- Home
- »
- Pharmaceuticals
- »
-
U.S. Pancreatic Cancer Treatment Market Size Report, 2030GVR Report cover
![U.S. Pancreatic Cancer Treatment Market Size, Share & Trends Report]()
U.S. Pancreatic Cancer Treatment Market (2025 - 2030) Size, Share & Trends Analysis Report By Type (Exocrine, Endocrine), By Treatment (Chemotherapy, Radiation Therapy, Others), By Distribution Channel, And Segment Forecasts
- Report ID: GVR-4-68040-499-6
- Number of Report Pages: 130
- Format: PDF
- Historical Range: 2018 - 2024
- Forecast Period: 2025 - 2030
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
Market Size & Trends
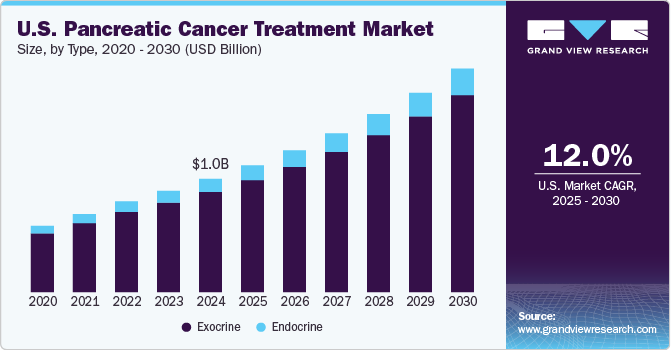
The U.S. pancreatic cancer treatment market size was estimated USD 1.02 billion in 2024 and is projected to grow at a CAGR of 12.02% from 2025 to 2030. The growth of the market is primarily driven by the rising incidence of pancreatic cancer, advancements in treatment modalities, and increasing awareness about early detection and treatment options. According to the American Cancer Society, in 2023, approximately 64,050 new cases of pancreatic cancer were reported in the U.S., with an estimated 50,550 deaths attributed to the disease. The grim prognosis and aggressive nature of pancreatic cancer are spurring demand for innovative treatment options, contributing to the market's growth.
Technological advancements and the development of novel therapeutics, including immunotherapy and targeted treatments, are significantly impacting the market. For instance, the adoption of nanotechnology-based drug delivery systems has improved treatment efficacy and reduced side effects. Furthermore, ongoing clinical trials for combination therapies, involving chemotherapy, radiation, and immunotherapy, are showing promising results, driving the demand for advanced pancreatic cancer treatment methods in the U.S.
The increasing incidence of pancreatic cancer in the U.S. is a major factor propelling market growth. Risk factors such as smoking, obesity, diabetes, and genetic predisposition are contributing to the rising cases. According to a study published by the National Cancer Institute in 2023, pancreatic cancer accounts for about 3% of all cancers in the U.S. and about 7% of all cancer deaths.
Additionally, the growing geriatric population is a significant driver, as the risk of developing pancreatic cancer increases with age. The U.S. Census Bureau projects that by 2030, nearly 20% of the U.S. population will be aged 65 and older, further fueling the demand for effective pancreatic cancer treatments. Government initiatives such as funding for cancer research, and partnerships between private and public sectors, are playing a critical role in market growth. For instance, the National Cancer Institute’s Pancreatic Cancer Detection Consortium (PCDC) supports research focused on early detection and treatment.
Innovations in treatment modalities are transforming the pancreatic cancer treatment landscape. Targeted therapies and immunotherapies, such as immune checkpoint inhibitors and CAR-T cell therapies, are gaining traction due to their ability to enhance survival rates with fewer side effects compared to conventional chemotherapy.
Radiation therapy has also seen advancements, with technologies like stereotactic body radiotherapy (SBRT) providing precise targeting of cancer cells while sparing healthy tissues. Additionally, liquid biopsies for early pancreatic cancer detection and monitoring have emerged as a promising diagnostic tool, enabling personalized treatment strategies. In June 2023, a major breakthrough was achieved when a leading biotech company announced FDA approval for its novel KRAS inhibitor, which has shown effectiveness in treating pancreatic tumors harboring specific genetic mutations.
Market Concentration & Characteristics
The U.S. pancreatic cancer treatment market is characterized by high innovation levels, driven by advancements in genomics, proteomics, and nanotechnology. Emerging therapies are being designed to address the unique challenges of pancreatic cancer, such as its resistance to conventional therapies and difficulty in early diagnosis. The development of combination therapies and targeted approaches is accelerating.
Mergers and acquisitions are shaping the competitive landscape, with major pharmaceutical companies acquiring smaller biotech firms to expand their pipeline of pancreatic cancer treatments. Such strategic moves allow companies to enhance their market share and access cutting-edge technologies.
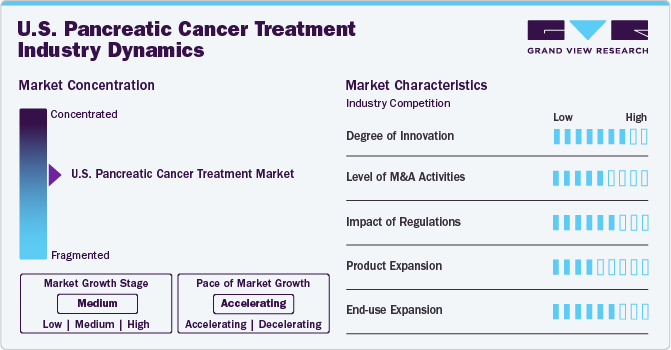
The U.S. Food and Drug Administration (FDA) plays a crucial role in ensuring the safety and efficacy of pancreatic cancer treatments. The regulatory process for approval of new drugs and therapies is stringent, involving rigorous clinical trials. However, initiatives such as the FDA’s Accelerated Approval Program are expediting the availability of innovative treatments for patients with advanced or metastatic pancreatic cancer.
Alternative treatment options, such as complementary therapies, dietary interventions, and palliative care, are often used alongside mainstream medical treatments. While these substitutes do not replace traditional therapies, they play a supportive role in improving patients' quality of life. Additionally, advancements in early screening technologies, such as imaging and biomarker-based diagnostics, are emerging as indirect substitutes by facilitating early detection and reducing reliance on aggressive treatments.
The application of pancreatic cancer treatments is expanding beyond oncology to other medical specialties such as palliative care and precision medicine. Precision medicine approaches, leveraging genetic and molecular profiling, are enabling the customization of treatments for individual patients, thereby improving outcomes. Hospitals, cancer centers, and research institutions are increasingly adopting these advanced therapies to enhance the standard of care for pancreatic cancer patients.
Type Insights
The exocrine segment dominated the U.S. pancreatic cancer treatment market, accounting for the largest revenue share of 88.39% in 2024. Exocrine tumors, particularly pancreatic adenocarcinomas, are the most common type of pancreatic cancers, comprising over 90% of all cases. The prevalence of exocrine pancreatic cancers is driving the demand for innovative therapies, including chemotherapy and targeted treatment options. The availability of advanced diagnostic techniques, coupled with increasing awareness about the disease, further propels growth in this segment.
The endocrine segment is anticipated to witness the fastest CAGR over the forecast period. Endocrine tumors, also known as pancreatic neuroendocrine tumors (pNETs), are less common but show unique treatment challenges and opportunities. Growing R&D efforts to develop therapies specifically targeting endocrine tumors, alongside advancements in precision medicine, are expected to contribute significantly to the growth of this segment. Increasing adoption of diagnostic imaging techniques, such as functional imaging, is also bolstering growth.
Treatment Insights
The chemotherapy segment dominated the U.S. pancreatic cancer treatment market in 2024 with a share of 43.25%, owing to its widespread use as the first-line treatment for both exocrine and endocrine pancreatic cancers. Chemotherapy regimens, such as FOLFIRINOX and gemcitabine-based therapies, are commonly prescribed due to their effectiveness in managing advanced cases. The high prevalence of pancreatic cancer and the lack of targeted therapies for many patients contribute to chemotherapy’s dominance in the market.
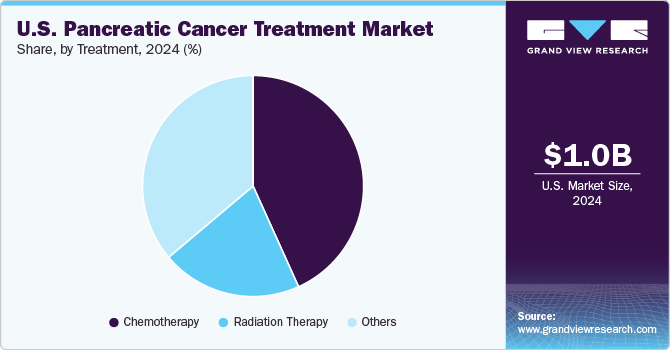
The radiation therapy segment is projected to grow significantly over the forecast period, driven by advancements in precision radiation techniques such as stereotactic body radiotherapy (SBRT). These technologies offer improved targeting of cancer cells while minimizing damage to surrounding healthy tissues. Radiation therapy is increasingly being used in combination with chemotherapy and surgical interventions to improve survival rates and patient outcomes.
Distribution Channel Insights
The hospital pharmacies segment held the largest market share of 46.01% in 2024. The dominance of this segment can be attributed to the high volume of inpatient and outpatient visits for pancreatic cancer diagnosis and treatment in hospitals. Hospitals are typically equipped with the infrastructure and skilled personnel required to administer complex therapies such as chemotherapy and radiation therapy. Additionally, the presence of specialized oncology departments in hospitals drives demand in this segment.
The retail pharmacies segment is the second-largest distribution channel and is expected to witness steady growth over the forecast period. Retail pharmacies play a vital role in providing oral chemotherapy drugs and supportive care medications, such as anti-nausea drugs and pain management therapies, to outpatients. Increasing accessibility and convenience for patients undergoing long-term treatment are key factors contributing to the growth of this segment.
Key U.S. Pancreatic Cancer Treatment Company Insights
Some of the leading players operating in the U.S. pancreatic cancer treatment market include Eli Lilly and Company,AstraZeneca, Novartis AG, Pfizer Inc., and Amgen Inc. These major industry participants leverage their extensive R&D capabilities and established customer networks to maintain significant market shares. Companies are heavily investing in developing targeted therapies and innovative treatment approaches to address the unmet needs in pancreatic cancer care.
Emerging players, such as Ipsen and Mirati Therapeutics, are making their presence felt by focusing on developing novel therapies and securing strategic collaborations to strengthen their market positioning. These companies emphasize gaining regulatory approvals for new treatments, which are crucial to capturing a share of the growing market.
Key U.S. Pancreatic Cancer Treatment Companies:
- Accuray Incorporated
- AstraZeneca
- Novartis AG
- Pfizer Inc
- Genentech, Inc (Roche Holding AG)
- Bristol-Myers Squibb Company
- Ipsen Pharma
- Eli Lilly and Company
- Elekta AB
- Siemens Healthineers AG (Varian Medical Systems, Inc., part of the company)
Recent Developments
-
In August 2024, the U.S. FDA had granted orphan drug designation to Exelixis, Inc.’s cabozantinib for treating pancreatic neuroendocrine tumors (pNET) and has set a Prescription Drug User Fee Act (PDUFA) target action date of April 3, 2025.
-
In February 2024, the U.S. FDA approved Ipsen’s irinotecan liposome (Onivyde) in addition to other drugs (oxaliplatin, fluorouracil, and leucovorin) for the first-line treatment of metastatic pancreatic adenocarcinoma.
-
In August 2024, myTomorrows, a global health technology company, partnered with Pancreatic Cancer Europe to support patients and caregivers in accessing and navigating clinical trials and find relevant treatment options.
U.S. Pancreatic Cancer Treatment Market Report Scope
Report Attribute
Details
Market size value in 2025
USD 1.14 billion
Revenue forecast in 2030
USD 2.01 billion
Growth rate
CAGR of 12.02% from 2025 to 2030
Historical data
2018 - 2024
Forecast period
2025 - 2030
Quantitative units
Revenue in USD million/billion and CAGR from 2025 to 2030
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Type, treatment, distribution channel
Country scope
U.S.
Key companies profiled
Accuray Incorporated; AstraZeneca; Novartis AG; Pfizer Inc; Genentech, Inc (Roche Holding AG); Bristol-Myers Squibb Company; Ipsen Pharma; Eli Lilly and Company; Elekta AB; Siemens Healthineers AG (Varian Medical Systems, Inc. part of the company)
Customization scope
Free report customization (equivalent up to 8 analysts working days) with purchase. Addition or alteration to country & segment scope.
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
U.S. Pancreatic Cancer Treatment Market Report Segmentation
This report forecasts revenue growth and provides an analysis of the latest industry trends in each of the sub-segments from 2018 to 2030. For this study, Grand View Research has segmented the U.S. pancreatic cancer treatment market report based on type, treatment, and distribution channel.
-
Type Outlook (Revenue, USD Million, 2018 - 2030)
-
Exocrine
-
Endocrine
-
-
Treatment Outlook (Revenue, USD Million, 2018 - 2030)
-
Chemotherapy
-
Radiation Therapy
-
Others
-
-
Distribution Channel Outlook (Revenue, USD Million, 2018 - 2030)
-
Hospital Pharmacies
-
Retail Pharmacies
-
Others
-
Frequently Asked Questions About This Report
b. The U.S. pancreatic cancer treatment market size was estimated at USD 1.02 billion in 2024 and is expected to reach USD 1.14 billion in 2025.
b. The U.S. pancreatic cancer treatment market is expected to grow at a compound annual growth rate of 12.02% from 2025 to 2030 to reach USD 2.01 billion by 2030.
b. Based on type, the Exocrine segment dominated the U.S. pancreatic cancer treatment market, accounting for the largest revenue share of 88.39% in 2024. Exocrine tumors, particularly pancreatic adenocarcinomas, are the most common type of pancreatic cancers, comprising over 90% of all cases. The prevalence of exocrine pancreatic cancers is driving the demand for innovative therapies, including chemotherapy and targeted treatment options.
b. Some of the key players in the U.S. pancreatic cancer treatment market are Accuray Incorporated, AstraZeneca, Novartis AG, Pfizer Inc, Genentech, Inc (Roche Holding AG), Bristol-Myers Squibb Company, Ipsen Pharma, Eli Lilly and Company, Elekta AB, Siemens Healthineers AG (Varian Medical Systems, Inc., part of the company)
b. The growth of the market is primarily driven by the rising incidence of pancreatic cancer, advancements in treatment modalities, and increasing awareness about early detection and treatment options. According to the American Cancer Society, in 2023, approximately 64,050 new cases of pancreatic cancer were reported in the U.S., with an estimated 50,550 deaths attributed to the disease.
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










