- Home
- »
- Clinical Diagnostics
- »
-
U.S. Traumatic Brain Injury Biomarkers Market Report, 2033GVR Report cover
![U.S. Traumatic Brain Injury Biomarkers Market Size, Share & Trends Report]()
U.S. Traumatic Brain Injury Biomarkers Market (2025 - 2033) Size, Share & Trends Analysis Report By Type (Protein Biomarkers, Genetic Biomarkers, Metabolomic Biomarkers), By Sample Type, By Application, By End Use, And Segment Forecasts
- Report ID: GVR-4-68040-622-2
- Number of Report Pages: 150
- Format: PDF
- Historical Range: 2021 - 2024
- Forecast Period: 2025 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
Market Size & Trends
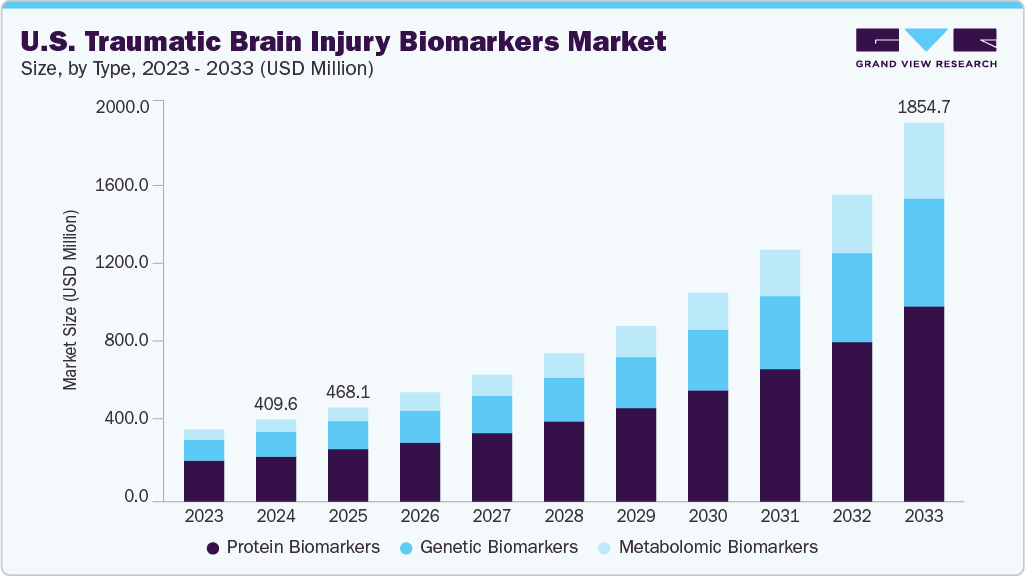
The U.S. traumatic brain injury biomarkers market size was estimated at USD 409.60 million in 2024, is projected to reach 1,854.73 million by 2033, and is forecasted to grow at a CAGR of 18.8% from 2025 to 2033. The U.S. traumatic brain injury (TBI) biomarkers market is expanding significantly due to increasing demand for pediatric TBI diagnosis, growing awareness and funding related to brain injury, and accelerating collaboration between academic institutions and industry.
TBI refers to an unintentional impact to the head that can lead to outcomes ranging from mild impairment to severe neurological damage or even death. According to a report by the Brain Injury Association America, 2025, the U.S. reported approximately 11.4 million individuals over the age of 40 who experienced a traumatic brain injury (TBI) resulting in loss of consciousness are currently living with a disability. In addition, around 2.9 million TBI-related emergency department visits are reported each year, indicating the significant healthcare burden associated with brain injuries.
Children and adolescents are particularly vulnerable to TBIs from falls, sports injuries, and accidents, yet symptoms can be subtle or delayed. Every year in the U.S., pediatric TBI leads to an estimated 500,000 emergency department visits. While children have a lower overall mortality rate, accounting for about 5% of all TBI-related deaths, the broader impact is more evident when considering years of life lost and years lived with disability. These long-term effects are notably greater in children than in adults, with the serious and lasting burden pediatric TBI poses on public health and quality of life. Pediatric care providers are increasingly turning to biomarker diagnostics to make safer and faster decisions, especially to reduce unnecessary radiation exposure from CT scans.
Blood-based biomarkers offer a non-invasive, objective method to assess injury risk and severity in young patients. A collaborative study conducted in June 2024, by renowned hospitals in Switzerland and Spain, discovered potential blood biomarkers that could significantly improve the diagnosis and management of mild traumatic brain injury (mTBI) in children. This multi-center research, involving ABCDx, the University of Geneva, and Vall d'Hebron Hospital, highlighted the potential of these blood biomarkers to accurately differentiate children with intracranial injuries (ICI) from those without. This reduced unnecessary CT scans and shorter hospital observation periods, improving efficiency in pediatric mTBI care.
Strong collaboration between research universities, biotech companies, and healthcare providers is advancing biomarker validation and commercialization. Institutions are working with biotech and diagnostics companies to translate laboratory discoveries into clinical tests. A new clinical trial holds significant promise for advancing the diagnosis and treatment of TBI. Focused on evaluating innovative diagnostic tools or therapeutic approaches, this trial could pave the way for more effective and timely interventions, potentially transforming patient outcomes in acute and long-term TBI management. These partnerships help accelerate clinical trials, FDA submissions, and market entry. In addition, shared data resources and biobanks support longitudinal studies that strengthen biomarker credibility. The U.S. ecosystem’s ability to foster innovation through public-private-academic collaboration is a major competitive advantage in expanding the TBI biomarker market.
A convergence of medical, social, and institutional factors drives rising funding for TBI biomarkers in the U.S. market. Growing public awareness of TBIs' long-term consequences, particularly in sports and military populations, has amplified demand for more accurate and timely diagnostic tools. Traditional imaging often falls short in detecting mild or early-stage injuries, creating a critical need for biomarker-based solutions to provide objective, rapid, and non-invasive assessments. In February 2024, government agencies such as the Congressionally Directed Medical Research Programs (CDMRP) and the U.S. Army Medical Research and Development Command (USAMRDC) have increased investments in initiatives like the Traumatic Brain Injury and Psychological Health Research Program (TBIPHRP). These efforts aim to advance early detection, treatment, and prevention strategies, while also addressing the psychological effects of TBI. Coupled with growing commercialization potential and venture capital interest, this influx of funding is accelerating innovation and reshaping the TBI biomarker landscape in the United States.
Market Concentration & Characteristics
The U.S. TBI biomarkers market is witnessing high innovation, particularly in blood-based and imaging biomarker technologies. Advances in proteomics, genomics, and metabolomics are enhancing early detection and monitoring. For instance, in April 2024, the U.S. Army Medical Materiel Development Activity (USAMMDA) and Abbott announced that the U.S. Food and Drug Administration (FDA) granted clearance for the traumatic brain injury (TBI) cartridge designed for use with whole blood on Abbott’s diagnostic device. These innovations accelerate non-invasive, rapid diagnostics, improving clinical decisions and patient outcomes across emergency and long-term care settings.
Mergers and acquisitions are moderately active in the TBI biomarker space, as companies aim to broaden diagnostic portfolios and gain access to proprietary technologies. Strategic partnerships between biotech firms and academic institutions further drive consolidation, fostering competitive advantages through integrated diagnostic platforms and expanding reach in trauma and neurology sectors. In October 2024, Roche partnered with the U.S. Biomedical Advanced Research and Development Authority (BARDA) to develop innovative blood-based biomarker assays for diagnosing and monitoring acute TBI.
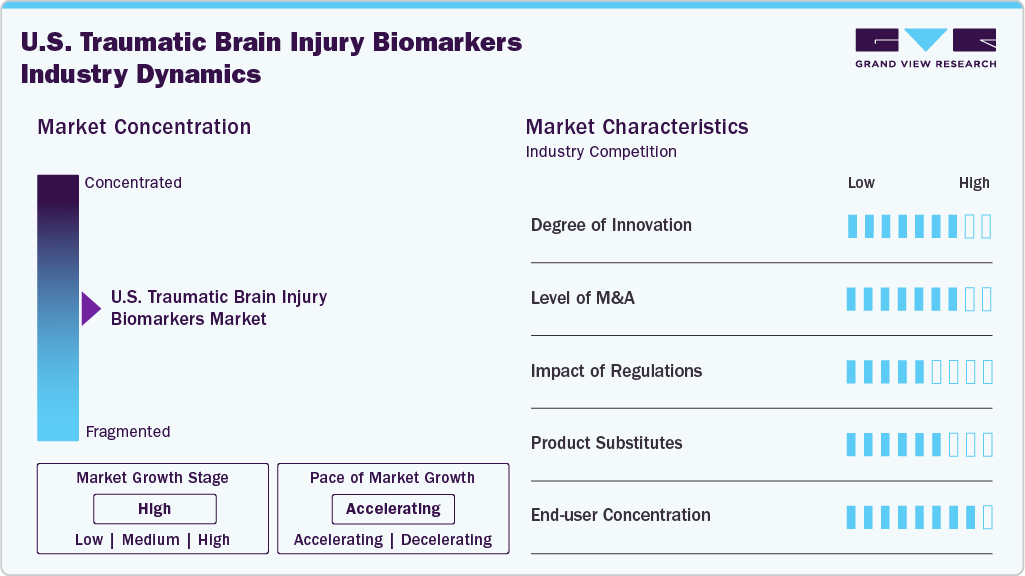
Regulatory oversight from agencies like the FDA plays a critical role in market dynamics. While stringent requirements ensure patient safety and test reliability, they can delay product launches. However, recent efforts toward accelerated approval pathways and breakthrough device designations are helping innovative TBI biomarker tests reach the market more efficiently.
CT scans and MRIs remain common for TBI assessment, and emerging biomarker diagnostics offer faster, less invasive alternatives. Biomarker tests provide valuable physiological insights that are absent from imaging. However, reliance on traditional imaging in clinical workflows can still limit immediate substitution, making integration essential rather than outright replacement in most care settings.
The market’s end users are primarily in trauma centers, emergency departments, and neurological research institutions. Academic hospitals and military healthcare facilities also form a major base due to the prevalence of veterans. This concentrated demand helps drive focused product development but may limit broader adoption without expanded clinical education.
Potential of Biomarkers in Paediatric Traumatic Brain Injury
-
Early and Accurate Diagnosis and Reducing Unnecessary Imaging: Biomarkers like GFAP and UCH-L1 can be detected in blood shortly after injury, facilitating swift identification of TBI.
-
Prognostic Value and Tailored Rehabilitation: Identifying children at risk for prolonged recovery can inform personalized rehabilitation strategies.
-
Monitoring Recovery: Serial measurements of biomarkers can monitor the healing process and effectiveness of therapeutic interventions.
-
Pathophysiological Insights: Studying biomarkers contributes to a deeper understanding of TBI mechanisms in the developing brain.
-
Facilitating Clinical Trials: Biomarkers can help categorize patients based on injury severity, improving the design and efficacy of clinical trials.
Type Insights
Protein biomarkers are represented as the dominant segment with a share of 55.72% in 2024, driven by their clinical utility, technological advancements, and growing adoption in research and clinical settings. Biomarkers, including glial fibrillary acidic protein (GFAP), S100 calcium-binding protein B (S100B), ubiquitin carboxy-terminal hydrolase L1 (UCH-L1), tau, and neurofilament light chain (NfL), offer rapid, non-invasive, and quantifiable insights into brain injury severity and prognosis. Their ability to detect neuronal and glial damage, which is often missed by traditional imaging, has made them indispensable in emergency care, sports medicine, and military applications.
Metabolomic biomarkers are emerging as the fastest-growing segment in the U.S. market. They are driven by their potential to provide comprehensive insights into the biochemical alterations following brain injuries. Unlike traditional protein biomarkers, metabolomic profiling captures dynamic changes in small-molecule metabolites, offering a more nuanced understanding of TBI pathophysiology. Recent studies have identified specific metabolite patterns associated with TBI severity, suggesting their utility in early diagnosis and prognosis. Advancements in analytical technologies have enhanced the sensitivity and specificity of metabolomic analyses, facilitating their integration into clinical settings.
Sample Type Insights
Blood-based biomarkers have emerged as the largest and fastest segment in the U.S. market, with a share of 63.69% in 2024, driven by their non-invasive nature, rapid turnaround times, and growing clinical utility. These biomarkers objectively measure brain injury severity and prognosis within minutes, facilitating timely decision-making in emergency settings and potentially reducing unnecessary CT scans. Research and development of blood-based biomarkers is accelerating, driven by increasing industrial collaborations to advance diagnostic precision and clinical utility. In May 2024, Danaher announced a collaboration with Johns Hopkins University to advance neurological diagnostics by evaluating novel blood-based biomarkers to improve the early and accurate detection of mild traumatic brain injury (TBI).
This growth is fueled by advancements in detection technologies, such as immunoassays and mass spectrometry, which have enhanced the sensitivity and specificity of biomarker analyses. The increasing prevalence of TBIs, coupled with the demand for personalized medicine and precision diagnostics, underscores the pivotal role of blood-based biomarkers in transforming TBI management and patient care in the U.S.
Application Insights
In the U.S. market, the diagnosis segment stood as the largest segment with a revenue of 56.25% in 2024, driven by the critical need for rapid, accurate, and non-invasive assessment tools. Traditional imaging methods like CT scans and MRIs, while standard, often fall short in detecting subtle or diffuse brain injuries. A research team from the University of Texas Medical Branch in October 2024 that was selected for the prestigious National Science Foundation’s National I-Corps program in recognition of their innovative work in traumatic brain injury (TBI) detection and monitoring. They developed a cutting-edge platform designed for the rapid detection and quantification of tissue and organ damage, potentially transforming the field of injury diagnostics. Adopting these diagnostic biomarkers is particularly significant in emergency and trauma care settings, where timely decision-making is crucial. Their integration into clinical practice not only enhances diagnostic accuracy but also reduces the need for unnecessary imaging, thereby improving patient outcomes and optimizing healthcare resources.
Monitoring treatment response is emerging as the fastest-growing segment in 2024, driven by the increasing demand for precision medicine and real-time evaluation of therapeutic efficacy. Biomarkers enable clinicians to assess how patients respond to treatment, track disease progression, and adjust care strategies promptly. This is particularly crucial in TBI, where recovery varies widely among individuals. Advancements in biomarker technologies, such as longitudinal blood-based assays, enhance the ability to monitor subtle neurological changes over time. As a result, this segment is gaining traction across clinical trials and personalized treatment planning.
End Use Insights
Research institutes have emerged as the largest segment in 2024, with a market share of 38.71%, driven by their pivotal role in advancing biomarker discovery and validation. Institutions such as the Center for BrainHealth at the University of Texas at Dallas are at the forefront, conducting extensive research on TBI, including studies on cognitive and social outcomes, memory deficits, and genetic factors influencing recovery. These centers are instrumental in developing and refining diagnostic tools and therapeutic strategies, often in collaboration with clinical partners. The substantial funding allocated to these research initiatives underscores their significance in shaping the future of TBI diagnosis and treatment. As the demand for precise and early detection methods grows, the contributions of research institutes remain central to the evolution of the TBI biomarker landscape.
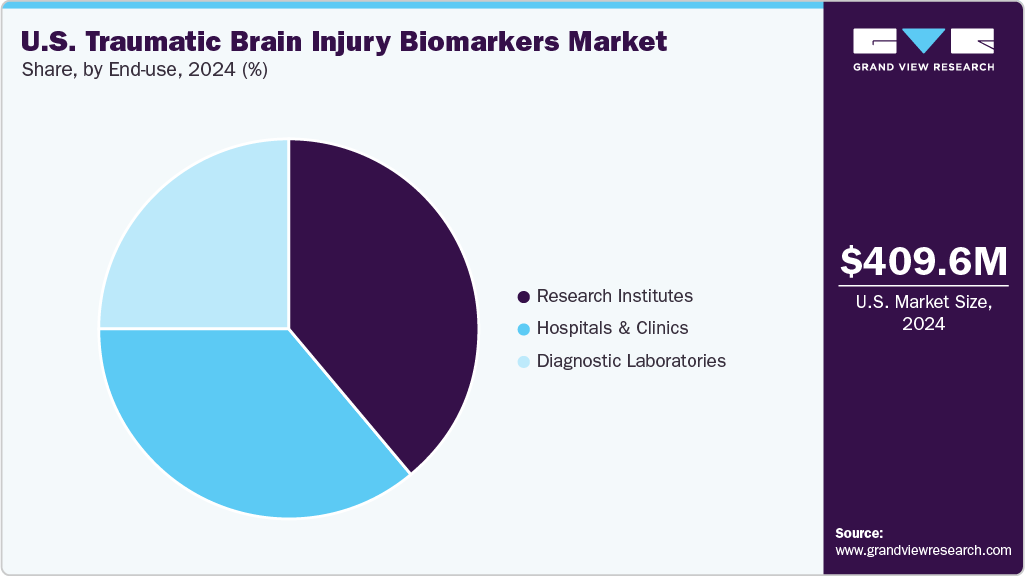
Hospitals and clinics are emerging as the fastest-growing segment in 2024, driven by their pivotal role in early diagnosis, acute care, and long-term monitoring of TBI patients. These healthcare settings are increasingly adopting advanced diagnostic tools, such as blood-based biomarker tests, to enhance the accuracy and speed of TBI assessments. For instance, in April 2024, the FDA-approved Abbott i-STAT Alinity test enables rapid detection of brain-specific proteins like GFAP and UCH-L1, facilitating timely decision-making in emergency departments and reducing unnecessary CT scans for bedside patients. Integrating such technologies in hospitals and clinics improves patient outcomes and streamlines clinical workflows, contributing to the segment's rapid growth.
Country Insights
The U.S. traumatic brain injury (TBI) biomarker market is experiencing significant growth, propelled by increasing TBI incidence, advancements in diagnostic technologies, and substantial investments in research and development. Key drivers include the demand for rapid, accurate diagnostics and the integration of artificial intelligence in biomarker analysis. Prominent players such as Abbott and BrainScope, offering EEG-based assessment tools, are leading innovations. Collaborations between research institutions, industry, and government funding further accelerate market expansion. These developments enhance early detection and personalized treatment strategies, positioning the U.S. as a pivotal contributor to TBI biomarker advancements.
Key U.S. Traumatic Brain Injury Biomarkers Company Insights
Leading U.S. traumatic brain injury biomarker market companies include Quanterix Corporation, Abbott Laboratories, Illumina, GE Healthcare, Qiagen NV, and Merck KGaA. Integrating advanced technologies such as artificial intelligence, next-generation sequencing, and liquid biopsy significantly improves diagnostic precision and speed. These advancements highlight the market's evolving landscape, where both established players and innovative solutions are driving future growth and transformation.
Key U.S. Traumatic Brain Injury Biomarkers Companies:
- Quanterix
- Banyan Biomarkers, Inc.
- Abbott
- GE Healthcare
- Thermo Fisher Scientific Inc.
- Immunarray
- Oculogica
- NeuroTrauma Sciences, LLC
- BRAINBox Solutions, Inc.
- Myriad Genetics, Inc.
- Abcam Limited
Recent Developments
-
In April 2025, Researchers at the University of Texas at San Antonio (UTSA) are developing a groundbreaking tool to enhance treatment plans for traumatic brain injuries (TBIs). It focuses on identifying TBI subtypes in veterans and analyzing their associations with biomarkers, behavioral patterns, and clinical outcomes to facilitate more personalized and effective rehabilitation strategies.
-
In April 2024, the USFDA announced the launch of Abbott's i-STAT TBI Plasma test. This test was the first rapid test on a portable analyzer to receive FDA 510(k) clearance and was developed in collaboration with the U.S. Department of Defense (DoD). It can help determine the need for a CT scan.
U.S. Traumatic Brain Injury Biomarkers Market Report Scope
Report Attribute
Details
Market size value in 2025
USD 468.06 million
Revenue forecast in 2033
USD 1,854.73 million
Growth rate
CAGR of 18.8% from 2025 to 2033
Actual data
2021 - 2024
Forecast period
2025 - 2033
Quantitative units
Revenue in USD million/billion and CAGR from 2025 to 2033
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Type, sample type, application, end use
Country scope
U.S.
Key companies profiled
Quanterix; Banyan Biomarkers, Inc.; Abbott, GE Healthcare; Thermo Fisher Scientific Inc.; Immunarray; Oculogica; NeuroTrauma Sciences, LLC; BRAINBox Solutions, Inc.; Myriad Genetics, Inc.; Abcam Limited.
Customization scope
Free report customization (equivalent up to 8 analyst’s working days) with purchase. Addition or alteration to country, & segment scope
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
U.S. Traumatic Brain Injury Biomarkers Market Report Segmentation
This report forecasts revenue growth at country levels and provides an analysis of the latest industry trends in each of the sub-segments from 2021 to 2033. For this study, Grand View Research has segmented U.S. traumatic brain injury biomarkers market report based on type, sample type, application, and end use:
-
Type Outlook (Revenue, USD Million, 2021 - 2033)
-
Protein Biomarkers
-
Genetic Biomarkers
-
Metabolomic Biomarkers
-
-
Sample Type Outlook (Revenue, USD Million, 2021 - 2033)
-
Blood-based
-
Cerebrospinal fluid (CSF)-based
-
Urine-based
-
-
Application Outlook (Revenue, USD Million, 2021 - 2033)
-
Diagnosis
-
Prognosis
-
Monitoring treatment response
-
-
End Use Outlook (Revenue, USD Million, 2021 - 2033)
-
Hospitals & Clinics
-
Diagnostic Laboratories
-
Research Institutes
-
Frequently Asked Questions About This Report
b. The U.S. traumatic brain injury biomarkers market size was estimated at USD 409.60 million in 2024 and is expected to reach USD 468.06 million in 2025.
b. The U.S. traumatic brain injury biomarkers market is expected to grow at a compound annual growth rate of 18.78% from 2025 to 2033 to reach USD 1,854.73 million by 2033.
b. Protein biomarkers represented as dominant segment with the share of 55.72% in 2024 in the U.S. traumatic brain injury (TBI) biomarker market, driven by their clinical utility, technological advancements, and growing adoption in both research and clinical settings. Biomarkers, including glial fibrillary acidic protein (GFAP), S100 calcium-binding protein B (S100B), ubiquitin carboxy-terminal hydrolase L1 (UCH-L1), tau, and neurofilament light chain (NfL), offer rapid, non-invasive, and quantifiable insights into brain injury severity and prognosis.
b. Some key players operating in the U.S. traumatic brain injury biomarker market include Quanterix, Banyan Biomarkers, Inc., Abbott, GE Healthcare, Thermo Fisher Scientific Inc., Immunarray, Oculogica, NeuroTrauma Sciences, LLC, BRAINBox Solutions, Inc., Myriad Genetics, Inc., Abcam Limited.
b. The U.S. traumatic brain injury (TBI) biomarkers market is expanding significantly due to increasing demand of pediatric TBI diagnosis, growing awareness and funding related to brain injury and accelerating collaboration between academic institutions and industry. Traumatic Brain Injury (TBI) refers to an unintentional impact to the head that can lead to outcomes ranging from mild impairment to severe neurological damage or even death
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










