- Home
- »
- Medical Devices
- »
-
U.S. Intravenous Solutions Market Size, Industry Report 2033GVR Report cover
![U.S. Intravenous Solutions Market Size, Share & Trends Report]()
U.S. Intravenous Solutions Market (2026 - 2033) Size, Share & Trends Analysis Report By Type (Total Parenteral Nutrition (TPN), Peripheral Parenteral Nutrition (PPN)), By Nutrients (Carbohydrates, Vitamins & Minerals, Single-dose Amino Acids), And Segment Forecasts
- Report ID: GVR-4-68040-286-4
- Number of Report Pages: 80
- Format: PDF
- Historical Range: 2021 - 2025
- Forecast Period: 2026 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
U.S. Intravenous Solutions Market Summary
The U.S. intravenous solutions market size was estimated at USD 4.9 billion in 2025 and is projected to reach USD 9.8 billion by 2033, growing at a CAGR of 9.4% from 2026 to 2033. The market growth is driven by an aging population needing medical interventions, a surge in chronic diseases requiring advanced treatments, and enhancements in healthcare technology that elevate patient care.
Key Market Trends & Insights
- Based on type, the total parenteral nutrition (TPN) segment dominated the market with a revenue share of 67.3% in 2025.
- Based on nutrients, the single-dose amino acid solution segment dominated the market with the largest share in 2025.
- Based on end use, the hospitals segment held the largest market share of 50.8% in 2025.
Market Size & Forecast
- 2025 Market Size: USD 4.9 Billion
- 2033 Projected Market Size: USD 9.8 Billion
- CAGR (2026-2033): 9.4%
This sector's growth reflects the critical need for innovative healthcare solutions to address the complex demands of modern medicine. The increasing incidence of cancer is a key driver for the U.S. intravenous solutions industry, as cancer patients frequently require IV therapy for chemotherapy administration, hydration, electrolyte balance, and supportive care. For instance, the American Cancer Society estimated that in 2026, around 60,480 new cases of oral cavity or oropharyngeal cancer and about 13,150 related deaths will be recorded, highlighting the growing patient pool requiring continuous IV-based treatment and nutritional support.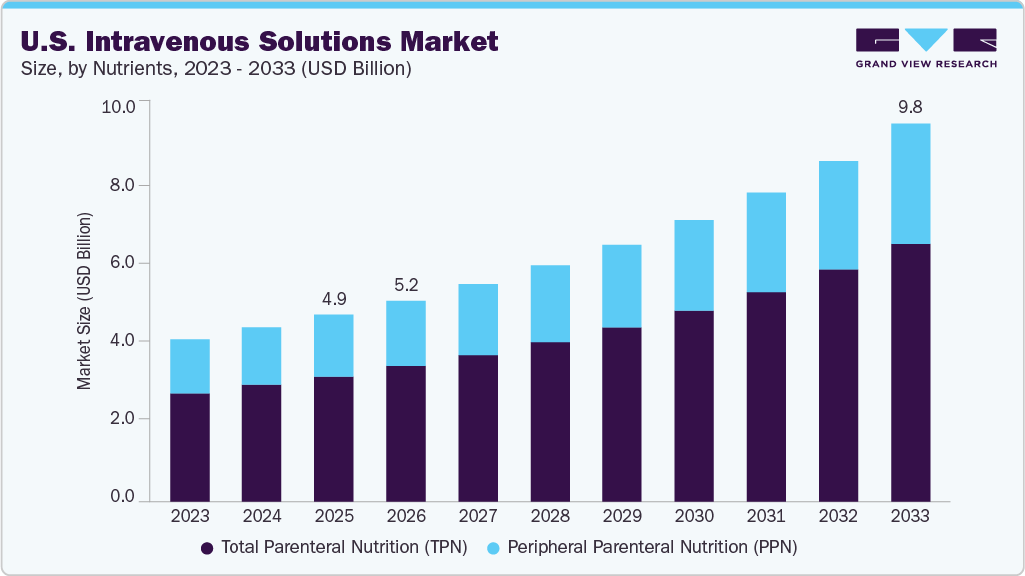
The rising use of immunonutrition formulas is driving growth in the U.S. IV solutions market, particularly in post-surgical recovery and critical care settings. These specialized parenteral formulations are designed to enhance immune response, reduce infection risk, and improve clinical outcomes in trauma, surgical, and critically ill patients. Products such as Elemune by Otsuka Holdings Co., Ltd., which contains L-alanyl-L-glutamine, are widely used for metabolic and immune support in infection and trauma cases. Similarly, SMOFlipid and Omegaven by Fresenius Kabi AG provide lipid-based nutrition enriched with omega-3 fatty acids, supporting energy requirements and reducing inflammation in ICU patients with limited enteral feeding. The growing clinical emphasis on improved recovery outcomes and reduced hospital stays is driving the adoption of advanced IV nutritional solutions across U.S. healthcare settings.
Market Concentration & Characteristics
The chart below illustrates the relationship between market concentration, industry characteristics, and industry participants. The x-axis represents the level of industry concentration, ranging from low to high. The y-axis represents various industry characteristics, including industry competition, regulatory impact, level of partnerships & collaborations, degree of innovation, and regional expansion. The U.S. intravenous solutions market is moderately concentrated, with a few established players holding a significant share, supported by strong manufacturing capabilities and regulatory compliance requirements. In contrast, smaller players operate in niche segments. The degree of innovation and the level of partnerships and collaboration activities are moderate, while the impact of regulations on the industry remains high. Regional expansion observes moderate growth.
The U.S. IV solutions industry is witnessing steady innovation focused on improving fluid composition, stability, and compatibility with advanced therapies. Manufacturers are developing customized electrolyte and nutrient formulations, ready-to-administer sterile solutions, and additive-free products to enhance safety and clinical efficiency.
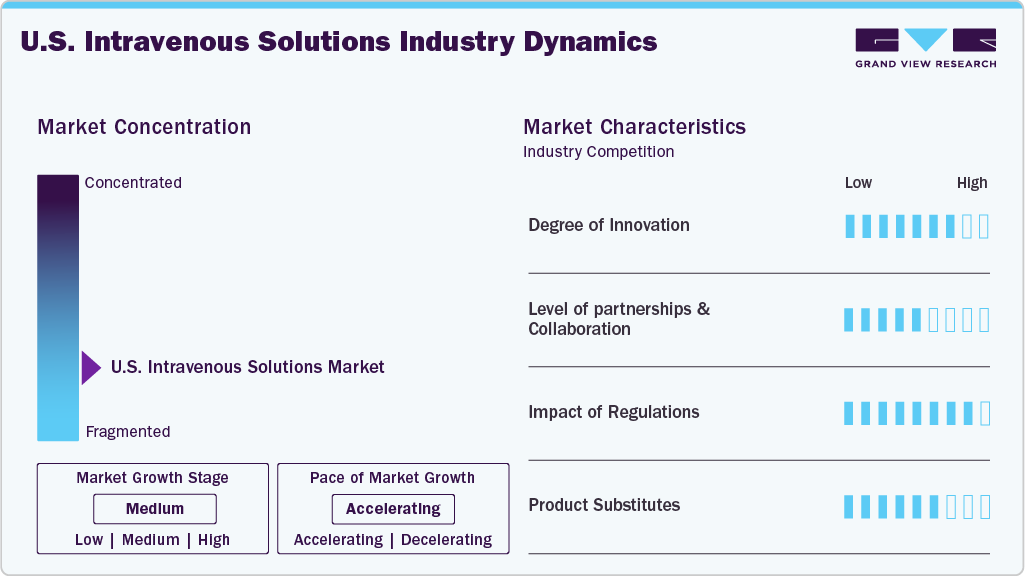
The IV solutions market sees moderate levels of partnerships and collaborations, primarily aimed at improving product formulation, quality, and clinical performance. Manufacturers are partnering with hospitals, research institutions, and pharmaceutical companies to develop advanced electrolyte blends, parenteral nutrition solutions, and therapy-specific formulations that improve patient outcomes and meet evolving clinical requirements.
For instance, in May 2025, Otsuka and ICU Medical announced a joint venture to strengthen IV solution supply in North America by combining global manufacturing scale with regional production and distribution capabilities. The partnership aims to address supply shortages, enhance supply chain resilience, and create one of the largest global networks for IV solutions manufacturing.
Regulations have a significant impact on the U.S. intravenous solutions market, as manufacturers must comply with stringent quality, safety, and labeling standards set by the U.S. Food and Drug Administration under the Federal Food, Drug, and Cosmetic Act. These regulations govern manufacturing practices, product approvals, and quality systems, ensuring patient safety while creating high entry barriers and increasing compliance costs for market participants.
The U.S. intravenous solutions industry is experiencing moderate expansion, primarily driven by efforts to strengthen domestic manufacturing capacity and address periodic supply shortages. Companies are focusing on expanding and upgrading production facilities, improving sterility and quality standards, and enhancing distribution networks. For instance, in October 2024, B. Braun Medical announced plans to increase its IV saline fluids production by 20% at its facilities in Irvine, California, and Daytona Beach, Florida.
Type Insights
Based on the product, the TPN segment accounted for the largest market share of over 67.3% in 2025 and is expected to grow at the fastest CAGR of 9.6% over the forecast period. The segment’s growth is driven by the rising prevalence of chronic diseases, increasing number of surgical procedures, and growing demand for clinical nutrition in ICU and neonatal care settings, where patients require specialized nutritional support. In addition, the shift toward advanced lipid emulsions and immune-supportive formulations is further supporting adoption. For instance, in May 2024, Baxter International Inc. received U.S. FDA approval for an expanded indication for Clinolipid (Lipid Injectable Emulsion),extending its use to pediatric patients, including neonates, thereby reinforcing the growing demand for specialized parenteral nutrition solutions and supporting segment growth.
The peripheral parenteral nutrition (PPN) segment is expected to grow significantly from 2026 to 2033. The segment's growth is attributed to its suitability for short-term nutritional support, the use of lower-osmolarity formulations that can be safely administered via peripheral veins, and a reduced risk of catheter-related complications compared to central parenteral nutrition. Moreover, PPN is widely adopted in post-operative recovery and mild-to-moderate malnutrition cases, where temporary nutritional supplementation is required, further supporting segment growth.
Nutrients Insights
The single-dose amino acid solution segment dominated the U.S. intravenous solutions market, accounting for the largest share in 2025, driven by its critical role in parenteral nutrition for patients unable to meet protein requirements orally or enterally. The segment’s dominance is supported by rising demand in clinical nutrition, especially among surgical, critical care, and malnourished patients, as well as by the increasing adoption of customized amino acid formulations to improve metabolic support and recovery outcomes.
The vitamins and minerals segment (salt and electrolyte) is expected to grow at the fastest CAGR over the forecast period. This growth is driven by the critical role of vitamins in essential physiological functions, including red blood cell formation, blood clotting, and maintenance of mucosal integrity. Deficiencies in these nutrients can lead to conditions such as anemia, beriberi, pellagra, and neurological disorders, thereby increasing the need for timely supplementation. As a result, the rising prevalence of such deficiencies and the growing use of parenteral nutrition in clinical settings are driving demand for vitamin- and mineral-based IV solutions.
End Use Insights
The hospitals segment dominated the U.S. intravenous solutions industry, accounting for a dominant share of 50.8% in 2025, primarily due to the high volume of inpatient procedures and the extensive use of IV therapies across emergency, surgical, and critical care settings. Hospitals represent the primary point of care for patients requiring rapid fluid resuscitation, electrolyte balance, parenteral nutrition, and intravenous drug administration, all of which rely heavily on IV solutions. Moreover, the growing prevalence of chronic diseases such as cancer, cardiovascular disorders, and renal conditions has increased hospitalization rates and the need for continuous fluid management and medication delivery through IV routes. Hospitals also manage many surgical procedures and intensive care admissions, where IV fluids are essential for maintaining hemodynamic stability and post-operative recovery.
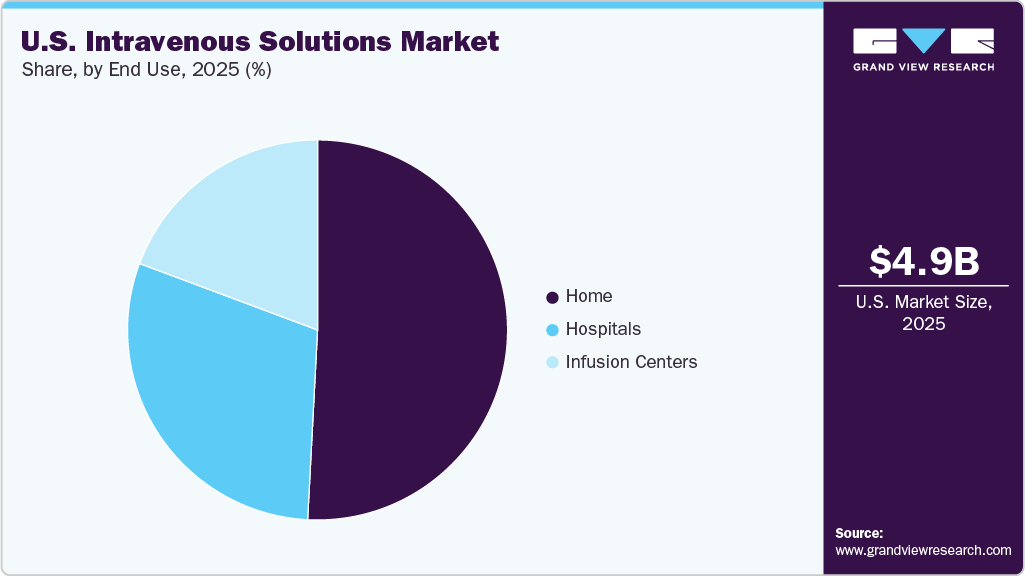
The home segment is expected to grow at a significant CAGR over the forecast period, driven by the increasing shift of healthcare delivery from hospitals to home-based settings. Rising demand for home infusion therapies, particularly for long-term treatments such as antibiotics, chemotherapy, hydration therapy, and parenteral nutrition, has strengthened the adoption of IV solutions in homecare environments. This trend is further supported by the growing geriatric population and the increasing prevalence of chronic diseases that require continuous or prolonged IV therapy.
Key U.S. Intravenous Solutions Company Insights
Product development, capacity expansion, strategic partnerships, and supply chain strengthening initiatives are the key strategic undertakings influencing the dynamics of the U.S. IV solutions market. Moreover, companies are focusing on regulatory compliance and domestic manufacturing expansion to address supply shortages and ensure consistent product availability across healthcare settings.
Key U.S. Intravenous Solutions Companies:
- Fresenius Kabi AG
- B. Braun SE
- Baxter
- Otsuka Pharmaceutical Co., Ltd.
- Pfizer, Inc.
- JW Life Science
- CSL Vifor (formerly Vifor Pharma)
- ICU Medical, Inc.
- Amphastar Pharmaceuticals, Inc.
Recent Developments
-
In April 2024, Baxter announced the further expansion of its pharmaceutical portfolio with the launch of five injectable products in the U.S., including IV solutions.
“Our Pharmaceuticals teams are relentlessly focused on bringing differentiated products to market that support our customers in helping to address vital patient needs, we look forward to further accelerating our impact with a robust innovation pipeline across our key therapeutic areas, including critical care, anti-infectives, pain and oncology.”
-Alok Sonig, executive vice president and group president, Pharmaceuticals, at Baxter
-
In December 2023, Fresenius Kabi entered into a multiyear supply and service agreement with the Mayo Clinic. Under this agreement, the Mayo Clinic is expected to purchase 10,000 Ivenix large-volume infusion pumps for its hospitals and clinics across Minnesota, Arizona, and Florida.
-
In March 2022, Fresenius Kabi disclosed that the U.S. authorities had approved an extended application for SMOFlipid Lipid Injectable Emulsion (ILE) for pediatric populations, encompassing both term and preterm infants
“There is a genuine need for alternative oil-based injectable lipid emulsions for patients of all ages, “Being able to meet these needs now with SMOFlipid for pediatric patients demonstrates our dedication to innovation when it comes to helping patients thrive on parenteral nutrition.”
- Pete Allen, senior vice president, Nutrition and Infusion Therapy, Fresenius Kabi USA.
U.S. Intravenous Solutions Market Report Scope
Report Attribute
Details
Revenue in 2026
USD 5.2 billion
Revenue forecast in 2033
USD 9.8 billion
Growth rate
CAGR of 9.4% from 2026 to 2033
Historical data
2021 - 2025
Forecast period
2026 - 2033
Quantitative units
Market Value in USD million/billion, volume in thousand units, and CAGR from 2026 to 2033
Report coverage
Revenue forecast, volume forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Type, nutrients, and end use
Key companies profiled
Fresenius Kabi AG; B. Braun SE; Baxter International Inc.; Otsuka Pharmaceutical Co., Ltd.; Pfizer, Inc.; JW Life Science; CSL Vifor; ICU Medical, Inc.; Amphastar Pharmaceuticals, Inc.
Customization scope
Free report customization (equivalent up to 8 analyst’s working days) with purchase. Addition or alteration to the country & segment scope.
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
U.S. Intravenous Solutions Market Report Segmentation
This report forecasts revenue & volume growth at the country level and provides an analysis of the latest industry trends in each of the sub-segments from 2021 to 2033. For this study, Grand View Research has segmented the U.S. intravenous solutions market report based on type, nutrients, and end use:
-
Type Outlook (Volume, Thousand Units; Revenue, USD Million, 2021 - 2033)
-
Total Parenteral Nutrition
-
Peripheral Parenteral Nutrition
-
-
Nutrients Outlook (Volume, Thousand Units; Revenue, USD Million, 2021 - 2033)
-
Carbohydrates
-
Vitamins & Minerals
-
Single-dose Amino Acids
-
Parenteral Lipid Emulsion
-
Others
-
-
End Use Outlook (Volume, Thousand Units; Revenue, USD Million, 2021 - 2033)
-
Home
-
Hospitals
-
Infusion Centers
-
Frequently Asked Questions About This Report
b. The U.S. intravenous solutions market size was estimated at USD 4.9 billion in 2025 and is expected to reach USD 5.2 billion in 2026.
b. The U.S. intravenous solutions market is expected to grow at a compound annual growth rate (CAGR) of 9.4% from 2026 to 2033 to reach USD 9.8 billion by 2033.
b. In terms of product, total parenteral nutrition (TPN) dominated the market with the largest share of 67.3% in 2025. The segment’s growth is driven by the rising prevalence of chronic diseases, increasing number of surgical procedures, and growing demand for clinical nutrition in ICU and neonatal care settings, where patients require specialized nutritional support.
b. Some key players operating in the U.S. intravenous solutions market include Fresenius Kabi AG, B. Braun SE, Baxter International Inc., Otsuka Pharmaceutical Co., Ltd., Pfizer, Inc., JW Life Science, CSL Vifor, ICU Medical, Inc., Amphastar Pharmaceuticals, Inc.
b. Key factors driving the market growth are the aging population needing medical interventions, a surge in chronic diseases requiring advanced treatments, and enhancements in healthcare technology that elevate patient care
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










