- Home
- »
- Clinical Diagnostics
- »
-
U.S. Filter Integrity Test Market Size, Industry Report, 2033GVR Report cover
![U.S. Filter Integrity Test Market Size, Share & Trends Report]()
U.S. Filter Integrity Test Market (2025 - 2033 ) Size, Share & Trends Analysis Report By Test Method (Forward Flow (Diffusion) Test, Bubble Point Test, Pressure Hold Test, Water Intrusion Test), By Mode, By Type, By End-use, And Segment Forecasts
- Report ID: GVR-4-68040-737-7
- Number of Report Pages: 150
- Format: PDF
- Historical Range: 2021 - 2024
- Forecast Period: 2025 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
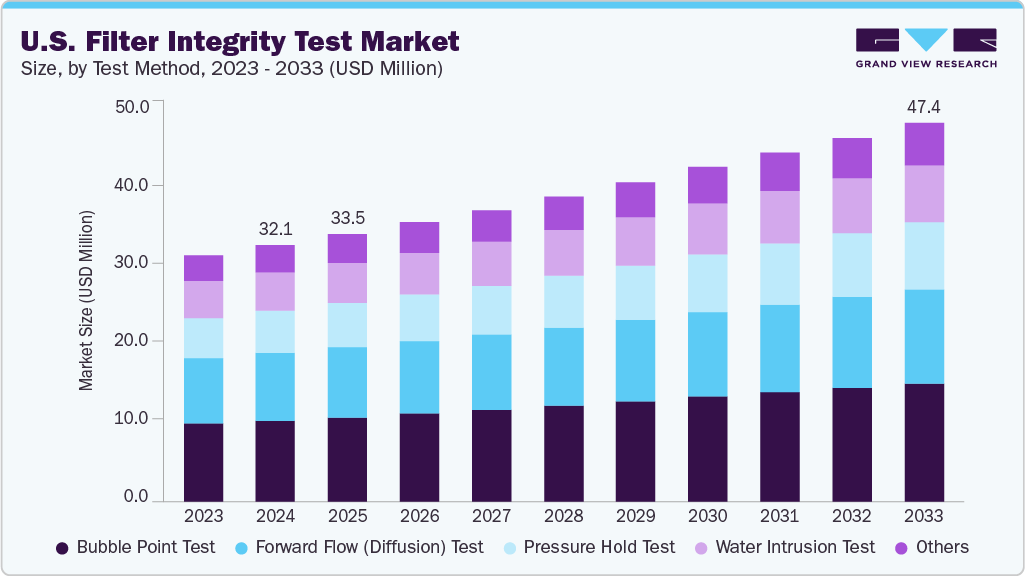
U.S. Filter Integrity Test Market Size & Trends
The U.S. filter integrity test market size was estimated at USD 32.12 million in 2024 and is projected to reach USD 47.36 million by 2033, growing at a CAGR of 4.44% from 2025 to 2023. The market is primarily driven by the rapid growth of the biopharmaceutical industry. Increasing demand for biologics, monoclonal antibodies, and advanced therapies has heightened reliance on sterile filtration processes, where integrity testing is essential for maintaining product quality and regulatory compliance. With strict FDA oversight and sterility assurance requirements across every stage of biologics production, companies are making significant investments in advanced manufacturing facilities and filtration systems, creating strong demand for integrity testing solutions nationwide.
A recent example underscores this trend. In April 2025, UCB, a biopharmaceutical company committed to treating severe diseases, announced plans to establish a new, state-of-the-art biologics manufacturing facility in the U.S. This expansion highlights the growing biomanufacturing footprint in the country and underscores the importance of U.S. filter integrity testing systems to support compliance and ensure product safety. As more organizations invest in biologics capacity and cutting-edge production capabilities, demand for FIT solutions is expected to rise further.
The market is also being propelled by technological innovation and evolving regulatory expectations. As the U.S. strengthens its position as a leader in biologics, vaccines, and cell and gene therapies, integrity testing-particularly Pre-Use Post-Sterilization Integrity Testing (PUPSIT)-is gaining traction as a critical quality measure. These tests help manufacturers meet global standards while minimizing contamination risks and avoiding costly recalls. The integration of automated testing solutions into Good Manufacturing Practice (GMP) environments further ensures efficiency, compliance, and reliability.
For instance, in May 2025, Parker Bioscience Filtration, a division of Parker Hannifin, launched the SciLog NFF+ PF, a compact, multipurpose normal flow filtration system equipped with onboard PUPSIT functionality. By addressing the complexities of PUPSIT-such as reducing manual handling, operator errors, and product waste-this system supports safer and more streamlined biopharmaceutical production. Such innovations are setting new industry benchmarks, positioning the U.S. FIT market for sustained growth in the years ahead.
Market Concentration & Characteristics
The U.S. market demonstrates a high degree of innovation driven by the country’s advanced biopharmaceutical and biotechnology industries. Key players such as Pall Corporation (part of Danaher), Thermo Fisher Scientific, and 3M continue to invest in advanced testing solutions, including automated bubble point, diffusive flow, pressure hold, and water intrusion tests. The U.S. is also witnessing the integration of automation, AI-based analytics, and IoT-enabled systems for real-time monitoring, which is increasingly adopted by pharmaceutical manufacturers to comply with FDA-mandated sterility assurance standards.
M&A activity is strong in the U.S., with companies using acquisitions to expand capabilities and product portfolios. For instance, in January 2025, Thermo Fisher Scientific announced its acquisition of Solventum’s purification and filtration business, further strengthening its leadership in integrity testing and bioprocessing solutions. This followed its consistent strategy of portfolio expansion in advanced laboratory testing and filtration systems.
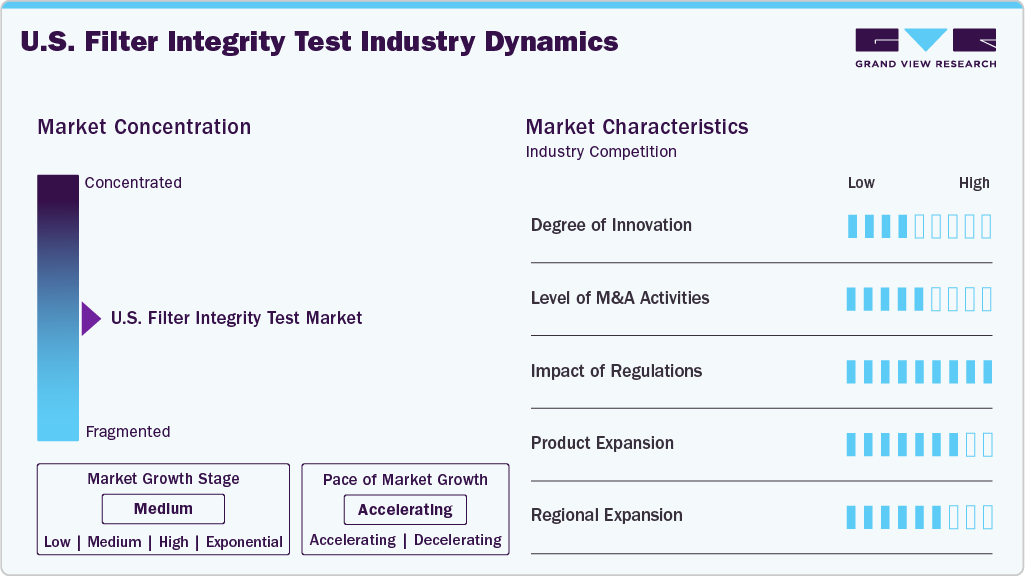
The U.S. Food and Drug Administration (FDA) plays a critical role in shaping the filter integrity tests market in U.S. Regulatory requirements such as current Good Manufacturing Practices (cGMP) and sterility testing guidelines mandate the use of validated U.S. filter integrity tests before and after sterile filtration in pharmaceutical and biotech manufacturing. This strict regulatory environment drives widespread adoption of integrity testing systems in the U.S., ensuring compliance and safeguarding patient safety.
Due to the specialized role of U.S. filter integrity testing in confirming sterility assurance, substitutes are very limited in the U.S. market. While visual inspection and microbial challenge testing exist, these are either insufficient or more resource-intensive compared to automated integrity tests. Hence, demand for specialized integrity testing systems remains inelastic and essential.
Within the U.S., the market benefits from the strong presence of multinational corporations alongside emerging domestic players. Expansion efforts are focused on collaborations with biopharma hubs such as Boston, San Diego, and North Carolina’s Research Triangle Park, as well as partnerships with contract development and manufacturing organizations (CDMOs). Companies like Pall Corporation and Sartorius have been scaling up operations and customer support centers in these regions to strengthen their U.S. footprint.
Test Method Insights
The bubble point test segment dominated the market in terms of revenue share in 2024 and is projected to grow at a CAGR of 3.74% over the forecast period. Bubble point remains the most widely applied integrity test because it is explicitly referenced in FDA aseptic processing guidance and embedded in pharmacy/biologics SOPs, making it the default benchmark for validating sterilizing-grade membrane performance. The FDA’s Sterile Drug Products Produced by Aseptic Processing - cGMP states that integrity testing must be performed post-use and recognizes bubble point and forward flow as acceptable methods, effectively cementing bubble point in batch release protocols across sterile fill-finish operations (injectables, vaccines, radiopharma). On the product side, leading platforms used by U.S. sites-Sartorius Sartocheck 5 Plus, Palltronic Flowstar family, and Merck MilliporeSigma Integritest 5-all implement automated bubble point with advanced anomaly detection and data-integrity controls (ALCOA+), which simplifies adoption and harmonization across multi-site quality systems. In beverages and life-sciences adjacency (e.g., breweries using sterile membrane cartridges), U.S. documents from suppliers continue to present bubble point as a primary method for confirming pore size conformance pre-service and for trending membrane health over time.
The pressure hold test segment in the market is anticipated to grow at the fastest CAGR over the forecast period, as manufacturers seek more objective, operator-independent readouts that minimize subjective visual assessments inherent to manual bubble detection. U.S. literature has long framed pressure hold as a robust alternative, particularly where diffusive flow can mask first-bubble visualization; peer-reviewed U.S. studies in radiopharma flagged pressure-hold’s advantages in reducing false outcomes compared with visual bubble point. Modern U.S.-market instruments-Sartocheck 5 Plus, Palltronic Flowstar IV/V, and Integritest 5-embed high-resolution pressure transducers and algorithmic leak diagnostics, making pressure-hold set-ups turnkey and compliant with 21 CFR Part 11 data controls. FDA aseptic processing thinking (and PDA technical practice) already recognizes post-use integrity testing by appropriate nondestructive methods, and operations teams increasingly choose pressure hold to standardize across hydrophilic filters where diffusion measurements can vary with temperature or wetting quality.
Mode Insights
The automated segment dominated the market in terms of revenue share of 88.79% in 2024. The segment addresses FDA expectations for data integrity, repeatability, and deviation control in cGMP. U.S. manufacturing and QA leaders prefer closed, automated testers that: enforce validated recipes; automatically correct for environmental variables (temperature/volume); capture tamper-evident audit trails (21 CFR Part 11); and (4) integrate with MES/LIMS for batch release. The broader U.S. tool landscape is also consolidating around automation: Thermo Fisher Scientific’s 2025 deal to acquire Solventum’s Purification & Filtration business strengthens an already deep life-sciences portfolio that surrounds integrity testing with upstream/downstream skids and analytics-further normalizing automated, networked QA workflows.
The manual segment is anticipated to grow at a considerable CAGR from 2025 to 2033. It is growing from a low base, as smaller aseptic compounders, academic core labs, craft beverage producers, and pilot-scale bioprocess units expand post-pandemic production and QA formality. The draw is cost and simplicity: handheld pressure sources and calibrated gauges let teams run bubble-point or pressure-hold checks without purchasing full-featured consoles. U.S. SOP content and method guides remain abundant and often teach first principles-e.g., performing bubble point with controlled pressure steps, or using pressure-hold to troubleshoot leaks-making manual testing a practical on-ramp while volumes are limited.
Type Insights
The liquid filter integrity test segment held the largest market share of 74.22% in 2024. High penetration of the forward flow (diffusion) test and bubble point test has led to the segment's predominant position in the market. These tests are crucial for evaluating the integrity and efficiency of filters used in liquid filtration systems across industries. Their rising adoption highlights the importance of ensuring effective removal of contaminants and upholding product quality and safety standards, cementing the dominance of liquid filter integrity testing in the market. In December 2021, Pall Corporation unveiled the Palltronic Flowstar V filter integrity test instrument, marking a significant advancement in automated filter testing for biopharmaceutical applications. Further, Integritest 5 by Merck is a lightweight, fully automated, and portable filter integrity tester designed for verifying filters and processing equipment.
The air filter integrity segment is expected to grow at the fastest CAGR over the forecast period. Air FIT primarily targets hydrophobic PTFE membranes used for sterile tank venting, lyophilizer vents, and process gas sterilization. Because hydrophobic media resist wetting, Water Intrusion Tests dominate; some applications use diffusive airflow or pressure-decay variants for small areas. Growth drivers include stricter environmental monitoring, increased use of single-use bioreactors requiring sterile gas feeds, and the updated Annex 1 emphasis on demonstrable sterility assurance for critical utilities.
End-use Insights
Biopharmaceutical & pharmaceutical industry dominated the market with the largest revenue share of 60.48% in 2024. Filter integrity testing is a cGMP-anchored control that bookends virtually every sterile filtration step in drug manufacturing. FDA guidance is explicit: post-use integrity testing is expected; methods (bubble point/forward flow/pressure hold) must align with bacterial retention validations; and results are part of batch records used to justify release. That compliance backbone-combined with intense quality-risk management across U.S. commercial and clinical facilities-creates high test frequency and broad installed bases of automated testers. Suppliers continue to invest around this demand: Thermo Fisher Scientific’s 2025 acquisition of Solventum’s purification & filtration business expands a U.S. giant’s footprint in bioprocess filtration, complementing platforms already in use across U.S. CDMOs and big pharma.
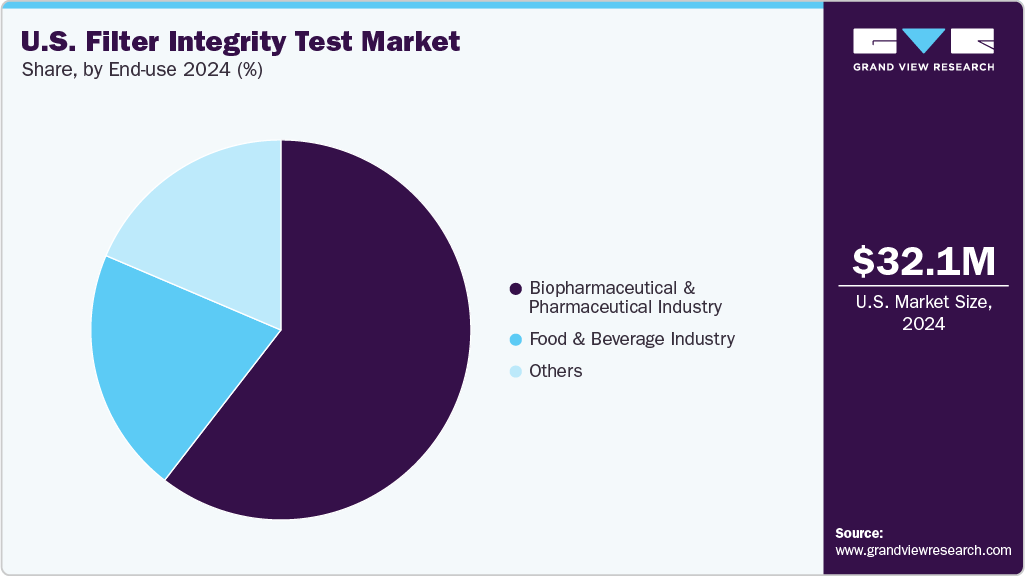
Food & beverage industry is projected to experience significant growth over the forecast period. Filters play a key role in eliminating impurities and contaminants from beverages and food products, ensuring consumer safety and satisfaction. Maintaining filter integrity is imperative to prevent contamination and preserve product freshness and taste. With increasing consumer awareness and regulatory scrutiny surrounding food safety, the food and beverage industry places a significant emphasis on stringent quality control measures.
Key U.S. Filter Integrity Test Company Insights
Key participants in the U.S. filter integrity test industry are concentrating on developing innovative testing solutions and obtaining essential certifications to expand their product portfolios. In addition, companies are engaging in partnerships, collaborations, mergers, and acquisitions to reinforce their market presence. These efforts aim to enhance technological capabilities, broaden regional reach, and improve access to rapid, accurate, and non-invasive drug testing methods across various workplace settings.
Key U.S. Filter Integrity Test Companies:
- Merck KGaA
- Sartorius AG
- Parker Hannifin Corp
- PALL Corporation (Danaher)
- Donaldson Company, Inc.
- Pentair Ltd.
- 3M
- Meissner Filtration Products, Inc.
- Beijing Neuronbc Laboratories Co., Ltd.
- Brother Filtration
- Analytical Technologies Limited
- Thermo Fisher Scientific, Inc.
Recent Developments
-
In May 2025, Parker Bioscience Filtration, a division of Parker Hannifin, launched the SciLog NFF+ PF, a compact, multipurpose normal flow filtration system equipped with onboard PUPSIT functionality. By addressing the complexities of PUPSIT-such as reducing manual handling, operator errors, and product waste-this system supports safer and more streamlined biopharmaceutical production
-
In April 2025, UCB, a biopharmaceutical company committed to treating severe diseases, announced plans to establish a new, state-of-the-art biologics manufacturing facility in the U.S. This expansion highlights the growing biomanufacturing footprint in the country and underscores the importance of U.S. filter integrity testing systems to support compliance and ensure product safety.
-
In July 2024, Parker introduced ASEPT-X2, a new sterilizing-grade gas filter with a hydrophobic/hydrophilic design capable of handling condensate and steam-in-place cycles (up to 150 SIP cycles) without blinding - ideal for aseptic food & beverage processing.
-
In November 2023, STEMart, a U.S.-based company specializing in medical device development, unveiled its latest offering: Filter Testing Services. Geared towards pharmaceutical and medical device manufacturers, these services assist in verifying filter performance during the manufacturing process, ensuring product safety and efficacy.
U.S. Filter Integrity Test Market Report Scope
Report Attribute
Details
Market size value in 2025
USD 33.47 million
Revenue forecast in 2033
USD 47.36 million
Growth rate
CAGR of 4.44% from 2025 to 2033
Actual data
2021 - 2024
Forecast period
2025 - 2033
Quantitative units
Revenue in USD million, and CAGR from 2025 to 2033
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Test method, mode, type, end-use
Key companies profiled
Merck KGaA; Sartorius AG; Parker Hannifin Corp; PALL Corporation; Donaldson Company, Inc.; Pentair Ltd.; 3M; Meissner Filtration Products, Inc.; Beijing Neuronbc Laboratories Co., Ltd.; Brother Filtration; Analytical Technologies Limited; Thermo Fisher Scientific, Inc.
Customization scope
Free report customization (equivalent up to 8 analyst’s working days) with purchase. Addition or alteration to country, regional & segment scope.
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
U.S. Filter Integrity Test Market Report Segmentation
This report forecasts revenue growth and provides an analysis on the latest trends in each of the sub-segments from 2021 to 2033. For this report, Grand View Research has segmented the U.S. filter integrity test market report based on test method, mode, type, and end-use:
-
Test Method Outlook (Revenue, USD Million, 2021 - 2033)
-
Forward Flow (Diffusion) Test
-
Bubble Point Test
-
Pressure Hold Test
-
Water Intrusion Test
-
Others
-
-
Mode Outlook (Revenue, USD Million, 2021 - 2033)
-
Automated
-
Manual
-
-
Type Outlook (Revenue, USD Million, 2021 - 2033)
-
Liquid Filter Integrity Test
-
Air Filter Integrity Test
-
-
End-use Outlook (Revenue, USD Million, 2021 - 2033)
-
Biopharmaceutical & Pharmaceutical Industry
-
Food & Beverage Industry
-
Others
-
Frequently Asked Questions About This Report
b. The U.S. filter integrity test market size was estimated at USD 32.12 million in 2024 and is expected to reach USD 33.47 billion in 2025.
b. The U.S. filter integrity test market is expected to grow at a compound annual growth rate of 4.44% from 2025 to 2033 to reach USD 47.36 million by 2033.
b. The bubble point test segment led the U.S. filter integrity test market in 2024 with a share of 31.55%. As it is explicitly referenced in FDA aseptic processing guidance and embedded in pharmacy/biologics SOPs, making it the default benchmark for validating sterilizing-grade membrane performance.
b. Some key players operating in the market include Merck KGaA, Sartorius AG, Parker Hannifin Corp, PALL Corporation, Donaldson Company, Inc., Pentair Ltd., 3M, Meissner Filtration Products, Inc., Beijing Neuronbc Laboratories Co., Ltd., Brother filtartion, Analytical Technologies Limited, Thermo Fisher Scientific, Inc.
b. Key factors that are driving the U.S. filter integrity test market growth include the rapid growth of the biopharmaceutical industry. Increasing demand for biologics, monoclonal antibodies, and advanced therapies has heightened reliance on sterile filtration processes, where integrity testing is essential for maintaining product quality and regulatory compliance
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










