- Home
- »
- Clinical Diagnostics
- »
-
U.S. Non-Invasive Cancer Diagnostics Market Report, 2033GVR Report cover
![U.S. Non-Invasive Cancer Diagnostics Market Size, Share, & Trends Report]()
U.S. Non-Invasive Cancer Diagnostics Market (2026 - 2033) Size, Share, & Trends Analysis Report By Product (Pathology-based Instruments, Imaging Instruments), By Technology, By Indication, By End User, And Segment Forecasts
- Report ID: GVR-4-68040-911-6
- Number of Report Pages: 100
- Format: PDF
- Historical Range: 2021 - 2024
- Forecast Period: 2026 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
U.S. Non-Invasive Cancer Diagnostics Market Summary
The U.S. non-invasive cancer diagnostics market size was estimated at USD 27.08 billion in 2025 and is projected to reach USD 47.52 billion in 2033, growing at a CAGR of 7.4% from 2026 to 2033. The growth is driven by the continuous advancements in diagnostic technologies, the rising prevalence of cancer, and increasing initiatives by both private and public organizations aimed at enhancing early detection and treatment.
Key Market Trends & Insights
- By product, the instruments segment held the largest revenue share of 64.60% in 2025.
- By technology, the imaging technology segment accounted for the largest revenue share of 39.69% in 2025.
- By indication, the liver cancer segment held the largest revenue share of 16.57% in 2025.
- By end user, the diagnostic laboratories segment accounted for the largest revenue share of 53.50% in 2025.
Market Size & Forecast
- 2025 Market Size: USD 27.08 Billion
- 2033 Projected Market Size: USD 47.52 Billion
- CAGR (2026-2033): 7.4%
The U.S. non-invasive cancer diagnostics industry’s growth is further driven by strategic expansion initiatives by companies to strengthen commercialization and innovation capabilities. For instance, in June 2025, Dxcover launched its U.S. headquarters in Nashville to accelerate the commercialization of its AI-powered PANAROMIC cancer test in the world’s largest life sciences market. The new facility will support partnerships, local manufacturing, and R&D, strengthening its non-invasive, cost-effective cancer screening platform and expanding early detection capabilities.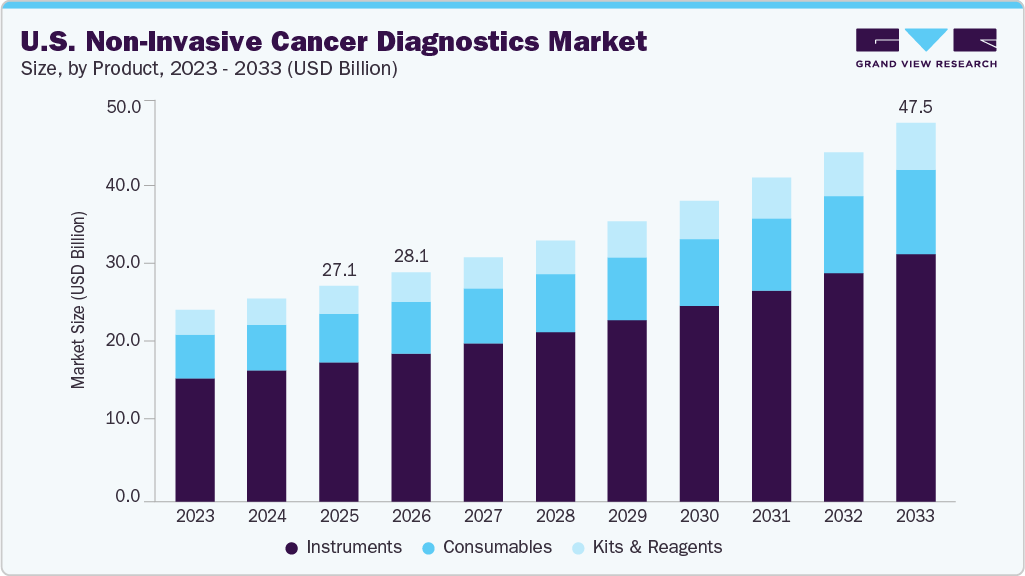
In addition, increasing demand for early, accurate, and minimally invasive detection methods that improve patient outcomes. Advancements in blood-based and multi-cancer screening technologies are further supporting timely diagnosis and wider clinical adoption. For instance, in September 2025, Exact Sciences launched Cancerguard, a first-of-its-kind multi-cancer early detection blood test in the U.S., capable of identifying over 50 cancer types through multiple biomarkers. The test aims to address gaps in early diagnosis, demonstrating strong sensitivity and high specificity, and is expected to improve outcomes by enabling earlier detection alongside existing screening methods.
Product Insights
The instruments segment dominated the U.S. non-invasive cancer diagnostics market, accounting for the largest revenue share of 64.60% in 2025. This dominance is supported by the growing adoption of advanced pathology tools, such as PCR, NGS, and microarrays, which enable faster, more accurate biomarker-based testing. Also, increasing use of imaging systems such as magnetic resonance imaging (MRI), CT, and PET, supported by AI integration and improved imaging capabilities, is enhancing early cancer detection and treatment monitoring. For instance, in March 2026, Perimeter Medical Imaging AI received USFDA approval for its AI-enabled imaging device Claire, the first in the U.S. for intraoperative breast cancer margin assessment. Technology aims to help surgeons detect residual cancer more accurately during breast-conserving surgery, potentially reducing repeat procedures and improving patient outcomes.
The consumables segment is expected to grow significantly over the forecast period. Consumables such as test strips, syringes & needles, vials & containers, and centrifuge tubes are typically single-use items, which means laboratories and healthcare providers must continuously purchase them to support ongoing diagnostic procedures. As the demand for early cancer detection and non-invasive screening methods increases, the volume of diagnostic tests performed is also rising, directly driving the demand for consumables.
Technology Insights
The imaging technology segment accounted for the largest revenue share of the U.S. non-invasive cancer diagnostics industry in 2025. This growth is driven by the increasing integration of advanced artificial intelligence (AI) solutions that enhance early cancer detection and diagnostic accuracy. For instance, in November 2025, DeepHealth unveiled next-generation clinical AI solutions at RSNA 2025, with a strong focus on improving cancer detection and diagnosis. The company introduced its AI-powered Breast Suite, designed to enhance early breast cancer detection and screening accuracy, with studies showing up to a 21% increase in detection rates.
The liquid biopsy segment is experiencing significant growth over the forecast period, due to the continuous technological advancements that significantly improve the accuracy, sensitivity, and scope of early cancer detection. Innovations in biomarker identification, particularly in circulating tumor DNA (ctDNA), circulating tumor cells (CTCs), and RNA-based markers, are enabling more precise cancer detection at earlier stages. For instance, in July 2025, Researchers at the University of Chicago introduced an advanced liquid biopsy test leveraging RNA modifications to detect early-stage colorectal cancer. The test achieves around 95% accuracy, offering improved early detection over current DNA-based non-invasive methods.
Indication Insights
The liver cancer segment accounted for the largest revenue share of the U.S. non-invasive cancer diagnostics market in 2025, driven by the growing shift toward non-invasive, early-detection technologies such as liquid biopsies and biomarker-based blood tests, which enable accurate diagnosis with simple blood samples without the need for surgical procedures. This growth is further supported by the rising incidence of liver cancer in the U.S. According to the American Cancer Society, the U.S. is expected to record about 42,340 new cases of primary liver and intrahepatic bile duct cancer in 2026, with approximately 30,980 deaths. The data highlights a significant long-term trend, with liver cancer incidence rates having tripled in the country over the past four decades, indicating rising disease burden.
The lung cancer segment is expected to witness significant growth over the forecast period, due to the high disease burden and mortality associated with lung cancer, which creates a strong need for early and accurate detection solutions. Lung cancer remains the leading cause of cancer-related deaths in the U.S., accounting for about 1 in 5 cancer deaths, highlighting its significant clinical priority.According to estimates by the American Cancer Society for 2026, the U.S. is expected to report 229,410 new lung cancer cases and about 124,990 deaths annually, highlighting a substantial disease burden and a large patient population requiring continuous diagnosis and monitoring.
End User Insights
The diagnostic laboratories segment accounted for the largest revenue share of the U.S. non-invasive cancer diagnostics industry in 2025. The increasing number of strategic partnerships and new test launches by diagnostic companies. Collaborations between diagnostic firms and insurers or healthcare providers are improving test accessibility, expanding distribution networks, and accelerating the commercialization of advanced multi-cancer detection solutions. For instance, in March 2026, Guardant Health introduced its Shield multi-cancer detection test in Asia through a partnership with Manulife. The test will be accessible to eligible customers in Hong Kong, Singapore, and the Philippines from April 2026, enhancing access to early cancer detection.
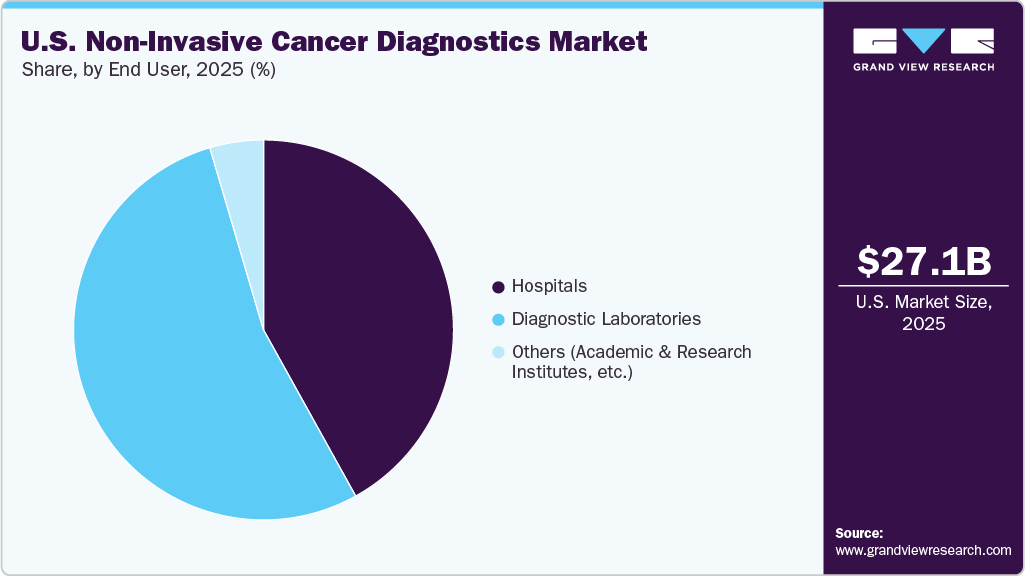
The hospitals segment is estimated to grow at the fastest CAGR over the forecast period. Hospitals in the U.S. are rapidly adopting non-invasive diagnostic solutions such as liquid biopsy, NGS, and advanced imaging systems to enable early and accurate cancer detection. These facilities offer centralized infrastructure and multidisciplinary oncology teams, making them the primary point of care for cancer patients. The availability of skilled professionals, strong reimbursement support, and continuous investment in advanced technologies further strengthen hospital dominance. In addition, the rising number of hospitals and increasing volume of cancer diagnostic procedures, along with a growing focus on precision medicine and patient-centric care, are accelerating the use of non-invasive diagnostics in hospitals, thereby driving segment growth.
Key Companies & Market Share Insights
Some key players operating in the market include Abbott Laboratories; GE HealthCare; Siemens Healthineers AG; Koninklijke Philips N.V.; Canon Medical Systems; and F. Hoffmann-La Roche Ltd.
-
GE HealthCare is a global healthcare solutions provider offering medical technology, pharmaceutical diagnostics, and AI-enabled digital solutions. The company serves healthcare providers and researchers across public, private, and academic institutions. It operates through four segments: Advanced Visualization Solutions, Imaging, Patient Care Solutions, and Pharmaceutical Diagnostics. GE HealthCare has a strong global installed base of imaging, ultrasound, and patient monitoring systems supporting patient diagnosis and care.
Key U.S. Non-invasive Cancer Diagnostics Companies:
- Abbott Laboratories
- GE HealthCare
- Siemens Healthineers AG
- Koninklijke Philips N.V.
- Canon Medical Systems
- F. Hoffmann-La Roche Ltd
- Illumina, Inc
- Guardant Health, Inc.
- Thermo Fisher Scientific Inc.
- FONAR Corporation
Recent Developments
-
In February 2026, Quest Diagnostics introduced a noninvasive blood test, Quest Flow Cytometry MRD for Myeloma, for monitoring measurable residual disease (MRD) in myeloma patients across the U.S. The test leverages advanced flow cytometry to identify minimal cancer cells, providing a convenient alternative to bone marrow biopsies and enhancing access to sensitive disease monitoring.
-
In February 2026, Integrated DNA Technologies announced a new NGS innovation roadmap aimed at advancing cancer research by improving library preparation, targeted enrichment, and whole-genome analysis.
-
In February 2026, Ultima Genomics, Inc. launches the NGS, UG200 Series, and Solaris 2.0 workflows, offering higher output, improved performance, and flexible wafer configurations at lower costs.
-
In November 2024, Canon Medical USA launched its AI-powered Vantage Galan 3T/Supreme Edition MRI system as competition grows in the medical imaging market. The system features a Japan-made magnet and fully in-house components.
-
In January 2024, DermaSensor Inc. received FDA clearance for its real-time, non-invasive skin cancer evaluation system, enabling U.S. primary care physicians to provide point-of-care testing for all skin cancer types. This innovation aims to improve early detection, speed up patient care, and address the growing U.S. skin cancer burden, which affects 1 in 5 Americans.
U.S. Non-Invasive Cancer Diagnostics Market Report Scope
Report Attribute
Details
Market size value in 2026
USD 28.79 billion
Revenue forecast in 2033
USD 47.52 billion
Growth rate
CAGR of 7.4% from 2026 to 2033
Base year for estimation
2025
Historical data
2021 - 2024
Forecast period
2026 - 2033
Quantitative units
Revenue in USD million/billion and CAGR from 2026 to 2033
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, trends
Segments covered
Product, technology, indication, and end user
Key companies profiled
Abbott Laboratories; GE HealthCare; Siemens Healthineers AG; Koninklijke Philips N.V.; Canon Medical Systems; F. Hoffmann-La Roche Ltd; Illumina, Inc.; Guardant Health, Inc.; Thermo Fisher Scientific Inc.; FONAR Corporation
Customization scope
Free report customization (equivalent up to 8 analysts working days) with purchase. Addition or alteration to segment scope.
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
U.S. Non-invasive Cancer Diagnostics Market Report Segmentation
This report forecasts revenue growth and provides an analysis of the latest trends in each of the sub-segments from 2021 to 2033. For this study, Grand View Research has segmented the U.S. non-invasive cancer diagnostics market report based on product, technology, indication, and end user:
-
Product Outlook (Revenue, USD Million, 2021 - 2033)
-
Instruments
-
Pathology-based Instruments
-
Polymerase Chain Reaction (PCR) Instruments
-
Next-generation Sequencing (NGS) Instruments
-
Microarrays
-
-
Imaging Instruments
-
Magnetic Resonance Imaging (MRI)
-
Computed Tomography (CT)
-
Positron Emission Tomography (PET)
-
Mammography
-
Others (Ultrasound, etc.)
-
-
-
Consumables
-
Kits & Reagents
-
-
Technology Outlook (Revenue, USD Million, 2021 - 2033)
-
Imaging
-
Liquid Biopsy
-
Immunoassay
-
-
Indication Outlook (Revenue, USD Million, 2021 - 2033)
-
Lung Cancer
-
Breast Cancer
-
Colorectal Cancer
-
Prostate Cancer
-
Blood Cancer
-
Liver Cancer
-
Cervical Cancer
-
Skin Cancer
-
Others (Thyroid Cancer, etc.)
-
-
End User Outlook (Revenue, USD Million, 2021 - 2033)
-
Hospitals
-
Diagnostic Laboratories
-
Others (Academic & Research Institutes, etc.)
-
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.









