- Home
- »
- Pharmaceuticals
- »
-
U.S. Lysosomal Storage Disease Treatment Market Report, 2033GVR Report cover
![U.S. Lysosomal Storage Disease Treatment Market Size, Share & Trends Report]()
U.S. Lysosomal Storage Disease Treatment Market (2025 - 2033) Size, Share & Trends Analysis Report By Type (Enzyme Replacement Therapy (ERT)), By Disease Type, By Country, And Segment Forecasts
- Report ID: GVR-4-68040-668-5
- Number of Report Pages: 150
- Format: PDF
- Historical Range: 2021 - 2023
- Forecast Period: 2025 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
U.S. Lysosomal Storage Disease Treatment Market Summary
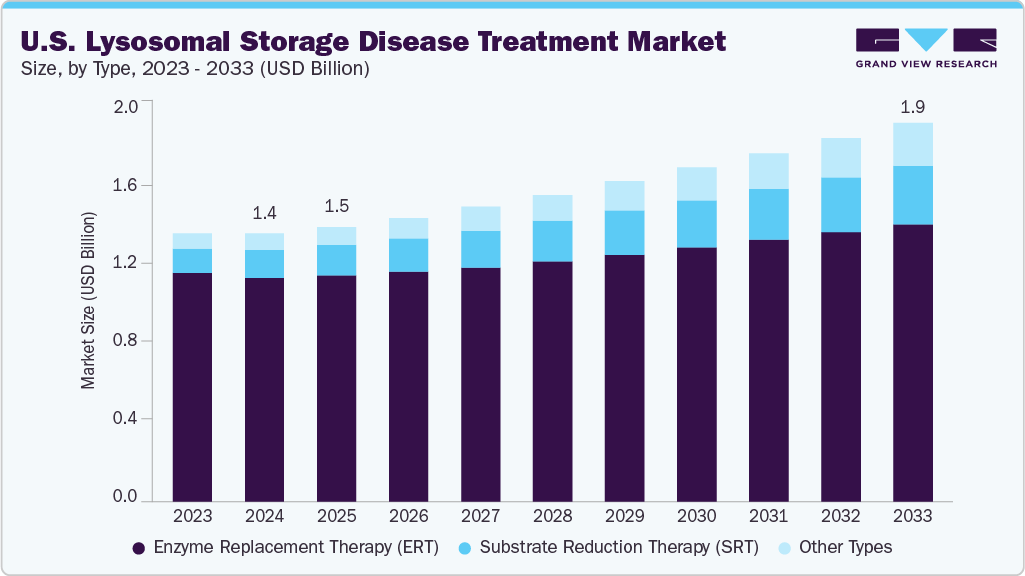
The U.S. lysosomal storage disease treatment market size was estimated at USD 1.41 billion in 2024 and is projected to reach USD 1.99 billion by 2033, growing at a CAGR of 4.10% from 2025 to 2033. The rising prevalence of rare genetic disorders, better diagnostic capabilities, and broader access to enzyme replacement and gene-based therapies drive this growth. In May 2025, the FDA accepted REGENXBIO’s BLA for RGX-121, a gene therapy for MPS II, showing 85% CSF heparan sulfate reduction and potential to replace long-term ERT. Increased awareness has led to earlier intervention and improved outcomes. Advancements in therapy design and strong industry-academic collaborations continue to drive innovation, supported by FDA incentives for orphan drug development.
One of the key drivers is the rapid evolution of gene therapy as a transformative approach for treating LSDs. Unlike enzyme replacement therapies, gene therapies target the root genetic cause, offering potential long-term or permanent benefits. In the U.S., several candidates for conditions such as MLD, Sanfilippo syndrome, and Gaucher disease are advancing through clinical stages. In March 2024, the FDA approved Lenmeldy (atidarsagene autotemcel), the first gene therapy in the U.S. for early-stage MLD, showing 100% survival at age 6 in treated presymptomatic patients versus 58% in untreated cases. Such milestones are increasing investment, accelerating adoption, and reshaping long-term LSD management.
Another major driver is the advancement of newborn screening programs across the U.S., which has led to earlier diagnosis of various LSDs. States are progressively expanding their recommended panels under the RUSP to include conditions such as Pompe disease and MPS I. Early detection enables the timely initiation of treatment, significantly improving long-term disease outcomes. For instance, in July 2024, Florida Newborn Screening added MPS II to its statewide panel using tandem mass spectrometry, enabling early diagnosis and clinical referral. As more states implement such measures, diagnosed cases are expected to rise. This trend supports proactive management and aligns industry strategies with early therapeutic intervention.
Market Concentration & Characteristics
The lysosomal storage disease (LSD) treatment market in the U.S. is marked by a high degree of innovation, driven primarily by advances in enzyme replacement therapies and gene therapies. Companies invest heavily in next-generation treatments with improved efficacy, reduced dosing frequency, and better patient compliance. Novel delivery systems and engineered enzymes are under continuous development. Research pipelines are robust, especially in addressing unmet needs in subtypes with limited options. Innovation remains essential to overcome disease complexity and heterogeneity across LSD types.
Entry into the U.S. LSD treatment space faces high barriers due to the need for specialized knowledge, extensive clinical validation, and regulatory hurdles. Developing therapies for rare diseases requires significant financial investment and long trial durations. A few large firms with established distribution and patient access programs dominate the market. In addition, orphan drug exclusivity and strong patent protection limit the space for new entrants. New players must also navigate complex reimbursement environments to gain traction.
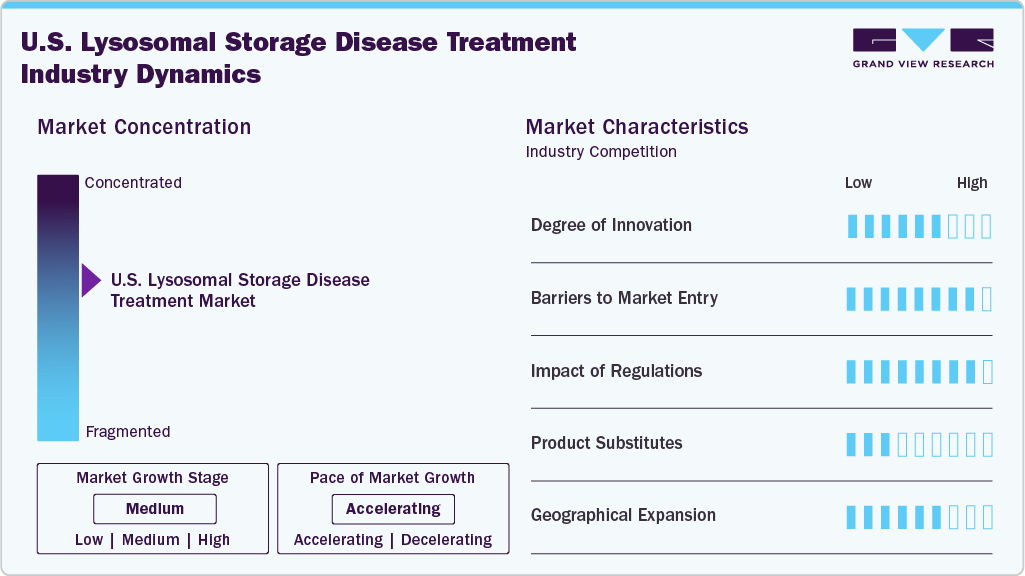
U.S. regulatory frameworks play a significant role in shaping the lysosomal storage disease (LSD) treatment market, offering both incentives and stringent oversight. The FDA provides accelerated pathways and orphan drug designations to encourage innovation for rare conditions. However, treatment approval still demands rigorous clinical evidence on safety and long-term efficacy. Post-marketing surveillance is also critical due to the chronic nature of LSDs. Regulations foster development but require high standards that only a few companies can consistently meet.
Product substitutes are minimally available in the LSD treatment market, owing to each therapy's highly specific nature. Most treatment regimens are tailored to particular enzyme deficiencies or genetic mutations. Supportive care options exist, but cannot replace the therapeutic role of enzyme or gene therapies. Switching between brands or treatment modalities is uncommon due to clinical risks and individual response variability. This limited substitution potential strengthens brand loyalty and long-term treatment adherence.
Geographical expansion within the U.S. lysosomal storage disease (LSD) treatment market primarily focuses on enhancing access in underserved and rural regions. Specialized treatment centers are often concentrated in major metropolitan areas, leading to disparities in care delivery. Companies are increasingly partnering with academic hospitals and specialty pharmacies to broaden their reach. Efforts are also being made to integrate telemedicine for follow-up care and treatment coordination. Expansion is crucial to ensure equitable access to advanced therapies nationwide.
Type Insights
The enzyme replacement therapy segment dominated the market with the largest revenue share of 83.7% in 2024, driven by the widespread use of approved therapies for Gaucher, Fabry, and Pompe diseases. These treatments offer targeted enzyme delivery that helps manage disease symptoms and improve patient outcomes. High physician familiarity and established dosing protocols have contributed to strong clinical adoption across treatment centers. Enzyme replacement therapies are widely reimbursed under private and public health plans, improving treatment accessibility. For instance, in February 2023, the U.S. FDA approved Lamzede (velmanase alfa), the first enzyme replacement therapy for alpha-mannosidosis. Developed by Chiesi Global Rare Diseases, Lamzede showed improved mobility and reduced serum oligosaccharide levels during a 52-week clinical trial. This approval expanded therapeutic options for patients with ultra-rare LSDs. Long-term efficacy data, infusion improvements, and multiple FDA-approved options have collectively ensured consistent supply, strengthened patient adherence, and supported wider market penetration.
The substrate reduction therapy segment is projected to grow at a significant CAGR of 8.51% over the forecast period, fueled by rising demand for oral alternatives that reduce treatment burden. These therapies offer convenience for patients who face challenges with frequent intravenous infusions. Substrate reduction therapies are being explored with enzyme replacement to enhance therapeutic impact. Clinical studies have shown promise in stabilizing disease progression in patients with limited response to standard treatment. Advancements in molecule design have improved specificity and tolerability, broadening the eligible patient pool. As new candidates progress through clinical stages, market growth is expected to accelerate through greater physician confidence and adoption.
Disease Type Insights
Fabry disease dominated the market with the largest revenue share of 29.3% in 2024, driven by the availability of multiple treatment options and a well-established diagnostic pathway in the U.S. Early recognition of symptoms by specialists has led to prompt initiation of therapy, improving clinical outcomes. Enzyme replacement and oral substrate reduction therapies are commonly used, supported by a growing body of long-term safety and efficacy data. In June 2023, the FDA cleared AceLink Therapeutics’ AL1211 for a Phase II trial in male patients with classic Fabry disease. AL1211, an oral glucosylceramide synthase (GCS) inhibitor, demonstrated approximately 78% reduction in glucosylceramide levels and significant decreases in Gb3 during Phase I healthy volunteer studies. Pharmaceutical companies continue to invest in Fabry-focused drug development to address unmet needs and expand treatment choices. Disease awareness efforts and expanded family screening have increased diagnosis rates. The strong presence of clinical infrastructure has enabled more consistent disease management across regions.
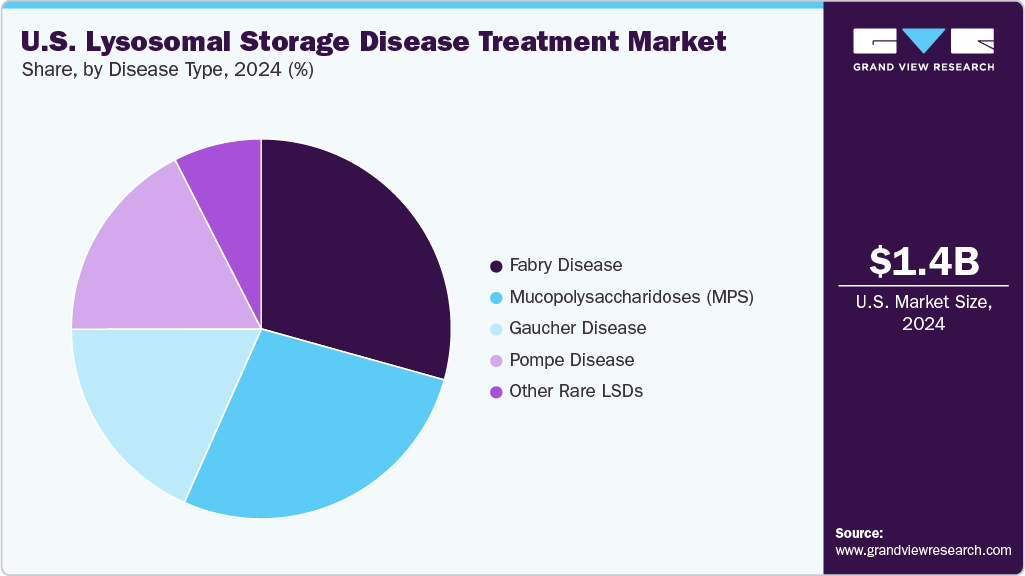
The mucopolysaccharidoses segment is projected to grow at the fastest CAGR of 5.11% over the forecast period due to the increasing focus on early intervention and the introduction of novel therapies for multiple MPS subtypes. Improved newborn screening initiatives have allowed earlier identification of affected individuals, enabling timely treatment. Enzyme replacement therapies and experimental gene-based solutions are actively being developed for conditions such as MPS I, II, and VI. In May 2025, the FDA accepted REGENXBIO’s Biologics License Application for RGX‑121 (clemidsogene lanparvovec), a potential first-in-class, one-time gene therapy for MPS II with Priority Review and a PDUFA target date of November 9, 2025. Several clinical trials are underway to evaluate long-term efficacy and cognitive outcomes in pediatric patients. Rising disease awareness among clinicians and caregivers translates into faster referral rates and diagnoses. The expanding treatment landscape and strong research pipeline will support sustained segment growth over the forecast period.
Key U.S. Lysosomal Storage Disease Treatment Company Insights
Pfizer Inc. and Takeda Pharmaceutical Company Limited contribute to the U.S. LSDs treatment market through enzyme replacement therapies and ongoing clinical research targeting rare genetic conditions. Sanofi (Genzyme Corporation) offers therapies for multiple LSD types, focusing on consistent treatment performance and patient support. BioMarin and Johnson & Johnson (Actelion Pharmaceuticals Ltd) support market growth with therapies addressing complex lysosomal disorders. Amicus Therapeutics, Inc. and Alexion Pharmaceuticals, Inc. expand options through specialized approaches to improve long-term disease management. Sigilon Therapeutics, Inc. and Orphazyme A/S explore novel delivery methods and focus on advancing pipeline candidates for underserved lysosomal disease subtypes.
Key U.S. Lysosomal Storage Disease Treatment Companies:
- Pfizer Inc
- Takeda Pharmaceutical Company Limited (Shire Plc)
- Sanofi (Genzyme Corporation)
- BioMarin
- Johnson & Johnson (Actelion Pharmaceuticals Ltd)
- Amicus Therapeutics, Inc
- Alexion Pharmaceuticals, Inc
- Sigilon Therapeutics, Inc
- Orphazyme A/S
Recent Developments
-
In May 2025, BioMarin Pharmaceutical Inc. announced its acquisition of Inozyme Pharma, Inc. to expand its enzyme replacement therapy pipeline. The deal includes INZ-701, a candidate for ENPP1 deficiency, with pivotal pediatric trial results expected in early 2026. This move strengthens BioMarin’s rare disease treatment portfolio.
-
In February 2025, Denali Therapeutics reported that its investigational therapy for Hunter syndrome, tividenofusp alfa (DNL310), reduced cerebrospinal fluid heparan sulfate by ~90% in 24 weeks and showed sustained cognitive and hearing benefits for over 4 years. A rolling BLA submission is planned, targeting a 2025-2026 launch.
U.S. Lysosomal Storage Disease Treatment Market Report Scope
Report Attribute
Details
Market size value in 2025
USD 1.45 billion
Revenue forecast in 2033
USD 1.99 billion
Growth rate
CAGR of 4.10% from 2025 to 2033
Base year for estimation
2024
Historical data
2021 - 2023
Forecast period
2025 - 2033
Quantitative units
Revenue in USD million/billion and CAGR from 2025 to 2033
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, trends
Segments covered
Type, disease type
Key company profiled
Pfizer Inc.; Takeda Pharmaceutical Company Limited (Shire Plc); Sanofi (Genzyme Corporation); BioMarin; Johnson & Johnson (Actelion Pharmaceuticals Ltd); Amicus Therapeutics, Inc; Alexion Pharmaceuticals, Inc; Sigilon Therapeutics, Inc; Orphazyme A/S.
Customization scope
Free report customization (equivalent up to 8 analysts working days) with purchase. Addition or alteration to country, regional & segment scope.
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
U.S. Lysosomal Storage Disease (LSDs) Treatment Market Report Segmentation
This report forecasts revenue growth at regional levels and provides an analysis of the latest industry trends in each of the sub-segments from 2021 to 2033. For this study, Grand View Research has segmented the U.S. lysosomal storage disease treatment market report based on type and disease type:
-
Type Outlook (Revenue, USD Billion, 2021 - 2033)
-
Enzyme Replacement Therapy (ERT)
-
Imiglucerase (Cerezyme)
-
Alglucosidase alfa (Myozyme/Lumizyme)
-
Idursulfase (Elaprase)
-
Velaglucerase alfa
-
Others
-
Substrate Reduction Therapy (SRT)
-
Other Types
-
-
Disease Type Outlook (Revenue, USD Billion, 2021 - 2033)
-
Gaucher Disease
-
Fabry Disease
-
Pompe Disease
-
Mucopolysaccharidoses (MPS)
-
Others
-
Frequently Asked Questions About This Report
b. The U.S. lysosomal storage disease treatment market size was estimated at USD 1.41 billion in 2024 and is expected to reach USD 1.45 billion in 2025.
b. The U.S. lysosomal storage disease treatment market is projected to grow at a CAGR of 4.10% from 2025 to 2033 to reach USD 1.99 billion by 2033.
b. Based on type, enzyme replacement therapy segment dominated the market with the largest revenue share of 83.7% in 2024, driven by the widespread use of approved therapies for Gaucher, Fabry, and Pompe diseases.
b. Key players in the U.S. lysosomal storage disease treatment market include Sanofi, Takeda Pharmaceutical Company, Pfizer Inc., Amicus Therapeutics, and BioMarin Pharmaceutical Inc. These companies offer enzyme replacement therapies and gene therapy solutions for conditions such as Gaucher disease, Fabry disease, and Pompe disease. Strategic collaborations, FDA approvals, and ongoing R&D in gene editing and novel biologics support their market positioning. Emerging players are also investing in targeted therapies and orphan drug development.
b. The U.S. lysosomal storage disease treatment market is driven by rising awareness and diagnosis rates of rare genetic disorders, favorable orphan drug legislation, and increased funding for rare disease research. Advances in gene therapy, enzyme replacement therapies, and personalized medicine also fuel growth. Strong regulatory support from the FDA and strategic collaborations between biotech firms further accelerate innovation. Additionally, patient advocacy groups and expanded newborn screening programs contribute to early detection and treatment adoption across the country.
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










