- Home
- »
- Pharmaceuticals
- »
-
U.S. Osteoarthritis Therapeutics Market, Industry Report 2033GVR Report cover
![U.S. Osteoarthritis Therapeutics Market Size, Share & Trends Report]()
U.S. Osteoarthritis Therapeutics Market (2025 - 2033) Size, Share & Trends Analysis Report By Product (Viscosupplements, Platelet-Rich Plasma Therapy, Adipose Tissue Therapy, Bone Graft Substitutes), By Application, By Primary User, And Segment Forecasts
- Report ID: GVR-4-68040-817-6
- Number of Report Pages: 190
- Format: PDF
- Historical Range: 2021 - 2023
- Forecast Period: 2025 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
U.S. Osteoarthritis Therapeutics Market Summary
The U.S. osteoarthritis therapeutics market size was estimated at USD 2.15 billion in 2024 and is projected to reach USD 4.50 billion by 2033, growing at a CAGR of 8.61% from 2025 to 2033. Growing geriatric population, rising prevalence of obesity and joint injuries, and increasing demand for minimally invasive and regenerative therapies are driving the osteoarthritis therapeutics industry.
Key Market Trends & Insights
- On the basis of product, the viscosupplements segment dominated the market with a share of 48.85% in 2024.
- On the basis of application, the knee osteoarthritis segment held the majority share of 59.82% in 2024.
- On the basis of primary user, the orthopedic surgeons segment held the majority share of 37.67% in 2024.
Market Size & Forecast
- 2024 Market Size: USD 2.15 Billion
- 2033 Projected Market Size: USD 4.50 Billion
- CAGR (2025-2033): 8.61%
The aging population is a major factor driving the growth of the market. According to the Centers for Disease Control and Prevention (CDC), in February 2024, the age-adjusted prevalence of arthritis among U.S. adults aged 18 and older was 18.9%, with the prevalence in adults aged 75 and older reaching 53.9%. As the U.S. population ages, particularly with the elderly demographic projected to represent nearly 20% of the population by 2030, the demand for osteoarthritis treatments tailored to the older age group is increasing. In September 2024, the CDC reported that 40.3% of U.S. adults were classified as obese between August 2021 and August 2023. Obesity is a significant risk factor for osteoarthritis, especially in weight-bearing joints, and its high prevalence is contributing to the rising number of osteoarthritis cases.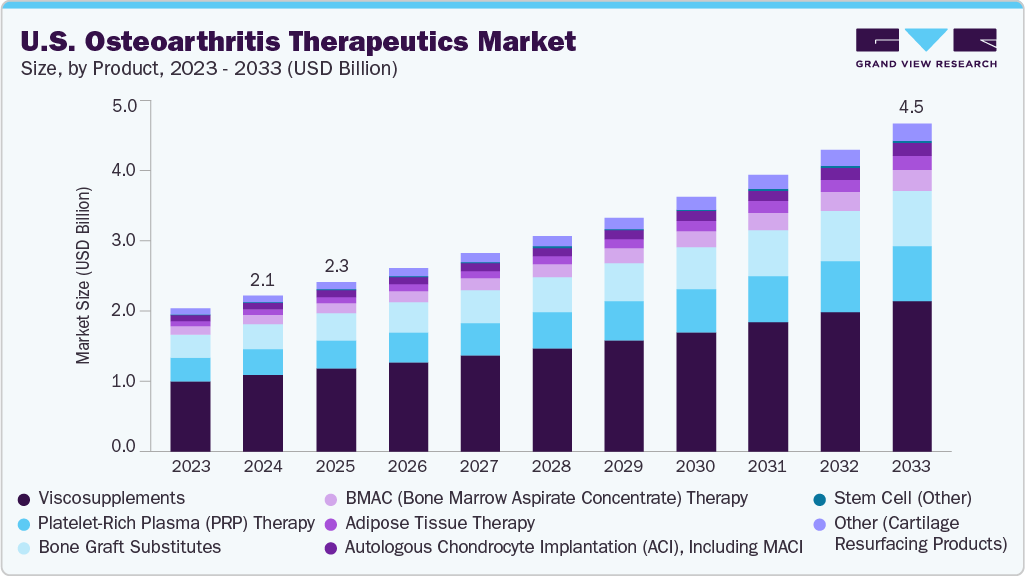
Additionally, the increase in joint injuries, particularly those related to sports and work-related accidents, is accelerating the onset of osteoarthritis. As more individuals engage in physical activities, the likelihood of joint damage and subsequent osteoarthritis increases. In February 2024, the International Labour Organization (ILO) estimated that approximately 395 million workers worldwide sustain non-fatal work injuries annually, many of which result in joint damage. This has increased awareness of the need for joint health management and has contributed to the rising demand for osteoarthritis therapeutics in the U.S.
Finally, the shift toward minimally invasive and regenerative therapies is playing a significant role in shaping the U.S. osteoarthritis therapeutics market. There is growing interest in non-surgical treatments, such as hyaluronic acid injections, platelet-rich plasma (PRP) therapy, and stem cell therapies, as they offer effective alternatives with shorter recovery times and fewer side effects compared to traditional surgical options. This shift in treatment preferences is expected to continue fueling market growth in the coming years.
The industry is driven by the aging population, rising obesity rates, increasing joint injuries, and a growing preference for minimally invasive and regenerative therapies. These factors are expected to support continued market growth, with advancements in treatment technologies playing a key role in meeting the demands of an aging population.
Market Concentration & Characteristics
The industry is evolving with notable innovation, particularly in disease‑modifying osteoarthritis drugs (DMOADs), biologics, and regenerative therapies. For example, the multicenter MILES Study compared mesenchymal stem cell (MSC) injections with corticosteroid injections for knee osteoarthritis across multiple U.S. sites. Additionally, biologics such as monoclonal antibodies targeting specific disease pathways are under investigation in OA. These innovations are still largely in the clinical‑trial or early‑stage phase, and long‑term efficacy and safety remain to be established. As a result, the U.S. OA therapeutics market is positioned in a high‐risk, high‐reward segment of clinical development.
Entering the U.S. OA therapeutics industry presents significant challenges due to high development costs, extended clinical trial durations, and complex regulatory processes. For new treatments, particularly those involving innovative biologics or regenerative therapies, navigating the FDA approval process can take years, requiring substantial investment in clinical trials to prove safety and efficacy. Beyond regulatory hurdles, market access is hindered by the need to build distribution networks and engage in negotiations with insurers to secure reimbursement coverage. Additionally, competition from existing players, including the increasing availability of biosimilars and generic treatments, intensifies pressure on new entrants. These products typically offer lower prices, making it difficult for new treatments to gain a foothold unless they present a significant clinical advantage or unique therapeutic benefit. With the market already populated by established therapies, new players must find ways to distinguish themselves through innovation or substantial improvements in treatment outcomes to successfully penetrate the U.S. market.
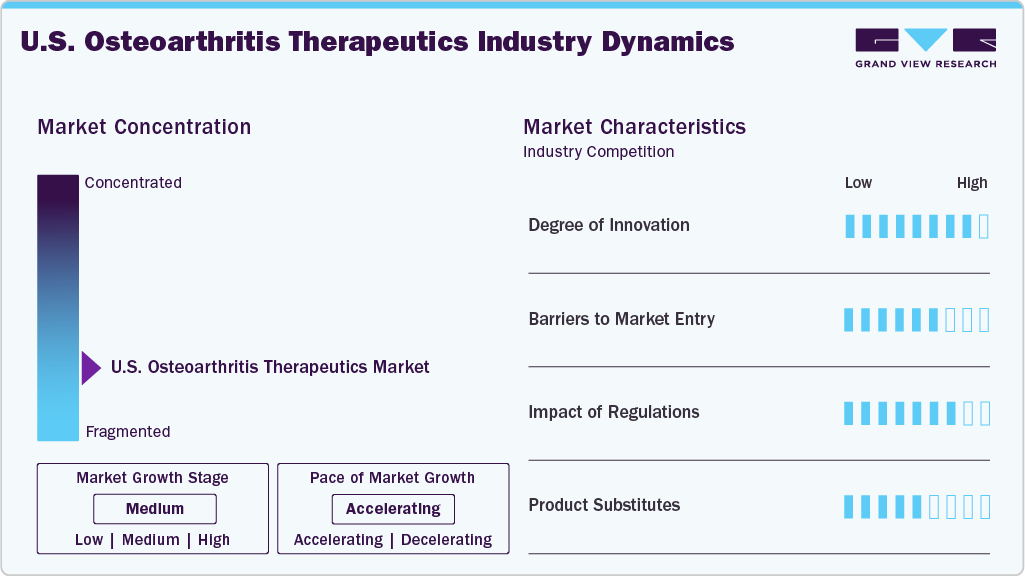
Regulations play a crucial role in shaping the OA treatment market. The FDA, EMA, and other regulatory bodies require extensive clinical trial data to approve new therapies, especially biologics and regenerative treatments. For example, Pfizer’s tanezumab faced rejection by the FDA in March 2021 due to concerns over its safety profile. Moreover, reimbursement agencies such as NICE in the UK have delayed coverage of some innovative OA treatments due to cost concerns. These regulations ensure safety but also slow the introduction of new therapies, increasing the development costs and limiting accessibility.
Regulations significantly influence the development and market entry of new osteoarthritis (OA) treatments in the U.S. The FDA and other regulatory bodies require comprehensive clinical trial data to approve new therapies, particularly biologics and regenerative treatments. For example, Pfizer's tanezumab, a monoclonal antibody aimed at treating OA, faced FDA rejection in March 2021 due to concerns over its safety profile, highlighting the stringent approval process for novel therapies. In addition to safety evaluations, regulatory requirements can lead to delays in market access, as seen with treatments facing reimbursement challenges. For instance, some innovative OA therapies have faced delays in gaining coverage from U.S. insurance providers, who scrutinize the cost-effectiveness of new treatments. These regulatory hurdles, while ensuring safety and efficacy, can slow the introduction of new therapies, increase development costs, and limit timely accessibility for patients. The need to meet extensive clinical data requirements can significantly extend the time to market, making it more difficult for companies to capitalize on emerging opportunities in the OA therapeutics space.
Product Insights
The viscosupplements segment dominated the market with the largest revenue share of 48.85% in 2024, driven by the use of hyaluronic acid (HA) injections in knee OA. In April 2025, the American Academy of Orthopaedic Surgeons (AAOS) consumer‑education website described viscosupplementation as HA injected into the knee joint acting as a lubricant and shock absorber. In September 2022, a systematic review and meta‑analysis published in The BMJ concluded that while HA injections for knee OA provide modest pain relief and functional improvement, the benefits are variable and the broad use remains debated. These findings reinforce the segment’s significance in the U.S. market, as clinicians and patients weigh HA products as non‑surgical options in mild to moderate knee OA to delay invasive interventions.
The Other (Cartilage Resurfacing Products) segment is projected to grow at the highest CAGR of 10.57% over the forecast period, focusing on younger or active patients with localized cartilage defects who are not yet candidates for total joint replacement. In March 2024, a report from UC Davis Health described the first U.S. clinical use of the Agili‑C implant (a resorbable device for knee cartilage repair), positioning it as a cartilage‑preserving alternative in U.S. practice. UC Davis Health Further, in 2023, a review in The Knee Journal reported on focal resurfacing implants, including systems marketed in the U.S., such as HemiCAP and BioPoly, with short‑term clinical outcomes supporting their use in delayed progression of OA. These validated instances indicate growing acceptance of cartilage resurfacing technologies in the U.S. market and suggest this segment may capture an increasing share by addressing treatment gaps before full arthroplasty is required.
Application Insights
The knee osteoarthritis (OA) segment dominated the market with the largest revenue share of 59.82% in 2024, driven by the high prevalence of the condition, particularly among aging individuals. According to CDC data from February 2024, approximately 33% of adults aged 60 and older in the U.S. are affected by knee OA, making it the most common joint disorder. This high prevalence is significantly influenced by factors such as obesity and sedentary lifestyles, which are contributing to the rise in knee OA cases. Treatments for knee OA vary from conservative approaches like physical therapy to more advanced options such as hyaluronic acid (HA) injections, which are widely used in the U.S. to alleviate pain and improve joint mobility in patients with mild to moderate OA. HA injections, including products like Synvisc and Monovisc, are increasingly popular as alternatives to nonsteroidal anti-inflammatory drugs (NSAIDs) and corticosteroids.
The ankle & foot osteoarthritis segment is projected to grow at the fastest CAGR over the forecast period, driven by increasing adoption of less invasive treatments for localized joint damage. Ankle distraction arthroplasty, a procedure that applies mechanical force to stimulate cartilage regeneration, has shown promising results in U.S. patients. A 2024 study published in Foot and Ankle International demonstrated significant improvements in pain relief and ankle function in patients undergoing ankle distraction arthroplasty using an Ilizarov external fixator. This procedure is gaining recognition as a joint-preserving alternative for young patients with moderate-to-severe ankle OA, delaying the need for total ankle replacement. Additionally, stem cell therapy is emerging as an innovative treatment option for ankle OA in the U.S., especially for younger, active individuals who wish to avoid major surgery. Foot OA, which affects 16.7% of U.S. adults aged 50 and older, is becoming more prominent, further increasing the demand for specialized therapies in this segment. Advances in biologic treatments, diagnostics, and patient-specific care plans are expected to continue fueling the growth of this segment within the broader U.S. market.
Primary User Insights
The orthopedic surgeons segment dominated the market with the largest revenue share of 37.67% in 2024, owing to significant adoption of advanced cartilage‑restoration devices. A landmark study published in May 2025 by Smith+Nephew revealed that patients treated with the AGILI‑C implant experienced an 87% lower relative risk of conversion to total knee arthroplasty (TKA) or osteotomy at four years compared with standard surgical care. Earlier, U.S. patient enrollment for the AGILI‑C IDE trial began with the first two cases in August 2018 at LSU Health New Orleans. These developments have positioned orthopedic surgeons as key prescribers and users of the device, which is integrated into operative workflows and patient‑education practices in U.S. orthopedic clinics.
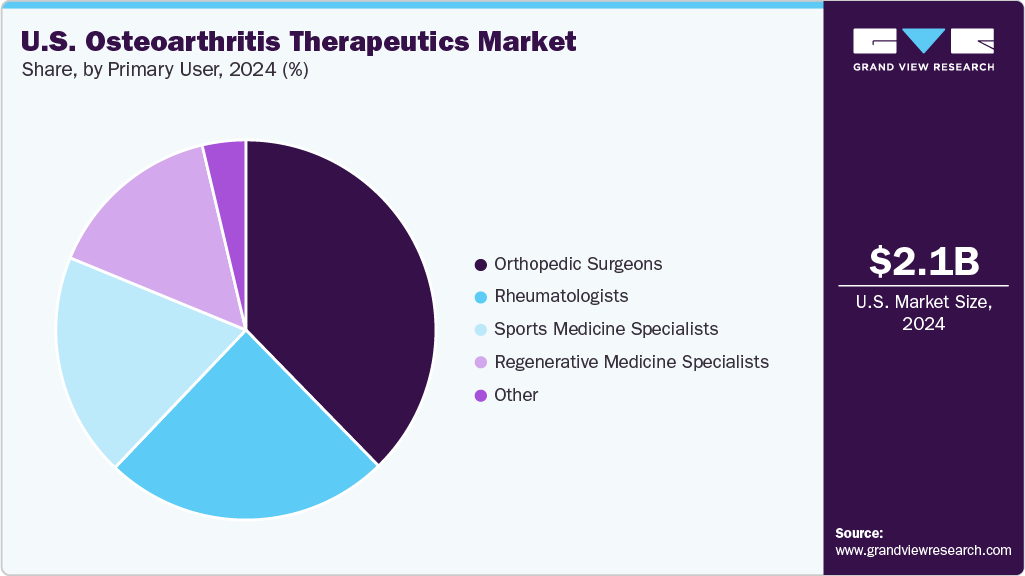
The regenerative medicine specialists segment is projected to grow at the fastest CAGR over the forecast period, driven by increasing uptake of biologic therapies in outpatient settings. A U.S.‑based retrospective study published in 2024 of leukocyte‑poor PRP for knee osteoarthritis showed favorable one‑year outcomes in pain and function. Additionally, a meta‑analysis published in March 2025 comparing LP‑PRP to hyaluronic acid injections confirmed superior WOMAC and VAS scores for LP‑PRP in knee OA management. Regenerative‑medicine specialists-delivering PRP, BMAC, adipose‑derived stem cells and exosome therapies-are increasingly operating in outpatient networks, thereby extending access outside the surgical channel and driving this segment’s accelerated growth.
Key U.S. Osteoarthritis Therapeutics Companies Insights
Key players operating in the U.S. osteoarthritis therapeutics market are undertaking various initiatives to strengthen their presence and increase the reach of their products and services. Strategies such as expansion activities and partnerships are key in propelling the market growth.
Key U.S. Osteoarthritis Therapeutics Companies:
- Stryker
- Zimmer Biomet Holdings, Inc.
- Medtronic
- Anika Therapeutics, Inc.
- Bioventus
- Vericel Corporation
- Smith & Nephew
- Fidia Farmaceutici S.p.A.
- SEIKAGAKU CORPORATION
- Sanofi
Recent Developments
-
In Jan 2024, Smith+Nephew announced completion of its acquisition of CartiHeal (developer of the AGILI‑C implant) for USD 180 million upfront (up to USD 330 million total) to expand its U.S. cartilage‑repair portfolio.
-
In Feb 2025, Doron Therapeutics announced that its lead intra‑articular biologic candidate MOTYS for knee OA had received RMAT (Regenerative Medicine Advanced Therapy) designation from the U.S. Food & Drug Administration (FDA), enabling an expedited review pathway in the U.S. market.
-
In Jul 2025, Genascence Corporation announced that its gene therapy candidate GNSC‑001 for knee OA received RMAT designation from the FDA.
U.S. Osteoarthritis Therapeutics Market Report Scope
Report Attribute
Details
Market size value in 2025
USD 2.32 billion
Revenue forecast in 2033
USD 4.50 billion
Growth rate
CAGR of 8.61% from 2025 to 2033
Base year for estimation
2024
Historical data
2021 - 2023
Forecast period
2025 - 2033
Quantitative units
Revenue in USD million/billion and CAGR from 2025 to 2033
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, trends
Segments covered
Product, application, primary user
Country scope
U.S.
Key company profiled
Stryker; Zimmer Biomet Holdings, Inc.; Medtronic; Anika Therapeutics, Inc.; Bioventus; Vericel Corporation; Smith & Nephew; Fidia Farmaceutici S.p.A.; SEIKAGAKU CORPORATION; Sanofi
Customization scope
Free report customization (equivalent up to 8 analysts working days) with purchase. Addition or alteration to country, regional & segment scope.
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
U.S. Osteoarthritis Therapeutics Market Report Segmentation
This report forecasts revenue growth at the country level and provides an analysis of the latest industry trends in each of the sub-segments from 2021 to 2033. For this study, Grand View Research has segmented the U.S. osteoarthritis therapeutics market report based on product, application, and primary user:
-
Product Outlook (Revenue, USD Million, 2021 - 2033)
-
Viscosupplements
-
Platelet-Rich Plasma (PRP) Therapy
-
Bone Graft Substitutes
-
BMAC (Bone Marrow Aspirate Concentrate) Therapy
-
Adipose Tissue Therapy
-
Autologous Chondrocyte Implantation (ACI), Including MACI
-
Stem Cell (other)
-
Other (Cartilage Resurfacing Products)
-
-
Application Outlook (Revenue, USD Million, 2021 - 2033)
-
Knee Osteoarthritis
-
Hip Osteoarthritis
-
Shoulder Osteoarthritis
-
Spine Osteoarthritis
-
Ankle & Foot Osteoarthritis
-
Other
-
-
Primary User Outlook (Revenue, USD Million, 2021 - 2033)
-
Orthopedic Surgeons
-
Rheumatologists
-
Sports Medicine Specialists
-
Regenerative Medicine Specialists
- Other
-
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










