enhance worker safety while minimizing complexity and cost compared with powered systems.
Extremity Insights
The lower body segment dominated the U.S. exoskeleton market, accounting for a revenue share of 41.11% in 2025, owing to rising investments, growing incidence of lower body disabilities, and increasing adoption of exoskeleton products by geriatric populations and paralyzed patients for weight-bearing capabilities and mobility. Moreover, technological advanced product launch drives the market growth. For instance, in November 2025, the expanded FDA clearance for Wandercraft’s Atalante X robotic exoskeleton, now approved for use in the rehabilitation of people with multiple sclerosis (MS) and a broader range of spinal cord injury levels, enables clinicians to use the device to help more patients stand and walk during therapy. This broadened approval reflects increasing clinical acceptance of advanced lower‑body wearable robotics in the U.S. healthcare system fuels the growth.
The upper body segment is anticipated to register the fastest CAGR over the forecast period due to the increasing need for devices that aid arm and shoulder mobility in rehabilitation for stroke survivors, neurological disorders, and other upper‑extremity impairments. For instance, in August 2025, the Utah NeuroRobotics Lab launched a large-scale clinical trial, securing nearly USD 940,000 in funding to evaluate a powered myoelectric upper-limb exoskeleton for stroke patients with hemiparesis. This study aims to assess improvements in upper-limb function, quality of life, and rehabilitative outcomes across a diverse patient group, highlighting active research and the growing clinical adoption of upper-body wearable robotics in rehabilitation programs.
End-use Insights
The healthcare segment dominated the U.S. exoskeleton industry, accounting for a revenue share of 48.95% in 2025, driven by the increasing prevalence of spinal cord injuries, the expanding use of exoskeleton systems across rehabilitation facilities, and the growing clinical use of wearable mobility systems in rehabilitation settings and broader access enabled by regulatory and reimbursement advances. For instance, in August 2024, Ekso Bionics’ Indego Personal exoskeleton securing dedicated Medicare reimbursement under HCPCS code K1007, with Medicare processing and paying initial claims at approximately USD 91,032, making the device more financially accessible for individuals with spinal cord injuries and enabling wider adoption of personal exoskeleton use at home and in the community a meaningful step toward integrating robotic mobility solutions into U.S. healthcare treatment plans and long‑term care pathways.
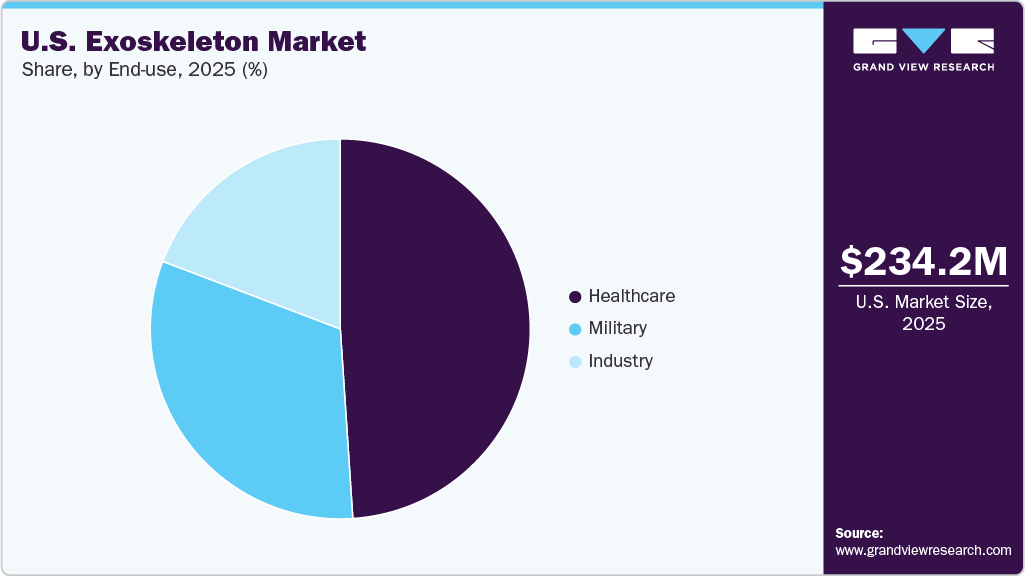
The industry segment is anticipated to register the fastest growth over the forecast period, owing to the increasing recognition of the advantages offered by exoskeleton technologies in physically demanding work environments and the rising prevalence of job-related musculoskeletal injuries. Employers across construction, manufacturing, logistics, and warehousing are progressively adopting wearable support systems to improve worker safety, reduce fatigue, and enhance operational efficiency. The growing demand for ergonomic innovations and injury-prevention solutions is further accelerating the penetration of exoskeletons across multiple industrial sectors. For instance, in August 2024, Festool introduced its ExoActive powered exoskeleton in the U.S. for construction professionals, a wearable support system designed to significantly ease shoulder and arm нагруз during repetitive overhead and forward-reach tasks. The device is engineered to lower physical strain by around 30% while delivering up to 11 pounds of active arm assistance, helping improve worker endurance, productivity, and injury prevention across construction and skilled trade environments.
Key U.S. Exoskeleton Company Insights
Key players who focus on innovation, product development, and strategic partnerships to maintain their market positions dominate the U.S. exoskeleton market. The market share is concentrated among established players with extensive research and development resources, while emerging players focus on cost-effective solutions and technological advancements.
Key U.S. Exoskeleton Companies:
- Ekso Bionics
- Lockheed Martin Corporation
- Suit X
- Rex Bionics Plc.
- ReWalk Robotics
- DIH Medical
- German Bionic
- Sarcos Technology and Robotics Corporation
- Parker Hannifin Corporation
- Bionik Laboratories
Recent Developments
-
In January 2026, Ascentiz unveiled a modular exoskeleton at CES 2026, featuring swappable hip and knee modules optimized with distinct drive technologies for natural movement assistance. The system integrates AI trained on over 690,000 gait cycles to deliver rapid mode switching and significant reductions in leg effort, knee cartilage pressure, and energy loss during dynamic and load-bearing activities.
-
In February 2025, Wandercraft launched a landmark clinical trial for the Personal Exoskeleton, the world’s first self-balancing device aimed at restoring natural standing, walking, bending, and reaching abilities for individuals with severe mobility impairments.
“The launch of this trial is a major milestone in our journey to redefine what mobility technology can achieve. We’ve seen the profound impact of our exoskeletons in rehabilitation centers, and now we are thrilled to explore how the Personal Exoskeleton could represent a significant leap forward in mobility and walking independence for people with severe walking impairment, starting with SCI, enabling them to walk hands-free without relying on crutches or walkers."
- Matthieu Masselin, CEO of Wandercraft
- In November 2024, Hippos Exoskeleton will release a knee brace featuring airbags designed to help prevent ACL injuries.
“The core technology we invented is not only on the hardware side, but also on the software side: the AI, the data processing capability, and the ability to use our AI model, which we designed by ourselves, to personalize the triggering threshold for every single individual, athletes, soldiers, and patients.”
- Kylin Shaw, Hippos CEO
U.S. Exoskeleton Market Report Scope
Report Attribute
Details
Market size value in 2026
USD 270.65 million
Revenue forecast in 2033
USD 640.25 million
Growth rate
CAGR of 13.09% from 2026 to 2033
Actual data
2021 - 2025
Forecast period
2026 - 2033
Quantitative units
Revenue in USD million, volume, 000' units and CAGR from 2026 to 2033
Report coverage
Revenue & volume forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Mobility, technology, extremity, end-use, region
Country scope
U.S.
Key companies profiled
Ekso Bionics; Lockheed Martin Corporation; Suit X; Rex Bionics Plc.; ReWalk Robotics; DIH Medical; German Bionic; Sarcos Technology and Robotics Corporation; Parker Hannifin Corporation; Bionik Laboratories.
Customization scope
Free report customization (equivalent up to 8 analysts working days) with purchase. Addition or alteration to country, regional & segment scope.
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
U.S. Exoskeleton Market Report Segmentation
This report forecasts revenue and volume growth at the country level and provides an analysis of industry trends in each sub-segment from 2021 to 2033. For this study, Grand View Research, Inc. has segmented the U.S. exoskeleton market report based on mobility, technology, extremity, end-use, and region:
-
Mobility Outlook (Volume, 000' Units; Revenue, USD Million, 2021 - 2033)
-
Mobile
-
Fixed/Stationary
-
-
Technology Outlook (Volume, 000' Units; Revenue, USD Million, 2021 - 2033)
-
Powered
-
Non-powered
-
-
Extremity Outlook (Volume, 000' Units; Revenue, USD Million, 2021 - 2033)
-
Upper Body
-
Lower Body
-
Full Body
-
-
End-use Outlook (Volume, 000' Units; Revenue, USD Million, 2021 - 2033)
-
Healthcare
-
Military
-
Industry
-
Frequently Asked Questions About This Report
b. The U.S. exoskeleton market size was estimated at USD 234.18 million in 2025 and is expected to reach USD 270.65 million in 2026.
b. The U.S. exoskeleton market is projected to grow at a compound annual growth rate (CAGR) of 13.09% from 2026 to 2033 to reach USD 640.25 million by 2033.
b. The mobile segment led the market in 2025 with the largest revenue share of 62.89% in 2025 due to a rising innovation and increasing consumer interest in wearable mobility aids.
b. Some of the key players operating in the market include Ekso Bionics: Lockheed Martin Corporation; Suit X; Rex Bionics Plc.; ReWalk Robotics; DIH Medical; German Bionic; Sarcos Technology and Robotics Corporation; Parker Hannifin Corporation; and Bionik Laboratories.
b. The key drivers of U.S. exoskeleton market are the rapidly growing geriatric population, rising adoption of medical devices across industries such as automotive, military, defense, and construction, and the rising incidence of stroke.
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










