- Home
- »
- Medical Devices
- »
-
U.S. Respiratory Durable Medical Equipment Market Report 2033GVR Report cover
![U.S. Respiratory Durable Medical Equipment Market Size, Share & Trends Report]()
U.S. Respiratory Durable Medical Equipment Market (2026 - 2033) Size, Share & Trends Analysis Report By Product Offering (Sleep Apnea Devices, Oxygen Therapy, Nebulizer Equipment), By Distribution & Service Channel, By Notable States (Midwest Only), And Segment Forecasts
- Report ID: GVR-4-68040-845-6
- Number of Report Pages: 330
- Format: PDF
- Historical Range: 2021 - 2025
- Forecast Period: 2026 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
U.S. Respiratory Durable Medical Equipment Market Summary
The U.S. respiratory durable medical equipment (DME) market size was valued at USD 4.01 billion in 2025 and is projected to reach USD 5.89 billion by 2033, growing at a CAGR of 4.93% from 2026 to 2033. This growth is attributed to the technological advances smaller, more efficient portable oxygen concentrators, improved non-invasive home ventilators and connected PAP machines.
Key Market Trends & Insights
- By product offerings, the oxygen therapy segment held the largest market share of 25.6% in 2025.
- By distribution & service channel, the National DME Networks (Lincare, Apria/O&M, AdaptHealth, Rotech) held the largest revenue share in 2025.
Market Size & Forecast
- 2025 Market Size: USD 4.01 Billion
- 2033 Projected Market Size: USD 5.89 Billion
- CAGR (2026-2033): 4.93%
In addition, higher chronic respiratory disease prevalence (notably COPD and related comorbidities) in many Midwestern states together with an older, Medicare-heavy population and large rural patient cohorts who prefer home-based care, creates enduring demand for concentrators, portable oxygen systems and home ventilators. The Midwest has a comparatively higher prevalence of chronic respiratory conditions such as chronic obstructive pulmonary disease (COPD), asthma, and sleep-disordered breathing, largely due to an aging population and higher smoking prevalence in certain states.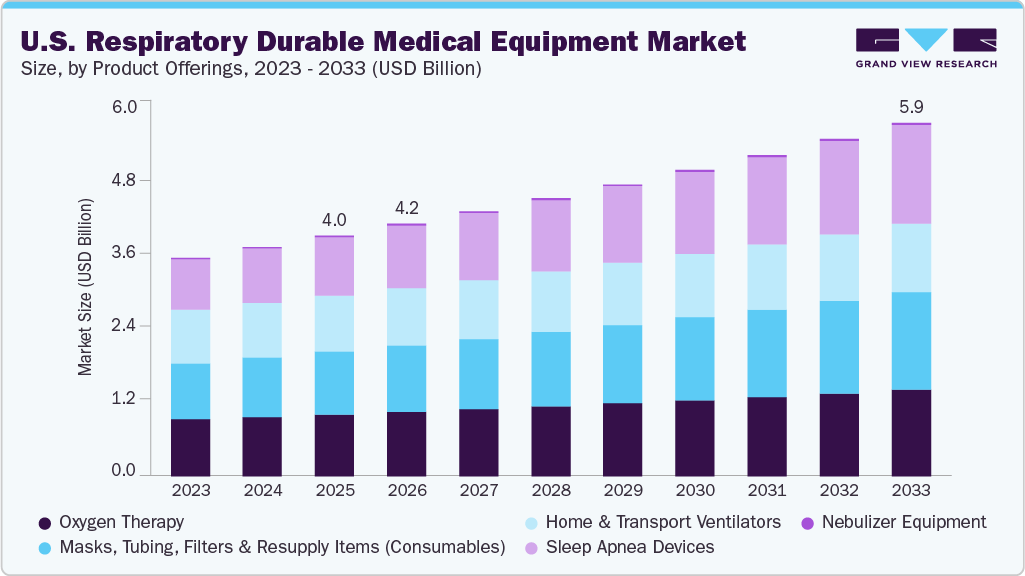
According to the American Lung Association, in 2023, approximately 11.1 million adults in the United States are living with COPD, which remains a leading cause of morbidity and mortality and is associated with an estimated USD 50 billion in annual healthcare costs. The increasing burden of COPD, combined with the need for non-invasive ventilation to manage hypercapnia, acute exacerbations, and sleep-disordered breathing in these patients, has fueled demand for home and hospital-based BiPAP/VPAP devices.
States such as Ohio, Michigan, Indiana, Illinois, and Missouri report a significant burden of COPD and obstructive sleep apnea (OSA), directly supporting demand for oxygen concentrators, CPAP/BiPAP devices, nebulizers, and ventilatory support equipment. The region’s aging demographic profile further accelerates adoption, as older adults are more likely to require long-term respiratory support and post-acute care equipment following hospital discharge.
Moreover, the strong emphasis on home-based care and cost containment across Midwestern healthcare systems, hospitals, and payers is actively shifting respiratory care from inpatient to home settings to reduce length of stay and readmission penalties, increasing reliance on DME providers for long-term oxygen therapy, sleep therapy devices, and remote patient monitoring-enabled respiratory equipment. This trend is fueled by Medicare and Medicaid reimbursement structures, which incentivize home oxygen and sleep therapy utilization, particularly in rural and semi-urban areas that characterize much of the Midwest. In addition, the Midwest has a relatively high proportion of Medicare beneficiaries, making federal reimbursement policies a critical demand driver for respiratory DME volumes.
For instance, Nebraska’s payer pattern reflects a mixed urban-rural respiratory profile, where PAP devices tilt toward Medicaid because sleep apnea incidence is higher in the adult, non-elderly population than in the 65+ group. Medicare still contributes substantially but remains secondary as its coverage primarily captures older COPD-dominant cohorts. However, oxygen concentrators, ventilators, and nebulizers lean strongly toward Medicare, consistent with Nebraska’s aging COPD and chronic respiratory population that relies more on federal senior coverage. Private plans track smaller shares due to lower employer-sponsored penetration in rural counties.
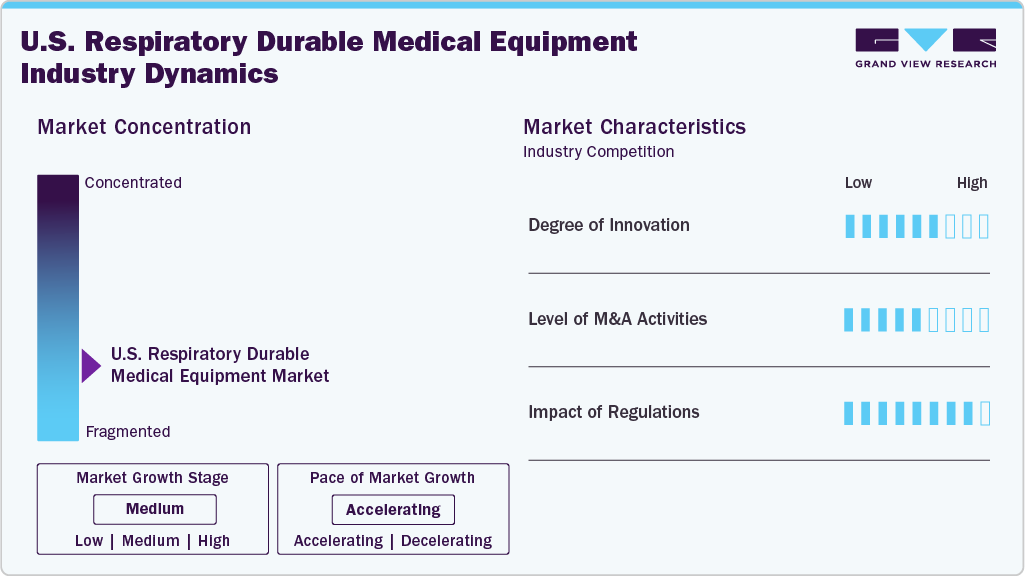
Market Concentration & Characteristics
The U.S. Respiratory Durable Medical Equipment (DME) market is characterized by moderate innovation, with companies developing advanced products to expand their offerings and stay competitive. For instance, in June 2025, Inogen launched the Voxi 5 stationary oxygen concentrator, delivering 1-5 L/min of continuous-flow oxygen in a compact, quiet, and durable design aimed at making long-term home oxygen therapy more affordable and accessible.
“With Voxi 5, we are fulfilling our mission to make respiratory care more accessible and empowering for patients, this launch exemplifies our commitment to innovation, affordability, and enhancing the quality of life for those dependent on oxygen therapy.”
- Kevin Smith, President and Chief Executive Officer.
The market players are using various strategies such as mergers and acquisitions, partnerships, collaborations, new product developments, and geographical expansion, which has significantly contributed to the level of M&A activities in the market. For instance, in July 2024 Owens & Minor, Inc. announced a definitive agreement to acquire Rotech Healthcare Holdings, Inc. for USD 1.36 billion in cash, aiming to expand its “Patient Direct” segment in home based care and broaden its respiratory, sleep apnea, diabetes and wound care offering.
“Rotech squarely fits into our existing Patient Direct segment and directly aligns with the strategy we outlined last December during our Investor Day, supporting our expansion in the very large and fast-growing home-based care space. We are excited to acquire a high-quality company like Rotech, an opportunity that doesn’t come along very often, and I look forward to welcoming the Rotech teammates into the Owens & Minor family”.
- Edward A. Pesicka, President & Chief Executive Officer of Owens & Minor.
Respiratory DME suppliers in Midwest must comply with comprehensive regulatory and licensing requirements. These include state-level licensing, facility and operational standards, alignment with federal FDA regulations, and specific obligations for home medical equipment, oxygen, and respiratory devices. Suppliers are also responsible for complaint handling, incident reporting, and biomedical service and maintenance, ensuring both regulatory compliance and patient safety.
Product Insights
The oxygen therapy devices segment accounted for the largest revenue share of 25.6%in 2025. The segment is further sub segmented by stationary oxygen concentrators, portable oxygen concentrators (POCs), cylinders and regulators, and conservers. The segment growth is driven by the devices such as stationary oxygen concentrators that play a crucial role in the oxygen therapy, with their growth driven by a growing shift towards home-based care to reduce hospitalization costs and enhance patient comfort.
Moreover, manufacturers are increasingly focusing on user-friendly designs and connected solutions. Features such as integrated alarms, remote monitoring capabilities, and compatibility with telehealth platforms enhance patient safety and allow healthcare providers to track therapy adherence in real time. For instance, in June 2025, Inogen has launched the Voxi 5 stationary oxygen concentrator, delivering 1-5 L/min of continuous-flow oxygen in a compact, quiet, and durable design aimed at making long-term home oxygen therapy more affordable and accessible.
With Voxi 5, we are fulfilling our mission to make respiratory care more accessible and empowering for patients. This launch exemplifies our commitment to innovation, affordability, and the enhancement of the quality of life for those dependent on oxygen therapy.
- Kevin Smith, President and Chief Executive Officer.
The sleep apnea devices segment is expected to grow at the fastest CAGR of 6.84% over the forecast period. This growth is driven by the adoption of Auto-titrating CPAP systems supported by the significant and growing prevalence of obstructive sleep apnea, which drives demand for effective and adaptive airway pressure therapy solutions. Recent technological advancements, including smart sensors, algorithm-driven pressure adjustments, and enhanced patient connectivity, are further shaping device offerings in the U.S. market.
Distribution & Service Channel Insights
The National DME Networks (Lincare, Apria/O&M, AdaptHealth, Rotech) segment held the largest revenue share in 2025. The growth of this channel is supported by the growing shift toward home-based respiratory care, demand for faster onboarding of sleep-therapy patients, and the need for scalable service models capable of managing large populations transitioning from acute settings. National DMEs also leverage digital tools such as remote compliance monitoring, automated resupply programs, and integrated patient-engagement apps to improve adherence and reduce administrative burden for providers.
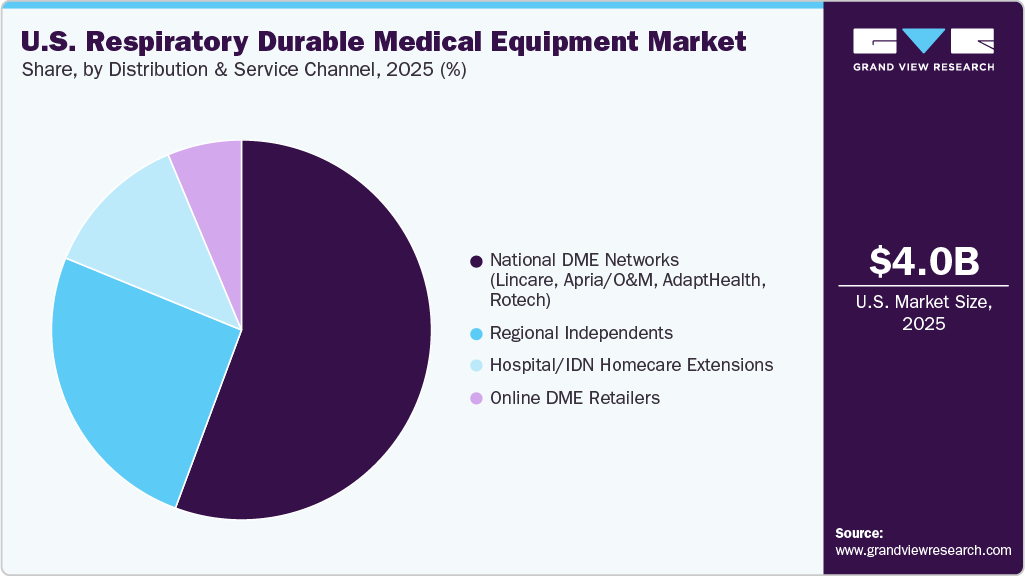
The online DME retailers’ segment is expected to grow at the fastest CAGR over the forecast period. The distribution and service channels in this Midwest market focus on balancing convenience and personal service, emphasizing 24/7 customer support, data-driven supply chain logistics, and outcomes-oriented respiratory care programs. This model enables better coordination with payers and health systems seeking to reduce total cost of care and improve patient experience amid diverse geographic challenges.
Key Midwest States Insights
Ohio (OH) in the U.S. held the largest revenue share of 17.5% in the U.S. DME market in 2025. Ohio’s respiratory DME market is driven by a high burden of respiratory diseases. The state consistently ranks among those with the highest COPD prevalence, especially in metropolitan areas like Cleveland and Cincinnati. This results in strong prescription volumes for long-term home oxygen therapy and related equipment. According to the American Lung Association (November 2023), 793,185 adults in Ohio had COPD, representing a prevalence of 8.6%, among the highest in the U.S. In 2023, approximately 11% of Ohioans had asthma. Moreover, county-level mapping from the Ohio Chronic Disease Atlas reveals dense pockets of chronic respiratory disease and hospitalizations across Appalachian and industrial counties, supporting the continued use of home oxygen, nebulizers, and airway-clearance devices to reduce readmissions.
Kansas (KS) is expected to grow significantly over the forecast period. According to the United Health Foundation, 9.9% of Kansas’s population has asthma, underscoring a substantial need for effective respiratory management. Along with high rates of tobacco use, obesity, and an aging population, these factors increase the risk of COPD and sleep-related breathing disorders, driving demand for home respiratory devices.
Key U.S. Respiratory Durable Medical Equipment Company Insights
Midwest respiratory DME companies benefit from dense hospital networks, aging populations, and strong Medicare utilization. Providers emphasize oxygen therapy, PAP devices, and home ventilation, competing through payer contracts, logistics scale, compliance expertise, and value-added patient support services across key states.
Key U.S. Respiratory Durable Medical Equipment Companies:
-
National DME Providers
-
Lincare Holdings Inc. (a subsidiary of Linde plc)
-
Apria Healthcare Group LLC (Owens & Minor)
-
AdaptHealth, LLC
-
Rotech Healthcare Inc.
-
-
OEM Manufacturers
-
ResMed
-
Koninklijke Philips N.V
-
Inogen
-
Drive Devilbiss International
-
Vyaire Medical
-
Breas Medical AB
-
-
Regional Independents
-
Home Oxygen Company
-
DME Supply USA
-
Kansas City Home Medical Supply
-
Integrated Respiratory Solutions
-
Recent Developments
-
In August 2025, AdaptHealth, LLC partnered with a large national healthcare system to become its exclusive provider of home medical equipment, covering over 10 million members across Medicare Advantage, Medicaid, and private insurance.
“We are excited to work with our new partner to help its members achieve their best health at home, we were able to demonstrate how our combination of talent, expertise, and tech-enabled patient experience aligned with the healthcare system’s innovative approach to serving its membership”
-Suzanne Foster. AdaptHealth CEO
-
In June 2025, Owens & Minor, Inc. and Rotech Healthcare Holdings Inc. mutually agreed to terminate their planned acquisition, with Owens & Minor paying an USD 80 million termination fee after regulatory clearance concerns made the deal unviable.
“For many months, our teammates, along with the Rotech team, have worked tirelessly in cooperation with the Federal Trade Commission to close this transaction, and while we believe there would have been ample benefits to patients, payors, and providers by adding Rotech to our Patient Direct business, the path to obtain regulatory clearance for this merger proved unviable in terms of time, expense, and opportunity.”
- Edward A. Pesicka, President & Chief Executive Officer of Owens & Minor.
U.S. Respiratory Durable Medical Equipment Market Report Scope
Report Attribute
Details
Market size value in 2026
USD 4.20 billion
Revenue forecast in 2033
USD 5.89 billion
Growth rate
CAGR of 4.93% from 2026 to 2033
Actual data
2021 - 2025
Forecast period
2026 - 2033
Quantitative units
Revenue in USD million/billion, and CAGR from 2026 to 2033
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Product offerings, distribution & service channel, Midwest States
Key companies profiled
National DME Providers: Lincare Holdings Inc. (a subsidiary of Linde plc); Apria Healthcare Group LLC (Owens & Minor); AdaptHealth, LLC; Rotech Healthcare Inc.; OEM Manufacturers: ResMed; Koninklijke Philips N.V; Inogen; Drive Devilbiss International; Vyaire Medical; Breas Medical AB; Regional Independents: Home Oxygen Company; DME Supply USA; Kansas City Home Medical Supply; Integrated Respiratory Solutions
By Notable States (Midwest Only)
Nebraska (NE); Iowa (IA); Kansas (KS); South Dakota (SD); North Dakota (ND); Missouri (MO); Minnesota (MN); Wisconsin (WI); Illinois (IL); Colorado (CO); Wyoming (WY); Ohio (OH); Indiana (IN)
Customization scope
Free report customization (equivalent up to 8 analysts working days) with purchase. Addition or alteration to country, regional & segment scope.
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
U.S. Respiratory Durable Medical Equipment Market Report Segmentation
This report forecasts revenue growth at country levels and provides an analysis of the latest industry trends in each of the sub-segments from 2021 to 2033. For this study, Grand View Research has segmented the U.S. respiratory durable medical equipment (DME) market report based on product offerings, distribution & service channel, and Midwest states:
-
Product Offerings Outlook (Revenue, USD Million, 2021 - 2033)
-
Sleep Apnea Devices
-
Auto-titrating CPAP (APAP)
-
Fixed CPAP
-
BiPAP / VPAP
-
Integrated Humidifiers
-
-
Oxygen Therapy
-
Stationary Oxygen Concentrators
-
Portable Oxygen Concentrators (POCs)
-
Cylinders & Regulators
-
Conservers
-
-
Nebulizer Equipment
-
Compressor Nebulizers
-
Mesh Nebulizers
-
Drug-specific nebulizers
-
-
Home & Transport Ventilators
-
Adult/pediatric long-term home ventilators
-
Portable Transport Ventilators
-
-
Masks, Tubing, Filters & Resupply Items (Consumables)
-
CPAP masks
-
CPAP cushions
-
Oxygen tubing & filters
-
Humidifier chambers
-
Bacterial/viral filters
-
Aerosol delivery accessories
-
-
-
Distribution & Service Channel Outlook (Revenue, USD Million, 2021 - 2033)
-
National DME Networks
-
Regional Independents
-
Hospital/IDN Homecare Extensions
-
Online DME Retailers
-
-
Notable States (Midwest Only) Outlook (Revenue, USD Million, 2021 - 2033)
-
Nebraska (NE)
-
Iowa (IA)
-
Kansas (KS)
-
South Dakota (SD)
-
North Dakota (ND)
-
Missouri (MO)
-
Minnesota (MN)
-
Wisconsin (WI)
-
Illinois (IL)
-
Colorado (CO)
-
Wyoming (WY)
-
Ohio (OH)
-
Indiana (IN)
-
Frequently Asked Questions About This Report
b. The global U.S. respiratory durable medical equipment market size was estimated at USD 4.01 billion in 2025 and is expected to reach USD 4.20 billion in 2026.
b. The global U.S. respiratory durable medical equipment market is expected to grow at a compound annual growth rate of 4.93% from 2026 to 2033 to reach USD 5.89 billion by 2033.
b. The oxygen therapy devices segment accounted for the largest revenue share of 25.6% in 2025. The segment growth is driven by the devices such as stationary oxygen concentrators that play a crucial role in the oxygen therapy, with their growth driven by a growing shift towards home-based care to reduce hospitalization costs and enhance patient comfort.
b. Some key players operating in the U.S. respiratory durable medical equipment market include National DME Providers: Lincare Holdings Inc. (a subsidiary of Linde plc), Apria Healthcare Group LLC (Owens & Minor), AdaptHealth, LLC, Rotech Healthcare Inc. OEM Manufacturers: ResMed, Koninklijke Philips N.V, Inogen, Drive Devilbiss International, Vyaire Medical, Breas Medical AB, Regional Independents: Home Oxygen CompanyDME Supply USA, Kansas City Home Medical Supply, Integrated Respiratory solutions
b. Key factors that are driving the market growth include technological advances smaller, more efficient portable oxygen concentrators, improved non-invasive home ventilators and connected PAP machines. In addition, higher chronic respiratory disease prevalence (notably COPD and related comorbidities) in many Midwestern states together with an older, Medicare-heavy population and large rural patient cohorts who prefer home-based care — creates enduring demand for concentrators, portable oxygen systems and home ventilators.
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










