- Home
- »
- Clinical Diagnostics
- »
-
U.S. Minimal Residual Disease Testing Market Report, 2033GVR Report cover
![U.S. Minimal Residual Disease Testing Market Size, Share & Trends Report]()
U.S. Minimal Residual Disease Testing Market (2025 - 2033) Size, Share & Trends Analysis Report By Technology (Flow Cytometry, PCR, NGS), By Cancer Type (Hematological Malignancy, Solid Tumors), By End Use, And Segment Forecasts
- Report ID: GVR-4-68040-651-7
- Number of Report Pages: 100
- Format: PDF
- Historical Range: 2021 - 2023
- Forecast Period: 2025 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
Market Size & Trends
The U.S. minimal residual disease testing market was estimated at USD 873.66 million in 2024 and is projected to grow at a CAGR of 9.38% from 2025 and 2033, driven by the country’s rising cancer burden and the healthcare system’s growing emphasis on precision medicine.

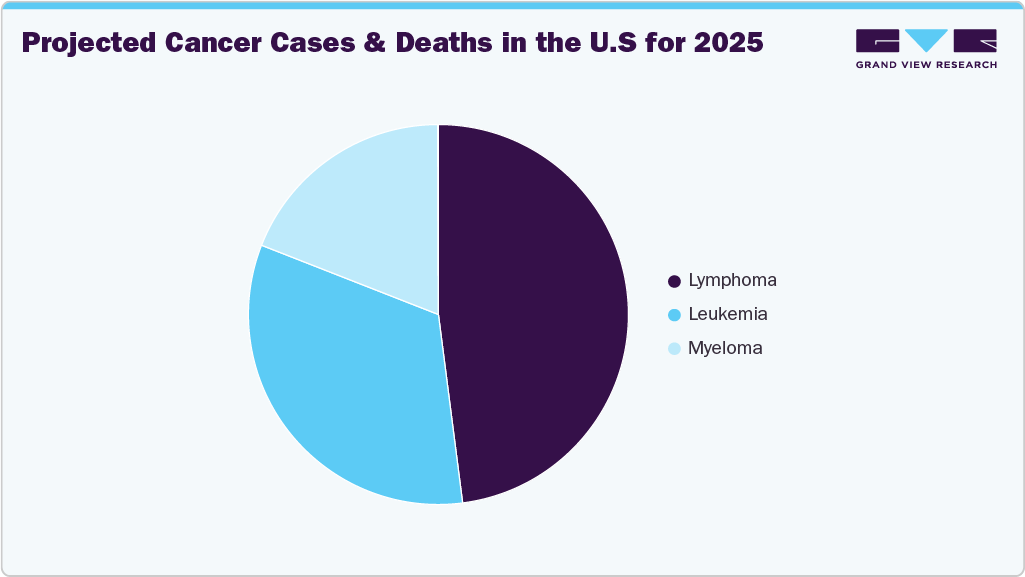
According to recent projections, about 89,190 people in the U.S. will be diagnosed with lymphoma in 2024, encompassing both Hodgkin and non-Hodgkin types. An estimated 21,050 deaths are anticipated, with non-Hodgkin lymphoma being both more common and deadlier, especially in men. These alarming statistics underscore the urgent demand for highly sensitive, early detection tools like MRD testing. MRD testing enables physicians to detect trace levels of cancer cells that persist after treatment, even when patients appear to be in remission. This technology plays a vital role in identifying patients at high risk of relapses, guiding further therapy, and improving long-term outcomes.
The shift toward value-based care and personalized treatment plans has elevated MRD testing from an optional research tool to a mainstream clinical necessity. As cancer therapies become more targeted and adaptive, the demand for precise disease monitoring tools like MRD testing continues to grow. This trend is especially pronounced in hematologic malignancies, where MRD status is increasingly being used as a surrogate endpoint in clinical trials and real-world treatment protocols.
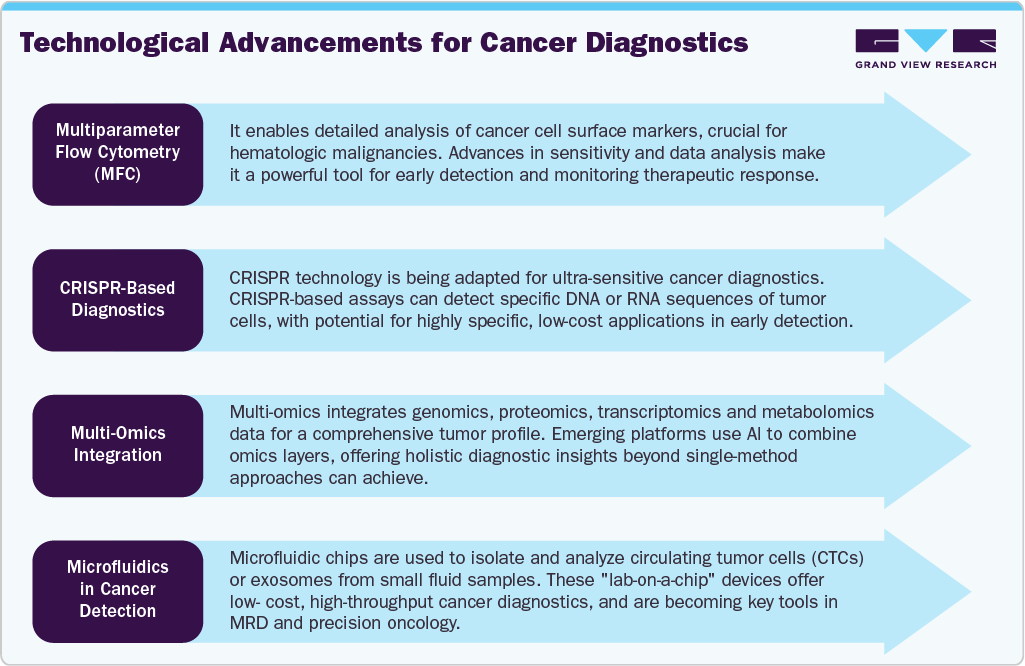
Recent years have witnessed rapid advancements in next-generation sequencing (NGS) and flow cytometry, making MRD detection more accurate, reproducible, and clinically actionable. FDA-cleared tests such as Adaptive Biotechnologies’ clonoSEQ for multiple myeloma, acute lymphoblastic leukemia (ALL), and chronic lymphocytic leukemia (CLL) have set new benchmarks for test sensitivity and adoption. Similarly, Natera’s Signatera, a tumor-informed ctDNA-based MRD test, is gaining traction in solid tumor management, including colorectal, breast, and bladder cancers. These assays allow for longitudinal disease tracking, personalized treatment decisions, and the early detection of recurrence-often months before symptoms or imaging changes appear.
Increased clinical utility has translated into broader payer acceptance; Medicare and major commercial insurers are expanding reimbursement policies, making these tests more accessible to patients nationwide. Simultaneously, growing oncologist awareness and training, along with inclusion in national clinical guidelines (such as NCCN recommendations), are normalizing MRD testing in community oncology settings. These forces collectively drive the integration of MRD into routine practice, transforming cancer follow-up care from reactive to proactive and establishing the U.S. as a global leader in MRD diagnostics.
Regulatory support from agencies like the FDA, including accelerated approvals and breakthrough device designations, has lowered barriers to market entry and clinical validation. Fourth, payer reimbursement, particularly through Medicare’s Local Coverage Determinations (LCDs), has made these tests more accessible. A pivotal moment came in June 2025, when Medicare approved coverage for Natera’s Signatera under LCD L38779, significantly enhancing patient access. Finally, clinical trial integration and guideline updates are accelerating adoption; more oncology trials now include MRD as a primary or secondary endpoint, and key societies are incorporating MRD testing into standard-of-care protocols. Together, these five factors ensure sustained double-digit market growth and signal a broader transformation in how cancer is monitored and managed in the U.S.
Market Concentration & Characteristics
The U.S. minimal residual disease testing industry is marked by a high degree of innovation, particularly in next-generation sequencing (NGS) and liquid biopsy technologies. Tumor-informed assays, AI-powered analysis, and ultra-sensitive ctDNA detection methods are transforming cancer monitoring. Ongoing research, biotech collaborations, and increasing FDA breakthrough device designations drive the continual evolution of MRD solutions across both hematologic and solid tumor applications, positioning the U.S. as a global innovation leader.
The U.S. minimal residual disease testing industry is experiencing moderate-to-high M&A activity as major diagnostics firms seek to expand capabilities and pipelines. Companies like Thermo Fisher, Illumina, and Exact Sciences have pursued acquisitions to bolster MRD assay portfolios. Strategic consolidations allow access to advanced molecular platforms and customer bases, while startups with novel MRD technologies become prime targets for integration into larger, scalable precision oncology ecosystems.

Regulatory frameworks in the U.S. are significantly shaping the MRD testing landscape. The FDA’s breakthrough device designations and accelerated pathways are expediting the clinical use of MRD assays. However, evolving guidelines around analytical validation and clinical utility present both opportunities and compliance challenges. Reimbursement policy alignment, especially from CMS, strongly influences adoption and access. Regulatory clarity continues to be a critical enabler for U.S. minimal residual disease testing industry expansion.
Product expansion in the U.S. minimal residual disease testing industry is robust, with companies broadening indications across cancer types. Originally focused on hematologic malignancies, MRD assays are now validated for colorectal, breast, lung, and bladder cancers. Firms are also developing pan-cancer MRD panels and multi-analyte assays. As test sensitivity improves, MRD products are extending into earlier-stage cancers and adjuvant treatment settings, reinforcing their clinical value and widening their market scope.
Product substitution risk is relatively low in the U.S. minimal residual disease testing industry due to the complexity, regulatory barriers, and clinical validation required. However, competition among testing platforms, such as flow cytometry, NGS, and digital PCR, can lead to substitution within specific use cases. Hospital systems and oncology practices may switch between platforms based on sensitivity, turnaround time, or payer coverage. Still, brand loyalty and clinical familiarity often reduce switching frequency.
Technology Insights
The flow cytometry segment led the market with the largest revenue share of 40.17% in 2024, due to its high sensitivity, speed, and ability to provide multi-parametric analysis of individual cells. This technique enables the precise detection and quantification of residual cancer cells in blood or bone marrow samples, even when present in extremely low frequencies down to one cancer cell among 10,000 to 1,000,000 normal cells. Technological advancements have further enhanced its capabilities, including next-generation flow cytometry with higher resolution and standardized panels. These improvements support its clinical adoption as a critical tool in guiding therapy decisions and improving long-term outcomes. Its cost-effectiveness compared to molecular methods and broad accessibility in clinical laboratories make it a preferred choice for MRD testing in both developed and emerging markets
The polymerase chain reaction (PCR) segment is expected to grow at the fastest CAGR over the forecast period, due to its exceptional sensitivity and specificity in detecting minute amounts of residual cancer cells. PCR-based techniques, including quantitative PCR (qPCR) and digital PCR (dPCR), can identify one malignant cell among a million normal cells, making them valuable for monitoring hematologic malignancies such as leukemia, lymphoma, and multiple myeloma. Research advancements in PCR technology, including real-time and digital PCR platforms, have improved accuracy, reduced turnaround times, and allowed for more precise quantification of MRD levels. Leading biotechnology and diagnostic companies continuously introduce advanced PCR platforms and assay kits designed specifically for MRD detection in hematologic cancers
Cancer Type Insight
The hematological malignancies segment led the market with the largest revenue share of 68.58% in 2024 and is anticipated to grow at the fastest CAGR over the forecast period, driven by high prevalence of blood cancers such as leukemia, lymphoma, and multiple myeloma. Minimal residual disease (MRD) testing has become an essential component in the management of hematological malignancies, significantly impacting prognosis, treatment decisions, and long-term patient outcomes. Hematological malignancies such as acute lymphoblastic leukemia (ALL), acute myeloid leukemia (AML), chronic lymphocytic leukemia (CLL) and multiple myeloma often involve the presence of residual cancer cells that remain after initial therapy, which can lead to relapse.
Meanwhile, Minimal residual disease (MRD) testing in solid tumors is an emerging and rapidly expanding segment within the oncology diagnostics market, driven by advancements in liquid biopsy technologies and the increasing need for early relapse detection. MRD testing is well-established, solid tumors such as colorectal, lung, breast, and pancreatic cancers pose unique challenges due to tumor heterogeneity and the lower concentration of circulating tumor DNA (ctDNA) in the bloodstream. A study published in the May 2024 issue of JAMA Health Forum highlighted the potential of circulating tumor DNA (ctDNA) testing in minimal residual disease (MRD) detection for stage II colorectal cancer patients. The research demonstrated that advanced blood-based liquid biopsies could accurately identify patients who might safely avoid chemotherapy after surgery by detecting early signs of residual or recurrent cancer.
End Use Insight
The hospitals and specialty clinics market segment led the market with the largest revenue share of 50.23% in 2024. driven by increasing demand for rapid, reliable quality control in biologics production. In January 2024, Charles River Laboratories advanced its portfolio with the launch of the Endosafe Trillium rCR cartridge. This innovative solution combines the proven Endosafe cartridge technology with a recombinant cascade reagent (rCR), offering a fully animal-free, efficient, and sustainable testing alternative. The Trillium rCR enhances speed and accuracy in endotoxin detection while aligning with regulatory expectations and Charles River’s 4Rs sustainability initiative-Replacement, Reduction, Refinement, and Responsibility further strengthening its market position.

The diagnostic laboratories segment is projected to grow at the fastest CAGR over the forecast period, which is critical for detecting tiny amounts of cancer cells remaining after treatment, primarily in hematologic malignancies such as leukemia and lymphoma. Laboratories employ highly sensitive techniques including flow cytometry, polymerase chain reaction (PCR), and next-generation sequencing (NGS). The choice of method depends on the specific type of cancer and its molecular characteristics.
In June 2023, NeoGenomics, Inc. announced that its RaDaR assay, a personalized liquid biopsy for detecting minimal residual disease (MRD) and cancer recurrence. The test is designed to identify extremely low levels of circulating tumor DNA (ctDNA) in the blood with high sensitivity and specificity. RaDaR can track up to 48 tumor-specific genetic variants unique to each patient’s cancer, providing valuable information and additional time to support critical treatment decisions. Achieving a low limit of detection is essential to accurately identify residual disease, which is often undetectable by conventional diagnostic methods. MRD positivity indicates the presence of remaining cancer cells and a higher risk of relapses, whereas MRD negativity suggests a better prognosis and longer remission.
Key U.S. Minimal Residual Disease Testing Company Insights
Leading players are focusing on expanding their product and service portfolios to increase revenue. Market players, such as Twist Bioscience, Thermo Fisher Scientific, Inc., Illumina, Inc., QIAGEN, Adaptive Biotechnologies, Natera, Inc., Guardant Health, Foresight Diagnostics, Inc., NeoGenomics, Inc., F. Hoffmann-La Roche Ltd., SOPHiA GENETICS, Sysmex, Personalis, Inc., Exact Sciences Corporation, Quest Diagnostics (Haystack Oncology), MedGenome, and Oncquest.
For instance, in March 2025, Adaptive Biotechnologies Corporation announced the launch of an enhanced version of its clonoSEQ assay. The upgraded assay is designed to detect measurable residual disease (MRD) in diffuse large B-cell lymphoma (DLBCL) using circulating tumor DNA (ctDNA).
Key U.S. Minimal Residual Disease Testing Companies:
- Twist Bioscience
- Thermo Fisher Scientific, Inc
- Illumina, Inc.
- QIAGEN
- Adaptive Biotechnologies
- Natera, Inc.
- Guardant Health
- Foresight Diagnostics, Inc.
- NeoGenomics, Inc.
- F. Hoffmann-La Roche Ltd.
- SOPHiA GENETICS
Recent Developments
-
In June 2025, QIAGEN expanded its oncology diagnostics portfolio through two strategic partnerships aimed at advancing MRD testing in clinical trials. These collaborations-with Tracer Biotechnologies and Foresight Diagnostics-enhance QIAGEN’s presence in the MRD space, supporting companion diagnostic co-development projects across both solid tumors and hematologic cancers.
-
In May 2025, QIAGEN announced a new commercial partnership and co-marketing agreement with ID Solutions, a French company specializing in high-quality digital PCR (dPCR) assays and customized molecular diagnostics for oncology and other disease areas. The collaboration aims to broaden access to dPCR assays for oncology research applications.
-
In April 2025, Natera, Inc., announced the nationwide availability of its ultra-sensitive Signatera Genome assay for physicians across the U.S. This launch is backed by a major genome-based molecular residual disease (MRD) study, which has been accepted for presentation at the 2025 American Society of Clinical Oncology (ASCO) Annual Meeting.
-
In March 2025, Adaptive Biotechnologies Corporation announced the launch of an enhanced version of its clonoSEQ assay. The upgraded assay is designed to detect measurable residual disease (MRD) in diffuse large B-cell lymphoma (DLBCL) using circulating tumor DNA (ctDNA).
U.S. Minimal Residual Disease Testing Market Report Scope
Report Attribute
Details
Market size value in 2025
USD 986.54 million
Revenue forecast in 2033
USD 2,021.08 million
Growth rate
CAGR of 9.38% from 2025 to 2033
Base year for estimation
2024
Historical data
2021 - 2023
Forecast period
2025 - 2033
Quantitative units
Revenue in USD million/billion and CAGR from 2025 to 2033
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Technology, cancer type, end use
Country scope
U.S.
Key companies profiled
Twist Bioscience; Thermo Fisher Scientific, Inc.; Illumina, Inc.; QIAGEN, Adaptive Biotechnologies; Natera, Inc.; Guardant Health; Foresight Diagnostics, Inc.; NeoGenomics, Inc.; F. Hoffmann-La Roche Ltd.; SOPHiA GENETICS
Customization scope
Free report customization (equivalent up to 8 analysts working days) with purchase. Addition or alteration to country, regional & segment scope.
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
U.S. Minimal Residual Disease Testing Market Report Segmentation
This report forecasts revenue growth at regional & country levels and provides an analysis on the industry trends in each of the sub-segments from 2021 to 2033. For this study, Grand View Research has segmented the U.S. minimal residual disease testing market report based on the technology, cancer type, and end use:
-
Technology Outlook (Revenue, USD Million, 2021 - 2033)
-
Flow Cytometry
-
Polymerase Chain Reaction (PCR)
-
Next Generation Sequencing (NGS)
-
Amplicon-based NGS
-
Hybridization Capture-based NGS
-
Others
-
-
Others
-
-
Cancer Type Outlook (Revenue, USD Million, 2021 - 2033)
-
Hematological Malignancy
-
Leukemia
-
Lymphoma
-
-
Solid Tumors
-
-
End Use Outlook (Revenue, USD Million, 2021 - 2033)
-
Hospitals and Specialty Clinics
-
Diagnostic Laboratories
-
Academic and Research Institutes
-
Others
-
Frequently Asked Questions About This Report
b. The global U.S. minimal residual disease testing market size was estimated at USD 873.66 million in 2024 and is expected to reach USD 986.54 million in 2025.
b. The global U.S. minimal residual disease testing market is expected to grow at a compound annual growth rate of 9.38% from 2025 to 2033 to reach USD 2,021.08 million by 2033.
b. On the basis of technology, the flow cytometry segment dominated the U.S. minimal residual disease testing with a share of 40.17% in 2024, due to its high sensitivity, speed, and ability to provide multi-parametric analysis of individual cells.
b. Leading players are focusing on expanding their product and service portfolios to increase revenue. Market players, such as Twist Bioscience, Thermo Fisher Scientific, Inc., Illumina, Inc., QIAGEN, Adaptive Biotechnologies, Natera, Inc., Guardant Health, Foresight Diagnostics, Inc., NeoGenomics, Inc., F. Hoffmann-La Roche Ltd., SOPHiA GENETICS, Sysmex, Personalis, Inc., Exact Sciences Corporation, Quest Diagnostics (Haystack Oncology), MedGenome, and Oncquest.
b. Key factors that are driving the market growth driven by the country’s rising cancer burden and the healthcare system’s growing emphasis on precision medicine.
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










