- Home
- »
- Medical Devices
- »
-
U.S. Small Molecule Drug Discovery Outsourcing Market, 2033GVR Report cover
![U.S. Small Molecule Drug Discovery Outsourcing Market Size, Share & Trends Report]()
U.S. Small Molecule Drug Discovery Outsourcing Market (2025 - 2033) Size, Share & Trends Analysis Report By Workflow (Target Identification & Screening, Target Validation & Functional Informatics), By Service, By Therapeutics Area, By End Use, And Segment Forecasts
- Report ID: GVR-4-68040-687-1
- Number of Report Pages: 120
- Format: PDF
- Historical Range: 2021 - 2024
- Forecast Period: 2025 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
U.S. Small Molecule Drug Discovery Outsourcing Market Summary
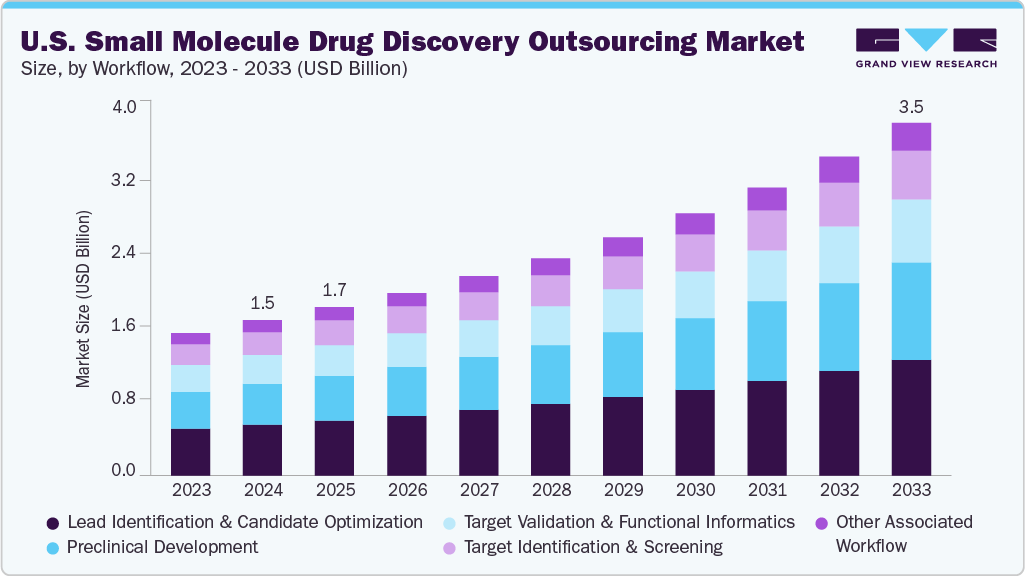
The U.S. small molecule drug discovery outsourcing market size was estimated at USD 1.52 billion in 2024 and is projected to reach USD 3.47 billion by 2033, growing at a CAGR of 9.7% from 2025 to 2033. Increasing R&D expenses, rising focus on early-stage research activities, and growing biotech companies with limited in-house resources drive the market.
Key Market Trends & Insights
- The U.S. small molecule drug discovery outsourcing in North America is expected to grow significantly over the forecast period.
- Based on workflow, the lead identification & candidate optimization segment held the highest market share of 32.68% in 2024.
- Based on service, the chemistry services segment held the highest market share in 2024.
Market Size & Forecast
- 2024 Market Size: USD 1.52 Billion
- 2033 Projected Market Size: USD 3.47 Billion
- CAGR (2025-2033): 9.7%
Besides, outsourcing has emerged as a strategic option for pharmaceutical and biotech companies aiming to accelerate the timelines, lower fixed overhead costs, and gain access to specialized expertise in areas such as medicinal chemistry, hit-to-lead optimization, ADME/PK studies, and IND-enabling activities.Innovations like AI-driven compound design, CRISPR-based target validation, high-throughput screening, and 3D cell models transform discovery processes, enabling contract research organizations (CROs) to provide more efficient and predictive results. Furthermore, rising R&D costs, increased demand for drug development, and a shift towards virtual pharmaceutical models support the market. In addition, an increase in registered clinical studies, a higher approval rate for small molecule drugs, advancements in medical technology, a rise in diseases being studied, and the urgent need for new treatments are anticipated to drive the market.
The landscape of small molecule drug development has evolved significantly in recent years. With an increased emphasis on molecularly targeted therapies, the formulations of small molecule active pharmaceutical ingredients (APIs) and drug products have become more complex and potent, necessitating specialized drug discovery, manufacturing processes & innovative drug delivery solutions. This has led to competitive demand for quicker entry into clinical development, coupled with review pathways leading to shortened timelines for clinical manufacturing.
Moreover, the growing adoption of integrated discovery platforms and innovative discovery partnerships, particularly in therapeutic areas such as oncology, CNS disorders, and rare diseases, is expected to drive market growth. Moreover, as the outsourcing landscape evolves, vendors offering flexible infrastructures, advanced technologies, and strong regulatory compliance are well-positioned to seize opportunities in small molecule drug discovery.
Moreover, the market is driven by growing interest in personalized medicine and a rising trend towards outsourcing drug discovery, development, and manufacturing to CDMOs, further supporting market growth. In addition, most companies are increasingly focusing on lower production costs and accelerating timelines from lab discovery to market, increasing the demand for small molecule drug discovery outsourcing services. Furthermore, expanding the generic drug market and favorable government support for CMOs and CDMOs have significantly increased collaborations between service providers and pharmaceutical companies.
Opportunity Analysis
The U.S. market for outsourcing small molecule drug discovery is experiencing growth driven by increasing complexity in R&D, rising pressure to manage costs, and an increased need for faster innovation. Biopharmaceutical companies, particularly smaller and virtual biotech companies, are increasingly utilizing outsourcing partners for tasks such as hit identification, lead optimization, and studies necessary for Investigational New Drug (IND) applications. Moreover, increased focus on precision medicine, rising research activities on rare diseases, and expanding oncology pipelines are expected to drive the demand for specialized CROs with specific expertise. Furthermore, technologies such as AI-based compound design, CRISPR for target validation, and 3D cell culture models are expected to provide CROs with new growth opportunities. In addition, the trend of reshoring, influenced by geopolitical uncertainties and tariff issues, benefits domestic CROs that offer comprehensive and localized services. Furthermore, long-term strategic partnerships are becoming more popular, fueling the continuous revenue and consistent service delivery. Thus, with the rising venture capital investment in early-stage drug development, the need for adaptable, technology-driven, and scientifically advanced outsourcing solutions is expected to drive the new market growth opportunities.
Impact of U.S. Tariffs on the U.S. Small Molecule Drug Discovery Outsourcing Market
U.S. tariffs have raised the costs of importing research chemicals, reagents, and laboratory equipment, particularly from China, impacting small molecule drug discovery outsourcing. These tariffs have put pressure on the profit margins of Contract Research Organizations (CROs) and have increased project expenses for sponsors, leading to a shift in sourcing strategies toward tariff-free regions such as India and Southeast Asia. While many smaller CROs face operational challenges, U.S.-based providers benefit from trends toward reshoring as sponsors prioritize domestic stability and regulatory compliance. In addition, the unpredictability of future tariff policies has encouraged the development of long-term, localized outsourcing partnerships. Thus, U.S. tariffs scenarios drive supply chain diversification, alter cost structures, and increase the demand for adaptable, U.S.-based discovery platforms that are prepared to handle geopolitical and economic fluctuations in early-stage pharmaceutical research.

Technological innovations are reshaping the landscape of small molecule drug discovery outsourcing in the U.S., facilitating faster and more precise R&D. Artificial intelligence and machine learning tools enhance target identification, virtual screening, and predictive toxicology, reducing reliance on trial-and-error methods. High-throughput screening (HTS) and automation improve the efficiency and consistency of screening extensive compound libraries. In addition, CRISPR and gene editing technologies enable accurate functional genomics and target validation, speeding up hit-to-lead processes.
Organoid and 3D cell culture models offer more physiologically relevant systems for screening and toxicity evaluations, resulting in better clinical translation. Cloud-based data integration and collaborative platforms allow for real-time sharing of complex discovery data, fostering seamless communication between sponsors and Contract Research Organizations (CROs). Thus, these advancements drive a move toward data-driven, modular outsourcing models prioritizing speed, scalability, and scientific rigor. CROs that embrace these innovations are positioned to gain a competitive edge by providing comprehensive solutions that enhance accuracy, decision-making support, and compliance with evolving regulatory standards.
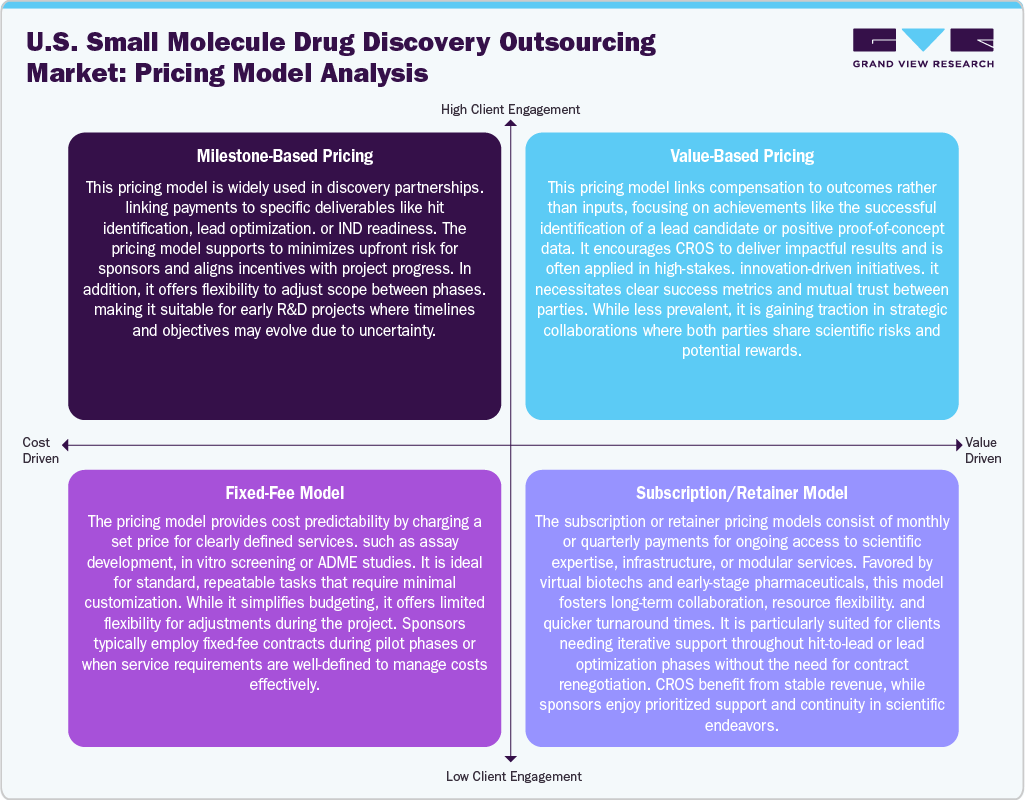
The U.S. market employs various pricing strategies designed to accommodate project complexity and client needs. Milestone-based pricing is widely used in the early discovery and preclinical stages, where payments are linked to specified outcomes such as target validation, lead optimization, or filing for an Investigational New Drug (IND). Fixed-fee arrangements are frequently used for routine services like assay development or in silico screening, providing clients with budget certainty, although they offer limited adaptability for changes in scope.
In addition, emerging trends indicate a growing interest in value-based pricing, especially in high-stakes partnerships where compensation is directly connected to results, such as the successful identification of clinical candidates or intellectual property creation, which is expected to support market growth. This approach promotes performance over quantity. Moreover, subscription or retainer models are becoming more popular with biotech and virtual pharmaceutical companies, enabling continuous access to scientific expertise, infrastructure, and modular services. This model fosters long-term relationships and facilitates better resource management. Furthermore, sponsors often utilize hybrid models, integrating fixed-fee or milestone-based pricing with advisory services on a retainer basis, allowing for cost optimization and quicker progression in early-stage development.
Market Concentration & Characteristics
The U.S. small molecule drug discovery outsourcing industry growth stage is moderate, and growth is accelerating. The market is characterized by the degree of innovation, level of M&A activities, regulatory impact, service expansion, and regional expansion.
AI and machine learning-enabled platforms, CRISPR-based target validation, and next-generation screening tools drive innovation. CROs incorporate predictive toxicology, organoids, and advanced analytics to enhance early discovery processes, minimize attrition, and accelerate timelines, particularly in oncology, neurology, and rare disease areas.

M&A activity is actively emerging due to rising CDMOs focused on acquiring niche discovery companies and AI-driven biotechnology companies. These activities aim to gain platform access, diversify pipelines, and improve early-stage chemistry capabilities. This strategic initiative is further transforming vendor ecosystems, particularly to provide comprehensive small molecule services from start to finish.
Regulations emphasize data integrity, early toxicology validation, and adherence to ICH and FDA guidelines for IND-enabling studies. Sponsors depend on CROs for audit readiness and proper documentation. Outsourcing partners must align with changing regulatory demands regarding traceability, reproducibility, and Good Manufacturing Practice (GMP) integration.
CROs and CDMOs are enhancing their upstream capabilities to incorporate target discovery, hit-to-lead optimization, and lead profiling. Besides, integrated discovery-to-IND models are gaining popularity, with advanced medicinal chemistry, ADME/PK, and early safety platforms being bundled together to accelerate small molecule programs seamlessly.
U.S.-based CROs are forming partnerships and setting up satellite labs in Canada, Europe, and Asia Pacific to facilitate cross-border discovery programs. The rise of nearshoring and dual-site models provides operational continuity, access to a diverse global talent pool, and quicker turnaround times for small molecule R&D services.
Workflow Insights
On the basis of workflow segment, in 2024, the lead identification & candidate optimization segment dominated the market, accounting for a revenue share of 32.68%. The segment growth is driven by rising demand for data-driven processes, higher R&D costs, growing requirement for faster timelines, the growing adoption of AI technologies, and the rising demand for high-quality preclinical candidates. Besides, most pharmaceutical and biopharmaceutical companies are shifting towards contract research organizations (CROs) to conduct high-throughput screening, structure-activity relationship (SAR) studies, and structure-based design through artificial intelligence and computational chemistry. Besides, the lead identification & candidate optimization support mitigating risks associated with early-stage assets, further propelling the outsourcing trend in the market. Moreover, growing focus on oncology and rare disease pipelines and a rising number of specialized partners with strong expertise in medicinal chemistry and in vitro pharmacology, essential for identifying, refining, and optimizing promising drug candidates, is expected to support the market growth.
On the other hand, the preclinical development segment is expected to grow at a CAGR of 10.37% over the forecast period. The preclinical development landscape in U.S. small molecule drug discovery outsourcing is driven by rising regulatory requirements for thorough safety and toxicology information and a growing demand for studies that support Investigational New Drug (IND) applications. Besides, sponsors are looking for CROs that can perform pharmacokinetic analyses, toxicology evaluations, and bioanalytical studies, utilizing Good Laboratory Practice (GLP) and non-GLP settings. With rising rates of drug pipelines, the precision in preclinical assessments is expected to drive the segment. Moreover, outsourcing offers access to scalable animal models, advanced equipment, and documentation that meets regulatory standards. Thus, the growing expansion of the IND process continues to drive collaborations with CROs that provide integrated workflows from discovery to preclinical phases and offer scientific advisory services. Such factors are expected to drive the market over an estimated period.
Service Insights
On the basis of service segment, the market is segmented into chemistry services and biology services. In 2024, the chemistry services segment dominated the market, attributed to increased focus on complex synthesis, scalable route development, and early formulation support. Besides, there is a growing demand for advanced medicinal chemistry, custom synthesis, and optimization of hit-to-lead compounds, including support for non-GMP and GMP-grade intermediates, which is expected to drive the segment growth over the estimated period. Moreover, high-potency compound handling, flow chemistry, and green chemistry techniques support market growth. Moreover, outsourcing enables biopharma companies to lower internal chemistry costs while gaining access to skilled chemists, diverse compound libraries, and flexible service offerings that aid early discovery and preclinical development, which is expected to drive the market growth over the estimated period.
On the other hand, the biology services segment is expected to grow significantly during the forecast period. Biology services in U.S. small molecule drug discovery outsourcing are increasingly focused on mechanistic validation, phenotypic screening, and disease-relevant models. Besides, most sponsors seek CROs that provide cell-based assays, target engagement studies, biomarker analysis, and functional genomics using CRISPR and RNAi. Moreover, using 3D cultures, organoids, and high-content imaging improves translational relevance. With the rise of personalized medicine, most CROs offering tailored biology platforms in oncology, CNS, and immunology are expected to witness rising growth over the estimated time period. Furthermore, outsourcing facilitates support for advanced validation, access to innovation, and cost-effective scaling of biological experiments.
Therapeutics Area Insights
On the basis of therapeutics area segment, the respiratory system segment accounted for the largest market share in 2024. This growth is driven by the increasing incidence of asthma, chronic obstructive pulmonary disease (COPD), and new post-viral lung conditions. Besides, growing demand for inhalable formulations, small molecule anti-inflammatories, and targeted therapies from sponsors supports the market. Furthermore, contract research organizations (CROs) offer a range of inhalation toxicology models, aerosol generation technologies, and preclinical lung imaging platforms, which are propelling the market growth over the estimated period. Moreover, outsourcing enhances the design and optimization of respiratory compounds, improving their bioavailability and selectivity for lung tissue. Furthermore, the regulatory focus on lung safety and the effectiveness of local delivery fuels the demand for specialized biological and pharmacokinetic evaluation services in respiratory drug discovery. Such factors are expected to drive the market over the estimated period.
The oncology segment is expected to grow significantly during the forecast period. Oncology continues to be the primary focus in outsourcing U.S. small molecule drug discovery, driven by a significant increase in targeted therapies, kinase inhibitors, and immuno-oncology treatments. Besides, most sponsors are shifting towards CROs for hit-to-lead optimization, biomarker identification, and efficacy evaluation in tailored cancer models. Moreover, growing demand for tumor-specific assays, 3D spheroid cultures, and patient-derived xenografts underscores the reliance on outsourcing for both biological & chemical processes, which is expected to witness rising growth over the estimated time period. Moreover, with the growing complexities of cancer biology and the necessity for accelerated timelines, collaborations with CROs are essential for reducing risks associated with oncology candidates and facilitating informed decision-making during the early stages of development. Thus, these factors support market growth.
End Use Insights
On the basis of the end use segment, the pharmaceutical & biotechnology companies segment accounted for the largest market share in 2024. Pharmaceutical and biotechnology companies are increasingly opting for outsourcing small molecule drug discovery in the U.S. to enhance cost efficiency, accelerate time-to-market, and tap into specialized scientific expertise. Besides, large pharmaceutical companies are increasingly adopting hybrid models, blending internal discovery efforts with external innovative solutions. Moreover, smaller and virtual biotech companies operating with limited resources, rely on outsourced end-to-end services ranging from hit identification to Investigational New Drug (IND) support are expected to support the market growth over the estimated period. Furthermore, other factors contributing to market growth are flexible pricing, quick scalability, and the incorporation of AI and automation. Moreover, collaborating with CROs enables sponsors to concentrate on their pipeline strategies and fundraising while utilizing external expertise in medicinal chemistry, biology, drug metabolism and pharmacokinetics (DMPK), and preclinical services that adhere to regulatory standards. Such factors are expected to support market growth.
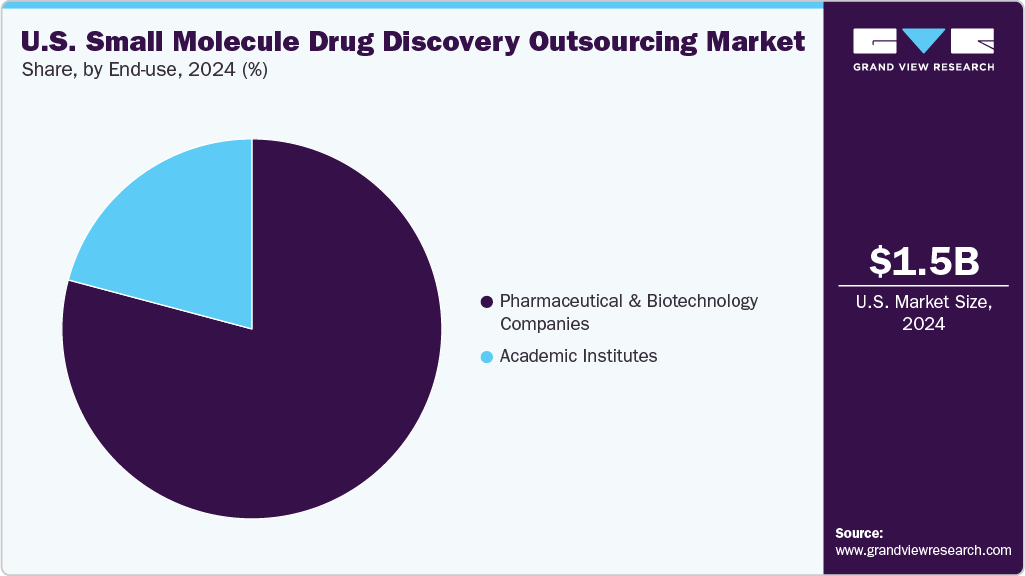
The academic institute’s segment is expected to grow significantly during the forecast period. Academic institutions are outsourcing small molecule drug discoveries to facilitate the translation of basic scientific research into potential therapeutic leads, all without the need to establish extensive internal infrastructure. Besides, with the rise in translational research funding, universities, and medical centers are turning to CROs for essential services such as compound screening, structure-activity relationship (SAR) studies, and early pharmacological validation. This outsourcing approach supports grant-funded initiatives, accelerates the achievement of proof-of-concept milestones, and encourages collaboration with biotechnology partners. Moreover, CROs are adept at navigating academic timelines and are open to intellectual property-sharing arrangements that support market growth.
Key U.S. Small Molecule Drug Discovery Outsourcing Company Insights
The key players operating across the market are adopting strategic initiatives such as service launches, mergers & acquisitions, partnerships & agreements, and expansions to gain a competitive edge. For instance, in June 2024, Veranova announced a significant expansion of its capabilities for developing and manufacturing antibody-drug conjugates (ADCs) and highly potent compounds at its facility in Massachusetts. This initiative involves approximately USD 30 million in investment focused on enhancing the site's current development and production capabilities to meet the increasing demand for U.S.-based capacity for ADCs and other potent small molecules.
Key U.S. Small Molecule Drug Discovery Outsourcing Companies:
- Charles River Laboratories
- Laboratory Corporation of America Holdings (Covance)
- Albany Molecular Research (Curia)
- Merck & Co., Inc.
- QIAGEN
- Eurofins
- Evotec
- GenScript Biotech
- Pharmaron
- Syngene International
- Dalton Pharma Services
- Oncodesign
- Jubilant Biosys
- Domainex
- Dr. Reddy Laboratories Ltd.
- Pharmaron Beijing Co., Ltd.
- TCG Lifesciences Pvt Ltd.
Recent Developments
-
In February 2025, Incyte and Genesis Therapeutics, Inc. formed a strategic partnership to research, discover, and develop innovative small-molecule therapies, initially concentrating on targets identified by Incyte. This collaboration will leverage the capabilities of the GEMS AI platform alongside Incyte’s extensive experience and success in drug discovery and development, with a mutual objective of advancing vital treatments for patients suffering from serious illnesses.
-
In April 2025, the Icahn School of Medicine, Mount Sinai, established the AI Small Molecule Drug Discovery Center to transform drug development through AI. This new center will combine AI techniques with conventional drug discovery processes to rapidly and accurately identify & design small molecule therapeutics.
U.S. Small Molecule Drug Discovery Outsourcing Market Report Scope
Report Attribute
Details
Market size value in 2025
USD 1.65 billion
Revenue forecast in 2033
USD 3.47 billion
Growth rate
CAGR of 9.7% from 2025 to 2033
Actual data
2021 - 2024
Forecast period
2025 - 2033
Quantitative units
Revenue in USD million/billion and CAGR from 2025 to 2033
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Workflow, service, therapeutics area, end use, country
Country scope
U.S.
Key companies profiled
Charles River Laboratories; Laboratory Corporation of America Holdings (Covance); Albany Molecular Research (Curia); Merck & Co., Inc.; QIAGEN; Eurofins; Evotec; GenScript Biotech; Pharmaron; Syngene International; Dalton Pharma Services; Oncodesign; Jubilant Biosys; Domainex; Dr. Reddy Laboratories Ltd.; Pharmaron Beijing Co., Ltd.; TCG Lifesciences Pvt Ltd.
Customization scope
Free report customization (equivalent up to 8 analysts working days) with purchase. Addition or alteration to country, & segment scope.
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
U.S. Small Molecule Drug Discovery Outsourcing Market Report Segmentation
This report forecasts revenue growth at the country level and provides an analysis of the latest industry trends in each of the sub-segments from 2021 to 2033. For this study, Grand View Research has segmented the U.S. small molecule drug discovery outsourcing market report based on workflow, service, therapeutics area, and end use:
-
Workflow Outlook (Revenue, USD Million, 2021 - 2033)
-
Target Identification & Screening
-
Target Validation & Functional Informatics
-
Lead Identification & Candidate Optimization
-
Preclinical Development
-
Others
-
-
Service Outlook (Revenue, USD Million, 2021 - 2033)
-
Chemistry Services
-
Biology Services
-
-
Therapeutics Area Outlook (Revenue, USD Million, 2021 - 2033)
-
Respiratory system
-
Pain and Anesthesia
-
Oncology
-
Ophthalmology
-
Hematology
-
Cardiovascular
-
Endocrine
-
Gastrointestinal
-
Immunomodulation
-
Anti-infective
-
Central Nervous System
-
Dermatology
-
Genitourinary System
-
-
End Use Outlook (Revenue, USD Million, 2021 - 2033)
-
Pharmaceutical & Biotechnology companies
-
Academic Institutes
-
Frequently Asked Questions About This Report
b. The U.S. small molecule drug discovery outsourcing market size was estimated at USD 1.52 billion in 2024 and is expected to reach USD 1.65 billion in 2025.
b. The U.S. small molecule drug discovery outsourcing market is expected to grow at a compound annual growth rate (CAGR) of 9.72% from 2025 to 2033 to reach USD 3.47 billion by 2033.
Which segment accounted for the largest U.S. small molecule drug discovery outsourcing market share?b. The lead identification & candidate optimization segment dominated the U.S. small molecule drug discovery outsourcing market in 2024, with a market share of 32.68%. Major factors boosting the demand for small molecule drug discovery outsourcing services include a rising demand for data-driven processes, higher R&D costs, growing requirement for faster timelines, the growing adoption of AI technologies, and the increasing demand for high-quality preclinical candidates, further fueling the segment's growth.
b. Some key players operating in the U.S. small molecule drug discovery outsourcing market include Charles River Laboratories, Laboratory Corporation of America Holdings (Covance), Albany Molecular Research (Curia), Merck & Co., Inc., QIAGEN, Eurofins, Evotec, GenScript Biotech, Pharmaron, Syngene International, Dalton Pharma Services, Oncodesign, Jubilant Biosys, Domainex, GenScript Biotech, Dr. Reddy Laboratories Ltd., Pharmaron Beijing Co., Ltd., and TCG Lifesciences Pvt Ltd., among others.
b. The increasing R&D expenses, rising focus on early-stage research activities, and growing number of biotech companies with limited in-house resources are key factors supporting the market growth. Besides, outsourcing has emerged as a strategic option for pharmaceutical and biotech companies aiming to accelerate timelines, lower fixed overhead costs, and gain access to specialized expertise in areas such as medicinal chemistry, hit-to-lead optimization, ADME/PK studies, and IND-enabling activities are expected to drive the market.
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










